Abstract
Substance use disorder (SUD) is a significant health problem, and trauma exposure is a known risk factor for the escalation of substance use. However, the shared neural mechanisms through which trauma is associated with substance use are still unknown. Therefore, we systematically review neuroimaging studies focusing on three domains that may contribute to the overlapping mechanisms of SUD and trauma—reward salience, negative emotionality, and inhibition. Using PRISMA guidelines, we identified 45 studies utilizing tasks measuring these domains in alcohol, tobacco, and cannabis use groups. Greater reward, lesser regulation of inhibitory processes, and mixed findings of negative emotionality processes in individuals who use substances versus controls were found. Specifically, greater orbitofrontal cortex, ventral tegmental area, striatum, amygdala, and hippocampal activation was found in response to reward-related tasks, and reduced activation was found in the inferior frontal gyrus and hippocampus in response to inhibition-related tasks. Importantly, no studies in trauma-exposed individuals met our review criteria. Future studies examining the role of trauma-related factors are needed, and more studies should explore inhibition- and negative-emotionality domains in individuals who use substances to uncover clinically significant alterations in these domains that place an individual at greater risk for developing a SUD.
Keywords
Introduction
A substance use disorder (SUD) develops upon continued substance use despite experiencing problems due to consumption. 1 SUD is considered a severe public health crisis. About 20.4 million people in the United States were diagnosed with a SUD in the past year. 2 According to the diagnostic criteria, there are a total of 11 symptoms a person may exhibit, including those associated with impairments in inhibitory control, cravings/urges to use the substance, needing more of the substance to achieve the desired effect (tolerance), and withdrawal effects that can only be improved by taking more of the substance. 1 SUDs may be associated with adverse physical and mental health outcomes on the individual level and exert large negative consequences on the societal level. For example, SUD patients experience an increased risk of suicidal outcomes 3 and social, academic, and work impairment. 4 Societally, the US suffers billions in annual productivity loss related to lost wages and productivity, crime, and healthcare expenses due to SUD. 5 Rates of SUD have dramatically increased following the onset of the COVID-19 pandemic, 6 especially in underserved communities. 7 Thus, it is vital, now more than ever, to better understand the mechanisms that potentially underlie the development of an SUD.
A neurobiological framework for the transition from controlled drug use to unhealthy use (ie, addiction) has been proposed that is comprised of three stages: binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation. 8 In their 2016 review, the authors build on this work by outlining a heuristic framework focusing on three functional domains: reward salience, negative emotionality, and executive function, which overlap with the three-stage addiction cycle, and identify neural circuits mediating each domain. 9 The binge/intoxication (reward salience) stage highlights regions of importance, including areas of the basal ganglia, such as the nucleus accumbens, ventral tegmental area (VTA), and ventral pallidum/substantia innominata. During the withdrawal/negative affect stage (negative emotionality), the extended amygdala is important, with projections to the hypothalamus and brainstem. The preoccupation/anticipation phase is defined as deficits in executive functioning and inhibition and recruits regions such as the anterior cingulate cortex (ACC) and prefrontal cortex. 9
Trauma exposure is a common risk factor for developing or worsening substance use and SUDs.10‐14 Indeed, trauma contributes to the onset of substance misuse,15,16 and substance misuse can lead to exposure to more trauma. 17 However, the underlying mechanism by which trauma contributes to substance misuse, and vice versa, is still largely unknown. 18 Some individuals who experience trauma may develop posttraumatic stress disorder (PTSD). PTSD is a debilitating disorder with a lifetime prevalence rate of 8% in the general population 19 and is highly comorbid with SUDs. 20
In the past three decades, much work has been done to elucidate the neural correlates of PTSD, and many reviews have been published on the topic.21‐30 Classical neurocircuitry models of PTSD highlight exaggerated activation of the amygdala, followed by poor regulatory control of the amygdala from prefrontal cortical regions. 31
There have been a handful of review papers that have explored the neural overlap between PTSD and SUD.32‐34 Hien et al 34 proposed a translational framework designed to advance the creation of interventions for co-occurring PTSD and SUD through understanding the overlapping mechanisms associated with each disorder. Building upon previous work on the Research Domain Operating Criteria (RDoC) 35 and its counterpart, Alcohol and Addiction Research Domain Criteria (AARDoC), 36 the authors highlight the functional domains proposed by Koob and Volkow, 9 providing an overview of studies that assess behavioral deficits and biological alterations associated with each domain separately for PTSD, SUD, and co-occurring PTSD + SUD. One limitation of Hien and colleagues’ review is the lack of an in-depth synthesis of the discussed neuroimaging studies exploring each functional domain. Given the many new developments in the neural mechanisms on this topic, a systematic review of neuroimaging findings related to the functional domains highlighted by Koob and Volkow 9 is warranted to explain the neural mechanisms underlying SUD development and explore their potential implications in trauma-exposed samples.
