Abstract
Changes in vision can have significant impact on health and quality of life. Differences between women and men have been identified in the incidence of several eye conditions. Some of these differences are a result of the greater longevity of women. However, the eye, like other organs, is susceptible to the impacts of inflammation and sex steroids. Conditions, such as thyroid eye disease, optic neuritis, and dry eye disease are related to autoimmune or inflammatory conditions and are, thus, more common among women. Idiopathic intracranial hypertension occurs disproportionately in women of childbearing age; the etiology of this condition appears to be related to both inflammatory and sex hormone fluctuations.
Eye Conditions in Women
Sex and gender influence the risk for and presentation of diseases in all organ systems. While more frequently recognized and discussed in such areas as musculoskeletal and cardiac conditions, the impacts of sex and gender differences are less commonly discussed in ophthalmologic diseases. The ocular surface is complex, and several structures are involved in maintaining tear film homeostasis. Sex-associated differences in ocular physiology have been identified in not only the meibomian glands 1 but also the lacrimal glands (differences in lacrimal gland size), 2 conjunctiva (women have fewer goblet cells than men), 3 and cornea (thickness, hydration, curvature, sensitivity, endothelial pigmentation, contact lens tolerance, and higher prevalence of decreased visual acuity during the menstrual cycle, pregnancy, and menopause), 4 as well as other ocular structures (Figure 1). 5
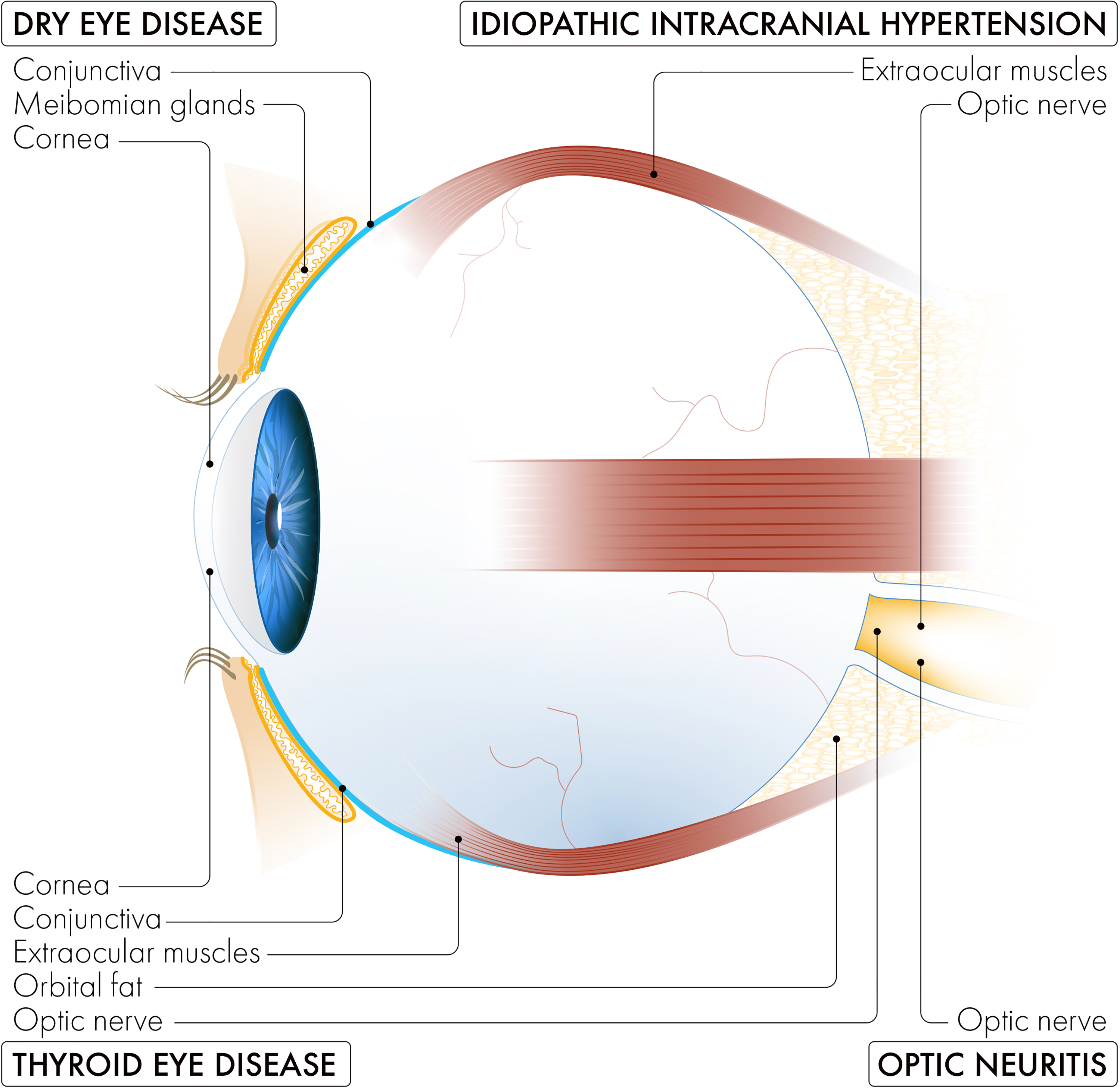
Sructures of the eye that are impacted by conditions that disproportionately affect women.
