Abstract
Background:
Acute kidney injury (AKI) is a common complication in patients with leukemia and lymphoma. In this single-center non-interventional retrospective study from 2010 to 2022, we aimed to evaluate causes, clinical features, and pathology finding in patients with Hodgkin lymphoma (HL), non-Hodgkin lymphoma/leukemia (NHL/CLL), or lymphoplasmacytic lymphoma (LPL) and AKI.
Methods:
We identified 101 patients with HL, NHL/CLL, and LPL. We analyzed AKI incidence and various clinical, laboratory, and pathology parameters and outcomes.
Results:
About 46.5% of patients with HL, NHL/CLL, and LPL presented with AKI. The incidence of AKI was the highest in the CLL setting (65.0%), it was lower and almost equal (48.0% and 47.0%) in NHL and HL, respectively, and the lowest (14.2%) in LPL. Study group comprised of 47 patients, M/F 31/16, median age was 64.0 [52.0; 71.0] years, and the patients with HL were significantly younger (p = 0.022). The median disease duration prior to the AKI presentation was 14 [5; 44] months. Pre-renal AKI was diagnosed in 48.9% of cases: tumor lysis syndrome (TLS), specific lymphoid infiltration (LI), and post-renal AKI presented in 12.7% each. Kidney biopsy in 49% of patients revealed LI in 39.1% of cases. Focal segmental glomerulosclerosis presented in 21.7% of cases, proliferative glomerulonephritis with monoclonal immunoglobulin deposits in 13.0%, AA amyloidosis, IgA nephropathy, and AA amyloidosis in 8.6% each. AKI recovered in 63.8% of patients and 59.5% of patients were alive and dialysis-free at last evaluation: 100% with HL versus 58.3%, 46.1%, and 0% with NHL, LL, and LPL, respectively (p = 0.025); and 8.5% remained alive on dialysis. About 25.5% of patients died after discontinuation or without need for dialysis; and 6.3% died on dialysis.
Conclusion:
AKI in patients with leukemia and lymphoma is heterogeneous and most commonly caused by volume depletion. AKI recovery, though not complete, occurred in 63.8% of cases with 68% survival.
Keywords
Introduction
Acute kidney injury (AKI) is one of the common complications in patients with cancer, recently reported cumulative incidence is 7.3%–9.3%, with the highest incidence in multiple myeloma, bladder cancer, leukemia, and lymphoma (>40%).1–3 AKI in the patients with hematological malignancies, such as multiple myeloma, leukemia, and lymphoma may be associated with tumor-related, treatment-related, and non-specific mechanisms, and in the vast majority of cases kidney biopsy is required to identify the etiology.3,4 In the setting of leukemia/lymphoma tumor- and treatment-related causes include tumor infiltration of the kidney, obstructive nephropathy due to retroperitoneal lymphadenopathy, disseminated intravascular coagulation, tumor lysis syndrome, glomerular diseases, and chemotherapy nephrotoxicity; non-specific causes include volume depletion, sepsis, and iodinated contrast nephrotoxicity.5 –7 For this particular study, we focused on causes, clinical features, and pathology findings in the patients with leukemia and lymphoma, presented with AKI in our center.
Methods
Patients
A single center non-interventional retrospective study using electronic database identified 101 patients ⩾18 years old, diagnosed with Hodgkin lymphoma (HL), non-Hodgkin lymphoma/chronic lymphocytic leukemia (NHL/CLL), or lymphoplasmacytic lymphoma (LPL), followed in 2010–2022.
Study group selection
We identified totally 17 cases of HL, 50 cases of NHL, 20 cases of CLL, and 14 cases of LPL. After evaluation of the clinical data, we excluded from further analysis patients without AKI, defined according to KDIGO Clinical Practice Guideline for Acute Kidney Injury 2012. 8 The study group comprised of 47 patients with HL, NHL/CLL, and LPL presented with AKI.
Study sub-groups selection according the main diagnosis
The main diagnosis was established before admission to nephrology unit in 26 (55.3%) patients, and 21 (44.7%) patients were diagnosed in site according to the WHO classification of tumors of hematopoietic and lymphoid tissues in collaboration with hematologists. 9 HL sub-group included 8 (17.0%) patients; NHL subgroup—24 (51.0%) patients; CLL subgroup 13 (27.6%); and LPL subgroup—2 (4.2%) patients (Figure 1).