This review systematically synthesizes neuroimaging studies focusing on three domains that may contribute to the overlapping mechanisms of SUD and PTSD—reward salience, negative emotionality, and executive function. Within this framework, we separately review studies that either used or did not use explicit measures of trauma exposure, enabling us to draw conclusions from and identify knowledge gaps for each population. Lastly, previous research has found alcohol, cannabis, and tobacco use to be the most used substances by trauma-exposed individuals. 37 As such, we will focus on these three substances within the review. After reviewing the neuroimaging literature, we suggest future research integrating these findings with trauma.
Methods
The Supplemental Materials provide a brief overview of the neuroimaging techniques used and an in-depth description of each pathway, including regions of interest (ROIs).
Eligibility
The systematic review was conducted according to the guidelines set by the Preferred Report Items for Systematic Reviews and Meta-analysis (PRISMA), without preregistration. We conducted two literature searches. The first search focused on individuals who use substances to investigate neurobiological mechanisms of the three domains irrespective of trauma exposure. The second search focused on trauma-exposed individuals who use substances to explore potential neurobiological characteristics of the three domains related to trauma exposure. Eligibility criteria for the first literature search included: 1) original empirical reports; 2) published in English; 3) used the following neuroimaging techniques: functional magnetic resonance imaging (fMRI)—with or without blood oxygen level-dependent (BOLD)—and positron emission tomography (PET); 4) included a control group in analyses. Exclusion criteria included: 1) meta-analyses/review articles; 2) studies in languages other than English; 3) studies containing only healthy subjects; 4) studies that did not contain a control group; 5) studies that did not make group comparisons; 6) studies that utilized long-term abstinent experimental groups; 7) studies using adolescent or child samples; 8) experimental group was a treatment-seeking sample; 9) intervention or substance was administered during imaging procedures; 10) structural imaging techniques were used; 11) family history of substance use was explored, but not personal substance use in the experimental group; 12) animal studies; and 13) tasks that do not measure any of the three domains was not used. Eligibility and exclusion criteria for the second literature search were like the first, including 14) a group of trauma-exposed individuals who use substances as an eligibility criterion.
Search Strategy
For the first literature search, we used PubMed, Web of Science, and Google Scholar and a combination of the following terms and Booleans in the title/abstract of articles: (inhibition OR impulsivity OR impulsiveness); (reward); (anxiety OR anhedonia OR fear OR threat) AND (substance use disorder OR substance abuse OR substance use OR substance misuse OR alcohol abuse OR alcohol use disorder OR alcohol use OR addiction) AND (neuroimaging OR functional magnetic resonance imaging OR fMRI OR functional MRI OR positron emission tomography OR PET). For the second literature search, we used the same databases, including the combination of the above terms, with the addition of AND (PTSD OR trauma OR posttraumatic stress disorder).
Study Selection
Four independent reviewers (CAH, JZ, JB, and SVR) screened study titles and abstracts for inclusion with a consensus on selection criteria. Data extraction forms were extracted by three reviewers (JZ, JB, and SS) and checked by two reviewers (CAH and SVR). All reviewers resolved any remaining inconsistencies.
The following information was gathered from the articles: sample size for each experimental and control group, the biological sex breakdown of each group, the mean age for each group, study design and methodology, and direction of activation for the brain regions of interest (ie, brain areas activated related to the three proposed domains).
Results
Study Characteristics
Refer to Figure 1 for the PRISMA diagram with a breakdown of the literature reviewed across all domains for the first literature search. In all, 45 articles met the inclusion criteria outlined above (n = 1195 substance use, n = 1291 controls), published from 2001 to 2022. Of the 45 studies, 30 were specific to the reward literature, nine to the inhibition literature, and six to the anxiety sensitivity literature. One study was included for both reward and inhibition domains. For the reward domain, of the 31 studies, 14 focused on alcohol (n = 412 individuals that use alcohol, n = 398 controls), 12 on tobacco (n = 224 individuals that use tobacco, n = 231 controls), and five on cannabis (n = 142 individuals that use cannabis, n = 192 controls). For the inhibition domain of the nine studies, five focused on alcohol (n = 151 individuals that use alcohol, n = 168 controls), and four on tobacco (n = 92 individuals that use tobacco, n = 86 controls) as the substance of interest. Lastly, for the negative emotionality domain, of the six studies, four focused on alcohol (n = 131 individuals that use alcohol, n = 177 controls), one on tobacco (n = 28 individuals that use tobacco, n = 28 controls), and one on cannabis (n = 28 individuals that use cannabis, n = 23 controls) as the substance of interest.

