Abstract
In this selective review, emerging literature linking biological sex, sociocultural gender, and sexual orientation to stress hormone functioning and multisystemic physiological dysregulations are summarized. Beyond sex as a binary biological variable, continuums of sex hormones, gender roles, gender identity, and sexual orientation each uniquely help delineate pathways and mechanisms linked to stress-related disease trajectories. This implicates glucocorticoid functioning and allostatic load, the “wear and tear” of chronic stress in synergy with unhealthy behaviors. Clinical considerations are also discussed for the field of gender medicine.
Keywords
Introduction
Every cell is sexed, every person is gendered, and every organism is stressed. 1 Whereas sex refers to a multidimensional construct that includes genes, anatomy, gonads, and hormones that collectively define us as male or female, gender refers to an array of socioculturally constructed roles, orientations, and identities that further influence within-sex variations in stress, coping, and resilience. For example, gender roles represent the social norms and stereotypes we assimilate and endorse through the enactment of masculine traits (eg, instrumentality) and feminine traits (eg, expressivity) that are relatively constant across generations and nations. 2,3 Historical changes in social pressures and scientific perspectives of sex and gender might incur health-promoting and/or health-damaging consequences. 4 In addition to biological sex and sociocultural gender, sexual orientation also comprises unique sets of sexual attractions, behaviors, and identities that also correspond with health inequalities. 5
In this selective review article, I will review highlights from my transdisciplinary research program that nuances sex, gender, and sexual orientation in relation to stress biology. By applying a sex- and gender-based analysis that appreciates individual variation beyond sex binaries, I will demonstrate how one’s sex, sex hormones, gender roles, gender identity, and sexual orientation uniquely influence functioning of the stress hormone cortisol and multisystemic physiological dysregulation known as allostatic load (AL) linked to physical and mental health.
Biopsychosocial Stress
Stress is broadly defined as a real or interpreted threat to an individual’s physiological and psychological integrity that results in biological and behavioral responses. 6 Three broad traditions for measuring stress and coping exist in the stress-disease literature: environmental, psychological, and biological perspectives. 7 As a multidimensional construct, stress involves interactions among inputs (environmental stressors), processes (subjective psychological distress), and outputs (objective biological stress responses) that are often investigated separately. 8,9
Exemplifying stress as a multidimensional construct is the release of stress hormones as part of the stress response. Situations that are novel, unpredictable, threaten self-preservation, and/or diminish one’s sense of control will additively contribute to biological stress responses. 10,11 Firstly, real or interpreted threats trigger the sympathetic–adrenal–medullary (SAM) axis to release catecholamines (eg, adrenalin) within seconds from the adrenal medulla surrounding the kidneys. Secondly, the hypothalamic–pituitary–adrenal (HPA) axis is activated within minutes to produce glucocorticoids (GCs). The paraventricular nucleus of hypothalamus activates the HPA axis by stimulating corticotrophin-releasing factor (CRF). The CRF then travels through a portal system linking the hypothalamus to the pituitary, where it signals the secretion of adrenocorticotropic hormone (ACTH) from the anterior pituitary rich with blood capillaries. Systemic ACTH then travels to the adrenal cortex surrounding the kidneys where it precipitates cellular activities in the zona fasciculata region of the adrenal cortex to produce GCs such as cortisol in humans. 12 Cortisol is catabolic and essentially transforms lipids into sugar as cellular fuel.
The SAM and HPA axes mobilize energy necessary for adaptation, but at the cost of recalibrating many biological functions in turn over time. 13 Compensatory alterations during acute stress include, for example, decreased digestion and bodily growth/repair that are counterbalanced to accommodate increased neurological, cardiovascular, respiratory, and immune activities that are metabolically taxing. Over periods of chronic stress, the HPA axis will eventually inhibit even more systems such as the hypothalamic–pituitary–gonadal (HPG) axis secretion of gonadal hormones (eg, testosterone, estrogen) at the long-term detriment of reproductive functioning. By contrast, stress-induced HPG axis activations have been shown in animals, 14 suggesting possible co activation with the SAM axis and HPA axis under conditions of acute stress. Similarly, the immune system is upregulated during acute stress, but then downregulated during chronic stress. Beyond peripheral effects, GCs access the brain where they modulate a wide variety of neural functions.
The brain’s ultimate role during stress is to detect threat and promote adaptation. In addition to the pituitary and hypothalamic control of the HPA axis, there are 3 major brain structures involved in the regulation of stress responses: (1) the hippocampus linked to memory and cognition, in addition to being implicated in negative feedback regulation of the HPA axis; (2) the amygdala responsible for fear conditioning and emotional processing with outputs to SAM axis and neuroendocrine regulatory systems; and (3) the prefrontal cortex involved in cognition, coping strategies, and exerting top-down control over subcortical structures. 15 -20 In general, GC receptors are involved in negative feedback of the HPA axis under stress reactive conditions, while mineralocorticoid receptors are traditionally thought to be involved in basal HPA axis regulation. 21
