Abstract
Advances in cancer therapies have allowed for improved cancer outcomes; however, with the benefits of novel treatments, clinicians must recognize associated side effects, including acute kidney injury (AKI). AKI and associated changes in kidney function are particularly consequential in patients with cancer, as these may lead to ineligibility for treatments, delays and dose reductions in therapy as well as exclusion from clinical trials. This in turn has a negative impact on cancer outcomes. In this review, we address anticancer drug-induced AKI, with specific focus on conventional cytotoxic chemotherapies and targeted therapies chemotherapies. We discuss frequently nephrotoxic chemotherapies, including: platinum-based agents (cisplatin, carboplatin, oxaliplatin); methotrexate and its derivative, pemetrexed; gemcitabine; and other systemic therapies frequently complicated by AKI. With respect to targeted therapies, we review both AKI and pseudo-AKI (i.e. serum creatinine elevation related to drug-mediated impairment of tubular creatinine secretion) from multiple classes, including: anaplastic lymphoma kinase (ALK) inhibitors; cyclin-dependent kinase-4/6 (CDK4/6) inhibitors poly-ADP ribose polymerase (PARP) inhibitors; vascular endothelial growth factor (VEGF) and multi-target tyrosine kinase inhibitors, among others. For each therapy, we review mechanisms of injury and associated renal lesions and preventive strategies, where available. Both oncology and nephrology clinicians must be aware of, promptly recognize, and appropriately manage AKI associated with cancer therapies to allow for optimal cancer outcomes.
Keywords
Introduction and overview of AKI in the patient with cancer
Considerable advances in cancer therapies have allowed for improved cancer outcomes; however, with the benefits of novel treatments, clinicians must recognize associated side effects, including acute kidney injury (AKI). Patients with cancer represent a particularly high risk population to experience AKI. Population-based data from Denmark indicates a one-year AKI risk of 17.5% post-cancer diagnosis. The highest risk estimates were seen in patients with kidney cancer, liver cancer or multiple myeloma. 1 A Canadian study observed 163,071 cancer patients initiating systemic therapy, of whom 9.3% experienced AKI. Increased risk of AKI was seen in patients with advanced cancer stage, chronic kidney disease, and diabetes mellitus (with adjusted hazard ratios [aHR] of 1.41, 1.80, and 1.43, respectively). 2 Kidney injury has a significant impact on hospital length of stay and increases the risk of in-hospital mortality by 4.7-fold. 3 Amongst patients with critical illness requiring renal replacement therapy, 6-month mortality has been estimated to be 73%. 4 AKI and associated changes in kidney function are particularly critical in cancer patients as these may lead to ineligibility for treatments, delays and dose reductions in therapy as well as exclusion from clinical trials. This in turn has a negative impact on cancer outcomes. Hence, it is of the utmost importance to prevent, recognize and manage AKI in this patient population.
The classic etiologic approach to AKI which includes assessment for prerenal, renal and post renal causes of kidney injury applies also to cancer patients (Figure 1). In this review we will focus on the intrinsic renal category, specifically anticancer drug-induced AKI. It should, however, be noted that the majority of AKI in the cancer population may often be due to reasons other than direct treatment-related toxicity (e.g. volume-depletion, sepsis, obstruction, etc.). Hence judicious attribution of AKI causes is critical to avoid interruptions or even cessation of beneficial cancer therapy.