Flow diagram of literature search results from identification to inclusion for studies where trauma exposure is not measured.
For studies with more than one comparison group,38–49 we report any significant findings between the substance use group and one of the control groups. One study in the reward domain examined twin pairs discordant for cigarette smoking. 38 For this study, we chose the comparison of individuals who regularly smoked versus those who did not regularly smoke but not their co-twins. One study in the negative emotionality domain included three groups, ie, individuals with co-occurring alcohol use disorder (AUD)+Anxiety, individuals with AUD-Anxiety, and healthy controls. 39 To prevent introducing a confound of psychiatric illness in our review, we excluded the findings between the AUD + Anxiety group and the other two groups. Thus, we present only findings from the AUD-Anxiety versus healthy control groups. 39
Refer to Figure 2 for the PRISMA diagram with a breakdown of the literature reviewed across all domains for the second literature search. No articles met the inclusion criteria to be included in the review. A breakdown of reasons we excluded articles is included in Figure 2.
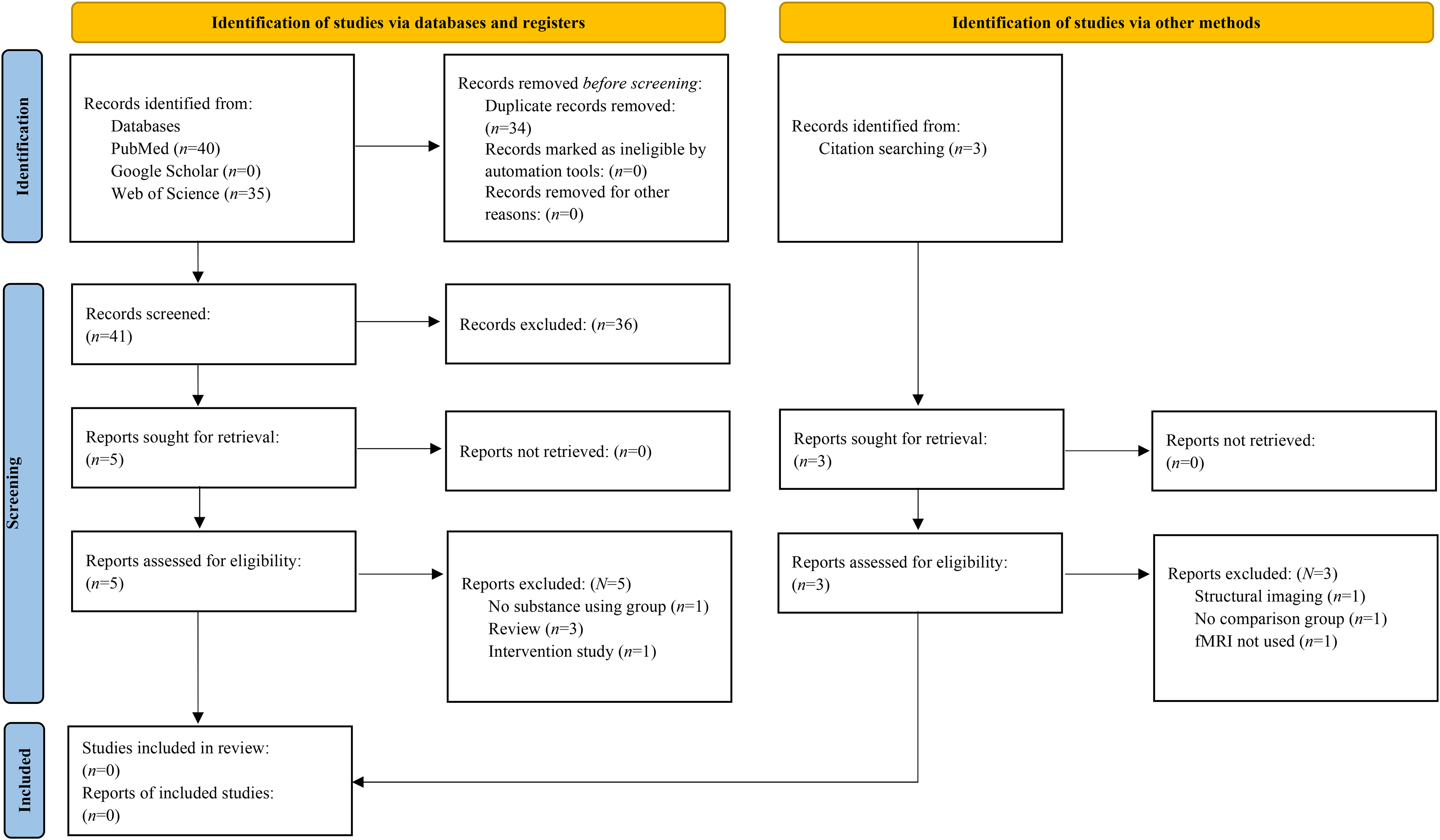
Flow diagram of literature search results from identification to inclusion for studies where trauma exposure is measured.
