Abstract
Mood disorders represent a pressing public health issue and significant source of disability throughout the world. The classical monoamine hypothesis, while useful in developing improved understanding and clinical treatments, has not fully captured the complex nature underlying mood disorders. Despite these shortcomings, the monoamine hypothesis continues to dominate the conceptual framework when approaching mood disorders. However, recent advances in basic and clinical research have led to a greater appreciation for the role that amino acid neurotransmitters play in the pathophysiology of mood disorders and as potential targets for novel therapies. In this article we review progress of compounds that focus on these systems. We cover both glutamate-targeting drugs such as: esketamine, AVP-786, REL-1017, AXS-05, rapastinel (GLYX-13), AV-101, NRX-101; as well as GABA-targeting drugs such as: brexanolone (SAGE-547), ganaxolone, zuranolone (SAGE-217), and PRAX-114. We focus the review on phase-II and phase-III clinical trials and evaluate the extant data and progress of these compounds.
Introduction
Mood disorders, including major depressive disorder, are now the number one cause of disability worldwide. 1 Traditionally, the pathophysiology of mood disorders was thought to be caused by a deficiency in monoamines in the synaptic cleft: serotonin, dopamine, and norepinephrine. While the monoamine-based hypothesis of mood disorders has led to significant advances and is the theoretical foundation for all approved standard oral antidepressants, it does not adequately explain several features of mood disorders, including the lag time to clinical efficacy for monoamine-based drugs. 2
The last two decades have seen significant advances in a hypothesis of mood disorders based on abnormalities in the amino acid neurotransmitter system, including glutamate (the primary excitatory neurotransmitter) and GABA (the primary inhibitory neurotransmitter) 3 , 4 (Figures 1 and 2). Neuroimaging work consistently documents abnormalities in these systems in key brain regions of patients with mood disorders. 2 Abnormal levels of these neurotransmitters have also been seen in plasma, serum and cerebrospinal fluid of patients with mood disorders. 5 These developments have been spurred by the discovery that ketamine, an antagonist at the NMDA receptor, can produce rapid antidepressant effects.
