Abstract
Background:
While hereditary renal cell carcinoma (RCC) syndromes account for up to 16% of RCC cases and have screening guidelines; no screening recommendations exist for individuals with family history of RCC and negative genetic testing (familial RCC risk).
Objective:
This study aimed to assess current practices regarding screening recommendations for patients with familial RCC risk, evaluate the perceived need for formalized screening guidelines, and identify optimal clinician involvement for developing such guidelines.
Methods:
We surveyed healthcare providers who provide medical care for RCC patients, assessing current familial RCC screening practices, the need for and process of developing empiric risk guidelines, and case-based scenarios to understand current practice. This IRB-approved survey was distributed using listservs, professional connections, and snowball methods. Descriptive statistics and qualitative coding were used to analyze the data.
Results:
Seventy healthcare providers participated, primarily genetic counselors (n = 46; 66%) and medical oncologists (n = 11; 16%). Respondents indicated that: 1) screening guidelines are needed (n = 64; 91%) for familial RCC risk; 2) RCC patients inquired about the risk of RCC to family members after receiving negative genetic test results (n = 59; 84%, sometimes, often, or always); and 3) asked about screening recommendations for family members with similar frequency (n = 56, 80%). Respondents varied on the optimal screening mechanism and indications for screening unaffected relatives of RCC patients.
Conclusions:
Our findings show providers need screening guidelines to address patient inquiries, guide clinical management, and improve insurance coverage. Respondents agreed that there was a need for a multi-disciplinary team to develop standardized recommendations.
Introduction
Renal cell carcinoma (RCC) represents approximately 4% of all newly diagnosed cancers in the United States. 1 Numerous studies have shown an increased risk for RCC in individuals with a family history of RCC, particularly affected siblings.2–4 While screening guidelines exist for monogenic hereditary RCC risk (e.g., von Hippel Lindau), guidelines have not been created for RCC screening for family members when germline testing is negative (familial RCC risk).
Familial RCC risk is defined as two or more individuals with RCC in a family without an identified hereditary cancer syndrome. 5 Up to 3.8% of individuals with RCC have one or more first-degree relatives (FDR) also diagnosed with the disease.6,7 Individuals with a FDR diagnosed with RCC have an increased risk up to 4.3-fold of developing RCC.3,4,6
Current National Comprehensive Cancer Network (NCCN)/American College of Medical Genetics (ACMG) guidelines recommend germline genetic testing for individuals with RCC based on certain personal or family history criteria, and screening for hereditary RCC including enhanced renal imaging such as MRI.8,9 Fewer than 20% of individuals who meet RCC genetic testing criteria are found to carry a pathogenic variant.10,11 For individuals where a single-gene cause for their disease is not identified, there are no clear screening guidelines for family members who may be at increased risk based on family history alone. In kidney cancer specifically, early detection can result in earlier stage disease, which can be treated with partial nephrectomy which typically has better outcomes and reduced mortality than late-stage disease. 12
Although data are limited on the effectiveness of kidney cancer screening, the impact of screening and early detection on reducing mortality in other cancers is substantial. 13 Yet, no guidelines for screening familial RCC cases exist. This study aimed to understand providers’ current practices regarding screening recommendations for familial RCC risk and their familiarity with hereditary RCC syndromes. It also sought to conduct a needs assessment for familial RCC screening recommendations and risk estimates, and to identify who should be involved in developing these screening guidelines.
Methods
A cross-sectional survey was nationally distributed to English-speaking healthcare providers who see patients with RCC in their clinical practice, particularly individuals with personal and/or family history of RCC. The study was approved by The Ohio State IRB 2023E0201, which was closed following an issue with bot responses to the survey and subsequently re-approved after modification The Ohio State IRB 2023B0338. No responses from the initial distribution were included in the study.