Main Findings
A pictorial overview of the main findings can be found in Figure 3.
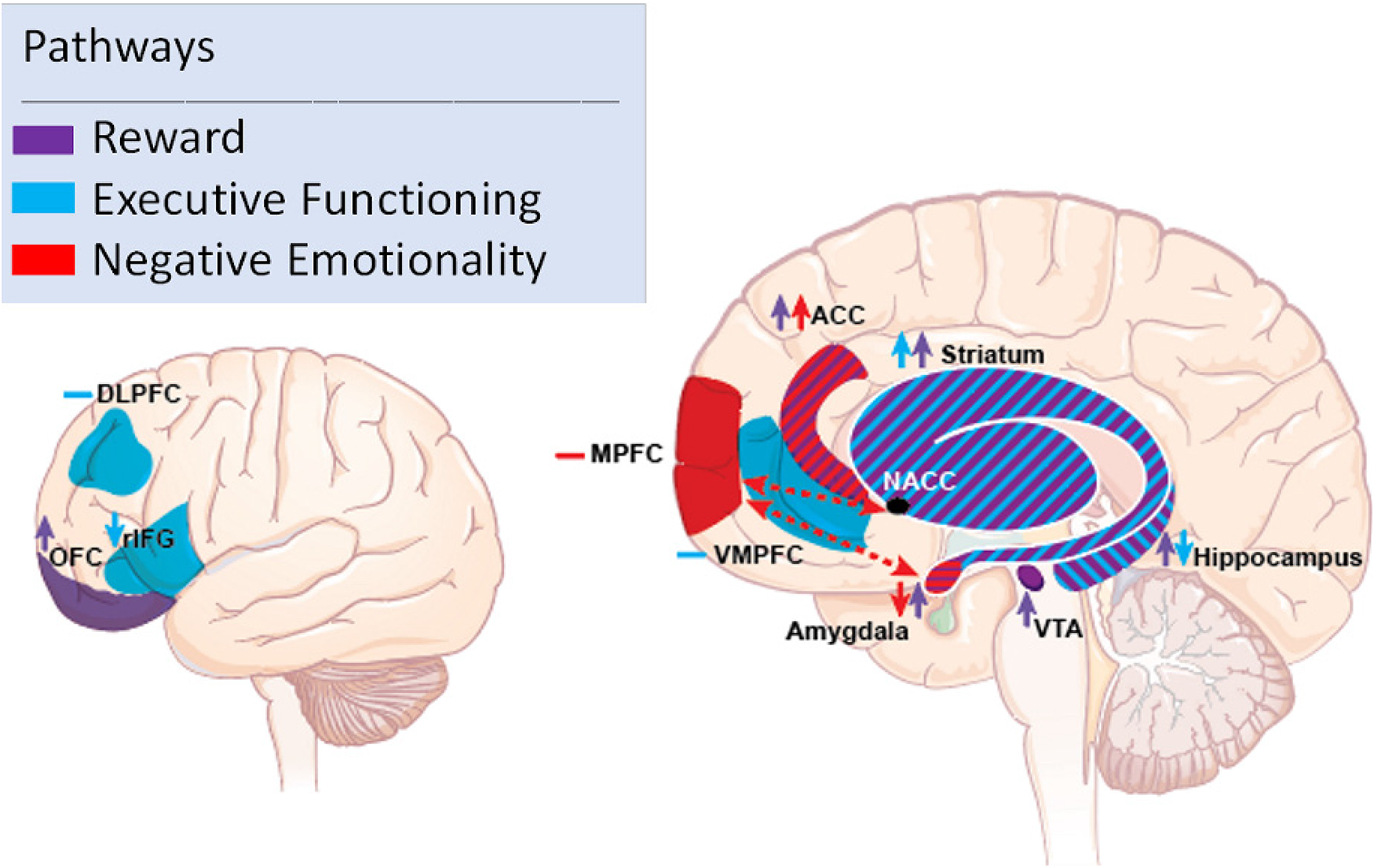
Overview of findings across each domain examined. Purple shading depicts activation in the reward domain, blue in the executive functioning (inhibition) domain, and red shading in the negative emotionality domain. Stripped shading highlights activation found in multiple domains for that respective region. Arrows depict greater or lesser activation in the respective region. Negative signs depict mixed activation found in the respective region. Abbreviations: DLPFC = dorsolateral prefrontal cortex; OFC = orbitofrontal cortex; rIFG = right inferior frontal gyrus; MPFC = medial prefrontal cortex; VMPFC = ventromedial prefrontal cortex; ACC = anterior cingulate cortex; NACC = nucleus accumbens; VTA = ventral tegmental area.