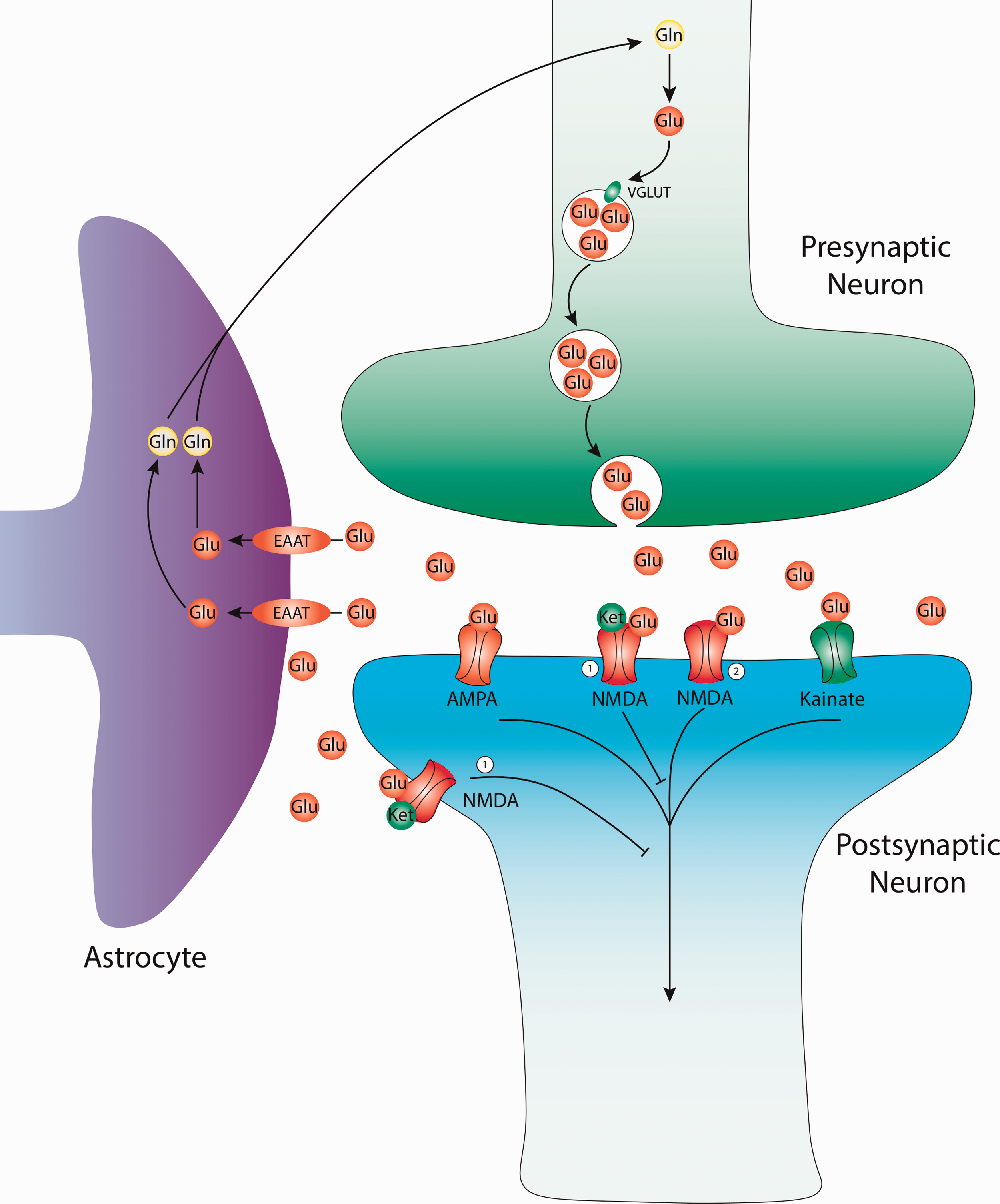
Glutamatergic neurotransmission and current targets for therapy. Glutamate (Glu) is derived from glutamine (GLN) in the presynaptic terminal. Alternatively, Glu can be created from the TCA cycle (not depicted). Glu molecules are then packaged into presynaptic vesicles by the vesicular glutamate transporter (VGLUT). The packaged vesicles then release their contents from the presynaptic neuron into the synaptic cleft. Within the cleft, Glu can bind various receptors including: α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA), Kainate, and N-methyl-D-aspartate (NMDA) receptors. Glutamate can be recycled from synaptic cleft by the excitatory amino-acid transporter (EAAT) of nearby astrocyte glial cells. Within glial cells glutamate is converted to glutamine before either being used by the glial cells or reused by nearby neurons for further glutamatergic transmission. 1) NMDA antagonist, site of action for: Ketamine, Esketamine, Lanicemine, AVP-786, AXS-05, Dextromethadone, MIJ821. 2) NMDA agonist, site of action for: Rapastinel, AV-101, NRX-100/NRX-101.

Gamma-aminobutyric (GABA) neurotransmission and current targets for therapy. GABA neurotransmitters are the predominant source of inhibition in the adult mammalian brain. GABA molecules bind two main types of receptors on neurons to promote inhibition: GABAAand GABAB. The GABAA receptors are ionotropic which function by increasing inward chloride ion flux to hyperpolarize neurons while GABAB receptors which use metabotropic signaling via G-proteins to open potassium channels and hyperpolarize neurons. Inhibition mediated by GABAA receptors can come in two distinct forms: phasic inhibition from intrasynaptic receptors following synaptic events or tonic inhibition from extrasynaptic receptors that provides more a more persistent source of inhibition. Allopregnalone (ALLO) is a GABAergic neurosteroid that acts as a positive allosteric modulator (PAM) at GABAA receptors to increase both of these forms of inhibition. 1) PAM of GABAA receptors, site of action for: Brexanolone, Ganaxolone, Zuranolone. 2) Protein Kinase C (PKC) mediated phosphorylation and increased extrasynaptic GABAA receptor trafficking, site of action for: Brexanolone, Zuranolone.