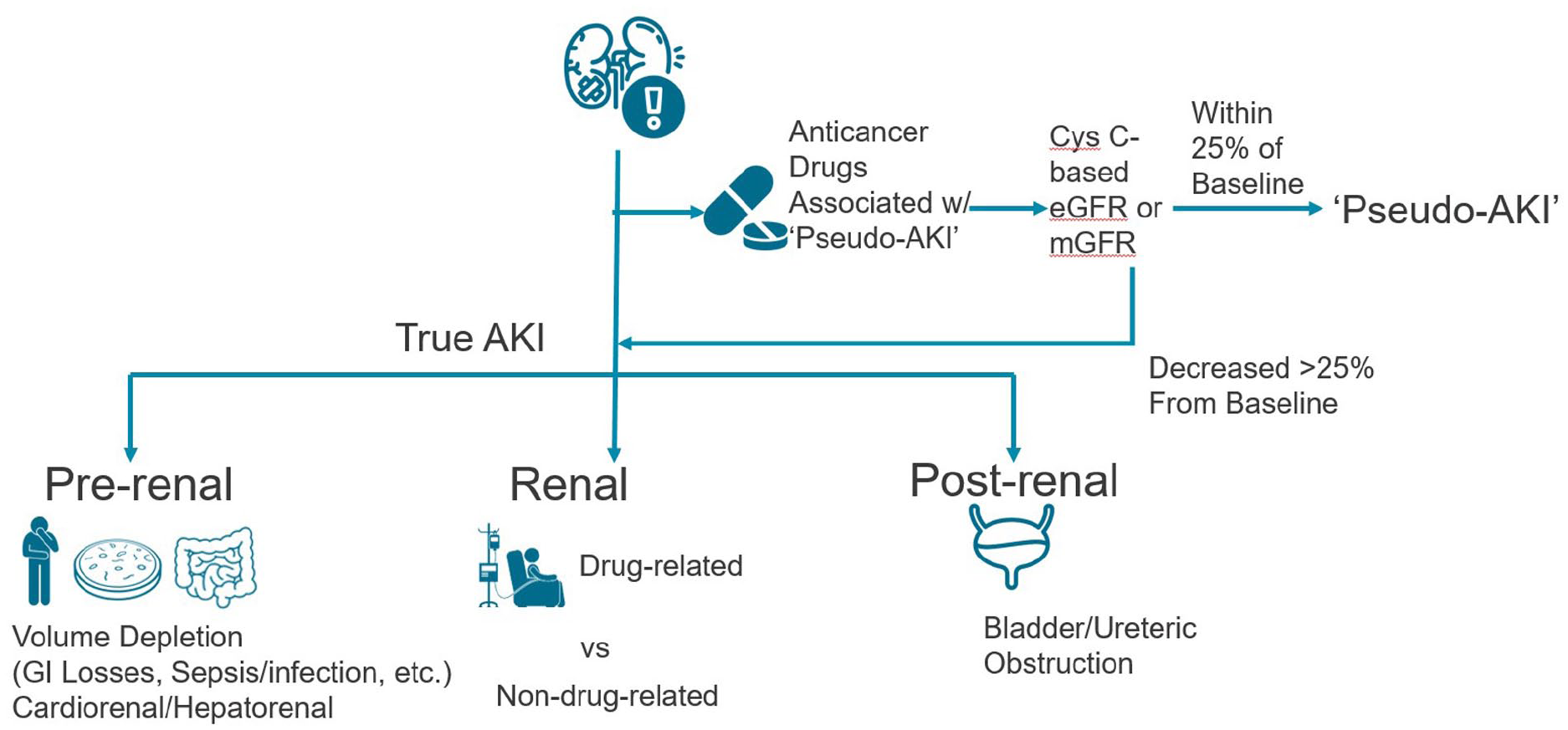
Simplified approach to acute kidney injury and pseudo-acute kidney injury in the patient with cancer.
In the current era of cancer management, systemic cancer therapy comprises: conventional/cytotoxic chemotherapy; immunotherapy, which utilizes the body’s own immune system to fight malignancy; and targeted anticancer therapies, which are directed to specific tumor mutations and susceptibilities. This review will address AKI associated with conventional cytotoxic chemotherapies and targeted therapies chemotherapies (as immunotherapies are discussed in a separate review).
Conventional chemotherapy
While targeted and immunotherapy have transformed oncology, conventional cytotoxic chemotherapy is still extensively used and remains the cornerstone of treatment for several malignancies.
Platinum-based agents
Platinum-based agents remain the mainstay of treatment for several cancers, including genitourinary, gynecologic, lung, and head and neck cancers. 5 Their antineoplastic effect is via DNA binding and inhibition of DNA replication, resulting in cell cycle arrest and apoptosis. 6 Cisplatin, carboplatin, and, to a much lesser extent, oxaliplatin are associated with direct tubular toxicity. Cisplatin-induced AKI (CP-AKI) incidence is dose-dependent and can be up to 20%–30% of recipients.7,8 In addition to AKI, these drugs are also associated with other features of tubular dysfunction, including hypomagnesemia, which is very frequent, in up to 50% of recipients. Hypokalemia, hypophosphatemia, kidney salt-wasting syndrome, and full Fanconi’s syndrome-like presentations have also been described. Rarely thrombotic microangiopathy been reported. 9 Although CP-AKI is common, fortunately, in a 5-year follow-up of 859 patients who received cisplatin showed that the median eGFR decline was 10 mL/min per 1.73 m², with fewer than 3% of patients progressing to eGFR <29 mL/min/1.73 m² and no cases of end-stage kidney disease. 10
Risk factors for cisplatin nephrotoxicity include advanced age (above 60), female patients, hypoalbuminemia (below 3.5 g/dL), hypertension, diabetes, hypomagnesemia, smoking status, higher doses (more than 100 mg), anemia (hemoglobin < 110 g/L) and leukocytosis (WBC > >12.0 ×109/L) have been established and even incorporated into risk prediction scores.11 –13 A recent important study by Gupta et al. demonstrated improved ability to predict cisplatin-associated AKI. The primary definition considered this as doubling or greater increase in serum creatinine or need for kidney replacement therapy within 14 days of cisplatin receipt. Risk factors for cisplatin associated AKI were assessed in a derivation cohort of 11,766 cisplatin recipients at Memorial Sloan-Kettering Cancer Center. Their simplified model included, age, hypertension, diabetes, selected lab parameters and cisplatin dose. Using these covariates they were able to discern risk categories well in both the in the derivation cohort and a similarly sized validation cohort from five other U.S. centers. This model performed well with AUC value in the range of 0.75, which was an improvement versus previously existing models and may allow for routine clinical prediction of high-risk cisplatin recipients.