Reward Domain
See Table 1 for an overview of findings for this domain.
Reward studies.

Notes. Abbreviations: SD = standard deviation; NR = not reported; OFC = orbitofrontal cortex; ACC = anterior cingulate cortex; VTA = ventral tegmental area
Regarding functional connectivity (FC), Strosche et al 42 found that individuals diagnosed with DSM-IV alcohol dependence exhibited decreased task-related FC between the ACC and insula and ACC and inferior frontal gryrus (IFG) while presenting alcohol-related cues than abstinent individuals. 42 During a feedback learning task, widespread greater FC in the dorsal ACC (dACC) and other target regions of interest, including the ACC, was found in individuals who use tobacco. 57 Lastly, one study found that during an unconscious presentation of tobacco smoking images versus the unconscious presentation of neutral images greater FC between the right amygdala and right ACC in individuals who use tobacco than those who did not use tobacco. 58
Regarding FC, during an alcohol cue reactivity task, greater FC between the OFC and insula and lesser FC between the OFC and striatum was present in individuals diagnosed with DSM-IV alcohol dependence versus individuals abstaining from alcohol. 42 When responding to feedback learning tasks, individuals who used tobacco showed increased connectivity between the ACC and OFC versus those who did not use tobacco. 57
Negative Emotionality Domain
See Table 2 for an overview of findings from this domain.
Negative emotionality studies.
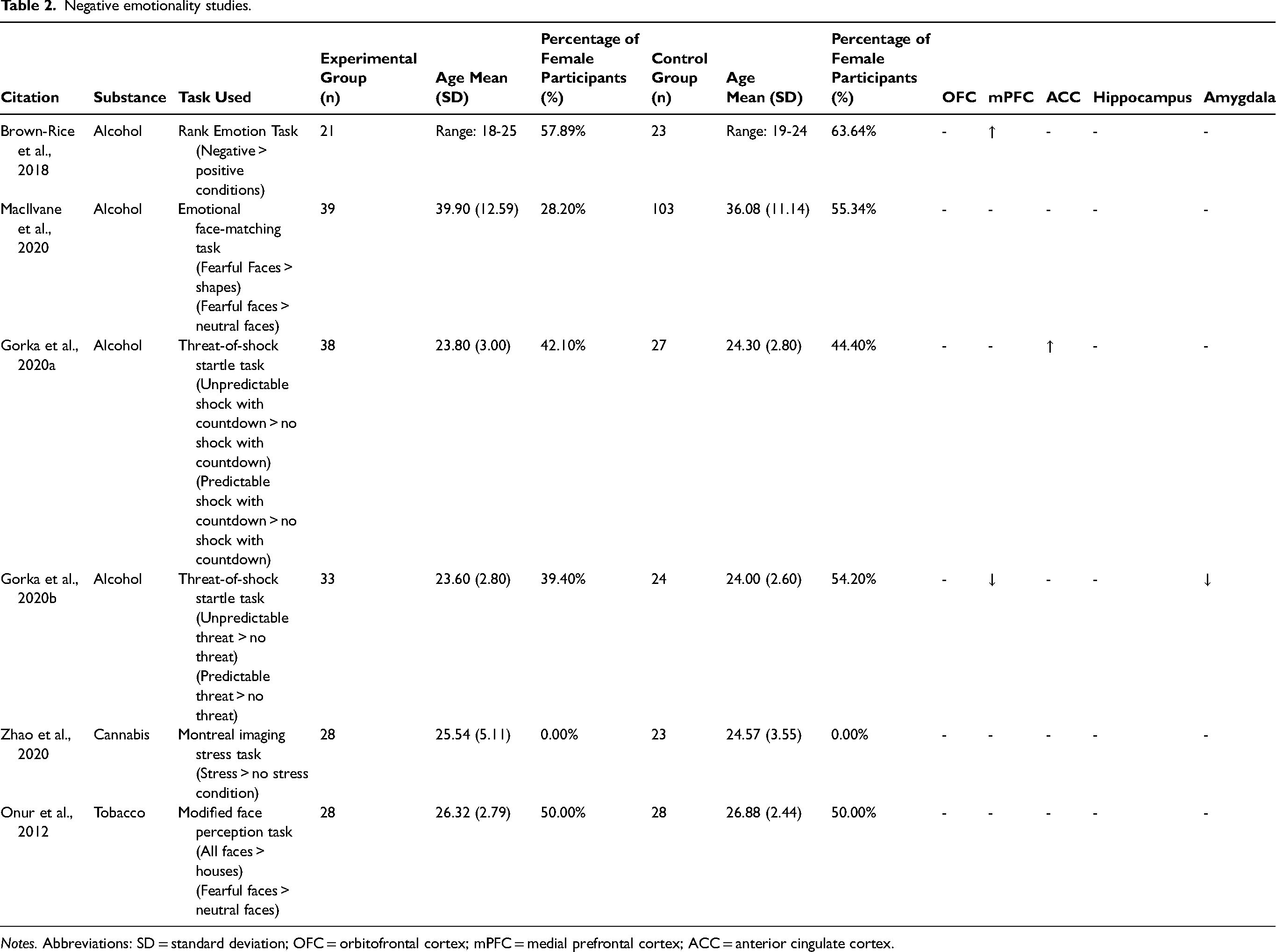
Notes. Abbreviations: SD = standard deviation; OFC = orbitofrontal cortex; mPFC = medial prefrontal cortex; ACC = anterior cingulate cortex.