Given the rapidly changing state of the field, we undertook an updated systematic review of key compounds in development with mechanisms of action based on the amino acid neurotransmitter systems. We systematically searched MEDLINE (no start date cutoff; end date cutoff of March 31, 2020), the US Clinical Trials Registry (www.clinicaltrials.gov), and Sponsor websites for the latest clinical trial updates for the following compounds: esketamine (Spravato), AVP-786, REL-1017, AXS-05, Rapastinel (formerly GLYX-13), AV-101, NRX-101, AGN-241751, brexanlone (Zulresso, SAGE-547), ganaxolone, zuranolone (SAGE-217), MIJ821, PRAX-114.
As no updates were found in the last two years, 6 the following drugs were omitted: EVT-101, Traxoprodil/CP-101,606, Lanicemine/AZD-6765, Rislenemdaz/CERC-301, basimglurant, riluzole.
We limit the scope of this review to compounds in Phase II or III human trials. Two compounds, esketamine (Spravato) and brexanlone (Zulresso) received FDA approval for treatment-resistant depression and post-partum depression, respectively, in March 2019. We also review what is known about the mechanisms of action of these compounds. Where reported in study results, we use Cohen’s d effect size measure (between group comparison). Where not given and where sufficient data were available, we estimated the effect size.
Ketamine
Ketamine is the prototypical rapid-acting antidepressant that was discovered to have powerful antidepressant effects over two decades ago. 7 Traditionally an anesthetic, its antidepressant effects are most pronounced at subanesthetic doses, most commonly 0.5 mg/kg given intravenously over 40 minutes. 8 There are many small, academic studies using this treatment paradigm, though the vast majority are single-dose studies. 7 ,9–11 Hence, there is little long-term data on the use of IV racemic ketamine as a treatment for psychiatric disorders. Given the lack of industry sponsorship, it is unlikely that ketamine will be approved by the US FDA.
Esketamine
Treatment-Resistant Depression
Esketamine is the S-enantiomer of ketamine. It was approved by the US FDA for treatment-resistant depression (TRD), in conjunction with an oral antidepressant in March 2019. In August 2020, it was also approved by the FDA for treatment of depressive symptoms in adults with MDD who have acute suicidal ideation or behavior.
The new drug application leading to approval for TRD consisted of 5 Phase-III trials and 2 Phase-II trials. The phase-III trials included a fixed-dose short-term trial in adults ages 18-64 (TRANSFORM-1), a flexible-dose short-term trial in adults ages 18-64 (TRANSFORM-2), a flexible-dose short-term trial in geriatric patients ages 65 or older (TRANSFORM-3), a randomized withdrawal study (SUSTAIN-1), and a long-term, open-label (non-randomized) study (SUSTAIN-2). Notably, all subjects, regardless of treatment assignment, started a new oral antidepressant on the first study day. Hence, the comparison was between esketamine nasal spray plus a new oral antidepressant versus placebo nasal spray plus a new oral antidepressant.
The short-term fixed-dose trial (TRANSFORM-1, n = 346) did not meet its primary outcome (change in depression severity at week 4, p = 0.088, d = 0.22 for 84 mg; d = 0.28 for 56 mg). 12 The short-term flexible-dose trial (TRANSFORM-2, n = 236) met its primary outcome (change in depression severity at week 4, p = 0.02), with an effect size of d = 0.30. 13 The short-term flexible dose geriatric study (TRANSFORM-3;n = 139) did not meet its primary outcome (p = 0.059, d = 0.31 in favor of esketamine). 14
In the randomized withdrawal study (SUSTAIN-1), patients who had achieved stable response (n = 121) or remission (n = 176) were maintained on a stable dose of esketamine dosing for 12 weeks. At this point, half were randomized to placebo nasal spray (plus antidepressant) in lieu of the esketamine (discontinuation), while the other half continued to receive esketamine (plus antidepressant). A significant difference in the primary outcome (time to relapse) was seen between those continuing esketamine and those who were withdrawn among both remitters (hazard ratio [HR] = 0.49, p<0.05) and stable responders (HR = 0.30, p<0.05). 15
The SUSTAIN-2 study was a long-term (up to 52 weeks) open-label, non-randomized study that included both new participants and those who previously completed the TRANSFORM-3 study. 16 Of 802 enrolled patients, 9.5% discontinued esketamine due to treatment emergent adverse events. The most common side effects included dizziness, dissociation, nausea, and headache. With regards to secondary outcomes, 76.5% maintained response and 58.2% maintained remission at study endpoint. 16 A long-term extension study of SUSTAIN-2 (SUSTAIN-3; NCT02782104) is ongoing, with plans to follow patients for several years.
