Abstract
Background
Fibroblast Growth Factor 2 (FGF2) is a neurotrophic protein that has been implicated as a biomarker for anxiety and depressive disorders, which comprise a significant component of the global burden of disease. Research using rodents has indicated that FGF2 is part of the stress response, but whether this translates to humans has yet to be investigated. In this study, we aimed to explore the potential role of FGF2 in the human stress response by examining its association with physiological and psychological processes during and following the Trier Social Stress Test (TSST).
Methods
Participants in the active stress experiment (N = 87) underwent the TSST, provided saliva samples to obtain levels of cortisol and FGF2, and reported on post-event rumination related to the TSST task over the following week. Participants in the no-stress experiment (N = 25) provided saliva samples for measurement of FGF2 and cortisol across a corresponding time period.
Results
Salivary FGF2 levels changed after the TSST and were associated with the pattern of change in salivary cortisol. Cortisol responses in the active stress condition were blunted in females (relative to males), however, sex did not interact with any other effect. FGF2 reactivity (ie, the magnitude of change over time) was not correlated with cortisol reactivity. Lower FGF2 reactivity following the TSST, but not overall FGF2 levels, or cortisol, was associated with higher fear of negative evaluation, repetitive negative thinking and post-event processing, as well as repetitive negative thinking in the week following the TSST. Participants in the no-stress experiment showed a decrease in cortisol, yet no change in their FGF2 levels.
Conclusion
These findings suggest that FGF2 is involved in the human stress response and higher levels of FGF2 reactivity may be associated with protective cognitive processes following stress exposure.
Introduction
Anxiety and mood disorders are among the top 25 leading causes of disease burden globally. 1 Identifying the individual factors that predict one's risk for these conditions could lead to earlier intervention, which improves prognosis. 2 One way to achieve this is by establishing biomarkers of mental illness. Fibroblast growth factor-2 (FGF2) is a neurotrophic protein and a potential biomarker for anxiety and depression. Variations in endogenous FGF2 are associated with anxiety behaviours in rodents. Rats that exhibited higher levels of conditioned fear expression had significantly lower hippocampal FGF2 protein and serum FGF2 after behavioural testing relative to rats that exhibited lower levels,3,4 an association which was evident even three months after behavioural testing. 5 Similarly, rats selectively bred for their high anxiety had significantly lower hippocampal FGF2 mRNA levels compared to rats selectively bred for low anxiety. The relationship between FGF2 and anxiety appears to be causal, as peripheral FGF2 administration in adulthood 6 or on the first day of life7,8 reduced anxiety in adult rats. In contrast, blocking FGF2 (either genetically or with short-hairpin mRNA silencing of hippocampal FGF2) increased anxiety behaviours in rodents.9–11
While the majority of research on FGF2 and anxiety has been conducted in rodents, a growing body of work has investigated the relationship between FGF2 and affective disorders in humans, with mixed results. Initial research showed decreased levels of FGF2 mRNA in the dorsolateral prefrontal cortex in post-mortem brain tissue from individuals with Major Depressive Disorder (MDD) relative to healthy controls. 12 Lower levels of serum FGF2 were found in adults with MDD 13 and children with anxiety and/or depressive symptoms, 14 while other studies reported higher serum FGF2 protein in adults with MDD, than healthy controls.15,16 We previously examined salivary FGF2 and anxiety responses during a laboratory fear conditioning task in healthy individuals. 4 We found that individuals with higher skin conductance responses during fear conditioning, and its subsequent recall, had lower salivary levels of FGF2 post-conditioning, consistent with prior findings in rats.3,5,17
One proposed mechanism by which FGF2 may regulate anxiety and depression is via the hypothalamic-pituitary-adrenal (HPA) axis. Stress is implicated in the onset and maintenance of psychiatric conditions, and anxiety and depression are associated with alterations in HPA axis activity.18–20 In rodents, acute stress increases astrocytic FGF2 expression in the hippocampus and chronic stress decreases hippocampal FGF2 mRNA expression.21–25 Changes in FGF2 following stress may be an adaptive neuroprotective response, as mice that demonstrated depressive-like behaviours and oxidative damage following chronic stress showed a reversal of these effects following peripheral FGF2 administration. 26 Glucocorticoid receptors (GRs) are involved in the relationship between stress and FGF2, as FGF2 knockout mice show reduced hippocampal GR expression and increased corticotropin releasing hormone (CRH) in the paraventricular nucleus compared to wildtype mice. 11 In wildtype rats, the administration of glucocorticoids increases FGF2 hippocampal mRNA expression and protein 24 and the administration of a synthetic corticosteroid increases FGF2 mRNA expression in the adrenal medulla and reduces GRs in the cortical neurons.27,28
While research to date has demonstrated that manipulating FGF2 influences the HPA axis, and manipulating the HPA axis influences FGF2, no study has examined changes in FGF2 and stress hormones concurrently following stress. Therefore, the precise relationship between cortisol and FGF2 following stress exposure remains unknown. More broadly, it is also unknown whether the relationship between FGF2 and stress seen in rodents translates to humans, and whether this potential relationship underpins the link between FGF2, and anxiety- and depressive-like features. In order to investigate these questions, in the current study we used the Trier Social Stress Test 29 (TSST) to elicit psychosocial stress in humans, and measured their salivary FGF2 and cortisol levels prior to and after stress exposure. FGF2 was measured in saliva as opposed to serum to enable multiple samples to be collected non-invasively. Past research has shown comparable sensitivity and specificity of the measurement of FGF2 in saliva and serum. 30 We aimed to investigate whether FGF2 levels change following stress, and whether the magnitude of potential changes in FGF2 are associated with stress-induced changes in cortisol. We also examined whether FGF2 levels after stress are related to repetitive negative thinking, a transdiagnostic factor in depression and anxiety disorders. 31 No research to date has assessed the link between FGF2 and cognitive risk factors for psychiatric conditions, and so inclusion of this measure allowed for the broader investigation of FGF2 as a biomarker.