Inhibition Domain
See Table 3 for an overview of findings for this domain.
Executive functioning (inhibition) studies.
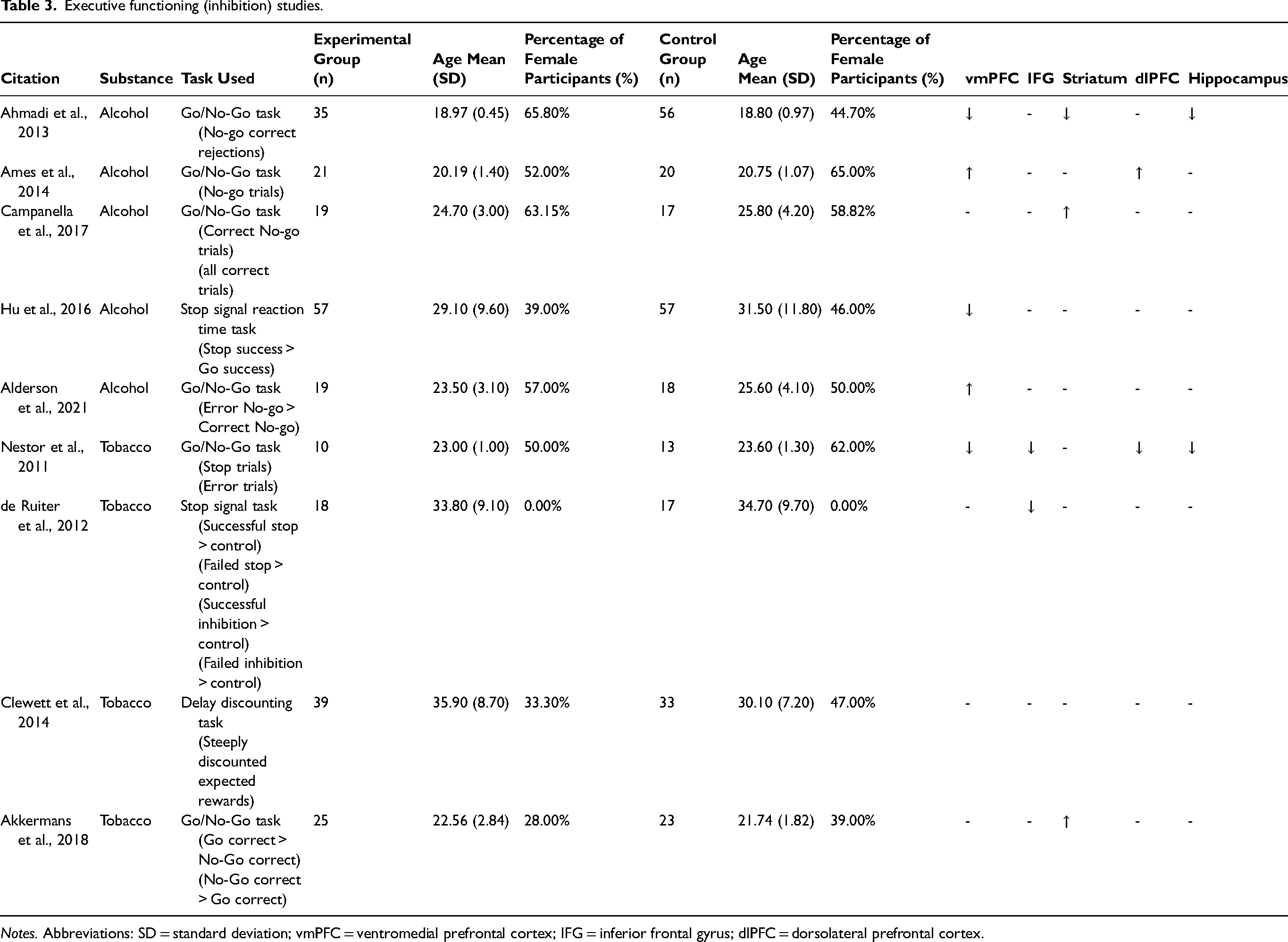
Notes. Abbreviations: SD = standard deviation; vmPFC = ventromedial prefrontal cortex; IFG = inferior frontal gyrus; dlPFC = dorsolateral prefrontal cortex.
