Abstract
Background
Depression, posttraumatic stress disorder (PTSD), and suicidality are more prevalent among people with seizures, but few studies exist in low-resourced minoritized populations. Existing knowledge of the overlapping neurocircuitry between seizure activity in limbic regions (eg, medial temporal lobe epilepsy) and PTSD also suggests that people with seizure histories may exhibit PTSD-like alterations in their fear-potentiated startle (FPS) responses. However, this hypothesis has yet to be systematically tested. Here, we leveraged a large study on stress and trauma to evaluate the prevalence of psychiatric conditions and FPS responses in a low-resourced minoritized population of individuals with a history of seizures.
Methods
Prevalence of self-reported PTSD symptoms, depression symptoms, suicidality, and history of suicide attempt were compared between people with and without self-reported seizures among a sample of 3012 predominantly racially marginalized and low-resourced civilians. We compared FPS responses in 13 people with a history of seizures to 13 rigorously matched seizure-free controls. FPS responses were measured using eyeblink data collected during a fear conditioning acoustic startle task.
Results
Participants with a history of seizures showed significantly higher prevalence of depression symptoms, suicidality, history of suicide attempt, PTSD symptoms, and probable PTSD diagnosis than seizure-free controls, even after controlling for trauma load. Those with a history of seizures displayed heightened FPS responses and impaired fear discrimination which mimics that of PTSD.
Conclusion
These data concur with prior literature on people with a history of seizure activity being at higher risk for psychiatric symptomatology. Additionally, this study provides novel insights on the psychophysiological fear response in trauma-exposed people with a history of seizures, which could inform the identification and treatment of psychiatric vulnerability in these individuals.
Introduction
An estimated one in ten U.S. adults will experience a seizure in their lifetime, and these periods of abnormal neural activity are associated with heightened prevalence of psychiatric comorbidities in people with seizures or epilepsy.1–4 However, many studies of these psychiatric comorbidities are limited in their generalizability due to small sample size and limited demographic representation. People with epilepsy are more likely to have experienced a traumatic event and exhibit posttraumatic stress disorder (PTSD) symptoms than the general population. 5 Greater trauma exposure for people with epilepsy may be attributable to the safety hazards that seizures pose, 6 as well as seizures themselves being traumatic and life-threatening experiences. 5 The greatest levels of trauma exposure and PTSD are reported for people who live in under-resourced, urban communities, 7 but this group has long been underrepresented in psychiatric and neurological research. Existing literature suggests that PTSD and epilepsy are interrelated, therefore it is critical to understand how these conditions interact in a demographic with high civilian trauma exposure and ongoing stressors. Prior work has also suggested heightened risk of depression and suicidality in people with epilepsy and similarly needs to be addressed in a large analysis of seizure history and psychiatric comorbidity in a low-resourced, minoritized population who experience chronic stress. 8
Seizure activity in temporal lobe regions may affect fear neurocircuitry and thus could be the cause for heightened PTSD diagnoses in people with medial temporal lobe epilepsy (mTLE), one of the most common forms of epilepsy. This is supported by evidence that psychiatric comorbidities are implicated most strongly in mTLE as opposed to other epilepsy types. 3 A case report of a patient whose temporal lobe seizures began to involve reexperiencing symptoms post-trauma hypothesized that the patient's limbic seizures were “exploiting” fear circuitry because of the overlap in neural networks responsible for producing temporal lobe seizures and PTSD symptomatology. 9 The neurobiological processes underlying PTSD symptomatology are interrelated; however, studies have consistently shown that the function and structure of medial temporal lobe regions, primarily the amygdala and hippocampus, are altered in PTSD.10–14 Other studies have documented differences in amygdala activation in response to fearful stimuli between people with mTLE and controls.15,16 Rodent studies also suggest that seizure activity in the amygdala impairs fear learning 17 and leads to heightened acoustic startle responses (ASRs) compared to control rodents. 18 Additional studies examining the fear-potentiated startle (FPS) response, which refers to the contraction of the orbicularis oculi muscle in response to sudden, loud acoustic stimuli during fear conditioning paradigms, 19 have observed hyperarousal and heightened FPS responses across various stimuli in people with PTSD.20–23
Based on the summarized findings, it is plausible that seizure activity, whether localized to medial temporal structures or more generalized, may alter connections to and from the amygdala as well as other brain structures responsible for vigilance and arousal, thus resulting in a FPS response that is similar to people with PTSD. However, few studies have examined the FPS response in individuals with a history of seizures. A better characterization of FPS responses in people with seizures, and how these FPS responses compare to those of PTSD, may provide insight on which underlying neurobiological processes may be shared by the two conditions.