Materials and Methods
Participants and Sample Selection
Active Stress Experiment
Participants were recruited from the University of New South Wales (UNSW) undergraduate and surrounding community through online advertising. Participants were reimbursed with either course credit or $20 per hour. Exclusion criteria included current/historical mental health difficulties, dental hygiene issues, hormonal contraceptive use (which blunt cortisol stress responses 32 ) and antidepressant use. Participants were required to be between 18–35 years old.
136 participants were eligible to participate in the experiment. 31 participants were excluded at the testing session, as they failed to follow the preparation instructions required for saliva. 104 participants completed the experimental session, and 1 participant dropped out. 17 participants were excluded due to contaminated or insufficient saliva samples. The final sample size was 87 participants (51 females), aged 18–28 (median = 20.4 years). Power analyses (G-power) indicated that a minimum of 74 participants was required to detect a small effect size (F = 0.1), with 80% probability (α = 0.05) in a repeated measures ANOVA analysis. Further, a meta-analysis found that N = 40 is the minimum sample size for detecting robust salivary cortisol responses in the TSST. 33
No-Stress Experiment
After the TSST study was completed, 27 additional participants, meeting the same inclusion/exclusion criteria as the TSST study, were recruited in a follow-up study to provide saliva samples over a corresponding timeframe to that of the TSST study. The purpose of this study was to verify that changes in FGF2 and cortisol observed were not merely due to the passage of time. Two participants were excluded prior to participating as they failed to meet the saliva screening requirements, resulting in a final sample of 25 participants (10 females), aged 18–26 (median = 18.96 years).
Measures
Repetitive Thinking Questionnaire-short form (RTQ-10)
The RTQ-10 is a measure of repetitive negative thinking (RNT) that asks participants to recall ruminative symptoms relating to the last time they felt distressed. 34 The RTQ-10 has good test-retest reliability (α = .76) and high internal consistency (α = .91). 35 The Cronbach's Alpha for RTQ-10 scores measured in session was α = .9.
State-Trait Anxiety Inventory-trait form (STAI-T)
The STAI-T is a measure of trait anxiety 36 with good internal consistency (α = .94) and test-retest reliability (α = 0.81). 37 The Cronbach's Alpha for STAI-T scores in the current experiment was α = .9.
Brief Fear of Negative Evaluation Scale (BFNE)
The BFNE is a measure of anxiety associated with social evaluation from others, 38 with high internal consistency (α = .96) and test–retest reliability (r = .75). 39 The Cronbach's Alpha for BFNE scores in the current experiment was α = .9.
Subjective Units of Distress (SUDS)
Participants in the active stress experiment provided a written SUDS score immediately after completing the TSST. This represents the level of subjective anxiety experienced during the TSST on a scale from 0 (no anxiety) to 10 (extreme anxiety).
Post-Event Processing Questionnaire (PEPQ)
Details of the PEPQ 40 are reported in the supplementary file.
Saliva Collection and Analysis
Participants were instructed to refrain from eating in the hour before saliva collection, consuming alcohol, nicotine or caffeine in the 12 h prior, and from intense exercise in the 24 h prior. Saliva was collected between 12:00 and 19:00 to control for diurnal effects of cortisol and potential diurnal effects of FGF2. 41 Samples of 1.5 mL were collected using the passive drool method, stored in a −30° freezer and were analysed within six months of collection. Samples underwent a single freeze-thaw cycle and were centrifuged at 1750 revolutions per minute at 4°C for 15 min prior to analysis. Salivary FGF2 and cortisol were analysed using commercially available enzyme-linked immunosorbent assays (ELISAs), as per manufacturer instructions (RDSDFB50, R&D Systems; S1-3002, Salimetrics, respectively). Integrated optical density for each sample was calculated using an iMark plate reader (Bio-Rad Laboratories, Hercules, CA) at 450 nm for FGF2 and 450 nm with a secondary filter correction at 490 nm for cortisol. Integrated optical density values for each were converted into pg/ml and μg/dL using standard curves of known quantities of FGF2 and cortisol.