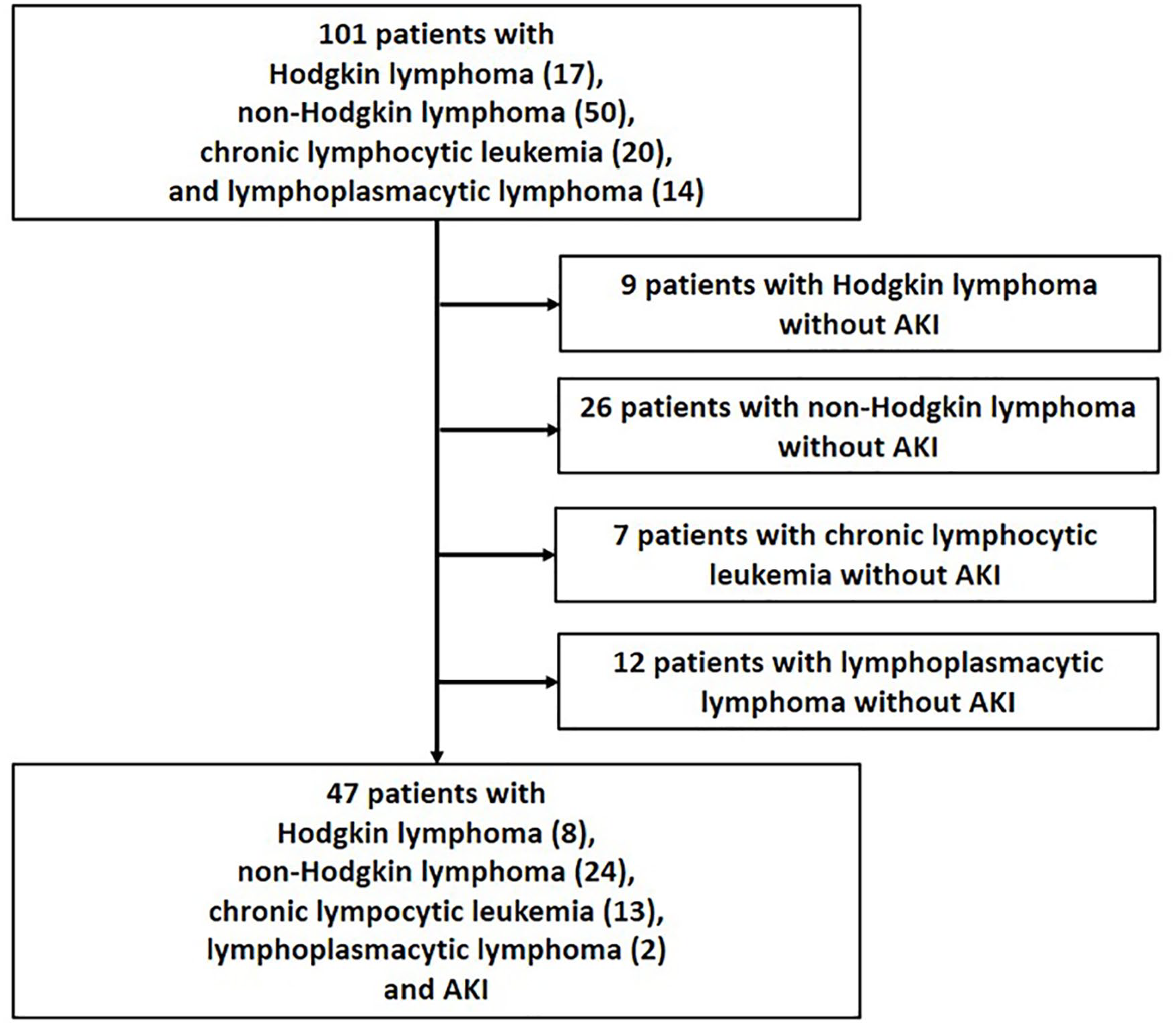
Study group selection.
Data
The main data collected were age, gender, medical history, drug history, blood pressure (BP), urine output, level of proteinuria (PU), hematuria, total blood count, blood chemistry parameters, serum electrophoresis, acid-base balance, kidney pathology findings, and results of hematologic work-up. Hematologic evaluation included bone marrow aspiration and/or biopsy, and/or lymph node biopsy with light microscopy and immunohistological study, peripheral blood or bone marrow cytometry, serum and urine immunofixation, abdomen, kidneys, and peripheral lymph nodes ultrasound, skeletal X-ray, and chest and abdomen CT.
For the purpose of this study, we analyzed the following parameters: demographics, disease duration, hematological diagnosis, previous chemotherapy, BP, anuric state, urinalysis and blood chemistry data, need for hemodialysis (HD) at presentation, AKI cause, kidney pathology findings, and AKI outcomes.