Individuals who experienced binge drinking episodes exhibited greater activation than individuals who drank light in the rACC and increased FC between the rACC and right lateral frontal cortex during the error No-Go > Correct No-Go responses. 74
Greater FC between the striatum (anterior putamen) and insula was found in individuals who used tobacco than in those who did not. 76
Discussion
This review systematically synthesized neuroimaging studies focusing on three domains that may contribute to the overlapping mechanisms of SUD and PTSD—reward salience, negative emotionality, and executive function (response inhibition). We focused on samples of individuals who use substances where trauma exposure is not measured and studies that explicitly measure trauma exposure, enabling us to draw conclusions from and identify knowledge gaps for each population. Our findings suggest altered activation and FC in reward, inhibition, and negative emotionality neural circuits in currently using, dependent on substances, or clinically diagnosed with SUD than controls. Notably, no studies on trauma-exposed individuals using, dependent on, or clinically diagnosed with SUD met the criteria for our systematic review. The implications of these findings are discussed below.
Reward Domain
Overall, the studies reviewed showed greater activation in individuals who use substances versus controls across many brain regions important in the mesocorticolimbic pathway, including the OFC, VTA, amygdala, and hippocampus, when using reward-related tasks such as cue-reactivity and monetary incentive tasks.
An increase in VTA and striatal activation during the presentation of cue-related activation is not surprising, given that the VTA is responsible for releasing dopamine to the striatum in response to rewarding stimuli. 77 However, even in individuals with substance use dependence, there was still greater activation in these areas compared to individuals without substance use dependence to cue-related stimuli. This was surprising given previous literature that suggests individuals dependent on a substance no longer exhibit similar increases in dopaminergic initiation given the tolerance created for the substance. 9 We excluded intervention studies that gave participants a substance inside the scanner; thus, the contextual cues associated with that substance rather than the actual substance could provide a greater release of dopamine-producing a rewarding effect. We did find greater activation in the hippocampus in individuals who use substances versus controls, providing evidence of the importance of context cue processing during reward processing. Thus, these findings highlight that activation in response to viewing reward-related stimuli leads to greater activation of brain regions underlying reward even during dependence.
The increased activation in the VTA and striatal findings may suggest that when passively viewing rewards without the ability to receive the reward leads to an increase in reward-related activation, whereas actively receiving the reward or consciously making decisions with outcomes that will directly affect the participant may lead to deficient regulation of reward in the VTA, striatum, and ACC.
Negative Emotionality Domain
The negative emotionality domain, whose phenotypical expression is related to the mesolimbic domain of the brain, encompasses the mPFC, ACC, and amygdala. Only 6 studies were eligible to be included in the review. Regardless, these results highlight greater dACC and lesser mPFC activation in individuals with hazardous drinking patterns and AUD compared to controls.
The dACC is important in the acquisition of conditioned fear. Thus, greater activation in the dACC could suggest that individuals who use substances have altered appraisal and expression of conditioned fear than controls. Interestingly, one study found greater MFG activation in response to negative images in individuals with hazardous drinking patterns versus individuals who do not exhibit hazardous drinking patterns. 68 This finding is interesting, given the regulatory role of the MFG in responding to potentially threatening stimuli. However, the MFG is potentially trying to hyper-regulate regions responsible for fear expression (ie, the amygdala), leading to maladaptive responses.
Inhibition Domain
Overall, studies found lesser activation in the IFG and hippocampus. In contrast, mixed findings in activation were present in the ACC, striatum, and dlPFC in individuals who used substances versus controls.
Given its important role in executing successful response inhibition, the findings of lesser IFG in individuals who use tobacco could correspond with the behavioral deficits seen in inhibiting their urge to use substances, even when the substance is causing functional difficulties in the person's life. Deficiencies in the brain regions responsible for inhibition may lead to impulsive behavior associated with initiating and maintaining substance use. 78 More studies are needed to determine better the role of the ACC, striatum, and dlPFC in inhibitory processes.