Regarding AKI prevention, given that the concentration of cisplatin in the kidney is about five times greater than its concentration in other tissues, intravenous volume repletion emerged as one of the earliest and most effective strategies for preventing CP-AKI.14,15 In a systematic review, different intravenous fluid protocols were reviewed with a final recommendation of 2–4 L over 2–6 h. 16 Magnesium supplementation is another preventive strategy for CP-AKI, as magnesium deficiency increases susceptibility for CP-AKI by downregulating renal efflux transporters in the renal proximal tubules and promoting inflammation, resulting in intracellular platinum accumulation. Intravenous magnesium helps restore transporter expression and enhances urinary platinum excretion. A recent study of more than 13,000 patients found that IV MG was independently associated with a lower risk of CP-AKI. 17 Split or fractionated dosing of cisplatin has been explored as a preventive strategy for CP-AKI, but it was not effective in AKI prevention, and the standard dose was associated with improved cancer outcomes. 18 Mannitol for forced diuresis is a preventive strategy that has shown conflicting results, with some evidence suggesting a higher risk of AKI among mannitol-treated patients, especially at higher cisplatin doses. 19 A recent target trial emulation study assessing the effect of mannitol on cisplatin associated AKI included 24,529 cisplatin recipients, of whom 9271 were treated with mannitol versus 15,258 who did not receive mannitol. They found that patients who received mannitol were at increased risk of AKI, with an adjusted odds ratio of AKI 1.66 times non-recipients. Recently, sodium-glucose co-transporter 2 (SGLT2) inhibitors have shown promise in preventing cisplatin-associated magnesium wasting and tubular dysfunction. In a rat model study, Jesus et al. 20 demonstrated that empagliflozin normalized serum magnesium in cisplatin-treated rats, which is suspected to be through reversal of Na+-K+-Cl- cotransporter 2 (NKCC2) inhibition and claudin-16 downregulation in the thick ascending limb and normalizing NaCl cotransporter (NCC) function and melastin six expression in the distal convoluted tubule (i.e. key sites of magnesium resorption). Canagliflozin was shown to reduce cisplatin-induced nephropathy in a mouse model (via reduced cisplatin uptake in kidney tissues). In vitro, canagliflozin was shown to suppress renal proximal tubular cell apoptosis in vitro through increased Akt activation, a signaling pathway involved in prevention of the mitochondrial pathway of apoptosis. 21
Carboplatin (10%–15%) and oxaliplatin (<5%) are also associated with nephrotoxicity, though the risk is significantly lower compared to cisplatin.9,22,23 Carboplatin is excreted primarily in the urine. Unlike cisplatin, carboplatin is not significantly secreted by the tubules; thus, kidney clearance is proportional to GFR. 24 Thus the area under the concentration (AUC) vs time curve for carboplatin will be dependent primarily on the baseline GFR and the dose administered. The Calvert formula was derived to calculate the dose of carboplatin necessary to achieve a particular AUC. 25 In a systematic review of elderly patients receiving platinum-based agents, the incidence of moderate or severe AKI with carboplatin was approximately half that of cisplatin. 22
Methotrexate
Methotrexate (MTX) inhibits folic acid metabolism and limits the cell’s ability to divide and produce proteins. It is used in hematological malignancies such as acute lymphoblastic leukemia (ALL), lymphomas, and sarcomas among others. 26 High-dose methotrexate is defined as a dose higher than 500 mg/m2. It is associated with AKI in 2%–12% of patients. Nephrotoxicity results from MTX and its metabolites’ accumulation as crystals in the renal tubules, which are poorly soluble at an acidic pH, and it also induces afferent arteriolar constriction, leading to a decline in GFR. 27 AKI increases the MTX concentration and leads to systemic toxicity, including mucositis, hepatitis, encephalitis, and myelosuppression. Nomogram-directed high-dose leucovorin or folic acid, given within 24–36 h post-methotrexate, helps prevent methotrexate toxicity. 