Several additional Phase-III studies are ongoing. One is comparing esketamine to quetiapine (50 mg, 100 mg, and 150 mg doses) in addition to SSRI/SNRIs (NCT04338321; expected n = 622). The third is similar in design to TRANSFORM-2 and is recruiting primarily from Chinese sites (NCT03434041).
Major Depression With Suicidal Ideation (MDSI)
Esketamine is FDA approved for the rapid reduction of depressive symptoms in adults with major depressive disorder who have active suicidal ideation with intent. This application is based on one positive Phase-II trial and two positive Phase-III trials, all of which had similar designs. In these studies, patients with MDD who were judged to be at imminent risk of suicide and in need of hospitalization were recruited from emergency departments or similar settings. All subjects received standard of care (including hospitalization and medications, but excluding ECT), with half receiving esketamine nasal spray and the other half receiving placebo nasal spray. The primary outcome was the change in depression severity (MADRS) at 4 hours following the first dose. The Phase-II trial (n = 68) (NCT02133001) demonstrated an effect size of d = 0.61 while the Phase-III trials (ASPIRE-I, N 226; ASPIRE-II, N = 230) demonstrated effect size of d = 0.33 and d = 0.30, respectively). 17 , 18,40
AVP-786
AVP is an oral formulation that combined dextromethorphan and quinidine. Dextromethorphan is an antagonist at the NMDA receptor that, similar to ketamine, may have antidepressant properties. Quinidine is added to this formulation to increase the bioavailability by inhibiting the action of cytochrome P450-2D6 and thereby blocking metabolism of dextromethorphan. 19 This compound is similar to dextromethorphan HBr and quinidine sulfate (manufacturer: Avanir), which is approved for the treatment of pseudobulbar affect. AVP-786 differs because it contains deuterium, a heavier isotope of hydrogen, which may further extend the half-life of dextromethorphan. 20 A single Phase-II study in patients with MDD or TRD (n = 206) was completed by February 2016 but results are not published (NCT02153502). The compound isnow in development primarily for agitation in Alzheimer’s dementia or for schizophrenia (see NCT03896945, NCT04464564, NCT04408755, NCT03393520, NCT02477670, NCT02446132). As of March 2021, no additional trials for mood disorders are registered in the US.
AXS-05
AXS-05 (Axsome) is a formulation of dextromethorphan and bupropion. In addition to its antidepressant effects,bupropion is added to increase the bioavailability of dextromethorphan through inhibition of CYP2D6. The compound is currently being developed as a potential treatment for both MDD and TRD.
Major Depressive Disorder (MDD)
Several trials were recently completed with a positive Phase-II and a positive Phase-III trial investigating AXS-05 for the treatment of MDD. The phase-II ASCEND (NCT03595579) trial compared the effects of AXS-05 with the active comparator, bupropion (210 mg) for 6 weeks (n = 97). Results indicated that there was a statistically significant improvement (p < 0.001, d = 0.50) in the depression severity scores between the AXS-05 group and the comparator group (primary endpoint). Additionally, remission was attained in 47% of AXS-05 patients, compared to 16% of the bupropion group (p = 0.004). 21
The phase-III GEMINI (NCT04019704) study showed a statistically significant difference between AXS-05 and placebo after 6 weeks (p = 0.002, primary endpoint), with an effect size of d = 0.38. 22 Additionally, the phase-III COMET (NCT04039022) open-label, long-term safety study of AXS-05 in patients with MDD or TRD found safety profiles consistent with these previous trials. In this trial, 40% of patients met response criteria by week 2 and 73.2% by week 6. This initial response was retained by 84.6% of patients at 6 months and 82.8% of patients at 12 months. Similarly, remission criteria were met by 21.5% of patients by week 2 and 52.5% of patients by week 6. Remission was retained in 68.7% of patients at 6 months and 69% of patients at 12 months.