Survey design
A cross-sectional survey was nationally distributed via Qualtrics to healthcare providers who see patients with RCC in their clinical practice, particularly individuals with personal and/or family history of RCC. The survey included demographic questions, current practice with recommendations for familial RCC risk, interest in and/or need for empiric risk guidelines, and methods for creating these guidelines. Additionally, participants were presented with case scenarios involving familial RCC and asked to provide hypothetical recommendations for kidney screening via multiple choice and free response questions. The study team's genetic counselors (LB, RM, PM, KP, SEG) designed the survey. The survey was piloted by multiple clinical cancer genetic counselors not involved with the study. The purpose of the pilot survey was to assess content, flow and comprehension of the questions, as well as to identify the estimated time spent completing the survey. The pilot survey answers were not included in the results, and the feedback was incorporated into the current format of the survey.
Survey distribution
Survey distribution utilized listservs and the snowball method. The survey was distributed directly to known individuals in the field, and they were requested to share with additional colleagues who fit the inclusion criteria (snowball method). Additionally, the survey was distributed via NSGC research listserv in August of 2024. It is unknown how many prospective respondents the survey reached and how many declined to participate. The study had a total of 70 participants who met the inclusion criteria and completed the survey.
After completing the survey, participants were given the option to provide their email address to enter an opportunity drawing for one of twenty $50 Amazon Gift cards.
Data cleaning and analysis
Initial analysis utilized published protocols for ensuring data integrity given the rise of internet bots. 14 A total of 108 responses were submitted during the study period and analyzed for the following: 1) Minimum completeness of 60% – eliminated 22 responses; 2) A reCAPTCHA score greater than 0.5 - eliminated four responses; 3) Outlier response time, as calculated by previously reported survey completion times based on length – eliminated 0 responses; and 4) Evaluation of qualitative and quantitative discrepancies – eliminated 0 responses. Of the 82 remaining responses, 12 individuals reported not caring for patients with a personal or family history of RCC and were removed. A total of 70 individuals were included in the final analysis.
Multiple-choice and other discrete questions that generated quantitative data were analyzed using descriptive statistics using R*Stats for frequency calculations. Open-ended responses were analyzed via coder pairs (KP/PN and LB/RM) who independently coded the questions and then compared responses and codes to finalize the overall themes and codes.
Results
Demographics and characteristics of respondents
In total, 70 individuals who met inclusion criteria responded to the survey. Table 1 summarizes the characteristics of these respondents, who were primarily female (89%), between ages 20–60 years (96%), and White (84%). The majority of respondents (46 individuals, 66%) were genetic counselors followed by 11 (16%) medical oncologists. Respondents had variable years of experience, geographical location, and practice setting.
Respondents’ reported demographics.

RCC clinic load, current practice, and familiarity with hereditary RCC
Among all respondents, 93% (n = 65) identified clear cell carcinoma as the most frequently observed type of RCC in their clinics. A large proportion of respondents (84%) indicated that less than 10% of their new cancer patients were diagnosed with RCC. Despite this, 79% reported being moderately or extremely familiar with hereditary renal cancer syndromes (Table 1). Of the non-genetic specialists (n = 10), most reported completing at least one aspects of genetic-adjacent tasks in clinic, such as ordering genetic testing themselves, making risk recommendations for family members, and/or completing a family history to determine patients who need additional hereditary cancer risk evaluation.
Screening recommendations based on specific indications
When asked whether they would recommend screening for family members of RCC patients with various personal and family histories (Figure 1; n = 69 due to one ‘Prefer not to Answer’ for all scenarios), providers varied on their certainty of providing recommendations based on indication. The indication with the highest agreement was for family members of patients with RCC and 2 FDR with RCC, where 76% of providers indicated they would offer screening. In contrast, the only scenario that elicited mostly uncertain responses was for family members of patients with multifocal RCC.