Maintenance of vision has a significant impact on quality of life, especially among an aging population, and women make up two-thirds of the blind and visually impaired in the United States. 6 This may not entirely reflect the direct impact of sex on eye function and has been attributed, for some conditions, to the fact that women live longer than men. Cataracts, glaucoma, and macular degeneration, in particular, have an enormous impact on the quality of life of women and are examples of age-related eye diseases that disproportionately affect women simply because more women than men live long enough to develop them.
However, some conditions of the eye demonstrate sex-based differences beyond that attributable to differences in life span. Similar to what is seen with conditions in other organ systems, the heightened inflammatory response and susceptibility of women to the development of autoimmune diseases make them more vulnerable to certain eye conditions. As with other tissues, those of the eye are affected by the exaggerated immune response seen with autoimmune diseases, resulting in damage and dysfunction. Tissues of the eye are also affected by lifelong sex hormone fluctuations, independently as well as through secondary influences on inflammatory processes (Table 1).
How estrogen and testosterone impact ophthalmic structures.
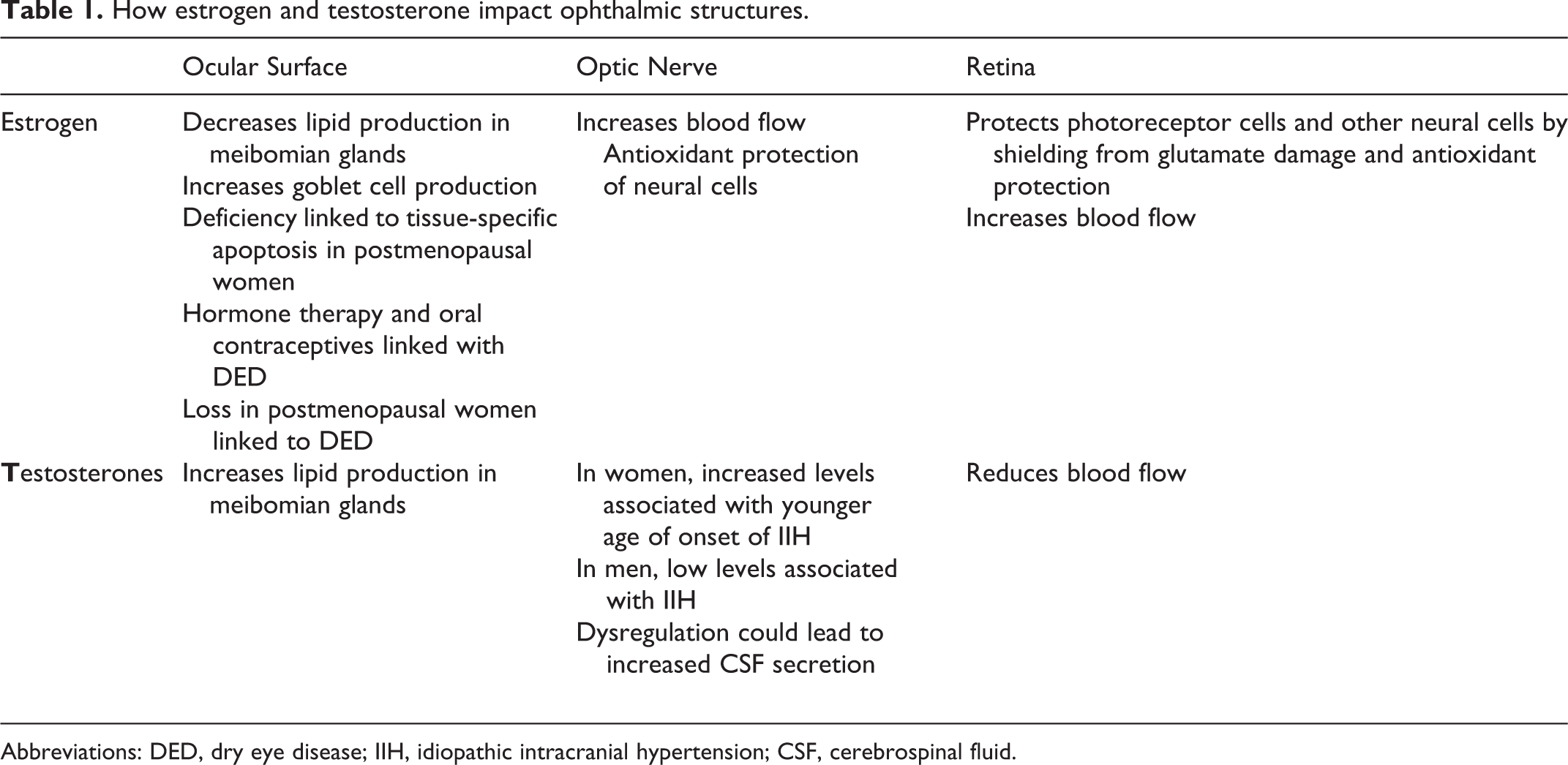
Abbreviations: DED, dry eye disease; IIH, idiopathic intracranial hypertension; CSF, cerebrospinal fluid.