Diurnal Cortisol
Stress hormones can be measured diurnally to capture naturalistic variation. Upon awakening, the cortisol awakening response (CAR) represents a normal surge in cortisol levels reaching maximal concentrations approximately 30 minutes upon awakening. 22 This surge is followed by gradually declining cortisol concentrations throughout the day as pulsatile secretion decreases in amplitude and frequency. 23 The nadir usually occurs around midnight, and cortisol levels start to rise again in the early morning hours. 24
Repeated measures spread throughout the day can help establish the circadian rhythm of the HPA axis. Five samples provide sufficient information to establish the slope and amplitude of the diurnal curve. 25 Since the CAR is marked by a single pulsatile secretion, repeated assessment over the course of the first hour after awakening is a more reliable marker of the circadian peak and the basal reactivity of the axis because it is sensitive to subtle HPA axis dysregulations. 26,27 Note, however, that morning cortisol and the CAR may represent an HPA axis mechanism that is distinct from diurnal variation thereafter. 23 For example, flattened diurnal cortisol patterns have been linked to increased evening levels that are related to distress and fatigue. 28
Diurnal HPA axis functioning can be used to identify disease vulnerabilities (Figure 1). A meta-analysis of 62 studies concluded that while the CAR is positively associated with workplace stress and general life stress, it is negatively associated with symptoms of burnout, fatigue, and exhaustion. 30 Hypocortisolism is a phenomenon that occurs in approximately 20% to 25% of patients suffering from stress-related diseases like chronic fatigue syndrome, fibromyalgia, post-traumatic stress disorder, burnout, 31,32 and atypical depression to name a few. 33 By contrast, increased HPA axis functioning during the afternoon and evening has likewise been associated with depressive symptoms. 34,35
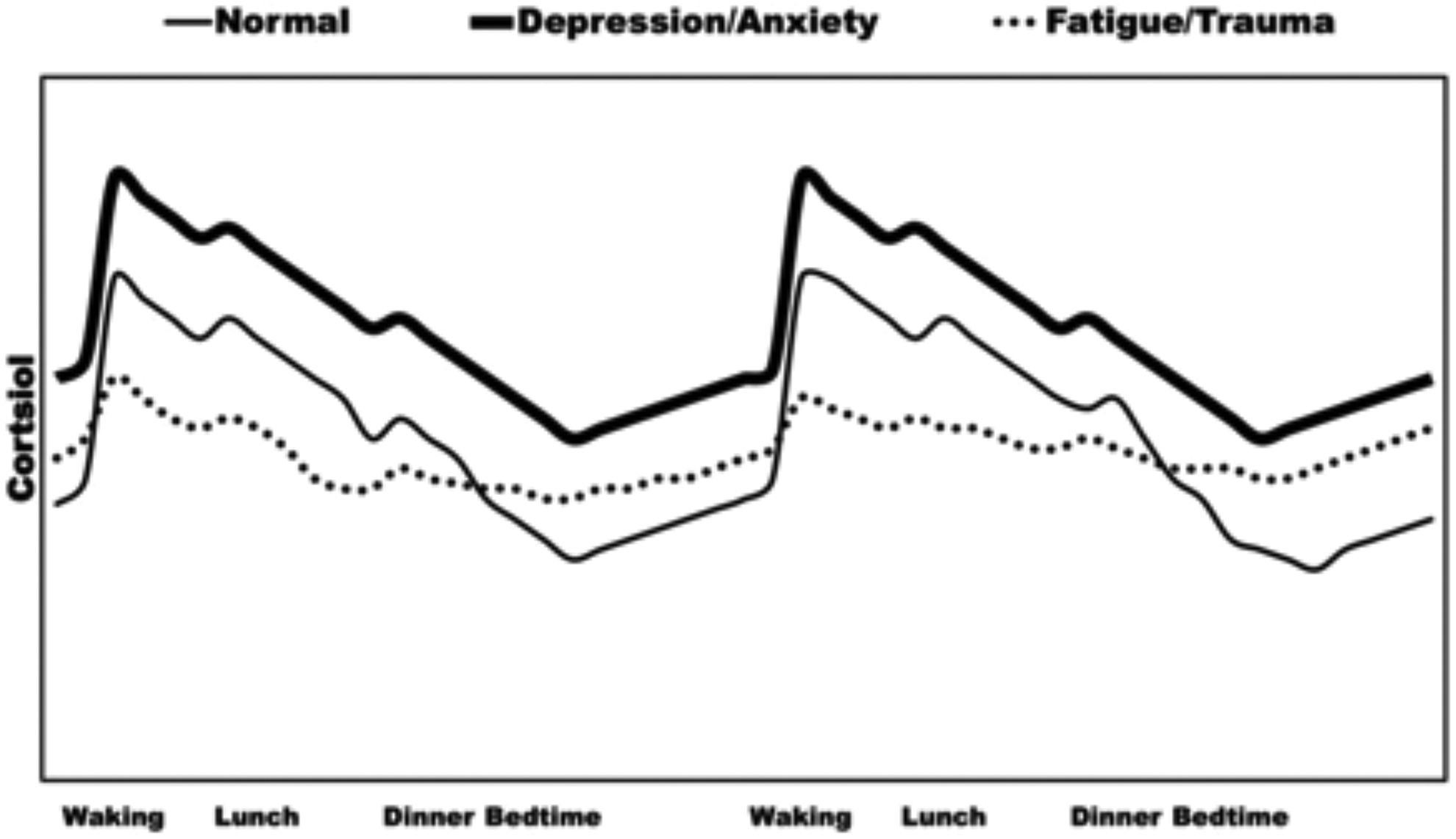
Hypothetical diurnal cortisol profiles in normal and pathophysiological conditions. 29
Identifying distinct cortisol profiles therefore requires multiple diurnal time points. To demonstrate this, one study found that low overall cortisol concentrations and small differences between morning and evening concentrations predicted depression 2 years later. 36 This underlines the need to assess afternoon and evening levels in addition to morning concentrations and the CAR. Finally, since individual pulses in response to stress can occur at any time during the day, a single sample is essentially useless for the assessment of baseline regulation of the HPA axis for either diurnal or reactive paradigm. 24
Reactive Cortisol
Numerous stress induction paradigms elicit social evaluative threat to trigger stress responses. 37 This occurs in laboratory situations that diminish one’s control as well as where the prospect of being negatively evaluated, rejected, and shamed is contextually manipulated. 37 -39 The motivation toward preserving one’s social-self vis-à-vis others’ evaluations involves complex interactions among cognitive appraisals, affective processes, and physiological responses. 40 -42 Those who experience social-evaluative threat more saliently fundamentally differ in their sensitivity to relative stressors and toward their need to preserve their self-esteem that can be deleterious to an individual’s health and well-being when it preoccupies them too much. 43
In concert with the autonomic nervous system, short-term adaptations during stress responses are dynamic biological processes that ensure survival. Stress responses are therefore adaptive in the short term, while long-term activations can result in physiological dysregulation. The reactivity hypothesis 44 proposes that exaggerated physiological and behavioral reactivity to stressors are risk factors for stress-related diseases like cardiovascular disease (CVD) among others. 45,46 In this perspective, pathophysiological reactivity is discernable by examining the magnitude of stress responses. Beyond focusing solely on increases from baseline to stressor exposure, researchers have been urged to also assess the duration and prolongation of stress responses after the stressor. 47,48 Indeed, the reactivity hypothesis has been criticized for often ignoring or dismissing physiological recovery, a period after exposure that is characterized by much individual variability. 49 -51
Noninvasive measures of salivary cortisol are advantageous in stress reactivity paradigms for similar reasons as those discussed for diurnal collection strategies. 52 Investigating stable and situational factors might strengthen our understanding of adaptive and maladaptive response patterns. 53,54 Figure 2 illustrates stress reactive cortisol and dynamic calculations used to capture responses to a popular laboratory-based psychosocial stressor. While stress induction paradigms attempt to understand disease mechanisms, it is important to highlight that responding to a stressor is adaptive and as a preserved mechanism has ensured our survival throughout evolution.
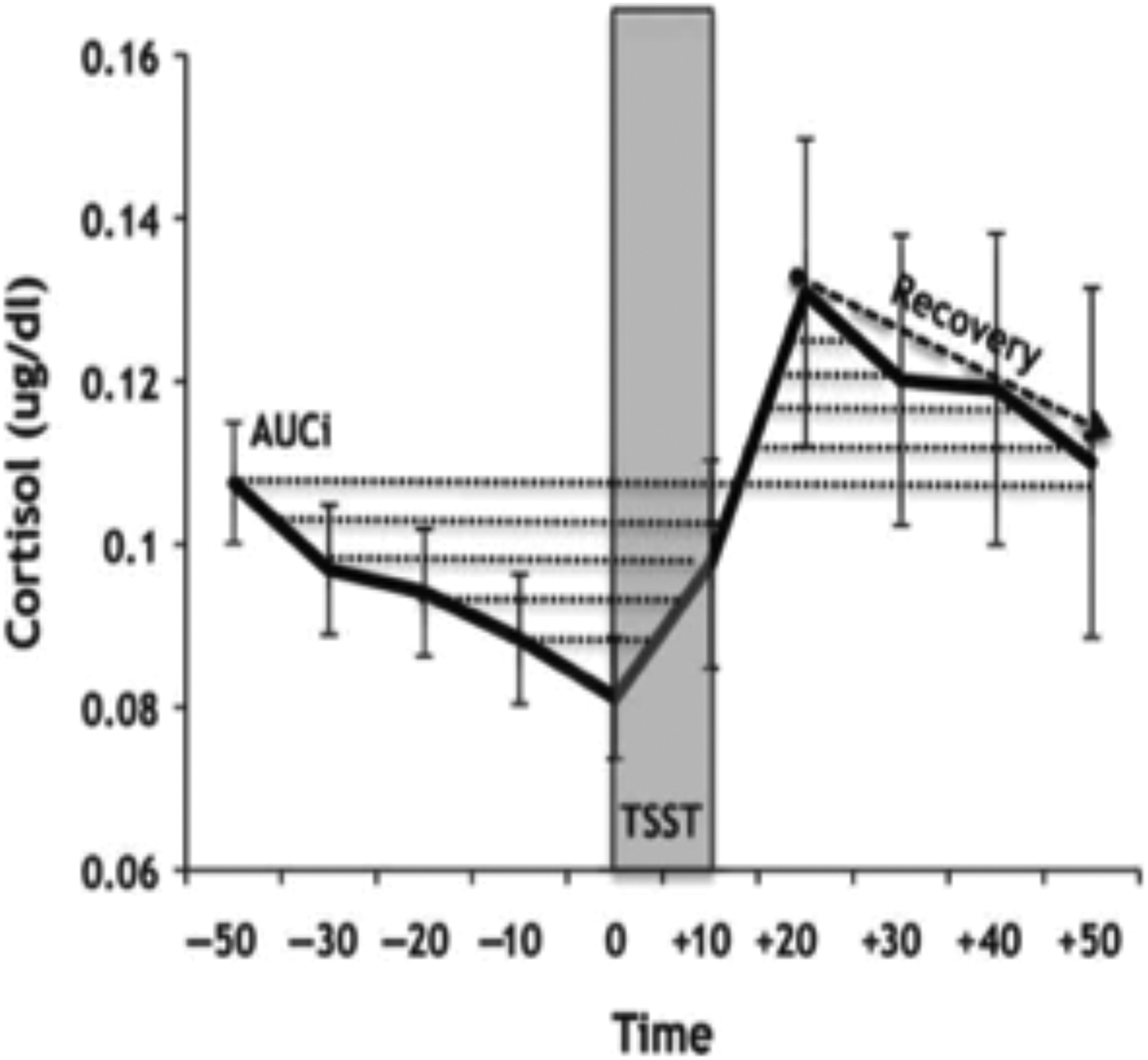
Schematic of stress reactive cortisol and dynamic calculations. 54 The Trier Social Stress Test (TSST) induces social-evaluative threat that modulate physiological responses. Mean salivary cortisol concentrations throughout a testing session (solid black line) can be easily transformed into stress reactivity and stress recovery measures. Here, the area under the curve with respect to increase or AUCi 55 is indicated as the area with dotted lines throughout the session). AUCi is a common formula used in psychoneuroendocrinology to summarize dynamics of stress reactive biomarkers. In addition, recovery percent slope (dashed line indicates recovery) represents linear decreases post-TSST.