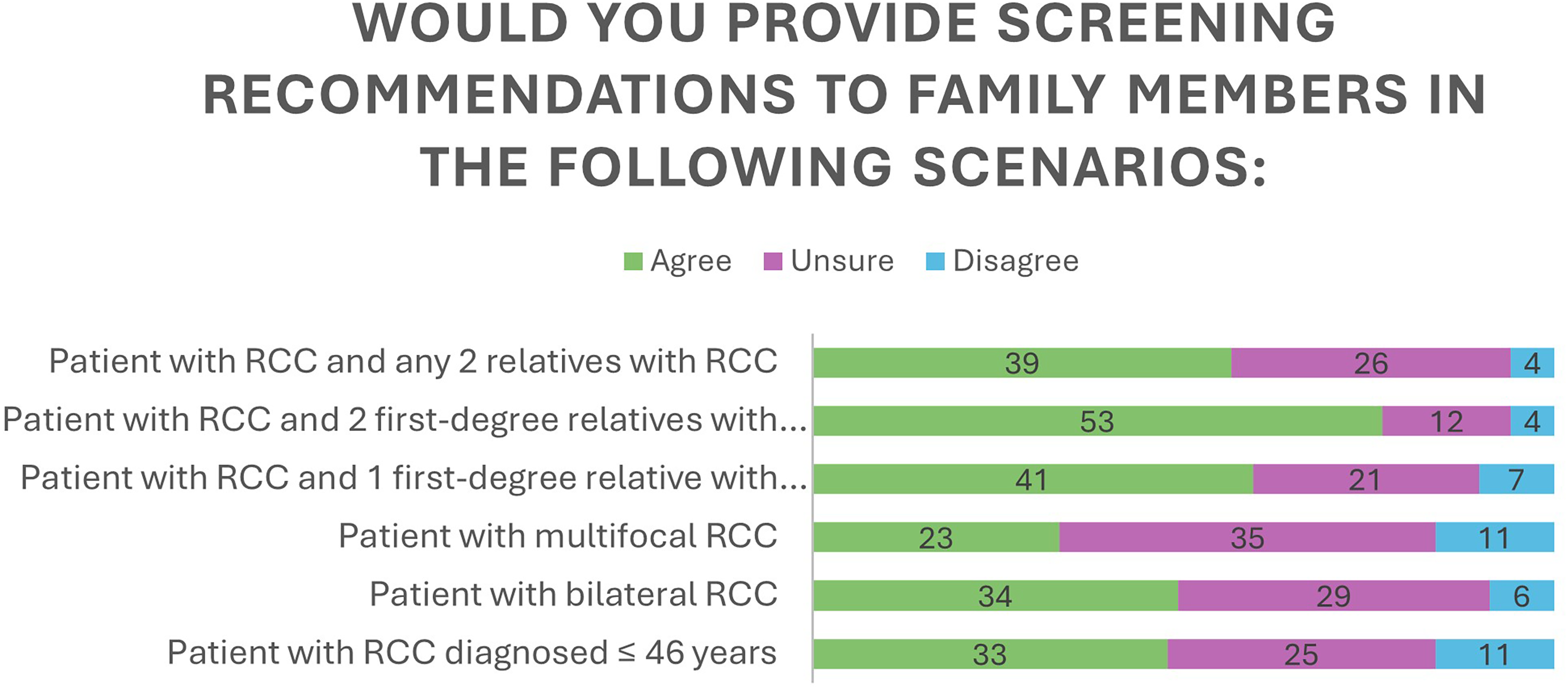
Recommendations for screening given specific combinations of personal and/or family history.
For broader scenarios, where patients had RCC and negative genetic testing (Table 2), five (7%) providers reported they would consistently recommend screening for RCC in family members, while (n = 20; 29%) would not. Approximately 64% of respondents reported they would consider screening on a case-by-case basis. The most common reason for consideration of screening for those who selected the case-by-case approach included a family history of kidney cancer (49%) and proband's age of diagnosis (21%). Additionally, 19% (n = 13) selected to defer to other health professionals, all but two of whom were genetic counselors. When asked why they do not provide screening recommendations for family members, 43 (62%) providers cited the lack of guidelines for determining when empiric risk recommendations/screening are warranted, despite the presence of familial risk.