The development of autoimmune diseases is multifactorial, with estrogens acting as potent stimulators of autoimmunity. 7 Specifically, 17-β estradiol and prolactin act as enhancers of humoral immunity and testosterone and progesterone act as immune suppressants. 8 However, some studies have shown that estrogen may have a protective effect on the eye, especially the optic nerve and retina. The protective effect can occur both directly, such as shielding retinal photoreceptor cells from glutamate damage 9 or antioxidant protection of neural cells, 10 and indirectly, by modulating blood flow to the optic nerve and retina. The vasodilatory properties of estrogen have been shown to improve ocular blood flow, while androgens impede retinal blood flow. 11 This protective effect of estrogen is reflected in the differing presentations between males and females at younger ages, compared to similar presentations in men and postmenopausal women. This may also explain why, although many of these pathologies are rarer in men, men suffer worse outcomes. 12
Thyroid eye disease, optic neuritis, and dry eye disease (DED) are ophthalmic manifestations of autoimmune disease and, as such, are more prevalent in women than in men. Idiopathic intracranial hypertension (IIH) is a potentially devastating disease that disproportionately affects women of childbearing age. Idiopathic intracranial hypertension has no known autoimmune link, and the etiology is unclear, although inflammatory and hormonal causes have both been implicated. These 4 diseases of the eye affect millions of women of all ages and have a significant impact on quality of life and health.
Thyroid Eye Disease
Thyroid eye disease (TED) is a self-limited orbital inflammatory condition which can be sight-threatening, debilitating, and disfiguring. 13 The most widely established risk factors for development or worsening of TED include female sex, older age, and smoking. 14 The incidence of TED is 16 per 100 000 females and 2.9 per 100 000 males with an approximate prevalence of 0.25%. 15
The most common presenting sign of TED is lid lag (occurring in 90% of patients with TED), followed by exophthalmos (60%) and restriction of eye movements (40%). 16 Exposure keratopathy and optic neuropathy may develop, which can lead to blindness. Diplopia has been reported in over 50% of patients with TED and up to 26% report low vision. 17
Most patients with TED present with a current or past medical history of hyperthyroidism (Graves’ disease), but TED can also be seen in hypothyroid or euthyroid patients. 18 In patients with TED, activation of fibroblasts in the retro-orbital tissues results in accumulation of hydrophilic glycosaminoglycans. 19,20 Although the precise mechanism of development of TED is not well understood, overexpression of thyroid-stimulating hormone receptors (TSHR) has been found in the retro-orbital tissues in patients with TED, and autoantigens to TSHR are found in patients with Graves’ disease. It has been proposed that stimulation of these receptors by autoantigens results in activation of fibroblasts. 21 Orbital fat and extraocular muscles enlarge, displacing the eye forward, restricting ocular movements and occasionally leading to compression of the optic nerve.
While the impact of sex has been studied extensively in the development of Graves’ disease, the study of how sex plays a role specifically in TED is limited. The prevalence of TED, although higher in females than in males, occurs at a lower rate in females when compared to Graves’ disease alone. Marcocci et al found a female:male ratio of Graves’ disease with orbitopathy of 2.1:1, but that ratio rose to 3.4:1 in patients with Graves’ disease without orbitopathy. 22
There is emerging evidence that development of TED occurs in both sexes, 23 although mechanisms may differ. Schlüter et al developed a mouse model of Graves’ orbitopathy. The model was used to study manifestations of ophthalmopathy in both sexes in the absence of additional human risk factors, such as advanced age, genetic variabilities, or smoking. Human TSHR A-subunit encoding plasmid was introduced to male and female mice, and both sexes comparably developed autoimmune hyperthyroidism. Both males and females developed inflammatory eye symptoms and proptosis at equal rates, although males developed ophthalmopathy earlier. Histologic differences were noted between the sexes, with infiltration of T cells, extension of brown fat and collagen deposition seen in males, and predominantly macrophage infiltration in muscles and connective tissue and muscle hypertrophy in females. The widespread overall collagen deposition found in males was thought to be evident of earlier acute inflammation and faster progression to chronic inflammation. The study of pathologic samples was potentially limited by subjective judgment during simple visual assessment and by use of middle sections of the orbit only, limiting the ability to examine the full extent of disease. 23 However, these data from an animal model may not translate to what is seen in humans. Kendler et al found that men were more likely to have limitation of upward duction and increased intraocular pressure on upgaze when compared to women. This finding is consistent with extraocular muscle hypertrophy, which contradicts the histological finding of predominant muscle involvement in female mice and lack of extraocular muscle hypertrophy in male mice. 23,24 More research is needed in this area.