Kidney pathology indications and assessment
Indications for kidney biopsy were the absence of clinical explanation for AKI and/or presence of nephrotic syndrome. Pathology evaluation of kidney tissue included light microscopy and immune staining. For light microscopy, sections of formalin fixed paraffin-embedded tissue stained with H&E, Masson’s trichrome, periodic acid-Schiff and Congo Red. Immunofluorescence was performed on frozen sections with FITS-conjugated antibodies to IgA, IgG, IgM, C1q, C3, fibrinogen, lambda, and kappa light chains.
Statistical analysis
Statistical analysis performed using SPSS 13.0 program package. Normally distributed variables presented as the mean ± standard deviation (Χ ± σ). Comparison between mean data performed using Student criteria or ANOVA test. For abnormally distributed variables median value and interquartile range were calculated, non-parametrical Kruskal–Wallis test and Wilcoxon signed-rank test were used for comparison between these variables. p-Value <0.05 was defined for statistical significance.
Results
Incidence of AKI
Among all patients with HL, NHL/CLL, and LPL totally 46.5% presented with AKI. The incidence of AKI was the highest in the CLL setting (65.0%), it was lower and almost equal (48.0% and 47.0%) in NHL and HL, respectively, and only 14.2% patients with LPL developed AKI (Figure 2).
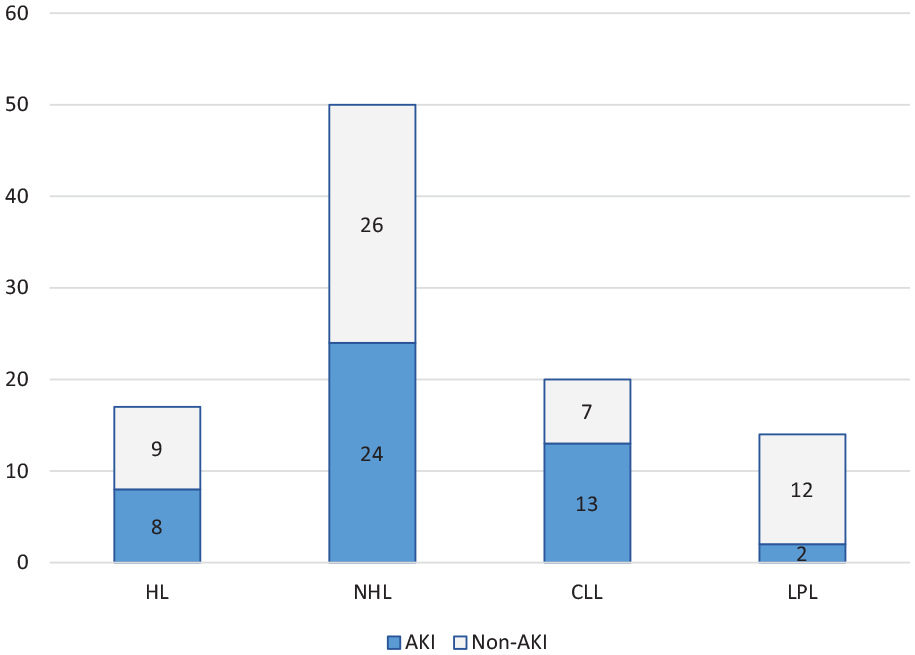
The incidence of AKI in the patients with HL, NHL/CLL, and LPL.
Demographic data and clinical presentation
Study group comprised of 31 (66.0%) males and 16 (34.0%) females, with the median age 64.0 [52.0; 71.0] years; the main demographic data and clinical presentation in the main group and in the sub-groups presented in the Table 1. We did not find significant differences in the gender distribution between the sub-groups; however, the patients with HL were significantly younger than in the other sub-groups (p = 0.022; Table 1).
Main demographic data and clinical presentation in the main group and in the sub-groups.