Procedures
Active Stress Experiment
Participants completed a saliva pre-screening questionnaire, then provided two baseline saliva samples, spaced 15 min apart. After the second sample, participants completed baseline trait questionnaires (ie, STAI-T and BFNE). Participants then completed the TSST following the standardised protocol of administration. 42 This involved preparing a 5-min speech and completing an oral arithmetic task, in front of the experimenter and a video recorder. Participants provided saliva samples immediately after the stressor, 15 min post-stress and 45 min post-stress, as well as completing psychological measures and watching clips from a nature documentary (see Figure 1). As part of the battery of questionnaires, participants also completed a measure of depression, anxiety, and stress, the results of which will be reported in a different manuscript as part of a longitudinal analysis. Participants also completed the RTQ-10 and PEPQ at home on days one, three and five after the experiment, with altered instructions to report on RNT specifically related to the TSST task. Participants were debriefed at the conclusion of the lab session about the aims and methodology of the experiment.
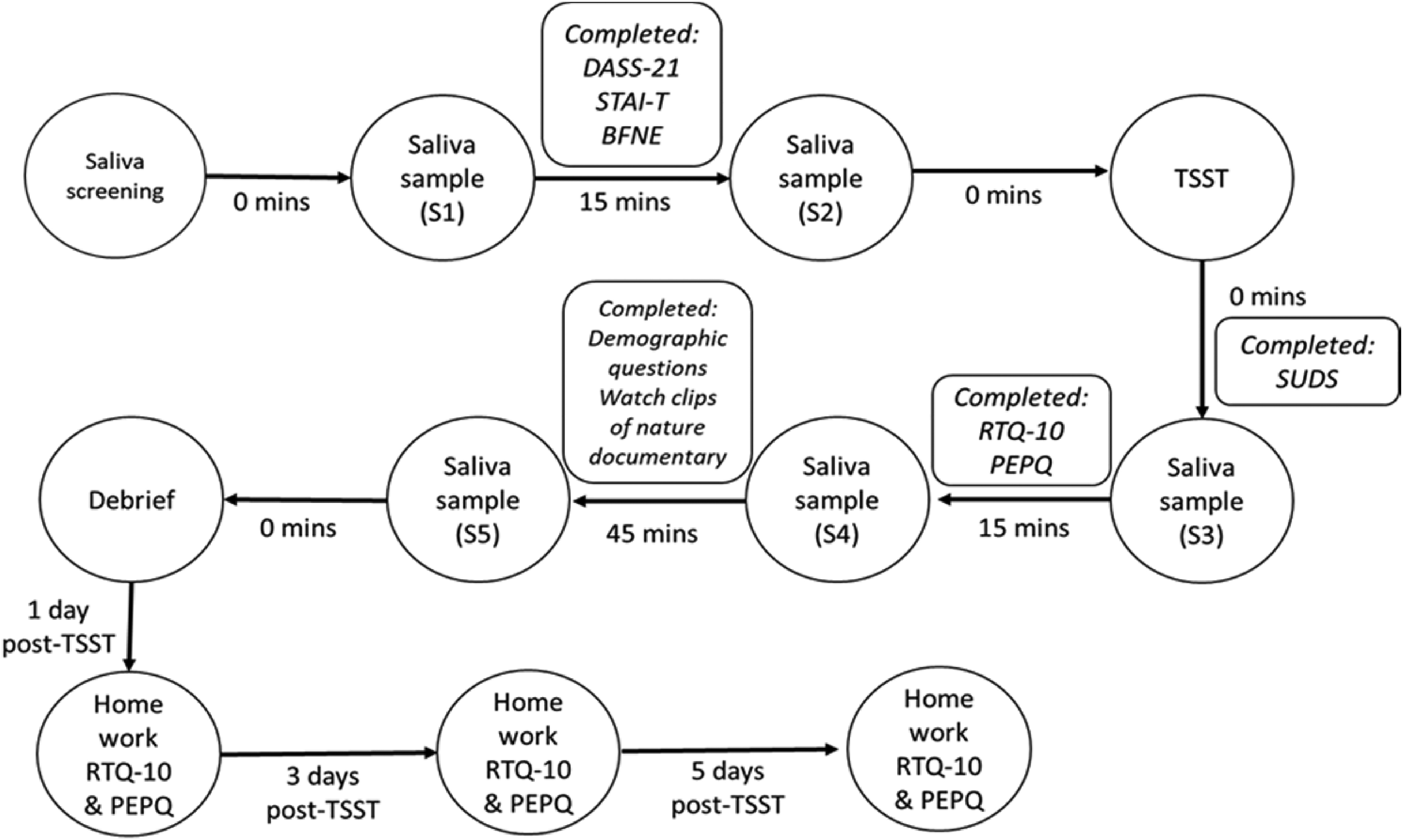
Timeline of the procedure for the active stress experiment.
No-Stress Experiment
Participants completed a saliva pre-screening questionnaire and provided five saliva samples at the equivalent timings of the active stress experiment participants. In place of the TSST, participants completed questionnaires and watched clips from a nature documentary (see Figure 2). Participants did not complete any follow-up measures after the session.