Responses to questions: RCC screening guidelines for family members.

Perceived patient interest in screening for familial RCC risk
Overall, providers reported that patients frequently inquire about their families’ risk for RCC and associated screening recommendations. Forty-four percent of respondents reported patients “sometimes” inquire about familial RCC risk, while 40% indicated patients ask “often” or “always” do so (Table 2). Similarly, 47% of providers noted that patients “sometimes” ask about screening recommendations, and 33% reported patients inquire “often” or “always” (33%) (Table 2).
Respondent interest in empiric risk screening guidelines and reasoning
Overall, 91% of providers responded “yes” when asked whether empiric risk screening guidelines are needed for family members of patients with RCC; and the other six respondents were unsure (Table 2). A free-text box invited providers to explain the need for such guidelines, and responses were coded into five themes (Table 3). A majority of respondents felt that “There appears to be empiric risk, and other cancers have empiric risk guidelines” (63%); “Providers want clear evidence-based guidance/guidelines” (62%); and “Guidelines provide a benefit to patients and family” (51%).
Core themes highlighting the necessity of RCC screening guidelines.

Considerations for RCC empiric risk screening guidelines
Participants were asked to select all specialists who should be involved in developing empiric/familial risk screening guidelines for RCC. The top three selected were genetic counselor (94%), urologist (93%), and medical oncologist (84%). The most commonly selected mechanism for RCC screening in this population was renal ultrasound, chosen by 55 respondents (79%), followed by biochemical screening (51%) (Table 2). Sixteen participants (23%) selected all three options (renal ultrasound, renal MRI, and biochemical screening), while over 45% of participants (32) selected two of these three options. (Table 2).
Discussion
This study assessed current practices and provider perspectives on RCC screening for unaffected family members that have a relative with RCC and negative germline genetic testing. Almost all respondents (91.4%) expressed a need for empiric risk screening guidelines. This sentiment was driven by the existence of similar guidelines for other cancer types, challenges in determining appropriate clinical screening options, inadequate insurance coverage, and frequent patient inquiries regarding managing familial risk. Respondents differed in their preferred mechanism for RCC screening, though most respondents suggested imaging as a primary tool for screening. Additionally, respondents felt that a multi-disciplinary team should be convened to develop empiric risk screening guidelines for RCC, including oncologists, urologists, genetic specialists, and surgical oncologists.
These results underscore the critical need for screening guidelines for individuals with a family history of RCC in the absence of a pathogenic variant conferring hereditary RCC risk. Nearly 70% of respondents who supported risk screening guidelines reported that these guidelines are necessary to mirror other cancer screening recommendations and guidelines. This indicates that providers are familiar with and utilize empiric risk screening guidelines for other cancer types; as such, they would be likely to also implement empiric risk guidelines for RCC in their clinics. Quantitative risk estimates for individuals with a family history of kidney cancer have varied in previous literature15–17; however, recent data suggests a minimum two-fold increase in RCC risk for individuals with at least one first-degree relative with RCC.4,6,18 This risk estimate is similar to the general population risk for colorectal cancer, a disease with well-established population screening guidelines. As such, given the comparable absolute risk number, it is not unreasonable to consider screening guidelines for RCC in individuals with a family history of RCC.