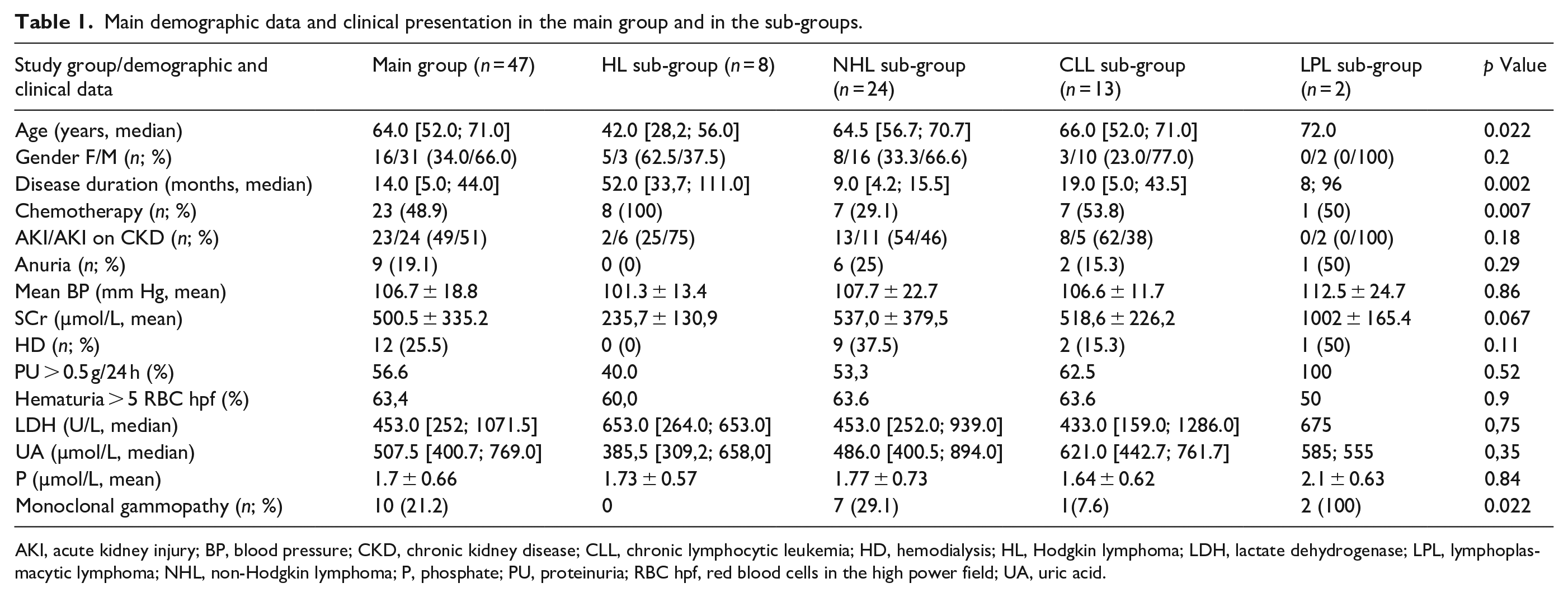
AKI, acute kidney injury; BP, blood pressure; CKD, chronic kidney disease; CLL, chronic lymphocytic leukemia; HD, hemodialysis; HL, Hodgkin lymphoma; LDH, lactate dehydrogenase; LPL, lymphoplasmacytic lymphoma; NHL, non-Hodgkin lymphoma; P, phosphate; PU, proteinuria; RBC hpf, red blood cells in the high power field; UA, uric acid.
The median disease duration prior to the AKI presentation was 14 [5; 44] months; it was significantly longer in the HL sub-group (p = 0.002); and 23 (48.9%) of patients previously received or were on specific treatment; 100% in the HL sub-group versus 29.1%–53.8% in the other sub-groups (p = 0.007; Table 1). A half (49%) of cases were diagnosed with de novo AKI, while another half (51%) developed AKI on the top of CKD.
The mean value of mean BP was 106.7 ± 18.8; 9 (19.1%) patients presented with anuria; and 12 (25.5%) demanded urgent HD at admission. We did not find significant differences between the sub-groups for these variables (Table 1).
In the main group mean serum creatinine (SCr) at presentation was 500.5 ± 335.2 µmol/L, with the markedly higher level in the LPL sub-group, even though the difference did not reach statistical significance (p = 0.067; Table 1).
In the patients with preserved diuresis PU > 0.5 g/24 h and/or microhematuria were detected in the main study group in 56.6% and 63.4%, respectively. Median lactate dehydrogenase (LDH) level was 453.0 [252; 1071.5] U/L, median uric acid (UA) level 507.5 [400.7; 769.0] µmol/L, and mean phosphate level 1.7 ± 0.66 mmol/L, without significant differences between the subgroups (Table 1).
Monoclonal gammopathy was detected in 10 (21.2%) cases (in eight patients in the serum and in two patients in the urine), with significant preponderance in the LPL sub-group (p = 0.002). Serum paraproteins were presented with IgGκ, IgGλ, IgMκ, and IgMλ (two cases of each), and we found urinary excretion of Bence-Jones κ in two cases. Paraprotein-related kidney damage was proven in five cases only—two patients had AL amyloidosis (AL) and three patients—proliferative glomerulonephritis with monoclonal immunoglobulin deposits (PGNMID). Out of the other five patients with monoclonal gammopathy, kidney biopsy was performed in three patients cases and showed lymphoid infiltration (LI) in two, and diabetic nephropathy (DN) in one case, without any features of paraprotein deposition (pathology findings presented in the section below).