Since prior literature suggests that seizures originating in limbic regions could impact neural networks involved in PTSD, our first aim was to determine whether there is an increased level of psychiatric comorbidities in people with seizures compared to people without seizures using self-reported seizure and psychological symptom data from a low-resourced, minoritized population with high levels of trauma exposure. Our second aim was to explore whether people with a history of seizures, when rigorously matched with people without seizures, would exhibit alterations in their FPS responses that mirror alterations present in PTSD. We hypothesized that people with a history of seizures would exhibit a significantly higher prevalence of psychiatric comorbidities like PTSD and heightened FPS responses compared to people without seizures.
Methods
Clinical Data Collection
Demographic and Clinical Data Collection
Data were collected as part of the Grady Trauma Project (GTP), a large ongoing study of the effects of civilian trauma on mental health in people accessing care at Grady Memorial Hospital, the largest and only safety-net hospital in Atlanta, GA. Self-report psychological symptom data were collected via interviews with patients receiving non-psychiatric care in medical clinics within Grady Memorial Hospital from 2005 to 2022. 24 These hour-long interviews were conducted by trained interns and participants were compensated between $15 and $40 (compensation changed over the course of the study). Approval of study procedures was provided by the Institutional Review Board of Emory University and the Research Oversight Committee of Grady Memorial Hospital (IRB# 00010651 and 00078593).
In participant interviews, a demographics questionnaire was used to determine participants’ age, sex, ethnicity, race, employment status, and pregnancy/breastfeeding status. The Beck Depression Inventory (BDI), a 21-item measure that queries depression symptoms over the two-week period prior to their interview (score range = 0 - 63), was utilized in the present study to measure depression symptoms (details in Supplementary Methods).25,26 The ninth item on the BDI asks participants to select the statement that best describes current suicidality from the following choices: “I don't have any thoughts of killing myself”, “I have thoughts of killing myself, but I would not carry them out”, “I would like to kill myself”, and “I would kill myself if I had the chance”. Responses to this question were used to measure the level of suicidal ideation (ie, suicidal wishes), whereas responses to the survey question “Have you ever attempted suicide?” were used to determine history of suicide attempt. The Modified PTSD Symptom Scale (mPSS) is an 18-item scale that was administered to participants to assess PTSD symptoms and probable diagnoses based on DSM-IV criteria (score range 0-54, details in Supplementary Methods).27,28 Participants were considered to have a probable, current PTSD diagnosis if they reported experiencing at least one item in the intrusions symptom cluster, at least three items in the avoidance/numbing symptom cluster, and at least two items in the hyperarousal symptom cluster for at least one month on the mPSS. Use of the mPSS to assess probable, current PTSD diagnosis has been validated in previous literature showing that these criteria correlate with diagnosis per the more extensive clinician-administered PTSD scale (CAPS) interview, with sensitivity and specificity rates up to 89% and 77%, respectively.29,30 The Traumatic Events Inventory (TEI), which is a 19-item questionnaire developed by our team to query exposure to a comprehensive range of traumatic experiences,31,32 was administered to measure lifetime trauma (score range = 0 - 19), representative of the total number of different types of traumas experienced, witnessed, or confronted. The Childhood Trauma Questionnaire (CTQ) is a 28-item measure that was administered to participants to approximate the level of exposure to traumatic events in childhood (score range: 25 - 125), specifically between the ages of 0-18.33,34
Large-Sample Psychological Symptom Data Collection and Analyses
Participants for Large-Sample Psychological Symptom Analyses
For our first aim, history of seizures was assessed during interviews using the yes/no question: “Have you ever had seizures, or have you ever been diagnosed with epilepsy?”. Interview data from all participants in the GTP dataset who had responded to the seizure/epilepsy status question and had completed at least one psychological symptom measure were utilized. Since our designation of seizure status relies on self-report which could include individuals with functional/psychogenic seizures and/or epilepsy, those who reported having a history of seizures/epilepsy will be referred to as the history of seizures group in our analyses. The demographic breakdown of all participants included in this aim is detailed in Table 1. Due to participant availability during interviews, certain demographic information collected may be incomplete. BDI data were available for N = 2959, suicidal ideation data (ninth item of BDI) were available for N = 2967, history of suicide attempt data were available for N = 3008, mPSS data were available for N = 2670, probable, current PTSD diagnosis data were available for N = 2675, TEI data were available for N = 2994, and CTQ data were available for N = 2984.