In the absence of formal guidelines, decision-making for who benefits from RCC screening can be highly variable. Respondents varied in whether age at diagnosis, family history, and/or type of kidney cancer played a role in recommending screening for family. This data suggests that multiple indications should be considered in determining optimal screening guidelines. Respondents also highlighted various mechanisms for screening, including renal ultrasound, MRI, and/or biochemical screening. As such, finding consensus on recommended imaging would standardize recommendations and patient messaging. It is possible respondents differ in their recommended screening modality based on clinical experience, insurance coverage, and institutional resources, which must be factored into the development of screening guidelines. Additionally, most respondents expressed that clear guidelines would also improve insurance coverage and subsequently, access to screening. While these screening approaches and indications have been discussed in previous literature for RCC, an expert-driven consensus guideline would provide universal recommendations, resulting in consistency and clarity for providers, patients, and payers. Other clinical guidelines exist, such as guidelines for genetic testing in RCC8,9,19 and empiric risk management recommendations for other cancers based on family history and/or germline testing.20–23 These guidelines suggest a similar benefit would be seen if RCC screening guidelines for individuals with a family history of RCC existed. 24
Multiple guidelines recommend inquiring about family history in patient care settings.25–27 Therefore, it is not surprising that over 80% of respondents receive questions from patients about RCC risk in family members and appropriate screening recommendations. Having standard and appropriate recommendations to screen for RCC allows for consistent conversations with patients and their families, among other stakeholders.
In order to develop consensus guidelines for empiric risk screening in RCC, a multidisciplinary gathering is required to discuss, evaluate, and identify standard recommendations. Over half of respondents recommended representatives from medical oncology, urology, surgical oncology, and genetics to participate in the development of screening guidelines. To ensure broad applicability, the diverse representation of healthcare providers should go beyond specialty to include individuals from a variety of practice settings, geographical location, training background, and years of experience. Consensus guidelines for other cancer types have used the above-mentioned demographics to define their expert panel membership, which can be mirrored in RCC. 20 28–30
Conclusion and future directions
This study is the first known evaluation of provider perspectives on kidney screening for family members based on personal and family history of RCC without a known hereditary cancer syndrome. The findings emphasize a strong desire among providers for empiric risk-based screening guidelines to address patient inquiries, guide clinical management, and improve insurance coverage. Given the comparable absolute risk of RCC among individuals with a first-degree relative to other cancers where screening guidelines exist, there is a compelling rationale to pursue consensus-based RCC screening recommendations. There was broad agreement on the need for a multi-disciplinary team—including oncologists, urologists, surgical oncologists, and genetic specialists—to develop standardized recommendations.
Guidelines for RCC screening due to a family history of RCC should consider:
Screening mechanisms (e.g., imaging, biomarkers), with specific guidance on optimal imaging (e.g., MRI vs. ultrasound) Relevant characteristics of the proband's RCC diagnosis (e.g., age at diagnosis, histology, laterality). Degree of familial relationship Appropriate screening frequency
The consensus panel should discuss specific case scenarios, as exemplified in this study, to determine what combinations of family history, age at diagnosis, and/or type of RCC warrants additional screening in family members. Additionally, members should consider if a specific specialist, or multi-disciplinary team, should manage this risk and cancer screening. The establishment of such guidelines would help address current variability in practice, provide clarity for providers and patients, and ensure equitable access to appropriate screening for individuals with a family history of RCC.
Limitations
The primary limitation of the findings is that the majority of respondents (n = 53 out of 70) reported being a genetics provider. The enriched sample could be subject to selection bias favoring those more engaged in hereditary cancer management. The survey format may also limit the depth of respondents’ rationale behind their screening preferences. Future research should include a broader sampling of providers across various specialties, practice settings, and regions. In addition, future research should incorporate qualitative approaches to better understand the complexities behind creating consensus guidelines for RCC screening in unaffected, germline-negative individuals with a family history of RCC.
Footnotes
Acknowledgments
The authorship is grateful to McKenna Lewis for her help collating demographic information from the data.
Ethical considerations
This study was approved by The Ohio State IRB (2023E0201and 2023B0338) on March 7th, 2023 and December 18th, 2023. All participants provided informed consent prior to participating
Author contribution statement
The following ICJME author roles were held by each author:
Funding
This research was supported by funding from National Society of Genetic Counselor (NSGC) Cancer SIG Grant Award. This funding was originally awarded in December 2022.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
This data is available upon request to the corresponding author.