Although TED is more common in women, men are 4 times more likely to suffer from severe TED 25 and are also more likely to have asymmetric disease. 24,26 No specific sex-based explanations have been noted for this. In addition, the vast majority of studies have not found sex differences in response to treatment of Graves’ disease or rates of remission. 27 Two studies have shown increased rate of relapse in males compared to females, but the definition of “relapse” in those studies did not meet the American Thyroid Association definition of relapse, which requires 1 year of euthyroidism before hyperthyroidism recurs. 27 Smoking is a well-established risk factor for progression and increased severity of TED. 28 The increased rate of smoking among men when compared to women is another proposed explanation for the increased severity of disease seen in men. However, Manji et al found that the more severe presentation in men persisted when controlling for smoking status. 29 There is some evidence that smoking carries a greater risk of disease progression for females than it does for males. 30
Gender may play a more significant role than sex in the development of more severe TED among men. Poorer compliance and follow-up in men could prevent or delay men from reaching a euthyroid state, thereby prolonging disease activity and increasing severity of ophthalmolopathy. 31 In addition, decreased sensitivity of some men to changes in their cosmetic appearance may result in delays in seeking treatment until the disease signs and symptoms become more obvious and seemingly urgent. 25,32
More research needs to be conducted into the mechanisms of development of TED, especially differences between the sexes and how that may manifest in clinical presentation and affect the optimal treatment regimen. It may be that sex does not play a primary role in the development of TED, although animal model studies would indicate some influence of sex, but that TED is seen more often in females because of the higher prevalence of Graves’ disease. Regardless, TED is still a major concern in women’s health. In a recent study of patients with hyperthyroidism, male sex and the absence of ophthalmopathy were associated with better quality of life. 33
Optic Neuritis
Optic neuritis (ON) is a condition that disproportionately affects young adult women. 34,35 The incidence of ON is 5.1 per 100 000 person-years, with a prevalence of 115 per 100 000 people, and two-thirds of those affected are women. 36 Patients most commonly present with monocular visual field deficits, pain that is exacerbated by eye movement, and an afferent pupillary defect. A swollen optic disc is only seen in about one-third of patients. 37 Although the pathophysiologic mechanism has not been definitively determined, it is thought to occur from an inflammatory event causing demyelination of the optic nerve possibly facilitated by dysregulation of T cells 38 and aberrant immune activation via B cells. 39
Optic neuritis is often the initial clinical event of multiple sclerosis (MS), with 15% to 20% of patients with MS having ON as their initial clinical finding. 40 As with other autoimmune conditions, MS affects a higher preponderance of females; some studies suggesting as many as 3 times more women are affected compared to men. The Optic Neuritis Treatment Trial showed that 72% of patients with acute ON and demyelinating lesions on MRI of the brain progressed to MS within 15 years, while 25% of patients without lesions evident on MRI progressed to MS within 15 years. 41 Patients who present without lesions at baseline are more commonly women, whereas there is no significant sex predilection among patients with lesions at baseline. 41 Since early initiation of treatment has been shown to be effective in slowing the progression of MS 42 and given the significant correlation of ON with MS, especially among women, a high index of suspicion to identify ON is critical. 43
The sex-based factors related to the increased prevalence of ON among women, specifically in relationship to MS, have not been extensively studied. Estrogen has been demonstrated to have a neuroprotective effect in a variety of conditions. 44 Pregnancy, especially in the third trimester when estriol levels are higher, has been linked to lower rates of relapse 45,46 and delayed onset of MS. 47 However, no studies have specifically looked at the impact of estrogen on the development of ON or its impact on the future development of MS.