Treatment Resistant Depression (TRD)
In addition to being pursued as a treatment for MDD, AXS-05 is also being pursued as a treatment for TRD. The STRIDE-1 phase-III study did not show a statistically significant difference between active (AXS-05) and control (bupropion) in depression improvement after 6 weeks (p = 0.12, d = 0.21). 23
Two additional phase-II trials for TRD are currently ongoing. The MERIT study (anticipated n = 50; NCT04608396) is a randomized withdrawal study that will investigate the effect AXS-05 on preventing relapse. Another ongoing study (NCT04634669) examines safety and adverse events over 12 months of treatment with secondary endpoints examining change in MADRS score in an open-label design.
Relmada-1017
Dextromethadone (REL-1017), the d-stereoisomer of methadone, acts as a noncompetitive NMDA receptor antagonist. A Phase-II fixed-dose study (NCT03051256) evaluated the safety and efficacy of 25 mgand 50 mg doses of REL-1017 over a 7-day period in individuals with TRD. The study was primarily a safety study and was thus not powered to detect efficacy outcomes. 24 Unlike ketamine, a drug that acts on the same binding site as REL-1017, there was no evidence of dissociative effects during treatment. The currently ongoing RELIANCE-I study (NCT04688164) is a Phase-III, two-arm RCT that will test for changes in MADRS at 4 weeks in a larger sample (anticipated n = 400). REL-1017 is also currently in the preclinical research stage for uses in treating Rett Syndrome, mitochondrial diseases/ALS, and various uses in ophthalmology.
MIJ821
MIJ821 is an NR2B inhibitor being developed by Novartis Pharmaceuticals. The intravenously-administered compound is currently in Phase-II clinical trials investigating the use of the drug in the treatment of treatment-resistant depression. According to clinicaltrials.gov, one 6-arm RCT (low dose weekly, high dose weekly, low dose bi-weekly, high dose bi-weekly, placebo, and ketamine (active comparator) 0.5 mg/kg weekly) has completed, however, results have yet to be made publicly available as of March 2021 (NCT03756129). As of January 2021, there is also a 7-arm RCT (very low dose, low dose, high dose, very high dose, placebo, high dose followed by placebo, and very high dose followed by placebo) described on clinicaltrials.gov that is not yet recruiting (NCT04722666). Both studies will use the MADRS at 24 hours as the primary endpoint.
AV-101
AV-101 (L-4-chlorokyurenine or 4-CI-KYN) is a prodrug converted via the kyurenine pathway to an antagonist (7-chlorokynurenic acid) at the glycine binding site of the NMDAR NR1 subunit that has been tested for the treatment of MDD. Results from a Phase-II study of AV-101 (ELEVATE) showed that the AV-101 treatment arm did not differentiate from the placebo group on the primary endpoint (MADRS at 2 weeks). 25 An additional small (n = 19) Phase-II crossover of AV-101 in individuals with TRD (NCT02484456) was also negative (p = 0.71, d = 0.22). 26
Rapastinel
Phase II and III trials of glutamate/GABA-based compounds in development for the treatment of mood disorders.

“*,” “**,” and “***” under the Esketamine section refers to the location/sponsor of the study: Janssen Pharmaceuticals*, Celon Pharma SA**, and China Medical University, China***.BPD – bipolar depression; MDD – major depressive disorder; PPD – post-partum depression.Unless otherwise noted, the data here is taken from www.clinicaltrials.gov. Compounds are grouped by purported mechanism of action and are listed in the same order they appear in Table 2 and different studies for the same compound are listed in chronological order of estimated or actual completion date. Some completed studies did not have publicly available data.
Potential antidepressant compounds in development.

Drugs are grouped according to purported mechanism of action. Abbreviations: AMPA – α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; GABA – gamma-aminobutryic acid; mGluR – metabotropic glutamate receptor; NAM – negative allosteric modulator; PAM – positive allosteric modulator; NMDAR – N-methyl-D-aspartate receptor; Route of administration: IV – intravenous; IN – intranasal. MDD – major depressive disorder; MDSI – major depression with suicidal ideation; IND – investigational new drug; PPD – post-partum depression; BPD – bipolar depression; TRD – treatment-resistant depression
*This data is still awaiting quality control at the US Clinical Trials Registry.