AKI causes
Distribution of AKI causes presented in the Table 2 and Figure 3. Pre-renal AKI due to the volume depletion dominated in the main group (48.9%), and in all sub-groups. Tumor lysis syndrome (TLS), LI, and post-renal AKI due to the ureteric compression by the enlarged lymphatic nodules presented in the main group in 12.7% each (Table 2).
AKI causes in the main group and in the sub-groups.
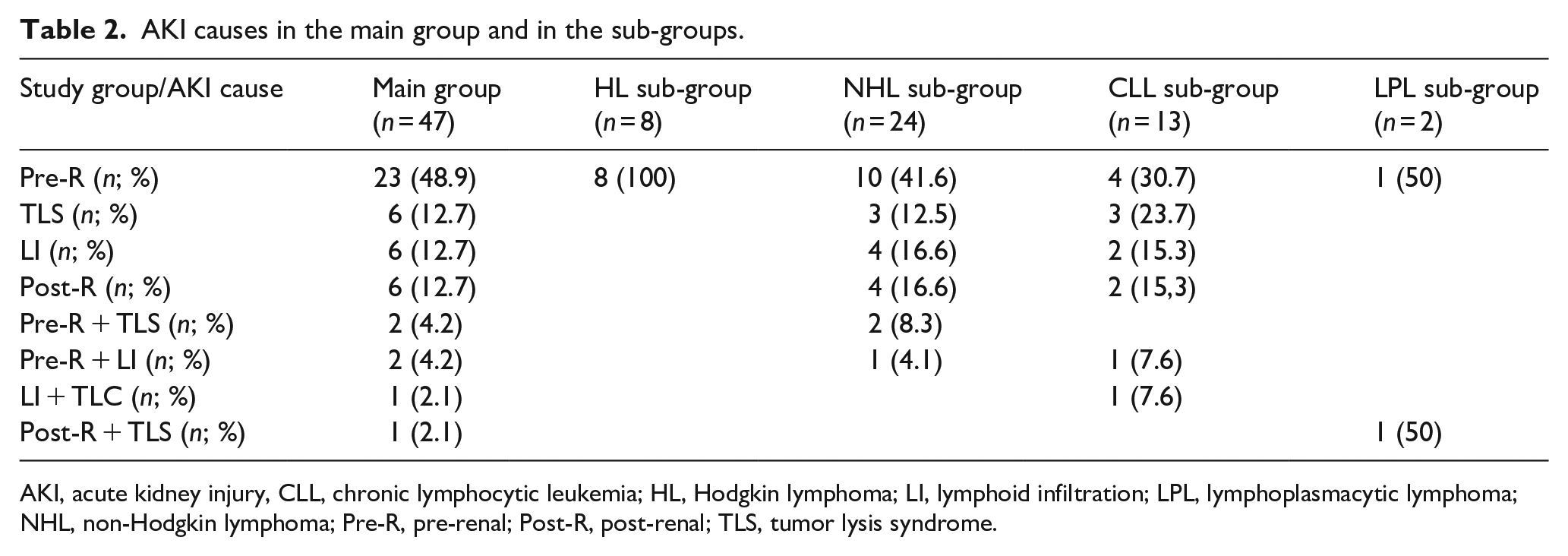
AKI, acute kidney injury, CLL, chronic lymphocytic leukemia; HL, Hodgkin lymphoma; LI, lymphoid infiltration; LPL, lymphoplasmacytic lymphoma; NHL, non-Hodgkin lymphoma; Pre-R, pre-renal; Post-R, post-renal; TLS, tumor lysis syndrome.