Burton et al looked at loss of nerve fiber layer and ganglion cell layer in patients with ON. Both cell types are essential for optic nerve function. Patients with vitamin D deficiency showed a higher rate of loss of both types of cells. Correcting that deficiency led to women having better visual acuity at the end of the study, likely related to the protective effect of vitamin D on the optic nerve. The authors theorized a synergistic effect between treatment with vitamin D and female sex. 48 Although the sample size was small, this study suggests that vitamin D treatment should be considered in female patients with ON, especially those who are vitamin D deficient, and at a minimum warrants further study. A randomized, placebo-controlled study of vitamin D treatment for patients with unilateral ON but without demonstrated vitamin D deficiency showed no protective benefit to the nerve fiber layer after 6 months of treatment. The sample size was small, but this may suggest that while correction of vitamin D deficiency protects the nerve fiber layer, supplementation with vitamin D in the setting of normal vitamin D levels offers no added benefit. 49
As with other ophthalmic diseases with demonstrated sex-based differences, ON is less commonly diagnosed in men, but they are more likely to have worse outcomes. Costello et al noted improved visual recovery in women: At 3 months after the onset of ON, 75% of women regained functional vision (based on results of visual field testing), compared to only 25% of men. 50 At 1 year, visual acuity is recovered in 90% to 95% of patients. 51 However, other visual functions may not be fully restored, such as contrast sensitivity and color vision. 52 These measures may not be as readily apparent but can negatively affect a patient’s quality of life. 53
Dry Eye in Women
Dry eye disease is a chronic condition of the ocular surface associated with ocular irritation, blurred vision, and eye pain. Women are disproportionately affected by DED, especially with increasing age, making postmenopausal women the most likely to suffer from DED. 5,54 -62 Dry eye disease not only causes ocular discomfort but also interferes with quality of vision. As vision is integral to performing almost every daily task, from eating to getting dressed to performing in a professional setting, poor vision can have a significant impact on capacity to function in daily life. The ability to read, work on a computer, or drive a car may be dramatically limited among those with DED. 63 Identifying and treating DED in women is critical to maintaining quality of life and the ability to live independently as women age.
Historically, DED has been overlooked as a serious condition. It was only formally defined as a disease in 1995 on the basis of consensus of a working group from the National Eye Institute (NEI)/Industry on Clinical Trials in Dry Eye. 64 The initial definition was “a disorder of the tear film due to tear deficiency or excessive tear evaporation which causes damage to the interpalpebral ocular surface and is associated with symptoms of ocular discomfort.” Our increased understanding of the complexity and impact of DED is illustrated by the Tear Film and Ocular Surface Society Dry Eye Workshop II updated definition created in 2017. A global consensus committee was created to create the expanded definition: “Dry eye is defined as a multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.” 65
Dry eye disease is an inflammatory condition that is chronic and may be progressive. Multiple factors can initiate an inflammatory cascade that causes damage to the ocular surface. 66 This ongoing damage then increases the inflammatory response, which perpetuates the “vicious cycle” of DED. Tear film instability may lead to visual changes due to the loss of light refraction provided by the aqueous layer. If allowed to progress, dry eyes can cause neurosensory damage that can lead to a less responsive tear reflex. 67 Dry eye disease can be further compounded by other lid or ocular surface abnormalities, creating further damage to the ocular surface. 5,68 Because sensory nerves can be damaged by the chronic inflammation associated with DED, dry eye can be considered a type of chronic pain condition.
Consistent with this inflammatory etiology, severe DED can be seen in patients with several autoimmune conditions that are much more prevalent in women, especially Sjögren syndrome (9:1 women:men), systemic lupus erythematosus (SLE) (7:1), rheumatoid arthritis (RA) (3:1), and TED (6:1). 7,69,70 About 20% of patients with SLE also have Sjögren syndrome. 71 In both Sjögren syndrome and SLE, the lacrimal glands are affected, contributing to DED. 71,72 Dry eye disease is common in patients with RA, reported in 15% to 25% of patients, and thought to be due to the inflammatory effects of RA. 73,74 In autoimmune TED, the resulting inflammation and swelling of orbital tissues leads to incomplete blinking and inability to fully close the eye. 70 This illustrates the importance of carefully monitoring patients with autoimmune disease for signs and symptoms of DED. 71
Reflecting the relationship to autoimmune diseases that are typically more common among women, DED has consistently been shown across epidemiologic studies and meta-analyses to be as much as twice as prevalent in women compared to men. The Women’s Health Study (2003) and the Physicians’ Health Studies (2009) estimated that 3.25 million women and 1.68 million men had been diagnosed with DED. 75,76 The National Health and Wellness Survey, published in 2017, showed that 6.8% of the US adult population (approximately 16.4 million people) have been diagnosed with DED. 77
Prevalence of DED increases with age in both men and women. 55–60,75,76,78 However, a meta-analysis of recent DED prevalence studies was conducted by the 2017 TFOS DEWS II researchers and showed that women had a higher prevalence of signs and symptoms of DED starting at a younger age, with a prevalence of 14% at the age of 50, which increases to 22% over the age of 80 years. For men, DED symptoms become more prevalent at 60 to 69 years of age, with a prevalence of 7%, which then increases to 13% at 80+ years of age. 54