NRX-100/NRX-101
This compound is a sequential treatment regimen, where a single-dose of intravenous ketamine (NRX-100) is followed by an oral formulation of D-cycloserine combined with lurasidone (NRX-101). This compound is being investigated for the treatment of bipolar depression with acute suicidal ideation or behavior. D-cycloserine is a partial agonist at the NMDA receptor and lurasidone is an antagonist at the serotonin 2a receptor as well as at the D2 dopamine receptor.
NRX-100/NRX-101 recently completed a Phase-II trial (STABIL-B), in which subjects with bipolar disorder and recent suicidal ideation or behavior who responded to an initial, single dose of intravenous ketamine were randomized to receive NRX-101 or lurasidone alone for 6 weeks. Preliminary data (posted on the US Clinical Trials Registry) demonstrate that patients who received NRX-101 exhibited lower levels of depression (MADRS, p < 0.02, d = 2.94) compared to those that received only lurasidone (NCT02974010). No participants in the NRX-101 group (n = 12) relapsed, while 2 lurasidone patients (n = 5) relapsed (NCT02974010).
NRX-101 is also currently in phase-IIb/III clinical trials (SBP-ASIB) for the treatment of severe bipolar depression and acute suicidal ideation and behavior, which is expected to complete in December 2021. One study currently recruiting is investigating NRX-100 versus placebo for the rapid stabilization of suicidal ideation and behavior in bipolar depression (SevereBD), while another phase-II/III study is investigating NRX-101 for the treatment of moderate bipolar depression and suicidal ideation (NCT03395392). Plans are also in place for a NRX-101 biomarker validation phase-II/III study investigating the effects of the compound on Glutamate+Glutamine (Glx) levels in the anterior cingulate cortex (ACC), an area that has been shown to have low levels of Glx in patients with depression and PTSD (NCT03402152). Researchers plan to compare Glx levels in patients with bipolar depression receiving lurasidone or placebo, and plan to study the effects of Glx modulation on depression.
AGN-241751 (Allergan)
AGN-241751 is an NMDA receptor modulator that is being pursued for the treatment of MDD. There is one 2-part (Part A: once daily dosing (3 mg vs. 10 mg vs. placebo) trial; Part B: twice daily dosing (3 mg vs. 25 mg vs. placebo) RCT) Phase-I/II study examining the change in MADRS score at 1-day post first dose (Part A), and at 7 days post first dose (Part B) (NCT03726658). Another Phase-II 5-arm, fixed-dose RCT is investigating AGN-241751’s effect on the MADRS at 1-day (NCT03586427). Both trials have completed, but no results have been posted as of March 2021.
Partial Allosteric GABAA Modulators
Brexanolone (SAGE-547/Zulresso™)
Brexanolone is an intravenous formulation of allopregnanolone, an endogenous neuroactive steroid, that acts as a positive allosteric modulator of GABAAreceptors through the δ subunit. 47 It is given intravenously over a 60-hour period. Allopregnanolone is the major metabolite of progesterone and its levels rise rapidly during pregnancy, followed by a precipitous drop after parturition. As the drop in progesterone levels following parturition has been linked with the pathophysiology of postpartum depression, 32 it is likely that brexanolone uses this pathway of GABAergic modulation as a therapeutic for PPD. 48
Following two positive phase-III trials, Brexanolone was approved by the FDA for the treatment of moderate to severe postpartum depression in 2019. At 60 hours, reductions in depressive symptom (HAM-D) scores in response to Brexanolone treatment were seen in the study investigating severe PPD (60 µg: p = 0.0013, d = 0.73; 90 µg: p = 0.0252, d = 0.48) and in the study investigating moderate PPD (90 µg: p = 0.0160, d = 0.43). 33 Brexanolone is the first FDA-approved compound for post-partum depression. 33 In the phase-III study of severe PPD (doses 60 and 90mcg), both doses retained statistical significance at 30 days; in the phase-III study of moderate PPD (90mcg), statistical significance was not retained at 30 days.