Demographics and Psychological Symptom Scores for Large-Sample Participants with and without a History of Seizures. Mean ± standard error of the mean (SEM) for continuous demographic variables (BDI total, mPSS total, age, CTQ total, TEI total), as well as proportions of categorical demographic variables (suicidal ideation, history of suicide attempt, PTSD diagnosis, sex, race, employment status) for participants included in psychological symptom data analyses, broken down by seizure/epilepsy status. F values (df) are reported for all continuous variables, and χ2 values are reported for all categorical variables. Some participants did not respond to all demographic questions. Number of respondents for each demographic variable are as follows: sex (N = 3009), age (N = 3010), race (N = 3008), employment status (N = 3008), CTQ (N = 2984), TEI (N = 2994), BDI (N = 2959), mPSS (N = 2670), suicidal ideation (N = 2967), history of suicide attempt (N = 3008), and PTSD diagnosis (N = 2675). Significant differences (P < .05,

Statistical Analyses of Large-Sample Psychological Symptom Data
Chi-square analyses were conducted to compare our categorical variables of interest (history of suicide attempt, PTSD diagnosis, depression diagnosis, suicidal ideation, race, sex, and employment status) between participants with and without self-reported seizures. One-way analysis of variance (ANOVA) tests were conducted to compare our continuous variables of interest (mPSS total score, BDI total score, CTQ total score, TEI total score, and age) between participants with and without self-reported seizures.
Sensitivity analyses were conducted to evaluate the impact of trauma exposure on psychiatric symptoms. For each of the clinical outcome measures (except for suicidal ideation), logistic or linear regression analyses were performed with seizure status as the predictor and childhood and lifetime trauma as covariates. For each analysis, only data from participants for whom we had both seizure status and the outcome or demographic variable(s) of interest were included.
Fear-Potentiated Startle (FPS) Response Data Collection and Analysis
FPS Participants
For our second aim, nine of the participants who reported experiencing seizures had also completed the FPS task. These nine participants were matched with nine participants who reported not experiencing seizures and had completed the FPS task. A rigorous matching procedure was used within our database of N = 251 with FPS procedures. Participants were matched on variables of race, sex, history of suicide attempt, PTSD diagnosis, pregnancy status, ethnicity, age, depression symptoms, childhood trauma exposure, and lifetime trauma exposure using successive runs of the case-control matching function in SPSS (Version 28.0.1.0) (details in Supplementary Methods). Additionally, four participants were recruited through a GTP study in collaboration with the Emory Comprehensive Epilepsy Program between 2017-2020 and included in our FPS analyses. Due to clinical circumstances, these participants required removal of the amygdalohippocampal complex via laser ablation to treat their mTLE. 35 Before surgery, these participants participated in the same FPS paradigm and clinical interviews as the history of seizures group described above and were matched to four seizure-free controls. Thus, 13 participants with a history of seizures (nine from the history of seizures cohort and four from the amygdala ablation cohort) and 13 matched participants without seizures were included in the FPS analyses. The demographic breakdowns of these 26 participants are detailed in Table 2. Across groups, probable, current PTSD diagnosis was determined for the history of seizures group (N = 9) based on the mPSS and DSM-IV criteria whereas the DSM-5 CAPS interview yes/no question for PTSD diagnosis was used for the amygdala ablation group (N = 4). To explicitly assess the effects of having a history of seizures on FPS responses, we excluded individuals who had current PTSD diagnoses from our analyses.