Timeline of the procedure for the no-stress experiment.
Statistical Analyses
Statistical analyses were conducted using SPSS Statistics Version 27 with values of p < .05 considered statistically significant. As the distribution of both FGF2 and cortisol values across the five time points were skewed (skewness = .788- 2.125 and 1.299- 2.004, respectively), values of FGF2 and cortisol were square root transformed prior to analysis, which substantially improved the distribution (skewness = .192-.893 and .603- 1.068, respectively).
Active Stress Experiment
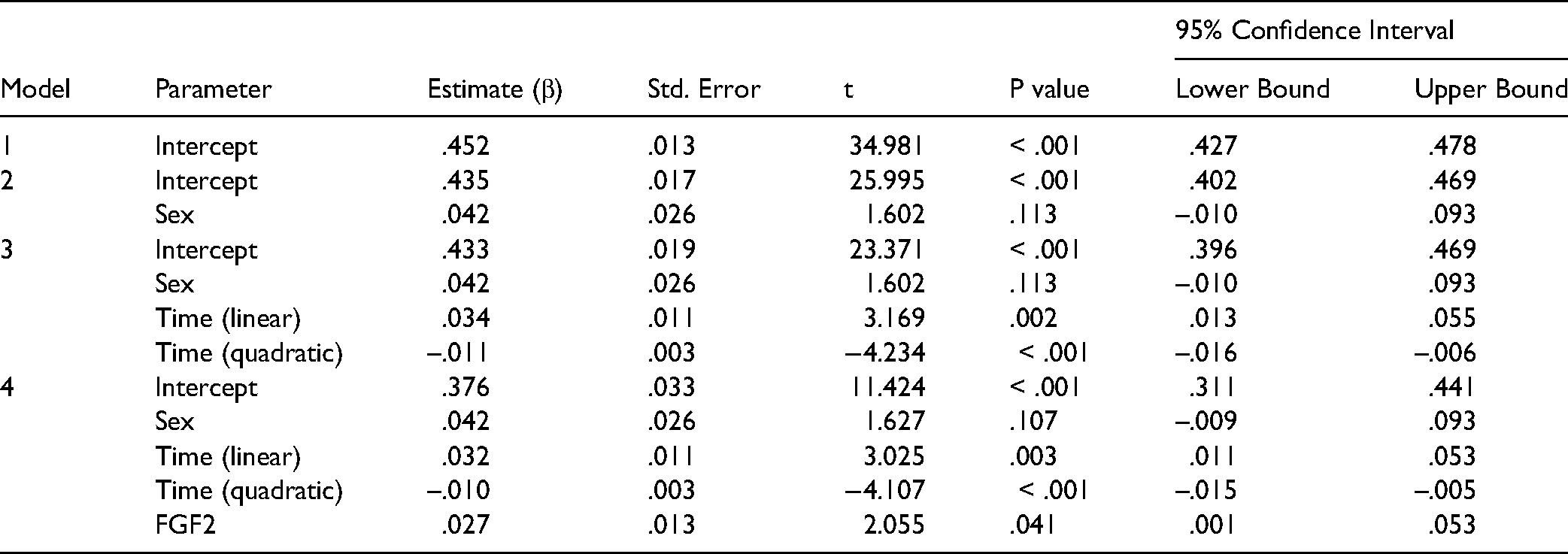
To determine whether cortisol and FGF2 levels changed significantly over time, repeated measures general linear models (GLMs), with time as the within-subjects variable and sex as a between subjects factor, were conducted on the five values of cortisol and FGF2. Greenhouse-Geisser correction was applied to account for violation of sphericity. To assess whether changes in FGF2 over time were associated with changes in cortisol over time, four stepped linear mixed models (LMMs) were conducted. Cortisol levels over time were the dependent variable, and participants were treated as a random effect in all models. The first model contained a random intercept, sex (coded female = 0, and male = 1) was added in the second model, time (linear), and time (quadratic) were added in the third model, and FGF2 levels over time were entered in the fourth model. All models contained an AR(1): heterogenous covariance matrix.
Area under the curve (AUC) calculations were conducted for each participants’ cortisol and FGF2 levels using formulae two and five from Pruessner et al (2003). 43 AUC in respect to increase (AUCi) from baseline represents the magnitude of change over time relative to a participant's own baseline, from pre- to post-TSST. We have termed these data points FGF2 reactivity and cortisol reactivity. AUC in respect to ground (AUCg) captures levels over time in relation to the distance from ground, or zero. Correlations were calculated to determine the relationship between FGF2 and cortisol levels over time (AUCg), and between the magnitude of stress-elicited change in FGF2 and that of cortisol, controlling for differences in basal FGF2 and cortisol levels (AUCi). We also correlated FGF2 and cortisol AUCg and AUCi with psychological variables, using bivariate correlations for single measurements and LMMs (with a scaled identify covariance matrix) for those with a time-varying component (ie, RNT).
No-Stress Experiment
Repeated measures GLMs with Greenhouse-Geisser correction were conducted on cortisol and FGF2, with time as the within-subjects variable and sex as a between subjects factor, to ascertain whether cortisol or FGF2 levels fluctuate over a corresponding timeframe to that examined in the active stress study.