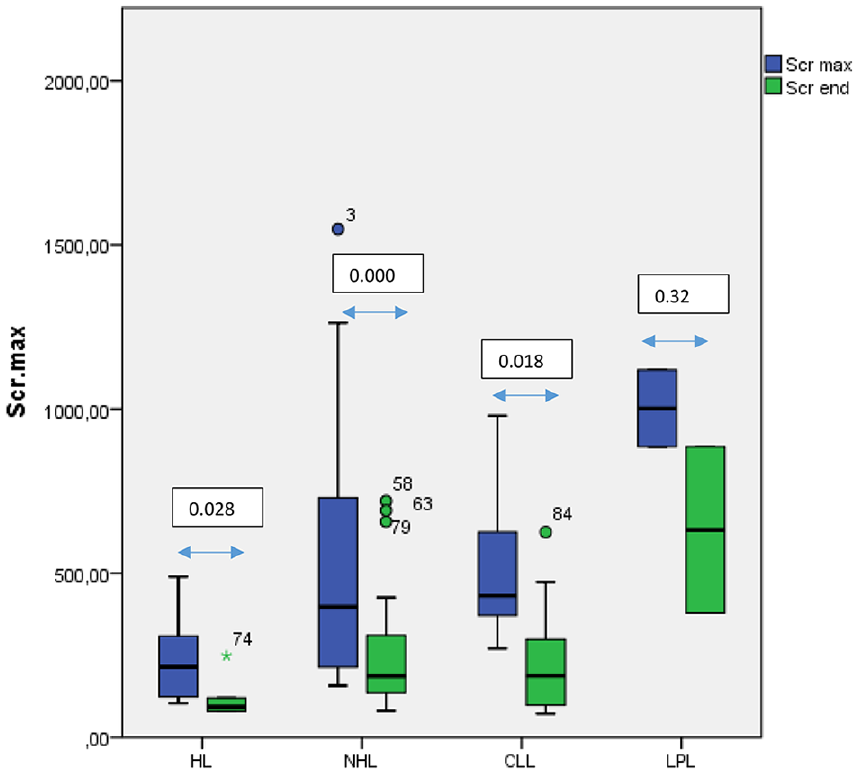
Serum creatinine levels in the sub-groups at presentation and at the latest assessment.
Pre-renal mechanism was the only cause of AKI in HL sub-group, in the other sub-groups pre-renal AKI was diagnosed only in 30.7%–50% of cases. TLS, LI, and post-renal AKI due to the ureteric compression with enlarged lymph nodes were found only in the patients with CLL or NHL; incidence of TLS was almost twice higher in CLL than in NHL (23.7% vs 12.5%), and LI and post-renal AKI presented in equal proportion (15.3% vs 16.6% for both). In the CLL and NHL sub-groups few patients presented also with various combinations of pre-renal AKI, TLS, and LI. A combination of post-renal AKI and TLS was found in one patient with LPL (Table 2).
Kidney pathology findings
In 24 (51%) cases, the cause of AKI was assessed on the clinical basis, and in 23 (49%) cases kidney biopsy was performed, kidney pathology data presented in the Table 3. Pathology evaluation showed LI in 9 (39.1%) cases—in 6 patients it was LI only, and in 3 patients a combination of LI and AL, IgA nephropathy (IgAN), or focal segmental glomerulosclerosis (FSGS) was found. PGNMID and FSGS presented in 3 (13.0%) cases each, and AA amyloidosis (AA) in 2 (8.6%) cases. AL, IgAN, membranous nephropathy (MN), membranoporliferative glomerulonephritis (MPGN), DN, and combination of FSGS and acute tubulonecrosis were found in 1 (4.3%) case each (Table 3).