Concept of Allostasis
Stress responses are adaptive and exemplify the concept of allostasis. Allostasis is defined as adaptive biological processes that preserve “stability through change”. 56 The neurobiologist Sterling and the epidemiologist Eyer coined the term allostasis to describe dynamic, multifaceted biological processes that maintain physiological stability by recalibrating homeostatic parameters and matching them appropriately to meet environmental demands. 56 Analogous to our understanding of resilient systems that have the capacity to dynamically adjust and stabilize to adapt to perturbations, 57 allostatic processes likewise alter metabolic functioning via compensatory as well as anticipatory mechanisms.
Homeostatic models generally define health as an internal milieu whereby all physiological parameters have relatively normal values. Disease, on the other hand, occurs when normal ranges fall into abnormal but pharmaceutically correctable ranges. In this view, Sterling and Eyer (1988) argued that biomedical models are in danger of inadvertently causing deleterious effects such as when correcting one parameter leads to another becoming dysregulated. These iatrogenic effects can arise when restoring one parameter to a normal range causes other parameters to become inappropriately high or low and cause altogether separate health issues. For example, hypertension medications can negatively alter interconnected biological systems (eg, potassium) when lowering blood pressure. In contrast, the allostatic model defines health as a state of responsiveness. A parameter with values outside the normal range is not necessarily considered inappropriate as each parameter is controlled by mutually reinforcing signals. 58
Allostasis fundamentally differs from homeostasis because of its emphasis on dynamic rather than static biological set points, considerations of the brain’s role in feedback regulation, 59 and its view of health as a whole-body adaptation to contexts. 60 According to allostatic theory, pathology itself is a form of adaptation where, for instance, an individual develops hypertension in order to conciliate environmental demands, even if these processes are ultimately pathophysiological. Health in allostatic theory is therefore defined as optimal predictive fluctuation, whereby shifts in the probability of demand should shift the response, and when the prediction reverses, so should the response. 61 It is when allostasis is chronically dysregulated that pathophysiological processes emerge and contribute to disease.
Allostatic Load Model
The multisystemic strain attributable to overactivated allostasis is referred to as AL. 62 Allostatic load is defined as the multisystemic “wear and tear” the brain and body experience when repeated allostatic responses exact their noxious toll under conditions of chronic stress. The AL is linked to brain changes that include synaptic and dendritic remodeling, suppressed neurogenesis, and structural atrophy/hypertrophy that further diminish the body’s ability to adapt. The AL model fuses multiple knowledge bases and disciplinary approaches, as coordination of allostasis depends on the brain’s evaluation of threat and execution of physiological responses. 63 -65 Indeed, the perception of threat and mobilization of allostatic mechanisms (eg, HPA axis reactivity) are happed by individual differences in constitutional (genetics, development, experience), behavioral (coping and health habits), and historical (trauma/abuse, major life events, stressful environments) factors that ultimately determine one’s vulnerability and/or resilience to stress. 66,67 Over time, chronic stress and AL can perpetuate unhealthy behaviors (eg, poor sleep, diet, smoking, drinking, physical inactivity) that eventually lead to allostatic overload, which in turn culminates in disordered, diseased, and deceased end points.
Allostatic Load Time Course
The AL model proposes that stress-related pathophysiology can be detected and objectively measured by assessing a battery of subclinically relevant biomarkers from multiple nonlinear and interconnected biological systems. 6 As illustrated in Figure 3, these contribute to pathophysiological allostatic states that reflect response patterns in which allostatic systems are dysregulated. There are 4 postulated physiological profiles that represent allostatic states: (1) repeatedly activated responses, (2) nonhabituating responses, (3) prolonged responses, (4) and inadequate responses. 67 Detecting allostatic states is possible by investigating repeated measures of specific biomarkers like cortisol dynamics. Because allostatic states prompt compensatory remediation among numerous biomarkers, delineating time courses of dysregulation in AL involves the challenge of determining which biomarkers falter first in the cascade. 65
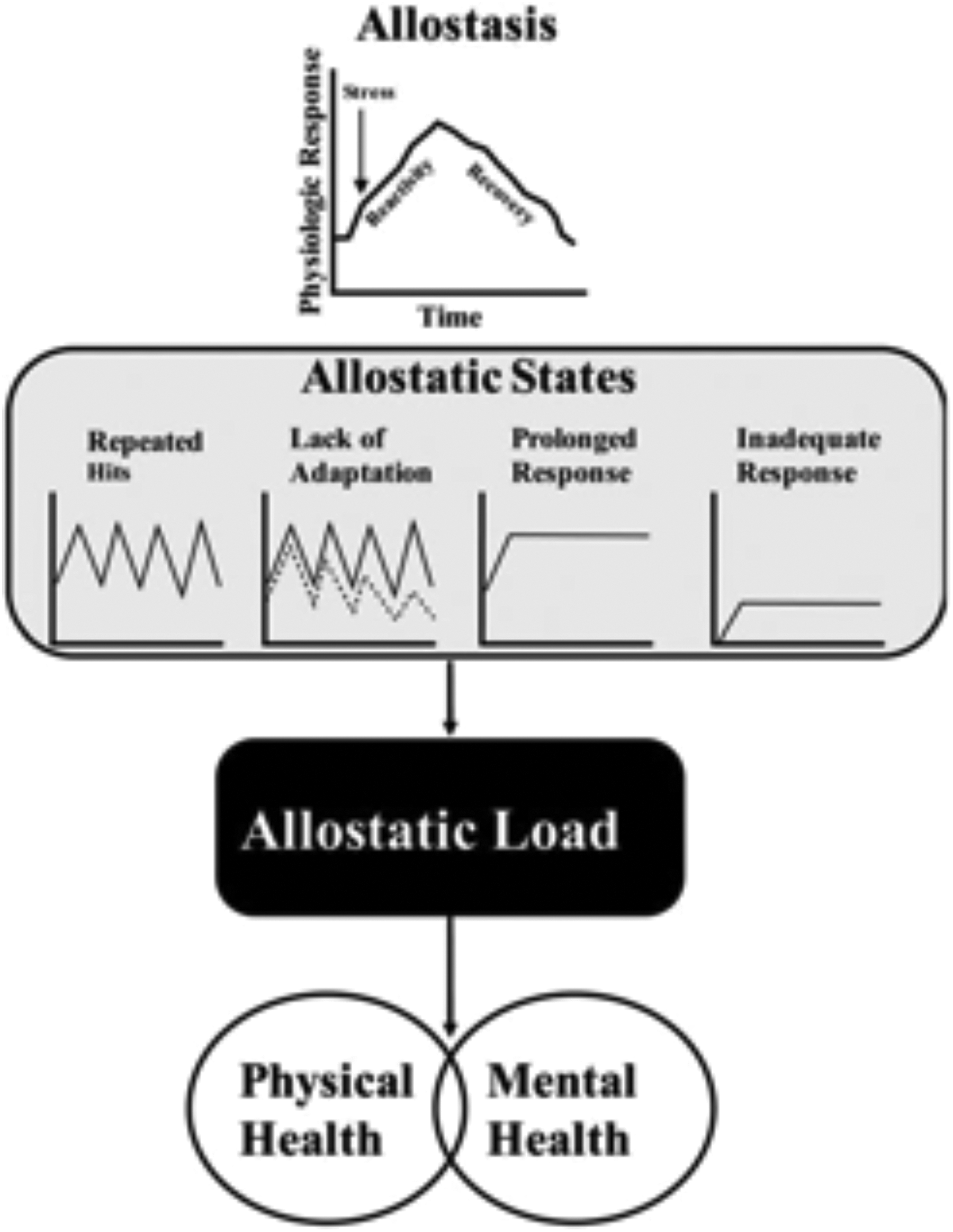
Allostasis, allostastic states, and allostatic load. 29
It has been proposed that under toxic stress, stress hormones like adrenaline and cortisol first become misbalanced and induce an interconnected “domino effect” on interdependent biological systems that collectively collapse as individual biomarkers “topple and trail” toward disease. 68 Numerous biomarkers contribute to further AL and eventually lead to disease in a theoretical sequence embodied in our “domino effect” analogy. We shall next explore this theoretical cascade in detail to highlight AL interconnectivity.