Results
Active Stress Experiment
Descriptive Statistics
Participant demographics and mean scores for the psychological measures are listed in Table 1. The mean and mode of SUDS ratings (5.6 and 7, respectively) indicate that participants reported a subjective experience of anxiety during the TSST task.
Participant Demographics, Mean Scores and Standard Deviations (in Parentheses) of Questionnaires in the Active Stress Experiment.

Repeated Measures GLM on Cortisol and FGF2 Levels
Cortisol levels and FGF2 levels changed over time, from pre- to post-stress (Figures 3 and 4). For cortisol levels, there was a main effect of time (F4, 340= = 9.930, p = <.001), but no between-subjects main effect of sex (F1,85 = 2.567, p = .113). There was a significant interaction between time and sex (F4, 340 = 3.993, p = .017). The trend of change in cortisol was primarily quadratic (F1, 85 = 20.219, p = <.001). Post-hoc pairwise comparisons were conducted separately for males and females. In females, sample 2 was significantly different from sample 5 (45 min post-TSST) only (p = <.001), with mean values indicating that cortisol decreased over time. In males, sample 2 was significantly different from sample 3 and 4 (p = .001; p = .009, respectively), with means indicating an increase in cortisol post-stress.

Mean cortisol levels (μg/dL) of saliva samples across the experiment for male and female active-stress participants. Samples were taken immediately on arrival (1), after 15 min (2), immediately after stress (3), 15 min post-stress (4) and 45 min post-stress (5).

Mean FGF2 levels (pg/mL) of saliva samples across the experiment for male and female active-stress participants. Samples were taken immediately on arrival (1), after 15 min (2), immediately after stress (3), 15 min post-stress (4) and 45 min post-stress (5).
For FGF2, there was a significant main effect of time (F4, 340 = 2.809, p = .042) but no between-subjects main effect of sex (F1,85 = .001, p = .971). There was no significant interaction between time and sex (F4,340 = 1.095, p = .351). The pattern of change in FGF2 was trending towards quartic, F1,85 = 3.839, p = .053. Pairwise comparisons on the main effect of time showed a significant difference between FGF2 levels in sample 2 and 3 (p = .015).
Linear Mixed Models- Relationship between FGF2 and Cortisol Over Time
The quadratic function of time was a significant predictor of cortisol levels across the course of the experiment (β = -.010; p = <.001). Higher FGF2 levels over time significantly predicted higher cortisol levels over time (β = .027, p = .041). See Table 2 for outcomes of the stepped models.
Linear Mixed Model with CORT as the Dependent variable.
Correlations between FGF2 and CORT AUCg and AUCi and Psychological Measures
FGF2 AUCi and cortisol AUCi were not correlated (r = .072, p = .507). FGF2 AUCi was significantly associated with scores on the in-session RTQ-10 (r = -.301, p = .005), in-session PEPQ (r = -.247, p = .021) and BFNE (r = -.279, p = .009). Cortisol AUCi was significantly associated with sex (r = .213, p = .047), with males showing a higher cortisol reactivity. FGF2 AUCg was not significantly associated with any measure, and cortisol AUCg was only associated with cortisol AUCi. Conducting partial correlations holding sex constant did not change the pattern of outcomes (data not shown). See Table 3 for all correlations.
Bivariate Pearson Correlation Matrix.

Note: * = p < .05; ** = p < .01.
Linear Mixed Models- Association between FGF2 AUCi, Cortisol AUCi and post-TSST RTQ-10
Time was a significant predictor of RTQ-10 scores (β = -1.123; p = <.001), showing that RNT decreased over time. FGF2 AUCi was also a significant predictor of RTQ-10 scores (β = -.099; p = .001), where higher FGF2 AUCi predicted less RNT about the TSST in the week following the experiment. FGF2 AUCg, cortisol AUCi, and cortisol AUCg, were not significant predictors of RTQ-10 scores over time (see Table 4). Identical results were obtained for the post-TSST PEPQ (see supplementary information Table S1).
Linear Mixed Models with RTQ-10 as the Dependent variable.

No-Stress Control Experiment
Descriptive statistics
Participant demographics and mean scores for the psychological measures are listed in Table 5.
Participant Demographics, Mean Scores and Standard Deviations (in Parentheses) of Questionnaires in the No-Stress Control Experiment.

Repeated Measures GLM on Cortisol and FGF2 Levels Over Time
Cortisol levels, but not FGF2 levels, changed over time (Figures 5 and 6). For cortisol levels, there was a main effect of time (F4,92 = 15.610, p = <.001), but no between-subjects main effect of sex (F1, 23 = .026, p = .873). There was no significant interaction between time and sex (F4,92 = 1.263, p = .292). Cortisol levels decreased linearly (F1, 23 = 24.299, p = <.001). Pairwise comparisons on cortisol levels at individual times points showed that sample 1 was significantly different from sample 2–5 (p's ≤.010). Sample 2 was significantly different from sample 4 and 5 (p's ≤.008). Sample 3 was significantly different from sample 4 and 5 (p's ≤.004). Sample 4 was significantly different from sample 5 (p = .016). Mean values indicate that cortisol levels decreased linearly across timepoints. There was no main effect of time for FGF2 levels (F4,92 = .534, p = .680). There was no between-subjects main effect of sex (F1,23 = 2.001, p = .171) and no significant interaction between time and sex (F4,92 = .759, p = .534).