Demographics and Psychological Symptom Scores for Participants Included in Fear

Startle Procedure
Startle procedures used in the present study follow prior work (Figure 1).35–38 Participants were presented with various stimuli throughout the startle procedure: an aversive air puff to the larynx (unconditioned stimulus “US”), a colored shape that was paired with presentation of the US (CS+, ie, threat cue) as well as a startling noise burst, and a different colored shape that was paired with the startling noise but not the US (CS-, ie, safety cue). Before the start of the startle procedure, two Ag/AgCl electrodes were attached over participants’ right orbicularis oculi muscle along with a ground electrode which was positioned at the mastoid bone. Participants were then instructed to sit in a booth facing a computer screen and wear both a set of headphones and a vest with an air puff dispenser apparatus strapped to their chest.

Schematic of fear-potentiated startle procedure. Participants first underwent a brief habituation phase, wherein two different shapes (the CS+ and CS-) were presented with the startle noise probe (106-dB [A] SPL, lasting 40 ms) without the US. Following habituation, participants were presented with a three-block series of CS+ (blue square) / CS- (purple triangle), startle tone, and US. Each of the three blocks consisted of four startle noise alone (NA) trials, CS+ trials, and CS- trials. During CS+ trials, participants were presented with the CS+ shape, then the startle probe, and then finally the US air puff (140 p.s.i) to their larynx lasting 250 ms. During CS- trials, participants were presented with the CS- shape and then the startle probe (no aversive air puff). During NA trials, participants were presented with only the startle probe (no shape or US). The shape remained on the screen for the entire duration of the trial (6 s). Participants thus went through 51 startle trials, with each trial being separated by a randomized time interval (9-22 s).
Participants first underwent a brief habituation phase, wherein two different shapes (the CS+ and CS-) were presented with the startle probe (106-dB [A] SPL, white noise burst lasting 40 ms) but without the US. Following habituation, participants were presented with a three-block series of CS+/-, startle noise, and US presentation. Each of the three blocks consisted of four startle noise alone (NA), CS+, and CS- trials. During the CS+ trials, participants were presented with the CS+ shape for six seconds, then the startle probe, and finally the US air puff (140 p.s.i) to their larynx lasting 250 ms. During CS- trials, participants were presented with the CS- shape for six seconds and then the startle probe (no aversive air puff). During NA trials, participants were presented with only the startle probe (no shape or US). Participants thus went through 51 startle trials, with each trial being separated by a randomized time interval (9-22 s).
Startle Data Processing
Biopac MP150 for Windows (Biopac Systems, Inc.) was utilized to collect participants’ electromyography (EMG) data. EMG data were sampled at 1000 Hz and amplified using the EMG module of the Biopac system. MindWare (MindWare Technologies, Ltd) was used to filter and rectify EMG data using a bandpass filter (28- 500 HZ). The peak EMG amplitude 20-200 ms following the onset of the probe was quantified as the ASR. EMG data for each participant were used to calculate the FPS response for each cue of each block of the startle task, calculated by taking the difference between participants’ startle response to CS trials and participant's startle response to NA trials in a given block, dividing that difference once again by their startle response to NA trials in that block, and then multiplying the resulting value by 100%: FPS Response = (EMG startle response during CS (+ or -) trials – NA startle)/(NA startle)×100. To assess differences in early versus late fear learning, we averaged FPS responses across blocks two and three (late acquisition, ACQ) and compared that to block one (early ACQ).23,37,38
Statistical Analyses for FPS Data
Repeated measures general linear model analyses were performed for the interaction between CS type (CS+, CS-), block (early vs late ACQ), and seizure status to determine if there were differences in FPS responses based on participants’ seizure status, task block, and/or cue type. Follow-up analyses were performed if statistically significant interactions were observed. Two-sided P < .05 was used for all analyses except CS type. Following the strong directional hypothesis that CS+ is expected to be greater than CS-, one-sided P < .05 was used for CS type post-hoc analyses.
Results
Psychological Symptoms, Trauma Exposure, and Seizure Status
Participants with self-reported seizures and controls did not differ significantly on the demographic variables of race, age, and sex (see Table 1). However, a significantly higher proportion of individuals with a history of seizures were unemployed than in the control group. Participants with self-reported seizures also had significantly higher childhood and lifetime trauma exposure than controls. History of suicide attempt, suicidal ideation, higher PTSD symptom scores, higher depression scores, and probable, current PTSD diagnosis were significantly more prevalent in people with self-reported seizures than controls (Figure 2).