Synergistic effects of primary mediators exact primary effects on cellular activities (enzyme, receptor, ion channel, genomic) that compromise the physiological integrity of allostatic mechanisms. For example, mitochondrial dysfunction, oxidative stress, and inflammation represent cellular consequences of the hyperglycemic effect that GC induce. Together, this leads to further AL at a cell–molecular level that we refer to as primary outcomes. 69 Over time, subsidiary biological systems compensate for the over- and/or underproduction of primary mediators/effect/outcomes and in turn shift their own operating ranges to maintain abated chemical, tissue, and organ functions. This prodromal stage is referred to as the secondary outcomes, whereby metabolic, cardiovascular, and immune parameters reach subclinical levels. Finally, AL progression leads to tertiary outcomes that include a plethora of diseases.
Medical professionals routinely incorporate many related biomarkers in standard blood tests. Unfortunately, clinical attention is largely placed on biomarkers values that reach cutoffs. The AL model proposes that by measuring the multisystemic, reciprocal interactions among primary mediators, primary effects, primary outcomes, and secondary outcomes together, individuals at high risk of tertiary outcomes can be identified. 67 This has important implications not only in terms of research but potentially also in clinical practice as detection and prevention strategies. For example, patients with severe mental disorders are prone to developing comorbidities that could be proactively prevented by accounting for subtle multisystemic dysregulations. 61
Indexing AL
Seeman and colleagues have pioneered the development of AL algorithms that are predictive of numerous tertiary/clinical outcomes and are predicted by diverse psychosocial factors identified in the stress-disease literature. Validation using longitudinal data from the MacArthur Studies on Successful Aging cohort led to a count-based AL index representing the following 10 biomarkers 70 : 12-hour urinary cortisol, adrenalin, and noradrenalin output; serum dehydroepiandrosterone sulfate (DHEA-S), high-density lipoprotein (HDL) and HDL to total cholesterol ratio; plasma glycosylated hemoglobin; aggregate systolic and diastolic blood pressures; and waist to hip ratio. Participants’ values falling within high-risk quartiles (clinical and preclinical ranges based on percentiles) with respect to the sample’s biomarker distributions are dichotomized as “1” and those within normal ranges as “0.” Once tabulated, these are summed to yield an AL index ranging from a possible 0 to 10 which can then be used to predict health outcomes.
Beyond these traditional biomarkers, many others have been incorporated in numerous analyses worldwide using similar formulations and/or more sophisticated statistical approaches (eg, multivariate reduction techniques). Notwithstanding, the simplicity of the count-based formulation makes it amenable to both empirical and clinical contexts. The thematic advantages of applying an elevated risk zone system when scoring AL are 5-fold as they represent: (1) early warning signals since cutoffs are anchored at subclinical thresholds, (2) multifinality in that similar AL algorithms predict different tertiary outcomes, (3) flexibility since calculations are based on different biomarker combinations, (4) synergism capturing the cumulative interaction of numerous biomarkers, and finally (5) antecedents that predict individual variation in AL. 71 In sum, AL algorithms are objective reflections of biological functioning that are intricately interconnected with genetic, neurological, developmental, behavioral, cognitive, and social factors.
Causes and Consequences of AL
By applying an AL index in analyses of the MacArthur cohort, researchers validated that increased AL levels are related to cognitive and physical impairments at baseline and associated declines over 3 years, as well as an increased risk of having CVD. 70 At 7-year follow-up, those with the highest AL levels were at greatest risk of cognitive and physical decline as well as risk of CVD and even all-cause mortality. Note that while differences were found when using neuroendocrine (stress hormones and DHEA-S) versus metabolic syndrome biomarkers (glycolipids, blood pressure, adiposity), the multisystemic AL index was better than using only single biomarkers or systemic clusters to predict outcomes. 72 In a similar cross-sectional analysis, it was shown that primary mediators contribute independently of cardiovascular and metabolic risk factors in predicting cognitive and functional declines. 73 These results were cross-culturally complemented using a Taiwanese cohort aged 54 to 71. 74 -78
As represented in Figure 4, AL indices correlate either cross-sectionally or longitudinally with a plethora of antecedents (eg, socioeconomic disadvantage, poor social networks, workplace stress, lifestyle behaviors) and consequences (eg, all-cause mortality, CVD, psychiatric symptoms, cognitive decline, physical/mobility limitations, neurological atrophy). Beyond the powerful effect of aging on AL, unique configurations of these antecedents appear to be experienced differently by each sex throughout life span development. As the following will demonstrate, a binary view of sex is insufficient in studies assessing AL.
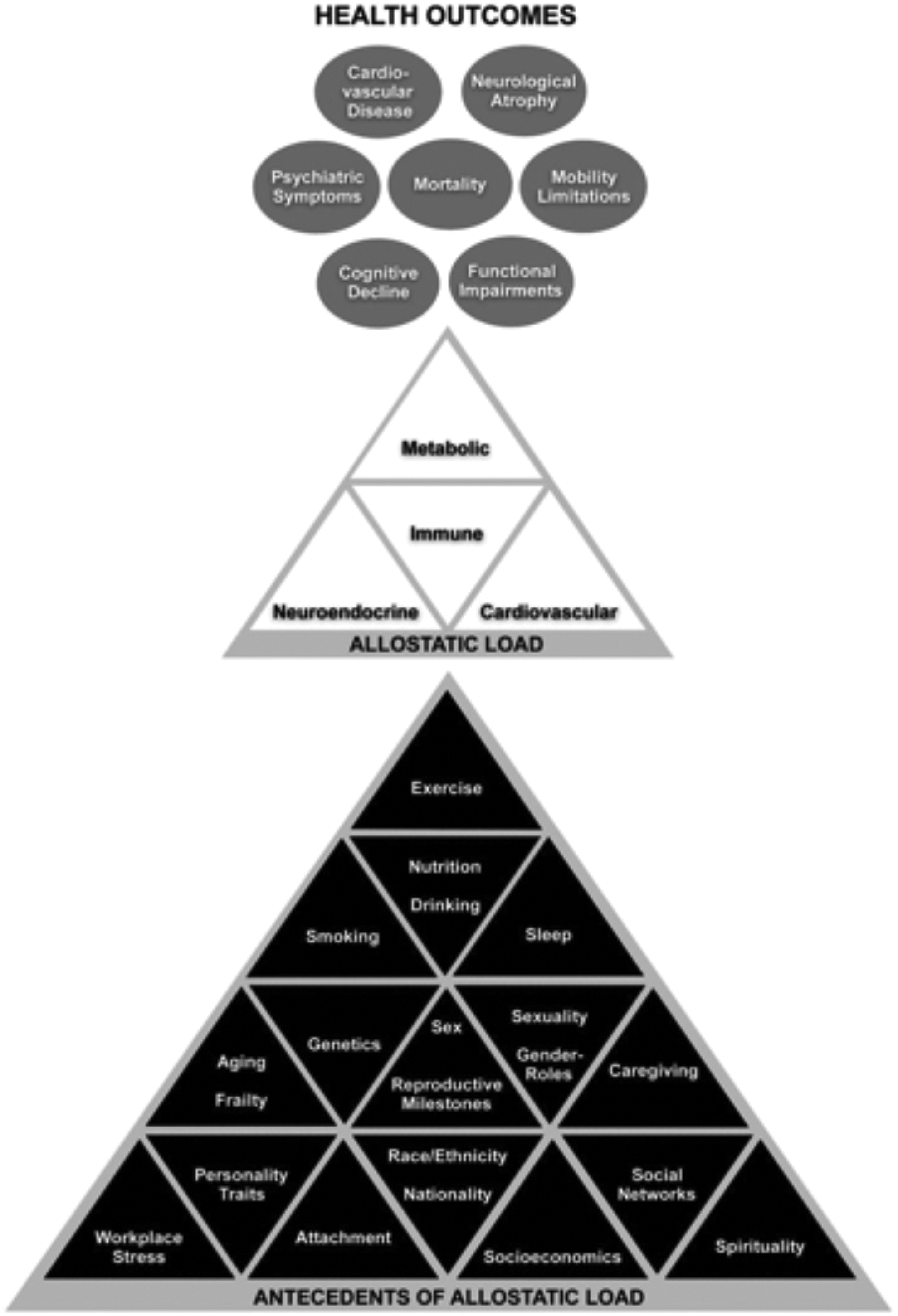
Transdisciplinary summary of the allostatic load literature. 29,68 In accordance with earlier review articles, 66,68,79 black triangles represent the identified antecedents of allostatic load, white triangles represent the biological systems that have traditionally been used to index allostatic load, and the gray circles represent health outcomes correlated or predicted by allostatic load.