9 Volume repletion and urinary alkalization, aiming to increase its solubility in the kidney tubules and enhance its elimination, are also effective in treating MTX nephrotoxicity. Glucarpidase is a recombinant bacterial enzyme that can reduce active MTX levels by 90%–95% within minutes of administration, which allows safe resumption of HDMTX following nephrotoxicity. However, it is not widely used because of its extremely high cost. Even though MTX is dialyzable, dialysis is insufficient for adequate clearance of MTX because of high protein binding and a significant post-dialysis rebound. EXTRIP Workgroup recommends against extracorporeal treatments, even if glucarpidase is not administered. 28 A recent target trial emulation study assessed receipt of glucarpidase within 4 days of MTX administration on kidney recovery (defined as survival to discharge with serum creatinine <1.5-fold baseline and without dialysis dependence). Glucarpidase receipt was associated with a 2.70-fold increase adjusted odds of kidney recovery, however there was no difference in time to death. Patients treated with glucarpidase also had lower risks of grade ⩾2 neutropenia (adjusted odds ratio [aOR], 0.50) and grade ⩾2 transaminitis (aOR, 0.50) by day 7. 29
Pemetrexed
Pemetrexed is an antifolate medication derived from methotrexate, treats several cancers, including non-small cell lung cancer and mesothelioma. Pemetrexed is contraindicated in patients with creatinine clearance < 45 mL/min. 30 It is usually given in combination with other therapies like platinum or immunotherapy, which makes it challenging to know the exact incidence of nephrotoxicity related to pemetrexed, but it is estimated to be in the range of 20%.9,31 It can cause renal injury through different mechanisms including: acute tubular (ATN), progressive interstitial fibrosis, nephrogenic diabetes insipidus, and distal renal tubular acidosis. 9 Nephrotoxicity treatment and prevention are mainly supportive and may include volume expansion, vitamin B12, and folate supplementation. Discontinuing drugs usually stabilizes or reverses AKI. In some patients, high cumulative drug dosing leads to CKD due to progressive interstitial fibrosis. 32
Gemcitabine
Gemcitabine is an antimetabolite used to treat a wide range of malignancies, including non-small-cell lung carcinoma, hepatobiliary and ovarian cancers, soft tissue sarcomas, and lymphomas.1.4 % of gemcitabine recipients may have an immune-mediated thrombotic microangiopathy (G-TMA), which can present as systemic or renally limited. Newly diagnosed or worsening hypertension is an early sign of G-TMA with or without AKI and proteinuria.9,33 Discontinuation of gemcitabine typically leads to stabilization or improvement in kidney function. While data on plasma exchange remain limited, complement inhibition with eculizumab has shown potential benefit in small case series. 33
Other nephrotoxic conventional chemotherapies
Multiple other conventional cytotoxic therapies have been associated with renal sequelae (Table 1). Ifosfamide may cause proximal tubular dysfunction with increased urinary phosphate excretion, AKI and progression to CKD in some cases. In a case series of 34 adults with renal dysfunction after ifosfamide treatment 29.4% progressed to Stage 5 CKD, 17.6% required haemodialysis and 17.6% patients died during a median follow-up period of 31 months. Kidney biopsies revealed acute tubular necrosis in 85.7% of cases and evidence of mitochondrial toxicity on electron microsopy. 34 Risk factors for ifosfamide-induced nephrotoxicity in children include cumulative drug dose, pre-existing kidney disease, and concomitant use of cisplatin or carboplatin. 35
Nephrotoxicities associated with selected coventional chemotherapies.
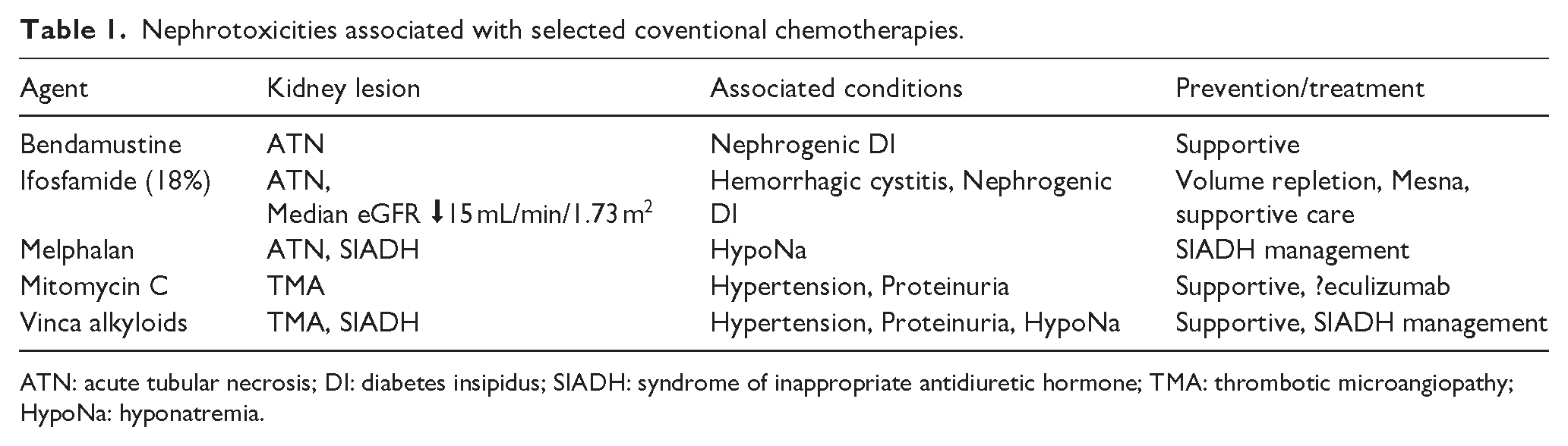
ATN: acute tubular necrosis; DI: diabetes insipidus; SIADH: syndrome of inappropriate antidiuretic hormone; TMA: thrombotic microangiopathy; HypoNa: hyponatremia.