Psychological symptom scores for large-sample participants with and without a history of seizures. Columns (± SEM) in gray represent the no history of seizures group and columns in blue represent the history of seizures group. Asterisks denote significant differences (P < .05).
Given greater levels of childhood and lifetime trauma levels in the history of seizures group (Table 1), sensitivity analyses were performed on psychiatric comorbidities with seizure status as a predictor and trauma measures (CTQ total and TEI total) as covariates. Seizure status still predicted PTSD symptoms, PTSD diagnosis, and history of suicide attempt after correcting for childhood trauma and lifetime trauma (Tables 3 and 4). After applying Bonferroni corrections, the increased prevalence of PTSD diagnoses was no longer significantly predicted by seizure status. Seizure status only significantly predicted BDI total when correcting for either childhood trauma (F(2, 2950) = 370.34, B = .035, P = .036) or lifetime trauma (F(2, 2950) = 328.76, B = .039, P = .021), but was not significant after controlling for both trauma exposures (Table 4).
Results of Logistic Regression Analysis for PTSD Diagnosis and Suicide Attempt History, with Seizure Status as Predictor and CTQ and TEI as Covariates. Two separate logistic regression analyses were conducted for PTSD diagnosis and suicide attempt history. χ2 (df) and Nagelkerke R² values are reported for both models. Beta values, standard errors, Wald statistics, odds ratios, and P-values are also reported for seizure status as well as both trauma measures (CTQ & TEI). Significant values (P < .05) bolded. Both CTQ (B = .02, P < .001) and TEI (B = .22, P < .001) scores were significant predictors of PTSD diagnosis; however, seizure status still significantly predicted PTSD diagnosis even with both trauma measures included in the logistic regression model (B = .35, P = .040). Both CTQ (B = .03, P < .001) and TEI (B = .14, P < .001) scores were significant predictors of suicide attempt history; however, seizure status still significantly predicted suicide attempt history even with both trauma measures included in the logistic regression model (B = .52, P = .004). *Did not survive corrections for multiple comparisons.

Results of Linear Regression Analysis for PTSD Symptoms (PSS Total Score) and Depression Symptoms (BDI Total), with Seizure Status as Predictor and CTQ and TEI as Covariates. Two separate linear regression analyses were conducted for PTSD symptoms and depression symptoms. F statistics (regression df, total df) are reported for both models. Standardized coefficients Beta and P-values are also reported for seizure status as well as both trauma measures (CTQ & TEI). Significant values (P < .05) bolded. Both CTQ (B = .25, P < .001) and TEI (B = .33, P < .001) scores explained a significant amount of the variance observed in PTSD symptom scores; however, seizure status still significantly predicted PTSD symptom scores even with both trauma measures included in the linear regression model (B = .04, P = .024). Both CTQ (B = .30, P < .001) and TEI (B = .25 P < .001) scores explained a significant amount of the variance observed in depression symptom scores. However, seizure status did not significantly predict depression symptom scores when both trauma measures were included in the same model.

Fear-Potentiated Startle Response in Individuals with a History of Seizures
Participants with self-reported seizures did not differ significantly from matched control participants on any demographic or psychological symptom variables, confirming successful matching (see Table 2). There was a significant interaction between CS type, block, and seizure status (F(24) = 7.51, P = .011). Post-hoc tests showed a significantly greater FPS response to CS+ compared to CS- for the history of seizures group during late ACQ (t(12) = 1.82, one-sided P = .047), but not early ACQ (t(12) = −.404, one-sided P = .347; d = .506), highlighting delayed discrimination of fear and safety signals (Figure 3). Correspondingly, the history of seizures group showed a significant increase in CS+ responses during late ACQ compared to early ACQ (t(12) = −3.98, P = .002; d = 1.10). Individuals with a history of seizures also exhibited larger responses to the CS+ during late ACQ compared to controls (t(24) = −1.81, one-sided P = .042; d = .709) whereas CS- responses did not significantly differ between the two groups. Control participants with no history of seizures displayed a significantly greater response to CS+ compared to CS- during early ACQ (t(12) = 1.85, one-sided P = .044; d = .514), highlighting the ability to successfully discriminate fear and safety signals during early stages of the FPS task. However, the control group did not display a significantly greater response to CS+ than CS- responses during late ACQ (t(12) = .992, one-sided P = .171; d = .275). After applying Bonferroni corrections, only the exaggerated CS+ during late compared to early ACQ remained.