Mean cortisol levels (μg/dL) of saliva samples across the experiment for male and female no-stress participants. Samples were taken on arrival (1), after fifteen minutes (2), 15 min after S2 (3), 15 min after S3 (4) and 45 min after S3 (5).
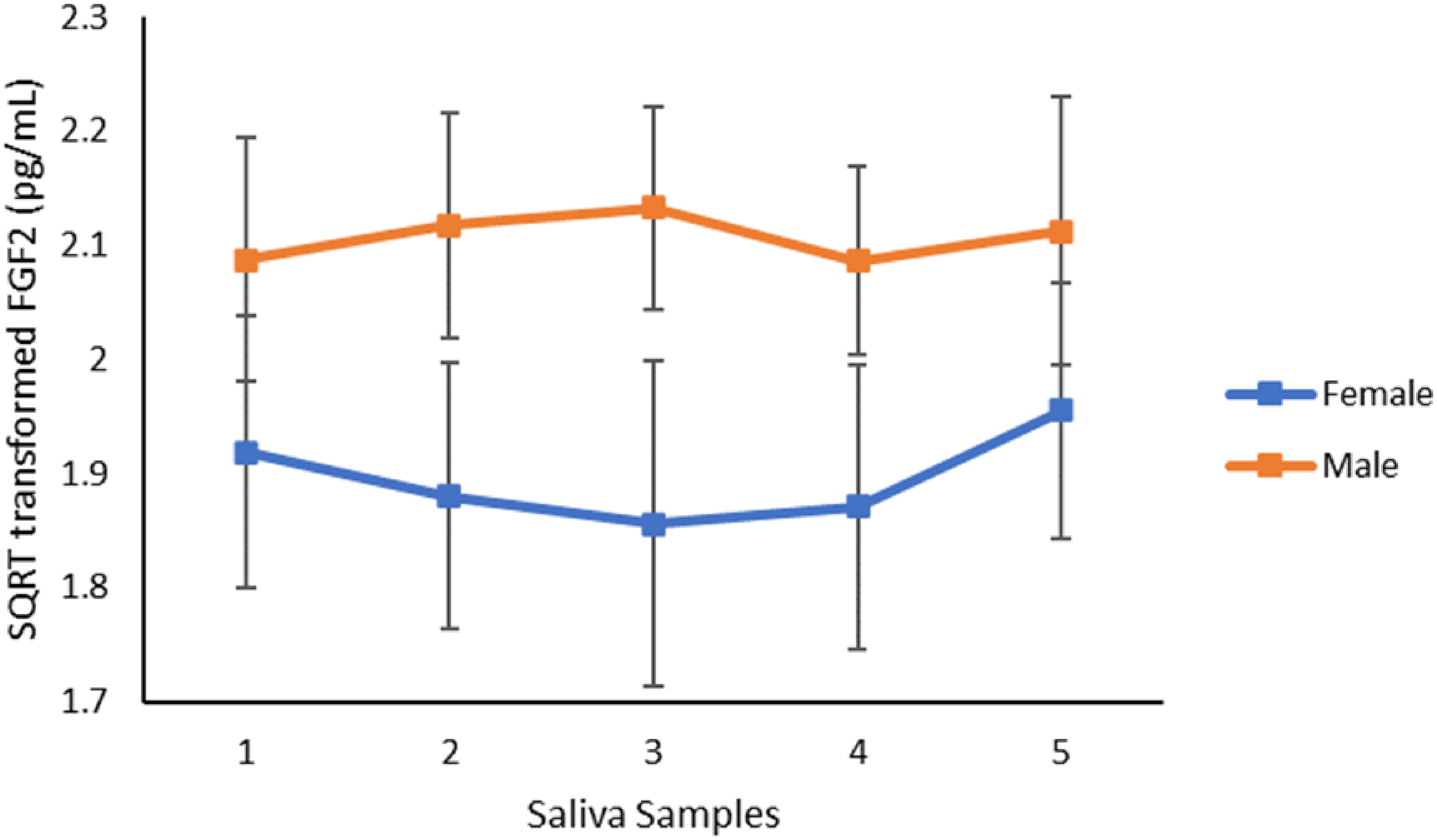
Mean FGF2 levels (pg/mL) of saliva samples across the experiment for male and female no-stress participants. Samples were taken immediately on arrival (1), after fifteen minutes (2), time equivalent to S3 of active experiments participant (3), 15 min post-S3 (4) and 45 min post-S3 (5).