Bendamustine has been shown low renal toxicity in phase II trials 36 but more recently nephrotoxicity attributed to acute tubular necrosis has been observed in 27.9% of patients in a retrospective analysis of 132 non-Hodgkin lymphoma patients treated with Benda-EAM [Bendamustine, Etoposide, Ara-C (cytarabine), and Melphalan] conditioning therapy before stem cell transplantation. 37
Vinca alkaloids such as vincristine and less commonly vinblastine can cause hyponatremia mediated by SIADH as a result of direct toxic effect on the osmoreceptor in the neurohypophysis and hypothalamus.38,39 There are also reports of TMA in context of use of these agents. 9
Targeted therapies and pseudo-AKI
Anaplastic lymphoma kinase (ALK) inhibitors
Anaplastic lymphoma kinase 1 (ALK 1) is a member of the insulin receptor tyrosine kinase family used to treat non-small cell lung cancers (NSCLC) that carry an ALK positive mutation (which applies to approximately 5% of all NSCLC) 40 (Figure 2). Several ALK-1 inhibitors are used in clinical practice, including first generation crizotinib, second generation ceritinib, alectinib and brigatinib, and third generation lorlatinib, ensartinib and entrectinib, all of which demonstrate varying profiles of renal side effects (Table 2). Peripheral edema is a common side effect of crizotinib and lorlatinib, affecting up to 50% of patients. 40 Electrolyte disorders such as hyponatremia and hypophosphatemia are also frequently described affecting patients receiving first generation agent more than second and third generation agents. 40 Development and progression of renal cysts have been reported for crizotinib requiring percutaneous drainage and interruptions in treatments in some cases. 41 One case of high grade proteinuria and presumed podocytopathy shortly after beginning of lorlatinib has also been described. 42 Increase in serum creatinine has been observed for treatment with crizotinib, ceritinib, alectinib, brigatinib, ensartinib entrectinib in 23%, 33%–58%, 26%, 2%, 19%, and 13%–15% of patients respectively (no data is currently available for lorlatinib).40,43 –45 Inhibition of multidrug and toxin extrusion protein (MATE-1) and organic cation transporter 2 (OCT2) at the basolateral and luminal surface of tubular epithelial cells that constitute the tubular creatinine secretion pathway is the presumed mechanism of ALK-1 inhibitor induced pseudo AKI. A study of 38 NSCLC patients demonstrated a mean decrease of 23.9% in eGFR using CKD-EPI most commonly observed in the first 2 weeks of treatment with crizotinib. After treatment cessation the average recovery of eGFR was 35.3% when compared to the last treatment day. 44 Chen et al showed similar results in over 800 lung cancer patients receiving MATEi tyrosine kinase inhibitors. In 15/17 cases where cystatin C was available the calculated GFR was higher using cystatin C versus creatinine. 46 Cystatin C can be useful to distinguish pseudo-AKI from true AKI as it is not subject to tubular secretion therefore its plasma levels are not increased as in the scenario of pseudo AKI. Alternatively an iothalamate or iohexol clearance study or the use of radionucleotides as exogenous filtration markers could be considered. 45 However, true acute renal injury has also been seen in patients treated with either crizotinib or alectinib with findings of acute tubular necrosis observed in kidney biopsies.47,48 Hence clinicians must be alert to not attribute all AKI to pseudo-AKI with these drugs. Importantly, data from Pinard et al. 49 suggests that AKI did not impact overall survival and most patients were able to continue ALK-1 inhibitor treatment.
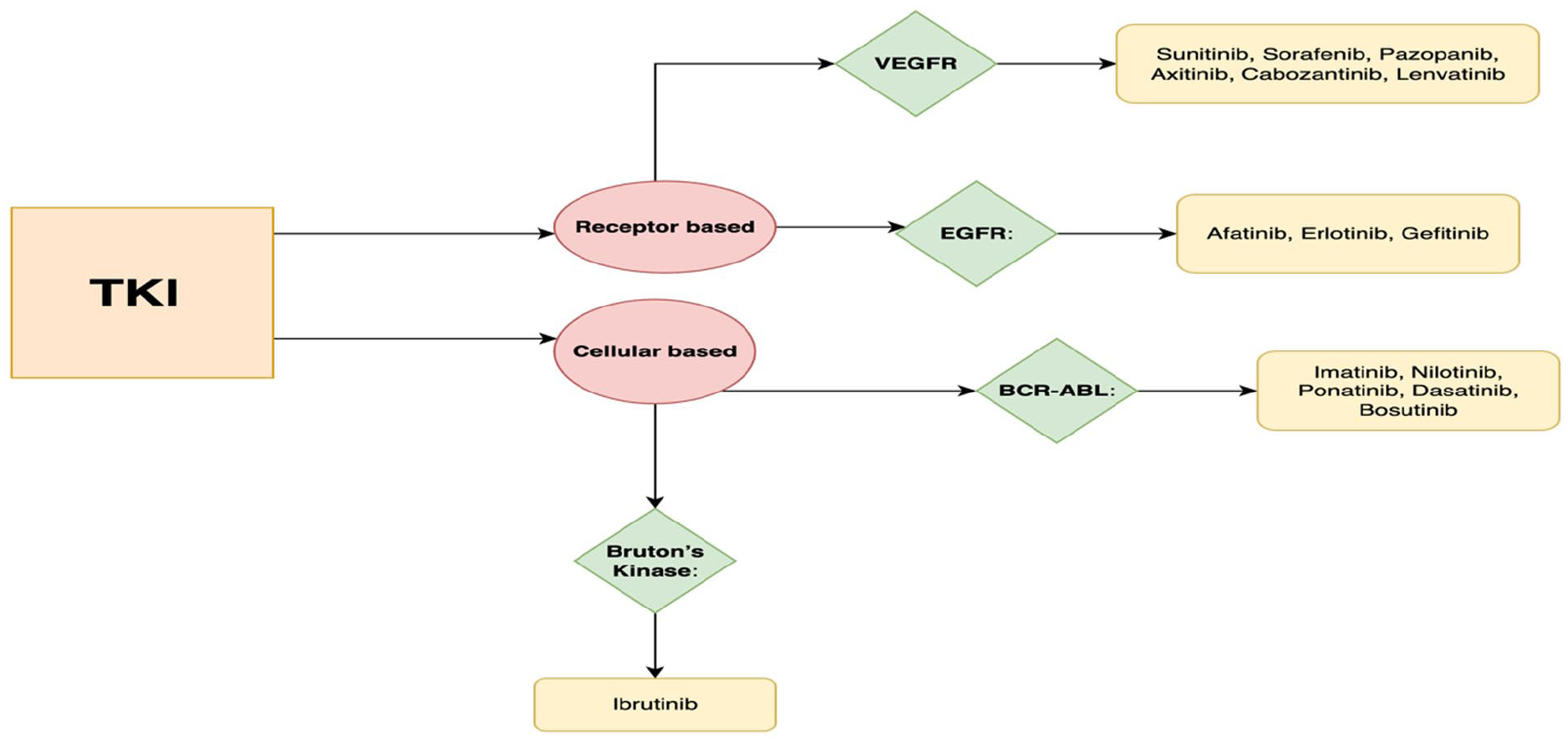