Trauma-Exposed Samples
No studies were identified that examined functional MRI correlates of reward salience, negative emotionality, or inhibition in substance use samples with trauma exposure that met the criteria of our PRISMA-guided systematic review. Given that trauma is a risk factor for the development of substance use and SUD, as well as the large co-occurrence of PTSD and SUD, it is likely that our sample of individuals who use substances has some proportion who were un-assessed trauma-exposed individuals. Therefore, studies are needed to examine the intersection of trauma history with functional correlates of reward salience, inhibition (or executive function), and negative emotionality in individuals who use substances. Determining how an individual responds to a stressor and how trauma-related factors may interact with neurobiological mechanisms to influence SU may facilitate screening individuals at risk and developing targeted clinical interventions to mitigate the escalation of substance use.
Limitations and Future Directions
There are limitations of the literature reviewed. First, this review highlights a lack of studies that have systematically measured trauma exposure in individuals who use substances when examining reward, negative emotionality, and inhibition neurocircuitry. We could not identify studies conducted in trauma-exposed samples; however, as noted above, it is plausible that participants in the studies where trauma exposure was not measured did indeed experience trauma, but trauma exposure was not assessed in these studies. It will be imperative to measure trauma exposure moving forward in studies using samples who use substances to start understanding the potential neural mechanisms associated with trauma that contribute to the development of substance use or vice versa. Second, while many studies examined neural correlates of reward-related neurocircuitry in SUDs, neuroimaging studies are only beginning to more thoroughly explore negative emotionality and negative emotionality in individuals who use substances79‐81 Future studies should extend this research to utilize tasks focusing on these domains to determine whether alterations are associated with substance use maintenance and development. Third of the studies reviewed, participants with varying levels of substance use were combined, including individuals who occasionally use, those who use heavily, and those diagnosed with a SUD. More studies should focus on each level of the substance use cycle to understand the neural correlates of early use, transitioning to heavy use, and later disorder. Lastly, many studies reviewed contained small sample sizes (n's < 100 per group). In the future, studies utilizing bigger samples should be published to increase the power to determine effects.
There are also limitations related to our systematic review. First, our systematic review focuses on alcohol, tobacco, and cannabis use, given that these substances have been reported most in trauma studies. 37 Other substances are important to explore, such as cocaine, heroin, and fentanyl. In addition, less research has been done on developing substance use post-trauma. Because our literature searches showed no studies examining trauma, we categorized the studies as not having measured trauma exposure. However, given that trauma exposure is a common risk factor for developing a SUD, such a sample is unlikely to be entirely unexposed to trauma. Therefore, the conclusions related to this sample should be interpreted with caution. It is important to note that the studies without trauma exposure do not mean the participants in these studies were not trauma-exposed, only that their trauma was not directly measured within the confines of the study. More neuroimaging studies should investigate the neural underpinnings associated with susceptibility to developing substance use problems post-trauma. Lastly, causality was not addressed in this review. It will be imperative to discover whether these findings result from using these substances or make an individual more prone to developing substance use issues. To answer this important question, longitudinal and twin samples should be used in the future.
Conclusion
This review systematically synthesizes neuroimaging studies focusing on three domains associated with substance use and may contribute to the overlapping mechanisms of SUD and PTSD—reward, negative emotionality and inhibition. More definitive research on individuals using substances, especially those with trauma exposure, and how these responses may contribute to the development and maintenance of substance use is needed. Consistent with existing literature, we have highlighted deficits in each domain, specifically greater activation in regions involved in reward processing, greater and lesser activation in regions involved in negative emotionality processes, lesser activation in regions involved in inhibition processing. Additionally, we propose that future studies should emphasize executive function and negative emotionality processes, investigate the role of trauma-related factors, and use longitudinal designs to better understand the underlying development of neurobiological alterations, providing mechanistic targets for preventative measures and treatment outcomes.
Supplemental Material
sj-docx-1-css-10.1177_24705470241258752 - Supplemental material for Functional Domains of Substance Use and their Implications to Trauma: A Systematic Review of Neuroimaging Studies
Supplemental material, sj-docx-1-css-10.1177_24705470241258752 for Functional Domains of Substance Use and their Implications to Trauma: A Systematic Review of Neuroimaging Studies by Cecilia A. Hinojosa, Siara I. Sitar, Joshua C. Zhao, Joshua D. Barbosa, Denise A. Hien, Justine W. Welsh, Negar Fani and Sanne J.H. van Rooij in Chronic Stress
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: C.A.H was funded under an NIH NIAAA Award (K99AA031333).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