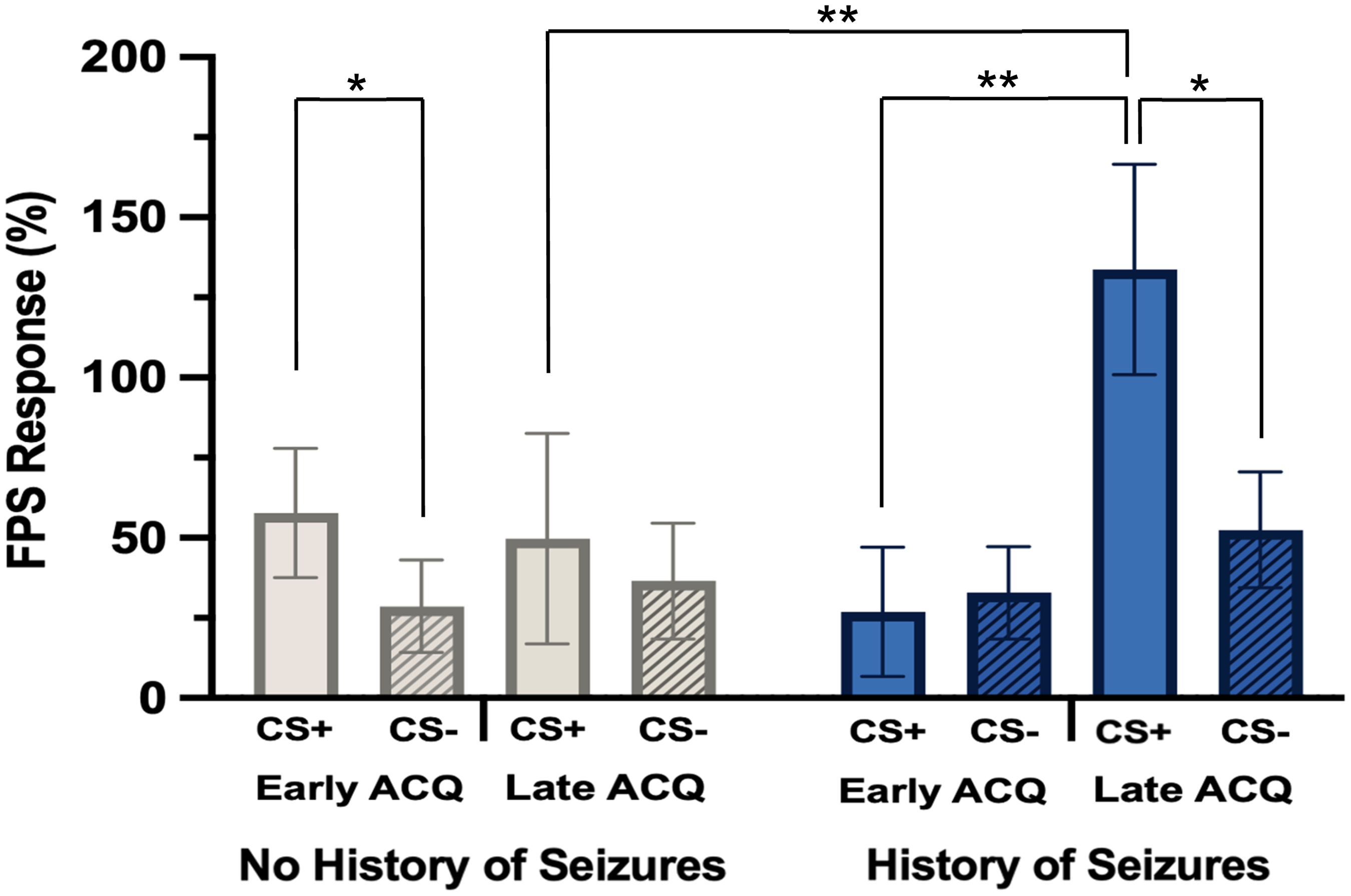
Fear-Potentiated startle response for participants with (N = 13) and without (N = 13) a history of seizures. FPS response for all participants separated by early (block one) versus late (average of blocks two and three) fear ACQ as well as CS type (CS+, CS-). There was a significant interaction between CS type, block, and seizure status (F(24) = 7.51, P = .011). The history of seizures group displayed significantly greater FPS response to CS+ compared to CS- during late ACQa (t(12) = 1.82, one-sided P = .047) and also showed a significant increase in CS+ responses during late ACQ compared to early ACQ (t(12) = -3.98, P = .002). Participants with no history of seizures displayed a significantly greater response to CS+ compared to CS- during early ACQa (t(12) = 1.85, one-sided P = .044). aDid not survive corrections for multiple comparisons. Columns represent mean FPS (%) for each condition ± SEM.