A phase-III study examining the drug’s safety, tolerability, and pharmokinetics in adolescent females (ages 15-17) with PPD has completed but has yet to post results as of March 2021 (NCT03665038). One planned phase-IV study was to evaluate the relationship between changes in depressive symptoms in response to brexanolone and related changes in resting state functional imaging (NCT04273191), but the company has decided not to proceed with the study at this time. Additionally, a compassionate use expanded access protocolfor brexanolone for adults with PPD was available until the drug was approved (NCT03924492).
Ganaxolone
Ganaxolone is a neuroactive steroid, similar to Brexanolone, that modulates the GABAA receptor. It is currently under development for the treatment of PPD. In one open label, pilot study in postmenopausal women, a significant decrease in MADRS score at week 8 (n = 10). 34 There are also two active Phase-II studies that have completed, but not yet published official results: one study is a multiple-dose escalation design in individuals with severe PPD that is utilizing the orally bioavailable form of the drug (NCT03460756); the other study is being conducted in individuals with moderate PPD and is utilizing the intravenous form of the drug (Magnolia study; NCT03228394). Some study results have been shared through press releases but, to our knowledge, the primary outcome data of both studies are not yet reported.
Zuranolone (SAGE-217)
Zuranolone is an oral formulation of a positive allosteric modulator of the GABAA receptor. The formulation is being investigated as a treatment for major depressive disorder, postpartum depression, and bipolar disorder. The treatment paradigm has been designed such that patients take the medicine for a 2-week course.
Major Depressive Disorder (MDD)
A Phase-II trial in patients with MDD (n = 89) achieved its primary endpoint (p = 0.0005, d = 0.81), measured as the change in HAMD-17 score at 2 weeks post randomization (NCT03000530). 35 There were no serious adverse events; those in the treatment group were more likely to experience headache, dizziness, nausea, and somnolence.
Sage has launched several Phase-III trials of zuranolone in MDD. A fixed-dose Phase-III trial (MOUNTAIN study, NCT03672175) in patients with MDD did not meet its primary endpoint (change in HAMD-17) at day 15 (p = 0.115). 36 A one-year, open-label study is ongoing and is assessing the safety, tolerability, and need for re-treatment with zuranolone in subjects with MDD (SHORELINE, NCT03864614).Interim results were released in October 2020 stating that 71.6% of patients responded and 39.8% of patients remitted following a 2-week course of treatment with 30 mg dose. Adverse event profiles were generally consistent with earlier trials. 37 Of responding patients who continued in the protocol, 44.5% did not receive an additional Zuranolone treatment course for up to one-year of follow-up. Two studies (RAINFOREST [NCT03771664] and REDWOOD [NCT04007367]) are listed as suspended to “evaluate potential amendments to the study” according to clinicaltrials.gov as of March 2021. Additionally, two new phase-III studies were recently launched in June 2020 that will evaluate the efficacy of Zuranolone in the treatment of MDD through the use of the 17-item HAM-D scale comparing Day 15 to baseline. One study will evaluate the efficacy of zuranolone monotherapy compared to placebo (NCT04442490), and the other will measure zuranolone + sertraline compared to placebo+ sertraline (NCT04476030). It should be noted that some later trials of zuranolone are looking at a higher dose (50 mg) than earlier trials (20 or 30 mg).
Sage Therapeutics also announced in a 2019 press release that zuranolone may be investigated as a potential therapy for treatment-resistant depression (TRD), however, no trial has been registered on clinicaltrials.gov as of the time of writing. 49
Post-partum Depression (PPD)
Zuranolone is also being developed as treatment for postpartum depression (PPD). According to a PowerPoint presentation at the International Marcé Society in October 2020, the Phase III trial, ROBIN (PPD-201, NCT02978326) achieved its primary endpoint of reduction in HAMD-17 scores at 2 weeks when investigating the effect of a 30 mg dose of zuranolone compared to placebo in women with severe PPD (Cohen’s d = 0.40; p = 0.0029). 38 A 2-arm, phase-III RCT investigating the effects of zuranolone on depressive symptoms in females with severe postpartum depression (PPD) as compared to placebo, is currently recruiting. This study will examine depressive symptoms using the 17-item HAM-D, with the primary endpoint being the change from baseline in the HAM-D total score at day 15 (NCT04442503).