Sex Differences in Human GC Functioning
Despite decades of research, it is not yet clear if diurnal HPA axis rhythmicity differs between the sexes and what other factors (eg, genetics, reproductive factors, age) might modulate functioning. 80 By contrast, the sexes show pronounced differences in their HPA axis responsivity to laboratory-based stressors. Kirschbaum and colleagues 81 first showed what appeared to be a sex-based difference in stress reactivity, with men showing 2-fold higher cortisol reactivity after public speaking and mental arithmetic in front of an audience compared to women.
In a follow-up study, women using oral contraceptives manifested further attenuation of cortisol reactivity. 82 Stress scientists reasoned that this was an estradiol-driven effect. Oral contraceptives contain high levels of estradiol that stimulate the production of cortisol-binding globulin, which in turn removes free cortisol from circulation, thus resulting in lower net levels of active cortisol. Consequently, studies compared cortisol reactivity between periods of the menstrual cycle phase. Results demonstrated that luteal women with low estrogen displayed reactivity patterns similar to men, while follicular women with high estrogen resembled women using oral contraceptives. 83 Acute stimulation of the HPA axis by either physical or psychosocial stressors suggests that estrogen regulation modulates differential reactivity between men and women. Yet, findings are mixed when physical stressors and pharmacological challenges are employed. 81,84,85 Another consideration is that HPA axis activities generally increase with aging, sometimes so much so that this effect can be 3 times stronger in postmenopausal women compared to older men. 86
Differential stress responses observed between the sexes and at specific hormonal states might further depend on the nature of the stressor. Estradiol levels rapidly and pervasively increase in response to acute stressors in animal models 87 ; however, interactions depend on the type, duration, and frequency of the stressor in a multiphasic manner. 88 One explanation is that each stressor has a different route by which it triggers the HPA axis. Psychosocial stressors involve the limbic system since it is based on the conscious interpretation of the situation. By contrast, physical stressors trigger the hypothalamus, while pharmacological challenges can modulate all levels of the HPA axis depending on the agent. 3
To complicate matters further, testosterone is converted into estradiol in the brain through aromatization that exerts HPA axis stimulation via mineralocorticoid and GC receptor neuromodulation that ultimately enhances CRF gene transcription. 89 In addition, enhanced testosterone production in the context of competition can amplify assertiveness and aggression when social status is threatened. 90,91 This is probable in the context of tasks that elicit social-evaluative threat; however, sex hormones are presumed based on self-reports and only rarely ever actually measured. Since intermitting within-sex differences influence crosstalk between the HPA axis and HPG axis in animals, 14 it is imperative that scientists take sex hormone variations into account when endeavoring to understand sex differences in human stress.
Sex differences in GC functioning are presumed to depend on sex hormone variations. And yet, surprisingly few psychoneuroendocrine studies have actually measured within-sex variations in testosterone, estradiol, and progesterone. In a study of healthy adults, we demonstrated that collecting saliva and adjusting for sex hormones alter diurnal and reactive cortisol profiles. 92 Specifically, controlling for testosterone, estradiol, and progesterone for both men and women altered “sex-specific” profiles. For reactive cortisol, men no longer showed higher cortisol production than women. For diurnal cortisol, we were able to unmask a sex difference whereby women showed higher cortisol production in the morning. It is especially interesting that testosterone covariation effects were consistently significant for both sexes. Moreover, women with higher testosterone showed higher cortisol concentrations in the morning. Future research will need to further determine the mechanisms whereby the HPG and HPA axes interact.
In summary, any robust study of sex differences in GC functioning should measure sex hormones to account for within-sex variations. Likewise, when endeavoring to understand gender-based modulation of GC and AL, accounting for the HPG axis is essential to rule out the role of biological sex.
Epigenetics Considerations
Biological sex at a genetic level (eg, XY and XX chromosomes) is shaped by numerous factors spanning preconception, gestation, childhood, adolescence, and beyond. Moreover, developmental processes are shaped by organizational milestones as well as (de)activation effects of sex hormones throughout the life cycle. In terms of the individual differences that transcend genomes, genetic sex and sex hormones are influenced by social and experiential circumstances that further alter the phenotypic expression. 93 This has influenced sex differences throughout evolution. For example, both genetic males and genetic females have sex hormone receptors throughout their brains that modulate structure and function. 94 These brain regions also respond differently to stressors with minimal overlap except to a subset of genes in critical stress regulatory regions like the hippocampus. 95 Interestingly, even identical twins show divergent epigenetic effects due to unique experiences throughout life. 96 In the context of stress responsivity, different genomes result in differences in how male and female genomes will generally respond to specific stressor as well as sex differences in these endophenotypes. 95,97 In considering epigenetics, it is important to emphasize that men and women (including gender diverse people) often process cognitive–affective information similarly, but they often use different neural circuits to do so. 98 Indeed, epigenetic regulation provides an insurance program that gonadal sex and brain sex are coordinated independent of genetic sex. 99
Sex Differences in AL
To date, it is unknown whether AL differs between men and women. With the exception of DHEA (an androgen antagonist of GCs), sex steroids have received no attention or inclusion in the AL literature. This lacuna is striking given the emphasis on understanding successful aging in comparison to senescence that is incomplete without considerations of sex hormone milieus. Based on an early review of the AL literature, 66 I identified that sex differences appear to depend on sample-based and age-related factors. For example, the MacArthur studies provided the first evidence for this observation. Specifically, for AL primary mediators, high cortisol levels were associated with memory impairments for women, 70 while elevated noradrenalin levels were related to cognitive declines in men. 100
Using AL algorithms, successive analyses of the MacArthur cohort revealed age-specific sex differences in the biological signatures linking AL to tertiary outcomes. An early analysis found that cardiovascular biomarkers were more often dysregulated for men, whereas neuroendocrine biomarkers were more dysregulated for women (Seeman et al, 2002). In a 12-year follow-up, high-risk pathways of AL biomarker clustering for men included adrenalin, noradrenalin, interleukin-6 (IL-6), C-reactive protein (CRP), and fibrinogen, while women included IL-6, CRP, glycosylated hemoglobin, and systolic blood pressure. 101 Interestingly, elevated systolic blood pressure occurred in 100% of high-risk pathways for women but only 17% for high-risk pathways among men principally driven by elevated fibrinogen, noradrenalin, and adrenalin levels otherwise absent for women.
These findings from the MacArthur cohort measured at different time points reveal that distinct sex differences in AL pathways emerge with advanced age. 66 This is consistent with numerous studies demonstrating links between chronic stress and advanced aging at molecular, social, and environmental levels. 102 -104 Identification of sex-specific thresholds in individual biomarkers and systemic clustering of AL at different ages should be a priority given increasing older populations worldwide. 71 Moreover, determining meaningful subclinical biomarker cutoffs between the sexes at different ages is essential and will be refined by accounting for sex hormones in AL studies. In so doing, sociocultural differences must additionally be considered.