Pathology patterns of kidney damage in 23 patients with AKI who underwent kidney biopsy.
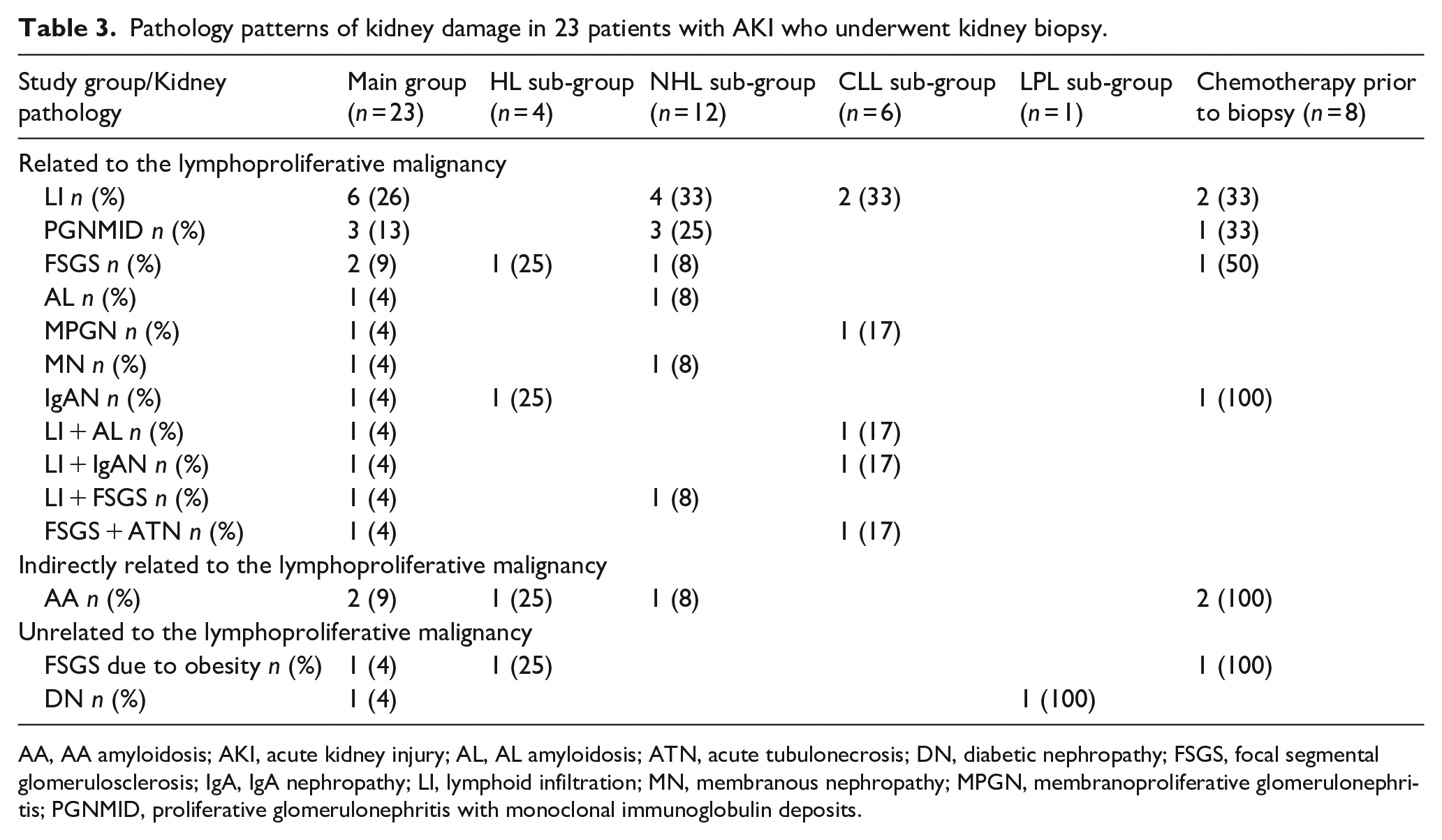
AA, AA amyloidosis; AKI, acute kidney injury; AL, AL amyloidosis; ATN, acute tubulonecrosis; DN, diabetic nephropathy; FSGS, focal segmental glomerulosclerosis; IgA, IgA nephropathy; LI, lymphoid infiltration; MN, membranous nephropathy; MPGN, membranoproliferative glomerulonephritis; PGNMID, proliferative glomerulonephritis with monoclonal immunoglobulin deposits.
LI, AL, MN, and MPGN were found only in NHL and CLL setting; PGNMID—in NHL sub-group only; FSGS, AA, and IgAN—in HL and in NHL/CLL sub-groups; and a single case of DN was revealed in a patient with LPL (Table 3).
AKI outcomes
AKI recovered in 30 (63.8%) patients in the main group, without significant differences between the sub-groups (Table 4). The mean SCr at the latest assessment significantly decreased—from 500.5 ± 335.2 to 238.4 ± 181.9 (р < 0.001); these significant changes were found in all sub-groups except LPL (Figure 3).
AKI outcomes in the main group and in the sub-groups.
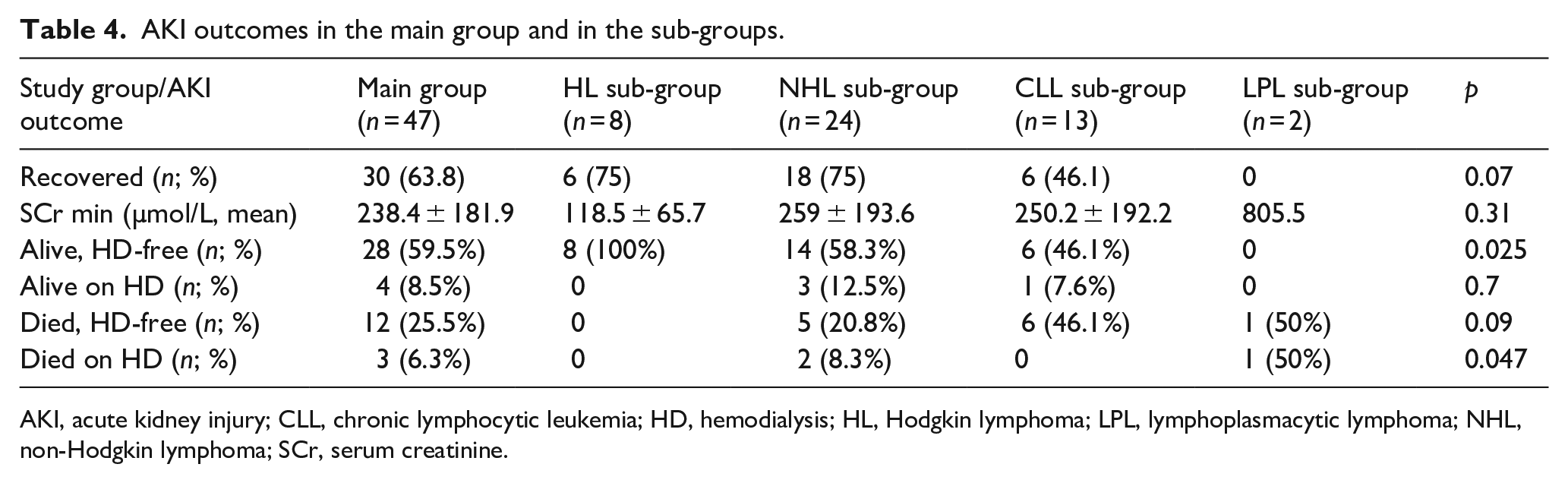
AKI, acute kidney injury; CLL, chronic lymphocytic leukemia; HD, hemodialysis; HL, Hodgkin lymphoma; LPL, lymphoplasmacytic lymphoma; NHL, non-Hodgkin lymphoma; SCr, serum creatinine.