While the differing inflammatory responses between the sexes and the increased incidence of autoimmune diseases among women may help to explain sex-based differences in prevalence of DED, other factors, such as the role of sex steroids, need to be assessed. The relationship between sex steroids and DED has been well established, but the specific role sex steroids play has yet to be fully determined. Hormones play an important role in tear film homeostasis, 79 and low androgen levels are the most consistent hormonal association with DED. 5,80 Estrogens suppress lipid production, while testosterone stimulates lipid production, which can affect the secretions of the meibomian glands. 1 Estrogen also stimulates goblet cell production. 81 In a study looking at DED symptoms in women from the beginning of pregnancy through 6 weeks postpartum, symptoms were at their highest level in the third trimester and lowest 6 weeks postpartum. 82 Estrogen deficiency has been linked to postmenopausal DED by initiating tissue-specific apoptosis. 83 Unfortunately, studies on the role of both systemic and ocular testosterone levels have shown conflicting and/or inconclusive results. Some of this may be due to the nonstandardized methods of studying DED. For example, one study based the determination of severity of DED on detailed eye exams, and levels of 17-β-estradiol, estrone, and total testosterone were shown to have an inverse correlation with tear film osmolarity (high osmolarity is a sign of poor tear film homeostasis). 84 Another study, using OSDI scores (which are based on survey results), found no significant difference between blood serum levels of testosterone and severity of DED. 85
Given the potential role of sex hormones in the development of DED, the use of hormones as treatment for DED has been proposed but not well studied. 79,83 In a 2001 case report, topical androgen therapy was used to successfully restore the lipid layer of the tear film, 86 but no further study has occurred. Several studies have looked at the effect of hormone replacement therapy on dry eye and have demonstrated no effect or even worsening of symptoms. 75,87 -89
In the Women’s Health Study, postmenopausal women treated with hormone therapy were at a 70% increased risk of DED when treated with estrogen alone and a 30% increased risk when treated with estrogen + progesterone/progestin. 75 Higher doses and increased duration of treatment were also associated with increased DED risk. 90 Oral contraceptive use in premenopausal women was also associated with increased severity of DED based on patient questionnaires. 91 The loss of estrogen receptors in postmenopausal women may explain why hormone replacement therapy is ineffective in this population. 81 More research is needed regarding the impact of sex hormones on DED in women of all ages.
Iatrogenic causes and gender-specific behaviors also contribute to the increased prevalence of DED in women. 5 For example, women make up two-thirds of contact lens wearers in the United States. 92 Contact lens wear has been associated with increased incidence of DED. 93,94 One study found that the incidence of discomfort related to contact lenses was higher in women than in men. 95 After laser vision correction, women are more likely to report increased level symptoms of DED 3 months after surgery when compared to men. 96
Cosmetic botulinum toxin type A (BTX-A) injections into the lateral canthal rhytids, a common technique for eliminating crow’s feet, results in temporary muscle weakness and incomplete blinking, which can lead to dry eye or exacerbate preexisting DED. 97,98 Interestingly, BTX-A injections have also been posited as a treatment of dry eye, as the incomplete blink can result in reduced lacrimal drainage. 99
Cosmetic eye tattooing, also known as dermapigmentation, can cause destruction of the meibomian glands, leading to tear film instability. 100 Cosmetics, such as mascara and eyeliner, and topical facial creams often contain ingredients that are harmful to the tear film. Retinoids, which are often found in facial creams, have been implicated in meibomian gland damage as well. 62
Sex-based differences in the usage of topical eye drops has not been studied, but because women are more likely than men to have DED, it can be theorized that women are more likely to use lubricant eye drops. Women also live longer than men and therefore have a higher incidence of age-related ophthalmic conditions, such as cataracts or glaucoma, that may require the use of eye drops to prevent postoperative infection, inflammation or pain, or to lower intraocular pressure. Most ophthalmic preparations contain preservatives in order to prevent bacterial contamination. Benzalkonium chloride is the most widely used preservative in ophthalmic drops and it has been shown to have pro-inflammatory and cytotoxic effects on the eye and to disrupt the tear film due to its detergent properties. 101 While preservative-free formulations of many eye drops are available, they are usually more difficult to find and are more expensive and may not be a practical alternative for all patients. In addition, many classes of systemic medications have been shown to increase risk of DED, including antidepressants, 102,103 anxiolytics, antipsychotics, and antihistamines. 101,104
Not only does DED affect the quality of vision, but it is also associated with increased levels of anxiety, depression, and sleep deprivation. 105 -109 Wan et al performed a systematic review and meta-analysis of studies that reported on the prevalence, incidence, and/or severity of depression and/or anxiety in patients with DED versus healthy controls. Patients with DED demonstrated a markedly increased prevalence of depression (summary odds ratio [OR] = 2.92, 95% confidence interval [CI]: 2.13-4.01, P < .00001) and anxiety (OR = 2.80, 95% CI: 2.61-3.02, P < .00001). Furthermore, the depression score (standardized mean difference [SMD] = 0.81, 95% CI: 0.48-1.15, P < .00001) and anxiety score (SMD = 0.37, 95% CI: 0.10-0.64, P = .007) were higher in DED patients than in controls. 108 Individuals with anxiety and depression are more likely to be more symptomatic from their DED. 107 For these reasons, DED can have a significant impact on quality of life. One study has correlated DED with an increased risk of suicidal ideation (OR = 1.24, 95% CI: 1.05-1.48). 110 It should be noted that the study did not control for the presence of depression and did not establish a causative relationship between DED and suicidal ideation.