We performed sensitivity analyses to assess whether the addition of the participants who would later undergo amygdala ablation in our analyses confounded our results. We analyzed FPS responses in the history of seizures cohort alone (N = 18). FPS response patterns were comparable to the large-sample cohort (N = 26) which included the history of seizures and amygdala ablation participants (Supplemental Figure 1). Repeated measures ANOVA analyses again revealed a significant three way interaction of CS×block×seizure status (F(16) = 8.27, P = .011). Post-hoc analyses showed a significant difference between paired CS+ responses across blocks (t(17) = −2.31, P = .034; d = .545), driven by the increase in FPS responses to CS+ from early to late ACQ in the history of seizures group (t(8) = −3.72, P = .006; d = 1.24), similar to what was found across both cohorts (Figure 3). Individuals with a history of seizures also exhibited larger responses to the CS+ during late ACQ compared to controls (t(16) = −2.14, one-sided P = .024; d = 1.01) whereas CS- responses did not significantly differ between the two groups.
Discussion
Psychiatric Comorbidities in Individuals with a History of Seizures
In the current study with a large group of low-resourced, racially marginalized civilians with high levels of trauma exposure and ongoing stressors, we demonstrate that depression, suicidality, and PTSD are all more prevalent among people with a history of seizures. This finding supports our hypothesis and expands on the existing literature by validating that heightened risk for psychiatric comorbidities in people with a history of seizures is also present in predominantly racially marginalized and trauma-exposed demographics with persistent stressors. Moreover, we demonstrate that for PTSD and history of suicide attempt specifically, this heightened risk is not explained by increased trauma exposure alone, although the heightened risk for PTSD in individuals with seizures did not survive corrections for multiple comparisons. Our findings highlight the intersection between seizure history and psychiatric risk which has important implications within epilepsy clinics and psychiatry. Novel detection practices, including automated screening assessments disseminated via patients’ electronic medical records, have shown promise for the time and cost-effective identification of psychiatric comorbidities in people with epilepsy. 39 In conjunction with these screening practices, our findings could improve timeliness and specificity in psychiatric screening of comorbidity in people with epilepsy.
Despite evidence that psychiatric comorbidities are more prevalent among people with seizures, recent studies have shown that psychiatric conditions, namely depression, are undertreated in racially marginalized and low-resourced populations. 40 Individuals within these communities tend to experience greater instances of trauma exposure and are at greater risk of developing psychiatric disorders. Thus, a greater effort needs to be made to implement these detection and treatment approaches in epilepsy clinics as well as other healthcare modalities through which people with seizures receive care in racially marginalized and under-resourced areas.
Fear-Potentiated Startle Response and Seizure Status
Our scientific premise is that seizure activity could exploit and affect fear neurocircuitry resulting in heightened FPS responses. FPS phenotyping in the subsample supported our hypothesis that people with a history of seizures would exhibit characteristics of FPS comparable to the well-established heightened FPS psychophysiological response in PTSD.35–37 Participants with no history of seizures discriminated fear and safety cues well during the FPS task and their overall differences in response to CS+ and CS- reduced during late acquisition in correspondence with other studies.20,41 In contrast, individuals with a history of seizures showed delayed fear discrimination, exaggerated responses to CS+ compared to CS- during late ACQ, and an overall pattern of greater fear reactivity as commonly observed in PTSD. Due to limited power, the post-hoc findings on delayed fear discrimination did not survive when accounting for multiple comparisons. These data suggest having a history of seizures could put individuals at risk for delayed or impaired fear discrimination, as well as heightened fear load, which is typically observed in individuals with PTSD.36–38
Our startle findings further emphasize the possible common risk between seizure status and PTSD. With additional validation, we speculate that abnormal FPS responses in epilepsy patients could contribute to seizure localization in limbic regions in a manner analogous to other neuropsychometric measures. Future studies of the overlapping and disparate neurobiological mechanisms of this unique psychophysiological signature may provide insight as to what brain areas are implicated in producing a shareable risk factor for the development and exacerbation of seizures and psychiatric conditions or explain the heightened risk that people with a history of seizures or epilepsy have for developing psychiatric conditions like PTSD.