Bipolar Depression (BPD)
In addition to being pursued as a treatment for MDD and PPD, Zuranolone is also being pursued as a treatment for bipolar depression in a two-part, phase-II study (NCT03692910). This study of zuranolone in subjects with Bipolar I/II Disorder with a current major depressive episode reported that all treatment-emergent adverse events were mild or moderate during the open-label trial (Part A), and that there was a 45% response rate by Day 15. The study has completed, but results from Part B of the study (2-arm parallel group) have not been reported as of March 2021.
PRAX-114
PRAX-114 is a GABAA positive allosteric modulator being developed by Praxis Precision Medicines that is currently in Phase-II trials for the treatment of MDD. One non-placebo-controlled study is investigating the safety and tolerability of PRAX-114 over a 14-day treatment period. According to the Australian New Zealand Clinical Trials Registry (ANZCTR), the study began enrolling in May 2019. As of January 2021, no official end date or results have been posted, but Praxis reported interim results for this study in their report to the SEC in September 2020. Secondary outcomes were improvement in depression severity; change from baseline in the Hamilton Depression Rating Scale were seen in the 45-mg, 60-mg, and 80-mg doses over an 8-day period. A reduction of 15 to 19 points from baseline were noted across the three dose groups. 39
In October 2020, Praxis submitted the Investigational New Drug (IND) submission to the US FDA for the initiation of a randomized, placebo-controlled Phase II/III clinical trial for PRAX-114 in MDD, but as of November 2020, this IND submission has been placed on full clinical hold by the FDA pending the resolution of nonclinical pharmacology and toxicology matters. 50
Future Challenges and Conclusions
Despite the fact that several large pharmaceutical companies have disinvested from the development of CNS drugs, 51 there have been several exciting developments in the treatment of mood disorders recently. Nonetheless, this novel approach targeting the amino acid neurotransmitter systems also brings new challenges. In contrast to traditional antidepressants that are generally designed to maintain a relatively steady-state serum drug level, several new antidepressants are designed to be delivered in a pulsatile fashion. Ketamine, for instance, which has a half-life of 2-4 hours, has downstream effects on the brain that outlast the drug’s persistence in the body. For many of these newer agents in the pipeline, questions of the optimal dose schedule remain unanswered.
Another challenge in the development of drugs targeting the amino acid neurotransmitter systems, many of which are purportedly rapid-acting, is the way that changes in mood are assessed. Most clinical trials rely upon the Hamilton Depression Rating Scale (HAM-D) 52 the Mongtomery-Asberg Rating Scale (MADRS) 53 to measure changes in symptom severity. Both of these scales were designed to evaluate symptoms over a period of weeks, not hours to days. For instance, both the HAM-D and the MADRS evaluate sleep and appetite as core features of depression, but it is unrealistic to expect these symptoms to change realibly over a period of hours. The development of improved measurements to assess changes in symptoms severity for rapid-acting agents should be considered.
Finally, the heterogeneity of mood disorders and our incomplete understanding of their pathophysiology makes it critical to recruit well defined clinical cohorts. Related to this, placebo response in clinical trials for mood disorders can be difficult to predict, rendering it challenging to show a placebo-adjusted improvement. Given that some agents still under investigation may be using a non-parenteral route of administration, these non-specific therapeutic effects can be even further enhanced in a given clinical context. Careful attention to these non-specific effects are critical to conduct successful clinical trials.
These challenges notwithstanding, it is an exciting time in the development of novel treatments for mood disorders. Two agents recently received FDA approval and several additional promising therapeutics are being evaluated in later stage clinical trials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Wilkinson has received contract funding from Janssen, Sage Therapeutics, and Oui Therapeutics for the conduct of clinical trials administered through Yale University; he has received consulting fees from Biohaven Pharmaceuticals, Sage Therapeutics, Janssen, Eleusis, and Oui Therapeutics. The other authors report no financial relationships with commercial interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: P.A.D was supported by an NIH Medical Scientist Program Training Grant T32GM136651. There was no direct support for this work.