When compared to men with DED, women with DED report worse health-related quality of life, worse perceived health, and worse quality of vision. 59,61,105,111 Women also report more limitations on their activities of daily living, such as reading, using the computer, or driving. 59 Miljanović et al found that those with DED were more likely to report problems with reading (OR = 3.64, 95% CI: 2.45-5.40, P < .0001), carrying out professional work (OR = 3.49, 95% CI: 1.72-7.09, P = .001), using a computer (OR = 3.37, 95% CI: 2.11-5.38, P < .0001), TV watching (OR = 2.84, 95% CI: 1.05-7.74, P = .04), driving during the day (OR = 2.80, 95% CI: 1.58-4.96, P < .0001), and driving at night (OR = 2.20, 95% CI: 1.48-3.28, P < .0001). 63
Dry eye disease has a significant impact on women’s health and well-being and should be considered and screened for in women as they age, women undergoing cosmetic procedures, women on medications associated with DED, and in any woman with an autoimmune condition. On a positive note, women are more likely than men to seek care for DED and to use treatments for DED, including lubricant drops and ointments, hot compresses, cyclosporine eye drops, and punctal occlusion. However, while women spent more money per month on dry eye treatments, they expressed greater dissatisfaction with treatment side effects and the amount of time before efficacy of treatment was seen. 59 Because DED is a chronic and progressive condition, it is critical that DED is recognized and treated in women in its early stages in order to maximize the quality of life and the ability to live independently.
Idiopathic Intracranial Hypertension
Idiopathic intracranial hypertension is a disorder that is more common in women, especially between the ages of 20 and 50; only 5% of those affected are men. 112 Idiopathic intracranial hypertension is characterized by elevated intracranial pressure of no known etiology when all other causes have been excluded. 113 Obesity is a risk factor; consequently, the most typical profile of a patient with IIH is an obese woman of childbearing age. 114 Among studies performed in the United States, the incidence of IIH was found to be 0.9 to 1.0 per 100 000 in the general population, increasing to 1.6 to 3.5 per 100 000 in women and to 7.9 to 20 per 100 000 in overweight women. 115 While this is a disease primarily of women of childbearing age, the etiology behind this sex-based difference has not been well studied.
Permanent vision loss occurs in up to 25% of patients with IIH, with a 1% to 2% incidence of blindness each year. 116,117 Although the disease is more common in women, men are twice as likely to sustain severe vision loss, similar to the worse outcomes seen in men with other eye conditions. Most patients also report debilitating headaches. 118 There is significant economic burden for individuals with IIH as well as for society. In the United States, the total economic costs are estimated at greater than US$444 million per year. The high costs are due to frequent hospitalizations of working-age individuals, leading to loss of productivity. A significant loss of earnings is reported in 57% of patients, with a forced change in occupation in 31%. 119
There are many proposed etiologies of IIH, several of which may be sex-related. Cerebral venous outflow abnormalities, increased cerebrospinal outflow resistance, increased abdominal and intracranial venous pressure due to obesity, altered sodium and water retention mechanisms, and abnormalities of vitamin A have all been posited. 120 Chronic inflammation has been implicated as well. 121
One possible mechanism for the role of chronic inflammation in development of IIH is dysfunction of 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1). 11β-Hydroxysteroid dehydrogenase type 1 can be found in a wide variety of tissues, including the choroid plexus, and plays a role in mediating local cortisol availability and activity. 122 Cortisol is an important anti-inflammatory mediator that in the choroid plexus is thought to drive epithelial sodium transport on the apical membrane of epithelial cells and establish an ionic gradient, promoting cerebrospinal fluid production. 123 Therefore, 11β-HSD1 may have a role in influencing the regulation of CSF production and, as a result, intracranial pressure. 124,125 Preliminary studies have shown that 11β-HSD1 activity is reduced in patients with IIH who undergo therapeutic weight loss. 121 An ongoing randomized clinical trial is assessing the effects of 11β-HSD1 inhibition in reducing intracranial pressure in patients with IIH. 126
Because IIH is found almost exclusively in females of reproductive age and no differences in incidence between males and female are noted before puberty, abnormalities in sex hormone regulation have been implicated in the development of the condition. 127,128 Sex steroids have been found to play a role in regulating 11β-HSD1, and there is reduced enzyme activity in women compared to men. 129,130 Sinclair et al theorize that in patients with IIH, abnormal sex hormone metabolism results in the loss of the normal suppression of 11β-HSD1 activity seen in women. 121
Klein et al used radioimmune assays to evaluate the roles of cortisol, testosterone, bioavailable testosterone, prolactin, dehydroepiandrosterone sulfate, androstenedione, insulin, aldosterone, estradiol, follicle-stimulating hormone, and luteinizing hormone in IIH and found that women with younger age of onset of IIH had elevated testosterone and androstenedione. However, a control cohort was not evaluated. 131
While androgen excess may be a risk factor for development of IIH in women, especially those with younger age of onset, it does not correlate with body mass index, waist to hip ratios, or duration of IIH. 131 Patients with IIH have increased adipose tissue, which plays a major role in peripheral androgen generation. 132 Increased intra-adipose androgen activation is observed in women with simple obesity. 133 There are case reports of transgender males who developed IIH after commencement of testosterone therapy. 134 -138 These results should be considered with caution as confounding factors such as weight gain may also have contributed elevated intracranial pressure. Furthermore, polycystic ovary syndrome, which has a cardinal defining feature of androgen excess, has a much higher prevalence in women with IIH (up to 57%) compared to the general female population (5%-10%). 139