Classification of receptor- and cellular-based tyrosine receptor kinase agents with renal adverse effects.
Targeted anticancer therapies and reported incidence of pseudo-AKI and reported forms of “true” AKI.

SCr: serum creatinine; AKI: acute kidney injury; ATI: acute tubular injury; ATN: acute tubular necrosis; AIN: acute interstitial nephritis; VEGF: vascular endothelial growth factor; TKI: tyrosine kinase inhibitor; TMA: thrombotic microangiopathy; CDK 4/6: cyclin-dependent kinase 4/6; PARP: poly (ADP-ribose) polymerase; BCR-ABL: breakpoint cluster region-Abelson; FSGS: focal segmental glomerulosclerosis.
Cyclin-dependent kinases 4 and 6 (CDK4/6) Inhibitors
Cyclin dependent kinase or CDK 4/6 inhibitors are used in hormone receptor positive metastatic breast cancer. Like ALK inhibitors, these drugs impair tubular secretion of creatinine via MATE-1 and MATE-2K transporters. A pharmacological study by Chappell et al. showed changes in creatinine-based eGFR with abemaciclib were not reflected in mGFR determined by iohexol clearance and neither in eGFR using cystatin C. Urinary biomarkers like NGAL and KIM-1 were also not affected. 50 A recently published retrospective cohort study found elevated creatinine levels in 17.5% of 234 patients treated with CKD4/6 inhibitors. Pseudo-AKI was diagnosed based on cystatin C measurements accounting for 16 of 41 patients (7/8 on abemaciclib; 8/11 on palbociclib, 2/4 on ribociclib). 51 Nevertheless, in a small case series of biopsy proven AKI episodes, three of six patients had severe stage 3 AKI with 1 requiring RRT. Each of the six patients had true AKI demonstrating acute tubular necrosis in five and interstitial nephritis in one case in kidney biopsy. Interestingly, the time of onset of AKI was prolonged in relation to initiation of CDK4/6 inhibitors with a median time-to-AKI of 278 days on therapy with no identifiable trigger for a second hit. 52 A large metanalysis involving 17 randomized controlled trials involving 19,638 patients found an elevated relative risk of nephrotoxic adverse events for all four currently approved CDK4/6 inhibitors with 9.94 for abemaciclib, 1.59 for palbociclib, 3.23 for ribociclib, and 3.04 for dalpiciclib. The authors also highlighted the challenge in distinguishing true versus pseudo-AKI. 53
Poly-ADP ribose polymerase (PARP) inhibitors
Poly ADP ribose polymerase inhibitors (PARPi) are approved as maintenance therapy following platinum-based chemotherapy for patients with ovarian, endometrial, breast, pancreatic and prostate cancer. Olaparib and rucaparib inhibits MATE-1 and MATE-2k transporters and represents another class of targeted agents that may cause pseudo-AKI. 54 Niraparib may have a hemodynamic effect on serum creatinine via dopamine and norepinephrine transporters. 55 Discordance between a calculated low glomerular filtration rate assessed by renal nucleotide scan (99mTc-DTPA) and an eGFR rate was observed in 63% of patients treated with PARPi (olaparib, rucaparib or niraparib). 56 A retrospective analysis of 269 patients showed AKI in 22.1% undergoing treatment with olaparib and niraparib. However only 2.2% of the cohort had sustained AKI that was directly attributable to PARPi. Other etiologies were hemodynamic-mediated AKI and postrenal obstruction. Most patients showed a dose-dependent transient decline of eGFR of about 10 ml/min within 30–60 days followed by stabilization in keeping with transient inhibition of tubular creatinine secretion. 54 In essence, although direct nephrotoxicity of PARP inhibitors has not been described, patients with these types of cancers are prone to other causes of AKI therefore creatinine changes must be worked up and not dismissed hastily as pseudo-AKI.