Sociocultural Gender in Relation to GC Functioning and AL
Our understanding of sex differences can be complemented by incorporating gender perspectives when interpreting the GC and AL literature. 105 Throughout history, men and women have differed in terms of their socialization, identity, and assigned roles. 106 Gender thus refers to the implicit and explicit dissimilarities in an array of socioculturally constructed roles and relationships as well as sex-typical personality factors. As an example of gender, gender roles refer to the behavioral norms endorsed and enacted by men and women within different societies. Self-identification as “man” or “woman” refers to one’s gender identity that is distinct yet related to gender roles shaped by social interactions and behaviors that influence how one accepts or rejects their body and place within society. 107 A central tenant of my research program is that gender allows insights into within-sex variation that binary male/female sex cannot.
Gender Diversity in GC Functioning
Sex differences in biological constitution in concert with individual differences in gender-based factors influence patterns of physiological stress responsivity. 3 It has been argued that sociocultural factors may be more important than biological factors in explaining sex differences in stress responsivity. 108 For instance, it appears that women demonstrate increased cortisol levels when facing social rejection, 109 whereas men tend to be more reactive when confronted with achievement-based stressors in laboratory paradigms. 37 These sex differences stem from, for example, sex steroids and body size, but also from sociocultural factors that construct one’s gender and ultimately drive diverse patterns of threat processing and coping mechanisms.
An evolutionary hypothesis put forth by Taylor and colleagues in 2000 states that men and women cope differently under stressful situations. Accordingly, the primary stress response pattern for men are “fight-or-flight” responses, whereas women might be more prone to engage in “tend-and-befriend” responses such as nurturing and affiliation-based behaviors that protect against the demands of pregnancy, nursing, and child care. 110 The central tenet of Taylor’s proposal is that instead of utilizing physical retaliation when faced with threats, women focus on behaviors that protect vulnerable offspring and maintain social bonds.
Such coping can also cross between the sexes in a gendered manner. A study found that men tend to benefit from their partner’s social support by secreting less cortisol when facing an acute stressor, whereas women had an increased cortisol response in the presence of their partner but not in the presence of a same-sex stranger. 111 This exemplifies a gender-based effect hidden within an apparent sex difference. Compelling animal and human studies have further substantiated Taylor’s “tend-and-befriend” hypothesis. It is not, however, clear whether these responses are due to sex, sex hormones, gender, or both—or perhaps even influenced by how our own gendered assumptions factor into our research designs and the interpretation of findings. 112 To further understand “sex” differences in GC functioning and AL, researchers must also endeavor to demarcate gender-based modulation further while accounting for sex hormones.
Gender Diversity in AL
It follows that individual differences in unique configurations of AL antecedents might differ according to gender and related behaviors that might ultimately contribute to AL health consequences. In accordance, the “tend-and-befriend” hypothesis sheds some light on sex and age differences found throughout the AL literature. For example, the salubrious effects of social networks and relational factors differ internationally according to sociocultural processes. Similar to stress reactivity, AL levels were shown to be lower only for Taiwanese elderly men with a spouse, while both sexes benefit from ties with close friends and neighbors in another analysis. 113 Interestingly, the perceived quality of these social relationships is not consistently linked to AL, suggesting that cross-cultural differences with Western societies may be influenced by sociocultural factors (eg, virtues of collectivism/interdependence, social obligation, and the absence of “self”). Such socialization is inherently influenced by gender-based constructs related to societal ideals and the enforcement of patriarchal and/or matriarchal virtues. 114
Collectively, one’s sex and gender roles might divergently influence biopsychosocial stress. In particular, extremely traditional or hegemonic masculine gender roles (eg, hostile, dictatorial) are strongly linked to coronary-prone behaviors (eg, inhibited emotional expression, lack of empathy, homophobia) that overlap with hostile personality traits that mediate the association between low socioeconomic status and increased AL levels. 115,116 The sociocultural amalgamation of such gender norms can lead to societies that oppress all forms of human variation that deviate from the enforced norms. An example that is relatively new development in Occidental societies is the near parity of women participating in the workforce. As women increasingly occupy nontraditional professions around the world, 117 changing gender roles might influence health and disease trajectories. 4
To date, little is known of the impact of gender on AL and how gender can interact with sex to influence AL. Various gender-specific and health-related behaviors confer a differential vulnerability and/or resilience to various diseases within and between societies that may be related to gender. 118 Many antecedents of AL such as social support, personality traits, occupational characteristics, and lifestyle behaviors function as part of a complex reciprocally interacting system. More specifically, retrospective reports of positive parental bonding, harmonious later life relationships, and the presence of a spouse are associated with lower AL in elderly men who benefit most from emotional support. 119,120 By contrast, social ties with close friends is protective for both elderly women and men. 119 Interestingly, decreased AL has been reported in women but not men who regularly attend religious services 121 and who have a greater sense of coherence and meaningfulness in life. 122 Taken together, these associations are clearly related more to sociocultural gender than to biological sex.
Sex × Gender Roles and AL Among Workers
Gender-based factors might influence changing demographics of wealth and health. The benefits that some women have gained in terms of socioeconomic independence as workers over the last decades 123 -125 have been countered by arguments stipulating that gender role conflicts can negatively affect health and well-being. For example, “masculine” working women like managers 126 may have had to struggle to achieve their status independently of their feminine identity. In the same vein, the greater implication of men into the organization of family life may not be well received by peers as this represents an appropriation of femininity. 127
In Canada and other industrial nations, differential exposures to workplace stressors are partially explained by an unequal sex distribution among occupations that often render women of diverse ages and social strata 128 more vulnerable to psychiatric disorders. 129 -131 These sex differences may be due, in part, to divergent gender roles and ranks held within occupational hierarchies. 132 In general, men are driven more by achievement motivations, while women are driven more by interpersonal motivations. 133 This parallels our understanding of masculine “agency” and feminine “affiliation” in the gender roles literature that have yet to be investigated in relation to AL.
In a study of 204 psychiatric hospital workers, we assessed the synergistic influences of sex and gender roles on AL and mental health while accounting for essential covariates like HPG axis functioning. We tested a popular psychological theory in gender studies developed by Sandra, Lipsitz, and Bem (1944-2014) in relation to AL. Our analysis assessed whether androgyny, that is, high self-endorsement of both masculinity and femininity, would be psychobiologically adaptive and related to lower AL. In contrast to androgyny, those scoring low on both masculinity and femininity are called undifferentiated and believed to be the most maladapted. Previous research had shown that gender roles are predictive of health outcomes, but not necessarily independently from one’s biological sex. 134 -136 Because women consistently report poorer health, 137 -139 visit physicians more often while taking more sick days off from work, 140 and are greater consumers of medications 139 compared to men, it could be presumed that women are more vulnerable to AL.
Our findings revealed that sex and gender roles interact in association with AL. 141 Specifically, androgynous men with both high masculinity and femininity showed low AL compared to undifferentiated men. In addition, undifferentiated men showed higher AL than undifferentiated women. Independent of sex, androgynous individuals showed better mental health as evidenced by higher self-esteem, well-being, and less depressive symptoms than undifferentiated men and women. 141 Future research is needed to identify who undifferentiated individuals are and how they develop in comparison to other gender role types. On a positive note, further understanding the salubrious nature of psychological androgyny—especially among men—could allow insights into protective pathways toward resilience. Finally, women as a group showed lower AL than men; however, AL did not differ among women by gender roles.
Why would androgyny not be protective against AL among women? The appropriation of multiple gender roles among working women has not been matched by appropriate social equity. In general, women still retain more “feminine” roles and responsibilities than the average man in both work and home life. 108,142,143 This is likely subjectively distressing for modern women, but perhaps this does not correspond to objective indicators like AL. Beyond perhaps representing sex/gender differences in subjective versus objective indices, it is possible that changing gender roles might also influence men in ways that have yet to be studied fully using sex- and gender-based approaches. For example, a Swedish study found that paternity leave—which is considered as departure from traditional masculinity—is associated with decreased mortality among fathers. 144 Also, how we deal with major stressful events in our lives is influenced by sexed/gendered coping strategies that interact with numerous additional factors that were not assessed.
Taken together, these findings force us to conceptually move beyond bipolar views of not only men and women but also masculinity and femininity. In developing her theory that androgynous individuals are more adaptively “flexible” in their gender expression, Bem 145 proposed that masculine and feminine gender roles are independent entities and therefore not binary. Yet, when interpreting gender roles along continuums, it is easy to resort to simplifications that can make masculine and feminine gender roles seem like oppositional constructs when they are in fact context-specific according to Bem.