In a recent case–control study, increased CSF androgen levels were found in patients with IIH compared to both obese and lean controls. However, controls were not carefully matched in either of the control cohorts, and only females were included in the study. In rat cell lines, expression of androgen-converting enzymes was found in the choroid plexus. Study of human choroid plexus samples showed increased activity of sodium–potassium pump in the presence of testosterone. Increased androgen levels in patients with IIH, the expression of androgen-converting enzymes in the choroid plexus, and the increased pump activity in the presence of testosterone support the hypothesis that androgen dysregulation could possibly lead to increased CSF secretion. 120
While women with IIH seem to have higher levels of androgens, men tend to have lower levels. Fraser et al found that men with IIH were more likely than case–controls to have signs and symptoms of testosterone deficiency. However, this was based on results of the ADAM questionnaire and not actual hormone levels. 140 Another study of 8 men with IIH, however, found low testosterone levels in 7 individuals, while 2 also had abnormal estradiol levels, and 4 had abnormal follicle-stimulating and luteinizing hormone levels. 141 There are also case reports of men treated with androgen deprivation therapy for prostate cancer who have developed IIH after induction of hypogonadism. 142
Idiopathic intracranial hypertension follows a paradoxical pattern similar to autoimmune disease: Women are affected in greater numbers, but men are affected more severely. 143 One possible explanation for this seeming paradox was posited by Hornby et al. There may be a window of abnormal serum testosterone levels that is shared by men with androgen deficiency and women with androgen excess and in which there is an increased risk of developing IIH. 138 In addition, testosterone has been shown to exhibit sexually dimorphic effects on human metabolism. 144 Differences in signaling or sensitivity to androgen receptors between the sexes or epigenetic modifications to local androgen action may help explain this sexually dimorphic response. 145 The role of other hormones in the development of IIH has not been well studied, or studies are limited by small sample sizes. Leptin is a hormone involved in hypothalamic control of weight regulation and satiety. 146 Increased leptin levels have been observed in the serum 147 and CSF 148 of obese women with IIH when matched with normal obese female controls, thus indicating that patients with IIH may have an abnormally elevated leptin level beyond what can be explained by increased adipose tissue. The presence of elevated leptin in both serum and CSF suggests that leptin crosses the blood–brain barrier freely in patients with IIH. Normally, leptin CSF levels are low in obese individuals. As a result, leptin resistance has been theorized as a potential causative factor in the development of IIH. 138,148
When sex-based differences exist in presentation or outcome of a disease entity, gender-based differences in health-care-seeking behavior must also be considered. Two studies looking at the differences in IIH between men and women have shown that the time from the onset of first symptoms to diagnosis was shorter in men. 143,149 Men are less likely than women to present with headache as an initial symptom; the most frequent presenting symptom in men is vision changes. Based on the observation that men are much less likely than women to complain of migraine or tension headache pain, 150 -152 Bruce et al theorized that men may have a greater threshold for headache pain or may be less likely to seek care for this. This is similar to findings from other studies of chronic pain that indicate either sex-based differences in pain perception or gender-based differences in expression of pain. As a result, men appear to be less symptomatic with headache at the onset of elevated intracranial pressure and only present once optic nerve damage has occurred and vision changes are evident.
Conclusion
Good vision and good health are closely intertwined and are essential for quality of life. While some eye conditions are more common in women due to their longer lifespan, there are also distinctive sex-based manifestations of ophthalmic diseases in women, related to increased inflammatory responses and the impact of sex hormones. This predilection for certain ophthalmic pathologies means that ocular health must be uniquely considered in women. In younger women of childbearing age, IIH and ON can have life-altering consequences as well as a profound impact on pregnancy and fertility issues. Thyroid eye disease can occur throughout life, and detecting an abnormal thyroid level early on can ameliorate the severity of disease and decrease permanent impact on quality of vision and life. Dry eye can occur throughout life but increases in prevalence and severity with age, limiting a woman’s ability to live independently as she ages. While complete eye exams with an ophthalmologist are an important part of routine health maintenance in all women, especially in women with autoimmune diseases, awareness of these disorders across all specialties can assist in getting women care early. Further study needs to be done on these 4 diseases to better understand their etiology and options for prevention and treatment. In addition, future research is needed to assess the impact of sex on other ophthalmic diseases in order to provide optimal care and maintain optimal quality of vision and life for women of all ages.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