BCR-ABL tyrosine kinase inhibitors (TKI)
Imatinib is the first FDA-approved TKI for chronic myelogenous leukemia (CML) therapy. It can cause a reversible increase in serum creatinine by inhibiting proximal tubular secretion of creatinine via blockade of OCT2, MATE1, and MATE2-K transporters. This results in pseudo-AKI, and in these cases, cystatin C can help distinguish it from a real renal injury. Because true AKI and CKD where also reported with imatinib in one study of 105 CML patients, 7% of patients developed AKI and 12% patients developed chronic kidney disease (CKD). The mean decrease in estimated glomerular filtration rate (GFR) was 2.77 mL/min/1.73 m2 per year. 57
Also, it is important to mention that newer generation agents like nilotinib and dasatinib do not appear to have the same effect. 58 Dasatinib is a second-generation BCR-ABL1 TKI associated with true AKI that is reported to cause TMA and podocytopathy. 59 In addition to proteinuria, which correlates with the dasatinib plasma concentration. 60
VEGF inhibitors and multi-target TKIs
Vascular endothelial growth factor (VEGF) is an important therapeutic target that showed benefit in randomised clinical trials in multiple cancer settings. Bevacizumab, ramucirumab, and aflibercept are monoclonal antibodies that bind to the VEGF molecule, preventing it from binding to the receptor, then inhibiting endothelial cell proliferation and vessel formation, whereas the small molecule TKIs (sunitinib, sorafenib, pazoponib, axinitib, cabozatinib, lenvatinib, regorafanib, and vendatinib) block the intracellular domain of VEGF. 61 VEGF and multitarget TKIs are associated with hypertension, proteinuria and TMA which may be dose dependent (Figure 3).61,62 Hypertension management should be individualized but generally angiotensin-converting enzyme inhibitor (ACEI) or angiotensin receptor blockers (ARB) would be considered as first-line options and calcium channel blockers as a reasonable second choice. 61
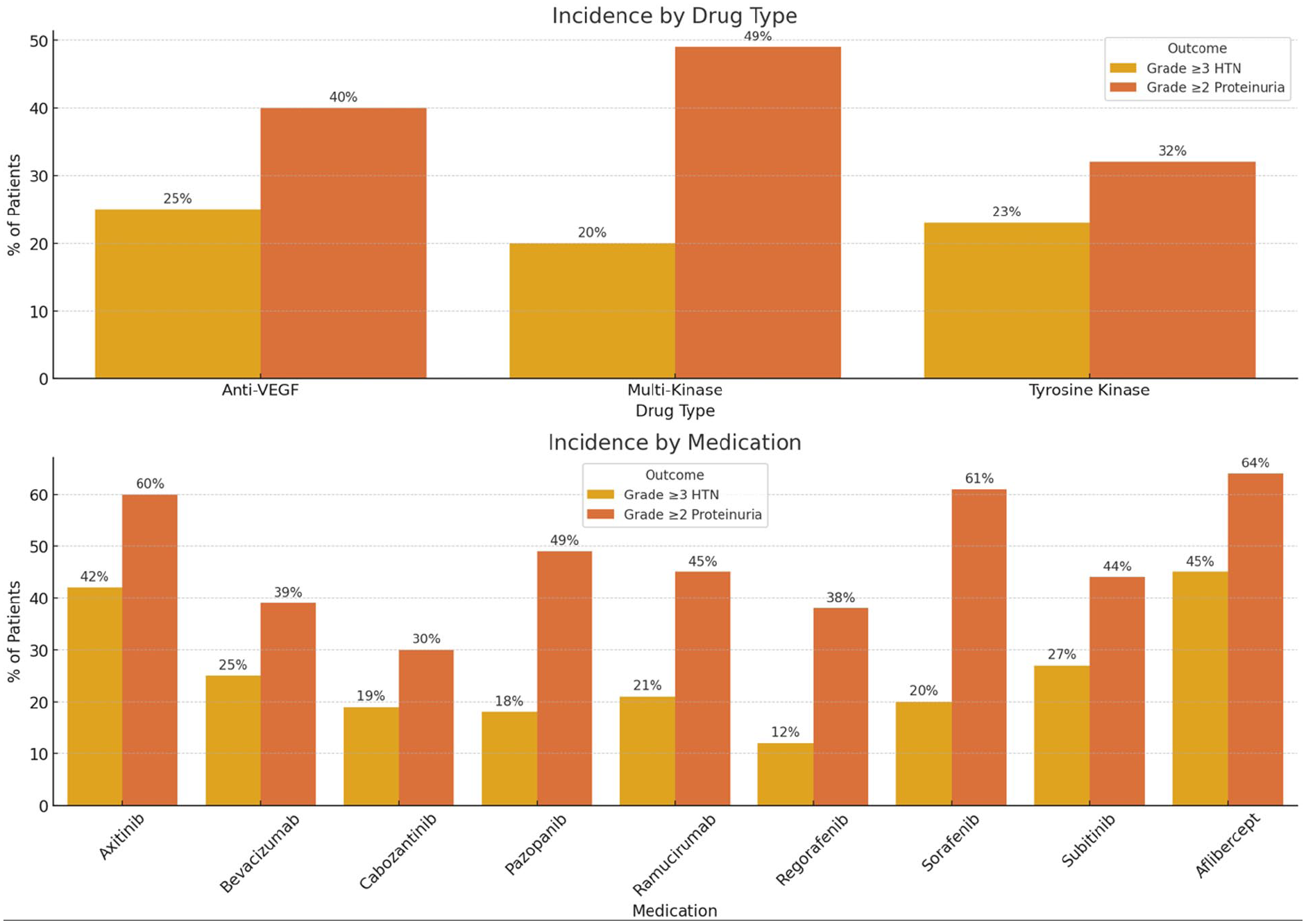
Incidence of hypertension and proteinuria associated with vascular endothelial growth factor signaling pathway agents. Data derived from Adegbite et al. 60
Mitogen-activated protein kinases inhibitors
Trametinib and cobimetinib are potent, highly specific inhibitors of MEK1/MEK2. It is another class of medication that can lead to rise in serum creatinine in up to 62% of patients in case of capmatinib, mimicking AKI without true renal injury. Pseudo-AKI is attributed to inhibition of renal tubular creatinine secretion via MATE1 and MATE2-K transporters, rather than actual nephron damage. 63
BRAF inhibitors (Vemurafenib, Dabrafenib, Encorafenib)
Usually used in combination with MET inhibitors. Vemurafenib inhibits the mutated BRAF V600E, blocking signal transduction trough the MAP kinase pathway and hence, proliferation of cancer cells. It is used to treat metastatic melanoma. In a study of 70 patients treated with vemurafenib (97%) had an immediate increase in serum creatinine level after initiation of vemurafenib that remained stable over time. Further work-up revealed that it was related to a decrease in tubular secretion of creatinine by 43%–81%. But ATN and AIN were also reported in patients treated with BRAF inhibitors. 45