Limitations
Our study leveraged large-scale self-report data to evaluate the presence or absence of seizures and to measure psychological symptoms, which may have introduced self-report bias or variability. First, data that were used to determine participants’ seizure status did not include detailed information about seizure semiology, consistent diagnostic confirmation by video-electroencephalography (EEG), and/or other specifications about their experience of seizures. Therefore, it is likely that our history of seizures sample contains some individuals with functional or psychogenic seizures, which develop as a result of trauma/stress, who are more likely to have elevated psychiatric symptomatology.42,43 Due to our inability to parse out such participants, it is possible that psychiatric symptomatology could be over-represented in our sample. Importantly, results remained the same after removing the amygdala ablation group from the FPS analyses. Future studies may use more detailed information on seizure or epilepsy diagnoses to clarify whether associations exist between psychophysiological responses and certain epilepsy types or characteristics. We further propose that patients with mTLE should be investigated specifically with respect to the presence and nature of PTSD symptoms. Regardless, our findings indicate the importance of seizures irrespective of its origin as a factor for greater risk for psychopathology. For the group with seizure history, PTSD diagnosis was based on the mPSS and DSM-IV while the DSM-5 CAPS interview yes/no question was used for the amygdala ablation group, which presents variability in PTSD diagnostic criterion. The number of participants included in our FPS analyses were relatively small and we may have thus been underpowered to detect potential differences between FPS responses of the history of seizures and control groups. Future studies should include more participants to clarify whether there are additional differences between the FPS of both groups. This limitation is mitigated by the rigorous matching procedure maximizing the power for the analyses. Lastly, our study population limits generalizability due to the focus on trauma-exposed, racially marginalized individuals. There are other populations that experience trauma and other chronic social stressors which could benefit from research in this realm. This limitation is offset to some degree by the high-risk nature of the sample and underrepresentation of minoritized populations in epilepsy and psychiatric comorbidity research.
Conclusions
Our study demonstrates that individuals with a history of seizures are at greater risk of psychiatric comorbidities and neurobiological characteristics of PTSD. In our large trauma-exposed, predominantly racially marginalized sample with ongoing stressors, depression symptoms, PTSD, suicidality, and heightened FPS responses are disproportionately prevalent among people with seizures. Our findings serve as a first step in characterizing the psychophysiological fear response of people with seizures, deepening our understanding of potentially overlapping clinical presentations between seizure activity and PTSD which could aid in improving screening to reduce trauma- and seizure-related risk for development and exacerbation of psychiatric disorders. Our novel findings utilizing a fear conditioning paradigm highlight the shared pathological presentation of heightened hyperarousal to acoustic stimuli observed in individuals with seizure histories and PTSD, which could be useful in defining targets for neuromodulatory treatment to downregulate these responses. Future studies should evaluate these characteristics in larger sample sizes with more robust clinical characterizations of seizure and epilepsy patients.
Supplemental Material
sj-docx-1-css-10.1177_24705470251321953 - Supplemental material for Characterizing the Prevalence of Psychiatric Conditions and Fear-Potentiated Startle Response in Civilians with a History of Trauma and Seizures
Supplemental material, sj-docx-1-css-10.1177_24705470251321953 for Characterizing the Prevalence of Psychiatric Conditions and Fear-Potentiated Startle Response in Civilians with a History of Trauma and Seizures by Trinidi Prochaska, Helena Zeleke, Sean T. Minton, Abigail Powers, Vasiliki Michopoulos, Jon T. Willie, Daniel L. Drane, Tanja Jovanovic and Sanne J.H. van Rooij in Chronic Stress
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (K01 MH121653, van Rooij, SJH; R01 NS088748, Drane, DL) and the Brain and Behavior Research Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