Hammarström 146 has noted that gender role theories have inherently assumed a complementary coexistence, for instance, between “active” men versus “passive” women. We must be cautious with such dichotomizations of stereotyped gender roles as we refine our conceptualization when researching stress phenomena. Indeed, Bem’s theory concerning psychological androgyny stands in stark contrast to oversimplified views of masculine versus feminine behaviors. 147 Indeed, we did not find marked differences when contrasting masculine and feminine individuals, but instead when comparing androgynous and undifferentiated individuals. This emphasis on the relative combinations of masculinity and femininity, and not their singular effect alone, provides compelling psychobiological support for Bem’s hypothesis.
Sexual Orientation and Gender Identity in Relation to Stress Physiology and AL
Lesbian, gay, bisexual, and transgender (LGBT) people are vulnerable to stress and disease because of stigma. In Canada, approximately 2% to 3% of the population identify as LGB. 23 While national statistics do not exist yet in Canada, approximately 0.6% of Americans identify as transgender. 24 Among surveys of LGB individuals, discrimination due to sexual orientation is reported by 29% to 78% of Canadians. 25 For transgender Ontarians, 67% report depression, 77% have contemplated suicide, and 43% have attempted suicide at least once. 26,27 As a whole, adolescent and adult LGBT people are anywhere from 1.5 to 4 times more likely to self-report depression, anxiety, suicidal ideation/attempts, substance abuse, eating disorders, risky sexual behaviors, homelessness, and victimization than heterosexuals. 21,28,30,31 Given these staggering statistics, the Institute of Medicine has mandated the health of LGBT people a research priority. 21
Sexual and Gender Minority Stress
Sexual minority stress models 21 propose that the stress of living in environments that stigmatize minority sexual orientations contributes to later health disparities according to 2 stress processes. First, proximal minority stress processes refer to individual-level experiences like internalized homophobia and concealment of one’s minority identity. 15 Second, distal minority stress processes refer to objective stressors like discrimination and violence experienced at the interpersonal level. At the macro-level, structural stigma represents the social conditions, cultural norms, and institutional policies that constrain the opportunities, resources, and well-being of the stigmatized. Sexual minority stress also applies to the understudied experiences of gender minorities like transgender and gender fluid people who face unique challenges in societies that often denigrate gender nonconformity. 31 Indeed, transgender people are at ultra-high risk of health problems, even more than LGB subgroups.
The accumulated findings over the past decades suggest that sexual and gender minority stress plays a role in the psychological, physical, and behavioral health of LGBT individuals. However, this rich body of psychological research is matched by a paucity of biopsychosocial research. Exceptionally, studies that use biological data have examined the relationship between psychological distress and physical health generally among HIV-positive gay and bisexual men. Beyond the HIV/AIDS pandemic, however, the health of LGB populations has yet to be studied extensively using GCs or AL. This may, in part, be because exploring the biological correlates of sexual orientation has been controversial. 148
Sexual Orientation and Sex Hormones
The HPG axis was once the topic of great research interest in the context of understanding the biological correlates of sexual orientation. While controversial, it was argued that anomalies in the organizational and activational effects of sex hormones could determine nonheterosexual proclivities. Based on animal sexual behavior, several eminent scientists proposed that prenatal androgen deficits could cause male homosexuality, while prenatal androgen excesses could cause female homosexuality. 149 During a critical period between 2.5 to 5.5 months’ gestation, it was argued that gestational triggers like maternal stress could expose the human fetus to hormonal imbalances that would sculpt hypothalamic and limbic brain regions. 150 A central tenant of this neurohormonal hypothesis is that differentially shaped brains of LGBT individuals correlate with reversals in sex hormones and sexual behaviors. 151
Throughout the 1970s, many studies published in prestigious journals focused on these hypothesized sex reversals in HPG axis functioning among sexual and gender minorities. In several studies using analytes extracted from either urine, serum, or plasma that were contrasted to secretory profiles of heterosexual controls, many mixed findings were reported. This inconsistent literature was critically reviewed by Meyer-Bahlburg. 152,153 He concluded that findings were overall inconsistent among sexual minority men, 152 although about one-third of sexual minority women manifested elevated androgen levels while otherwise showing no endocrine abnormalities. 153 Methodological differences between studies rendered comparisons and final conclusions impossible. Taken together, this body of research did not support a neurohormonal hypothesis of sexual orientation 154 despite more recent claims to the contrary. 155 In parallel, research among intersex that are born with ambiguous genitalia often show nonheterosexual proclivities in adulthood due in part of differential sex hormone sensitivities. 156,157 A critical factor ignored in these studies was whether sexual and gender minority stress could explain inconsistent findings.
Meyer-Bahlburg acknowledged that some early researchers of sex hormones and sexual orientation were rather cautious in their conclusions. 152 In particular, the inconsistencies in observed HPG axis patterns might not be the primary cause of sexual minority orientation, but rather a secondary consequence related to unmeasured factors such as psychosocial stress. 153,158 To exemplify this, critics considered a longitudinal study among military men undergoing stressful training that revealed plasma testosterone levels were lowest during the earlier novice phase compared to the later senior phase. 159 Unfortunately, this emerging knowledge from stress science was not applied to understanding the mixed HPG axis literature on sexual orientation and gender identity.
Sexual Orientation and GC Functioning
Despite speculation that psychosocial stress might be involved in study inconsistencies, 160 these studies of HPG axis functioning never took sexual or gender minority stress into account. Work by our group aimed to address this lacuna in a comprehensive psychoneuroendocrine study.
We provided the first evidence that sexual orientation modulates GC reactivity in a gender-based fashion. 161 That is, lesbian/bisexual women hyperreacted similar to heterosexual men, while gay/bisexual men hyporeacted similar to heterosexual women. In terms of cardiovascular parameters, we found that gay/bisexual men showed higher heart rate throughout in comparison to heterosexual men. 162 Studying sexual minorities represents a means to further understand the role of gender-based factors in stress reactivity paradigms that are otherwise believed to differ solely according to biological sex.
In addition to highlighting how one’s sexual orientation modulates HPA axis reactivity, we also identified a sex hormone covariation: cortisol systemic output was positively associated with estrogen and progesterone concentrations among lesbian/bisexual women but not among heterosexual women. This sex hormone covariation effect was controlled for in our main analyses. This being said, our curiosity led us to assess sex hormone variations further. Serendipitously, 2 additional saliva samples were collected 15 minutes before and 15 minutes after the psychosocial stressor separate from the 10 samples destined for cortisol. In analyses that adjusted for key covariates, we found that lesbian/bisexual women had higher overall testosterone and progesterone concentrations as a group than heterosexual women, while men showed no differences in HPG axis functioning by sexual orientation. 160
That lesbian/bisexual women would manifest higher overall levels of testosterone is in accord with some existing reports, 163,164 but also in disaccord with studies showing no differences. 165,166 As discussed earlier, a longstanding proposal of this mixed literature has been that elevated androgens among sexual minority women might be explained by environmental stress. 153 Consistently, lesbian/bisexual women had higher stress reactive cortisol 40 minutes after the stressor than heterosexual women. 161 Moreover, lesbian/bisexual women had higher progesterone concentrations than heterosexual women, something that has not been previously shown and is not easily explained. There is in fact very little literature on progesterone concentrations, which is somewhat surprising since it is a precursor of GCs and all sex hormones. Given that GCs, testosterone, and progesterone share common molecular precursors like pregnanolone, it is highly probable that coactivation of HPA axis and HPG axis is at play here.
To further triangulate methods and facilitate interpretation of these complex associations, we ran an analysis assessing intercorrelations among the HPG axis and HPA axis dynamics. 160 We found that cortisol dynamics (area under the curve formulas) were positively associated with testosterone and progesterone fluctuations while accounting for age, sex, gender roles, and sexual orientation. This supplemental result underlines the need to account for HPG axis functioning in studies of GC dynamics. By the same logic and as previously demonstrated, adjusting for sex hormones is necessary to represent biological sex modulation of GCs and is important in determining gender effects in stress reactivity.