Discussion
This experiment provides the first evidence that salivary FGF2 in humans changes in response to stress and that these changes are associated with those of salivary cortisol. However, neither FGF2 reactivity, nor absolute FGF2 levels, were associated with cortisol reactivity or absolute cortisol levels. While there were no sex differences in levels of FGF2 over time, females in the active stress experiment showed a blunted cortisol response compared to males. This is consistent with previous research indicating that only 25% of females showed a cortisol response to the TSST and Iowa Singing Social Stress Test, compared to 90% of males. 44 Furthermore, lower levels of FGF2 reactivity were associated with greater RNT, post-event processing and fear of negative evaluation, as well as future rumination (ie, the amount of RNT in the week following the experiment). Cortisol reactivity did not correlate with any cognitive outcomes.
Whereas FGF2 temporarily increased following stress, FGF2 levels did not change over the same time period in the no-stress experiment. This indicates that the observed changes in FGF2 were likely elicited by stress, rather than by time or another factor. Indeed, the finding that human salivary FGF2 levels change in response to stress is consistent with the existing rodent literature. Previous research has demonstrated that rats show an increase of hippocampal mRNA expression following exposure to stress.21,22 In rodents, the chronicity of stress differentially impacts FGF2 expression, with acute stress increasing hippocampal FGF2 mRNA expression23,24 and chronic stress decreasing hippocampal FGF2 expression. 24 The controllability of stress also differentially impacts FGF2 levels in rodents. Rats that were exposed to escapable tail shocks (ie, controllable stress) showed significantly higher FGF2 expression in the medial prefrontal cortex, dentate gyrus, and hippocampus, compared to rats that received inescapable tail shocks (ie, uncontrollable stress).21,22 Notably, the TSST is an acute stressor that is relatively controllable (ie, participants can terminate the experiment at any time point, and participants are given choices about the length and subject category of their speech). Future research in humans should consider whether the controllability of stress differentially impacts the direction or magnitude of salivary FGF2 release, and the relationship with physiological and psychological stress responses. For this purpose, the TSST could be adapted to decrease controllability, eg, by telling participants that their speech length/subject had been determined by another participant.
In addition to showing that FGF2 levels change following stress, this experiment demonstrated that stress-related changes in cortisol and FGF2 levels over time were related. This is comparable to rodent research showing that FGF2 knockout mice have higher serum corticosterone compared to wild type mice following stress 11 and modulating corticosterone levels leads to changes in FGF2 levels. 24 Although the results of the linear mixed model indicated that FGF2 levels over time were associated with cortisol levels over time, the temporal pattern of these changes were slightly different. That is, the trend of change in cortisol was primarily quadratic, whereas the trend of change for FGF2 was predominantly quartic. There was no relationship between FGF2 AUCg and cortisol AUCg, indicating that absolute FGF2 was not associated with absolute levels of cortisol. Moreover, there was no significant correlation between cortisol reactivity and FGF2 reactivity, indicating that the magnitude of change in FGF2 and cortisol over time (accounting for an individual's baseline) was unrelated. Differences were also seen between males and females in cortisol levels over time, yet there were no sex differences in FGF2 levels over time. Moreover, cortisol reactivity was not correlated with any cognitive measure of anxiety, despite FGF2 reactivity being associated with these measures. Finally, although cortisol levels decreased over time in the no-stress experiment, FGF2 levels remained stable and did not follow the pattern of change seen in cortisol. Combined, this suggests that FGF2 is likely not merely a proxy for cortisol or a biproduct of cortisol release, and that cortisol and FGF2 are potentially distinct aspects of the stress response and may be independently associated with stress-related psychopathology. It is possible that FGF2 is related to stress pathways independent of the HPA system. While there is no evidence to date linking FGF2 with the sympathomedullary (SAM) pathway of the autonomic nervous system, future studies could measure salivary alpha amylase to explore the potential association between the SAM pathway and FGF2.
This is also the first experiment to demonstrate that FGF2 reactivity is associated with cognitive processes involved in anxiety, demonstrating that lower levels of FGF2 reactivity were associated with higher levels of RNT, post-event processing and fear of negative evaluation by others. These findings complement and extend on past studies that have found a relationship between salivary FGF2 and physiological reactions to a physical stressor (ie, electric shock). 4 The current experiment used psychosocial fear, rather than fear associated with a threat of shock, to elicit a type of fear more generalisable to anxiety disorders. This also allowed for cognitive processes that are seen in anxiety disorders (eg, repetitive negative thinking) to be measured in relation to a stressful event and correlated with cortisol and FGF2 release following the same stressor. Although it is beyond the scope of the current experiment to ascertain mechanisms underpinning the relationship between FGF2 and psychological processes, there are several possibilities that may be considered. FGF2 regulates cell proliferation and differentiation in the central nervous system. 45 As neurogenesis plays a key role in physiological and behavioural stress resilience, 46 higher FGF2 reactivity may lead to better psychological coping due to increased neurogenesis and brain plasticity. FGF2 reactivity may also lead to more adaptive coping responses due to its impact on memory formation and consolidation. 47 FGF2 plays a crucial role in modulating molecular signals involved in fear acquisition and extinction, and higher exogenous levels of FGF2 result in enhanced memory formation. 17 As FGF2 facilitates the processing and consolidation of fear-related information, this may result in better integration of memories, preventing cognitive processes such as rumination.