Human epidermal growth factor receptor 2 (HER2) inhibitors
HER2 inhibitors target the HER2 receptor, a tyrosine kinase that promotes cell cycle progression survival through mTOR and MAPK pathways. HER2 overexpression is seen in different cancers, including 15%–20% of breast, 4%–53% of gastric, 2%–11% of colorectal, and 2%–14% of lung cancers. A study reported serum creatinine increases in 13.7% of patients receiving HER2 inhibitors (lapatinib, neratinib, tucatinib) indicating possibility of pseudo-AKI for which cystatin C-based eGFR may again help with diagnosis. 45
Epidermal growth factor receptor TKIs
There are two distinct structurally different classes of epidermal growth factor receptors (EGFR)-targeted therapies. Cetuximab and panitumumab are monoclonal antibodies that are mainly used to treat patients with metastatic colorectal or head and neck cancer. In contrast, erlotinib, afatinib, gefitinib are small molecule inhibitors of EGFR-TKIs used for treatment of non-small cell lung and other solid organ cancers. 64 The most common side effect of these agents is diarrhea, which can cause prerenal AKI. 65 EGF/EGFR is expressed in the ascending loop of Henle and the distal tubule convolute and involved in maintaining tubular integrity, hence mostly tubular side effects such as electrolyte disorders (hypomagnesemia, hypokalemia and hypophosphatemia) and acute tubular necrosis are reported in literature.66,67 Severe hypomagnesemia (grade 3–4) was found in 10%–36% of patients treated with cetuximab and less frequent with 3% in patients treated with panitumumab.66,68,69 Age, baseline magnesium levels and duration of treatment are recognized risk factors. 66 Also two cases of crescentic diffuse proliferative glomerulonephritis are reported with cetuximab of which one was associated with IgA immune-complex deposition70,71 and another case of pauci-immune crescentic GN associated with erlotinib. 72 A case of minimal change disease and a case membranous GN have been reported in association with gefitinib treatment.73,74
Conclusions
Anticancer therapies, including conventional therapies and targeted treatments are associated with multiple forms of AKI, in addition to serum creatinine elevation related to drug-mediated impairment of tubular creatinine secretion (pseudo-AKI). Oncology and nephrology clinicians must recognize the potential forms of kidney injury associated with specific agents and attribute AKI to drug-induced causes when the clinical presentation and known renal complications are congruent. Appropriate recognition and management of AKI associated with cancer therapies is necessary to improve cancer outcomes.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Abhijat Kitchlu administers funding from Amgen, Inc. for the University Health Network Onco-Nephrology Fellowship. The other authors have no conflicts of interest to declare.
Contributorship
Conceptualization: LA, NW, AK; Writing: LA, NW, AK; Figures and Tables: LA, NW, AK.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Abhijat Kitchlu