Sexual Orientation and AL
Our group has successfully applied the AL model to the study of LGB health among young to middle-aged adults. 167,168 Contrary to sexual minority stress theory, however, we were surprised to find that LGB people did not show higher AL indexing 21 biomarkers than heterosexuals. 167 In fact, gay/bisexual men had lower AL levels 167 and decreased cortisol reactivity in response to a psychosocial stressor 161 compared to heterosexual men. By contrast, lesbian/bisexual women showed tendencies toward higher cortisol reactivity compared to heterosexual women. 161 In addition, no differences in AL were found between women by sexual orientation. 167 Interestingly, LGB people who had fully “come out” to their family and friends showed lower levels of morning cortisol than those who had not completely disclosed their sexual identity. 167
A subanalysis of retrospective coping strategies during sexual identity formation revealed that LGB young adults who enacted less avoidance regarding their sexual orientation showed lower AL. In addition, those who sought social support reported less perceived distress. 168 Central to integrating one’s self-identity as a sexual minority is stigma management that occurs by developing strategies to help cope with the stressful realities of a marginalized identity. Taken together, individual (eg, coping strategies) and interpersonal (eg, social support) factors may promote resilience. Resilience is here defined as a dynamic process that promotes positive adaptation among individuals exposed to severe adversity, trauma, and stress. 169 -172
To confirm whether our AL findings would generalize in the United States, collaborators and I conducted an analysis of nearly 14 thousand people from the 2001-2010 National Health and Nutrition Examination Survey. 173 Consistent with our original findings from Canada, gay men showed lower AL compared to heterosexual men. By contrast, bisexual men showed the highest AL. Bisexual men are a minority within a minority that may not garner the same social supports as gay men. This replication expands our original AL findings that did not distinguish between gay and bisexual men due to restricted power. 167 Interestingly, again no differences in AL were detected for women by sexual orientation that we speculate may be due to gender inequalities in sociocultural and economic resources. 173
Existing AL studies among LGBT people highlight important subgroup differences that can only be demarcated in analyses that disaggregate by sex. It is clear that LGBT subgroups are heterogeneous and cannot be treated as a uniform group. Indeed, we speculate that each LGBT subgroup experiences unique combinations of risk and protective factors that have not been explored in AL studies. For example, subgroup behaviors toward diet, exercise, and body image may render gay men more physically fit, which could explain their lower AL compared to heterosexual men 173 ; however, this remains to be empirically tested or applied to an aging population. Among women, AL does not differ by sexual orientation or even by gender roles in separate research by our group 141 .
Critically, stigma has not yet been measured but is assumed to implicitly differ at the group level among LGBT people when compared to heterosexuals. This is a crucial limitation that we will need to be addressed in future research that accounts for more psychosocial factors. Lastly and perhaps most importantly, transgender individuals have yet to be included in published studies of AL. This omission is problematic given the exponentially higher rates of physical and mental health disparities among transgender people compared even to LGB subgroups. 174 Pioneering fieldwork by Zachary DuBois conducted among transgender men has shown that transgender-specific stressors (eg, transitioning stress, “coming out,” public restrooms) are associated with increased diurnal HPA axis production 175 as well as deleterious indices of cardiovascular and immune functioning. 176 More research using stress biomarkers are needed to understand the health needs of transgender and gender diverse people.
Clinical Considerations
The advancement of gender-based medicine recognizes nuances among men and women, boys and girls, and gender diverse people. Indeed, one glove does not fit all in medicine. Gender-specific medicine is concerned with informing scientist-practitioners on pathophysiology, clinical signs, prevention, and treatment of disease that are equally represented among men and women. 177 Refining our understanding of GC functioning as an integral component within the AL model shows promise as a tool tailored for gender medicine. The AL algorithms include subclinical thresholds for numerous biomarkers already routinely collected in medical practice. By identifying meaningful sex-specific preclinical values, establishing clinical norms, addressing gender-based variation, and combining disciplinary approaches, it will be possible to improve diagnostics, treatment, and prevention strategies. The transition from empirical to clinical application of stress biomarkers will need to be tailored with sex and gender considerations in mind. In particular, the development of clinical AL algorithms for the purposes of earlier detection of stress-related diseases or comorbidities 61 must consider sex differences in individual biomarker cutoffs. This could also be used in the context of interventions aimed at lowering stress pathophysiology.
To date, few treatment options aimed at correcting GC dysregulation or elevated AL have been investigated. Critically, interventions attempting to reverse deleterious pathways may be complicated by the fact that sex is so often totally disregarded in clinical research. 178 For example, a critique of 38 Cochrane systemic reviews on CVD identified that sex and gender were rarely considered despite mounting evidence that sex influences heart health. 179 Given that Cochrane guidelines are considered to employ rigorous standards for inclusion, this is particularly troublesome. More broadly in preclinical and clinical research (eg, randomized controlled trials), the disaggregation of findings on the basis of sex is fundamental. 144 Disregard for sex and gender perspective is especially problematic for the prevalence of numerous stress-related disease.
To complicate matters further, “sex-specific” diseases (eg, CVD, mood disorders, somatic complaints, muscular-skeletal disorders) can change throughout history. 180 For instance, CVD, once considered a predominantly male problem, now claims at least 41% of all deaths of Canadian women in comparison to 37% for men. 181 Rates of CVD have been increasing over the last 20 years for women, while those of men are declining or remaining stable. 182 The incidences of CVD are also increasing worldwide among women. 117,183 Could changes in heart health be explained by changing gender roles? Women have increased their participation in the workforce by 126% over the last 30 years 184 and represent 42% of the entire global labor force. 185,186 Yet, women around the world suffer more from competitive economic pressures, job insecurity, limited possibilities for training and promotion, and inadequate social benefits such as insurance and sick leave. 183
Measuring gender is complex as it represents a construct that includes, for example, social status, equality and access to resources, and behaviors related to sex that are not universal. 144 In an effort to pinpoint the influence of sex and gender factors in acute coronary syndrome, Pelletier et al innovated a “gender index” comprising various psychosocial variables such as primary earner status, personal income, number of hours per week doing housework, level of stress at home, and both Bem masculinity and femininity scores. They found that feminine gender is associated with the heaviest burden of CVD risk factors (eg, hypertension, depressive symptoms, smoking) independent of sex. Clearly, sex and gender overlap, but they represent distinct phenomena that must be parceled apart to better predict disease.
Conclusions
Sex, gender, and sexual orientation represent unique constructs that can help us better understand stress-disease processes. These constructs are similar in that individuals are, in general, gender socialized in accordance with their biological sex. By contrast, sexual orientation represents a sociocultural classification that overlaps with notions of gender but that nevertheless corresponds to unique experiences that are in addition to those attributable to one’s birth assigned sex, gender identity, and gender roles. The combination of these factors all fall along several spectrums that are likely further modulated by sex hormones. For example, lesbians who self-rate themselves as more masculine or “butch” show higher testosterone concentrations than their “femme” partners. 187 Ultimately, various sex- and gender-based factors interact to form a constellation of rich interindividual differences of great importance to health research.
Applying a sex- and gender-based analysis entails researching how variations in biological constitution (sex) and social experiences (gender) relate to health problems. 188 Relatedly, intersectionality is an inductive research framework that appreciates the reality that individuals are members of multiple social groups with diverse identities that will collectively determine their experiences, chances, opportunities, and ultimately their health. 189 For example, differences among specific LGB groups intersect according to age cohorts, race/ethnicity, socioeconomic status, culture, and geographic location. As an example, younger generations of LGBT individuals are 16 times more likely to have disclosed than older generations. 190 A sex- and gender-based analysis recognizes this plurality of individuality and can be viewed as an iterative process that evolves by refining research methodologies to ultimately promote equity in science, policy, and practice. 188 As a point of origin, the sexes share many similarities that should be remembered in our quest to delineate dissimilarities in stress biology and “sex-specific” disease susceptibilities. This approach provides a powerful framework to help solve health problems that cannot be easily explained by focusing solely on binary sex differences.
In summary, I have shown how sex, sex hormones, gender roles, gender identity, and sexual orientation are each uniquely related to GC functioning, AL, and/or mental health. While much more work is needed, my research program contributes to our understanding of within-sex variations in biopsychosocial stress that cannot be detected by solely focusing on sex as a binary variable. Indeed, gender and possibly even sex are continuous variables. Assessing interindividual differences among various sex × gender factors is needed to understand meaningful differences in stress biology that differs within sex.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