The present study found no evidence that FGF2 reactivity was associated with trait anxiety (ie, STAI-T) which deviates from the results previously described.6,14 This may be explained by methodological differences between the current study and previous research. For example, rodent experiments are limited to physical and behavioural observations and within human studies, and the age of the sample may influence outcomes as FGF2 protein and mRNA declines with age. 48 Additionally, different relationships with trait anxiety may be obtained depending on whether FGF2 was sampled peripherally (via saliva or blood serum) or centrally, and whether protein or mRNA expression was measured. However, the most notable difference between the present findings and past studies was that in our study the magnitude of change in FGF2 levels over time, from before to after a stressor, was correlated with psychological outcomes, but absolute levels of FGF2 over time were not correlated with any outcomes. In past studies, FGF2 levels were predominantly assessed at a single time point,4,14,16 and most prior studies did not include a stressor.14–16 In our earlier study, 4 saliva was collected at a single time point after fear conditioning (ie, after receiving electric shocks). It is therefore possible that the relationship between FGF2 levels and fear expression found in that study may reflect a relationship between changes in FGF2 levels due to the stress response and fear expression, mirroring the pattern of findings in the current study. The mechanisms contributing to the change in FGF2 levels following the TSST require further assessment in future research. It is possible that FGF2 protein is stored in the salivary glands, and the magnitude of release is increased when injury or stress occurs. Research has shown that FGF2 is expressed in the basal membranes of the salivary glands excretory ducts, 49 with the expression of both FGF2 and FGFR1 increasing in salivary gland tissue affected by lesions and tumours. 50 As these explanations are currently speculative, further research into the mechanistic release of FGF2 in the saliva following stress is needed.
The finding that salivary FGF2 changes in response to stress raises questions about salivary FGF2's utility as a biomarker, as it indicates that basal FGF2 may be difficult to accurately capture. For example, an individual's FGF2 levels may differ based on their pre-study experiences and acute stressors which precludes the development of normative data for FGF2 levels. Furthermore, exercise 51 and environmental enrichment 52 have been shown to increase FGF2 levels in rodents, which may also be the case in humans. This indicates it may be difficult to obtain a reliable basal measure for salivary FGF2 in humans. Notably, Lebowitz et al 14 reported that two measurements of serum FGF2, obtained six months apart, were highly correlated. It is therefore possible that serum FGF2 levels have greater temporal stability than salivary FGF2 levels, although whether serum FGF2 also exhibits stress-induced changes is yet to be investigated.
There are several limitations to the current study. Firstly, the findings are correlational. It is uncertain whether FGF2 directly mediates psychological outcomes or whether the association is underpinned by another mechanism. As FGF2 reactivity at the time of the stressor predicted future rumination, this potentially suggests that FGF2 contributes directly to these outcomes. However, it is also possible that this association merely arose because trait rumination is highly correlated with future rumination. In order to strengthen this body of research, future work should manipulate FGF2 indirectly (eg, via exercise) to demonstrate causal relationships between FGF2, stress and anxiety. Another limitation is the lack of a direct-comparison no-stress control group in the active stress experiment. Although this is in line with past studies examining changes in stress hormones and other neurotrophins following stress,43,53 running a control group concurrently would have allowed for the direct comparison between active- and no-stress participants.
Conclusions
The current study demonstrated that human salivary FGF2 changes in response to stress, and the magnitude of this change over time is associated with cognitive transdiagnostic risk factors for anxiety and depression . This study adds to an increasing body of work showing that the functional role of FGF2 in relation to stress and anxiety seems to be conserved across species. Although an isolated sample of salivary FGF2 may not be useful as a stand-alone biomarker, studying stress-induced changes in salivary FGF2 and its relation to coping after stress could yield valuable information about the biological mechanisms for adaptation to stress. This information could potentially be leveraged to develop novel treatments to boost resilience or identify individuals who are vulnerable to maladaptive stress responses involved in psychopathology.
Supplemental Material
sj-docx-1-css-10.1177_24705470221114787 - Supplemental material for The Association Between Salivary FGF2 and Physiological and Psychological Components of the Human Stress Response
Supplemental material, sj-docx-1-css-10.1177_24705470221114787 for The Association Between Salivary FGF2 and Physiological and Psychological Components of the Human Stress Response by Emma M. Bryant, Rick Richardson and Bronwyn M. Graham in Chronic Stress
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Discovery Project Grant from the Australian Research Council (DP180102485) awarded to BMG and RR and an Australian Government Research Training Program scholarship awarded to EB. The funding sources had no role in the study design, in the collection, analysis, or interpretation of data, in the writing of the report, or in the decision to submit the article for publication.
Ethical Approval
All procedures were performed at UNSW in accordance with the Declaration of Helsinki and with approval from the UNSW Human Research Ethics Committee (HC190368).
Informed Consent
Informed consent was obtained from all individuals prior to participating in the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