In the main group at the latest assessment, 28 (59.5%) patients were alive and dialysis-free, there were 100% in the HL sub-group versus 58.3%, 46.1%, and 0% in the NHL, LL, and LPL sub-groups, respectively (p = 0.025); and 4 (8.5%) remained alive on dialysis. Totally 12 (25.5%) of patients died from the complications of the main disease after discontinuation or without need for dialysis, and 3 (6.3%) patients died on dialysis—8.3% versus 50% in the NHL and LPL sub-groups, respectively, and nobody in the HL and CLL subgroups (p = 0.47; Table 4).
Discussion
The incidence of AKI in patients with lymphoma and leukemia is high, reaching 43% in the early studies 43%, and 31.8%–33.2% in more recent studies.10 –12 In our study AKI incidence was even higher—46.5%, most probably because we did not include in our study the patents with myeloid leukemia with the lesser AKI incidence.
The causes of AKI in the patients with HL, NHL/CLL, and LPL are heterogeneous; the most common is pre-renal AKI. 13 Lymphoid infiltration prevalence, according to the early autopsy series, is 63% for CLL and 34% for HL and NHL,14,15 and the incidence of LI in CLL and monoclonal B-cell lymphocytosis was 18% in the study from Mayo Clinic. 16 The incidence of tumor lysis syndrome varies from 42% in the early study 17 to 7.7% in the most recent study 11 ; and we could not find data, indicating the incidence of post-renal AKI. In our cohort, pre-renal AKI was also most common (48.9%); LI constituted 12.7%, which is close to the Mayo Clinic findings and significantly lower than in the early studies, because we diagnosed LI by indicated kidney biopsy, which is obviously not the same as the prevalence, estimated by autopsy. TLS presented in 12.7% of cases, which is in line with the recent findings; the same incidence we found for post-renal AKI. It is worthy to note, that in 12.6% of our patients AKI was associated with the combination of two mechanisms, which reflects the complicated nature of the disease. We could not estimate the statistical significance of the differences between HL, NHL, CLL, and LPL due to the small number of cases with different causes in each sub-group. However, we found that pre-renal AKI was the only type of AKI in the HL setting, probably due to the gastrointestinal complications of chemotherapy, which all patients in this sub-group received before or at admission.
There are some case reports, illustrating kidney pathology of AKI in lymphomas, and demonstrating LI in one case and light chain cast-nephropathy in another.18,19 In the study from China kidney biopsy was performed in 18 out of 20 patients with NHL, only 5 of them presented with AKI, and in these patients pathology showed MPGN, intraglomerular lymphoma and intracapillary monoclonal IgM deposits. 20 Study of 49 patients with CLL and monoclonal B-cell lymphocytosis from Mayo clinic 16 reports renal failure (without distinguishing acute or chronic) in 34 (69%) cases. Pathology findings in the renal failure setting were regarded either as directly/indirectly related to CLL, or as unrelated to CLL. Related to CLL included LI (six cases), thrombotic microangiopathy and MPGN (five cases of each), acute interstitial nephritis (four cases), light chain nephropathy (three cases), and MN and mesangioproliferative glomerulonephritis (one case of each). There were three cases, indirectly related to CLL, all of them infection-related; and five CLL-unrelated cases (diabetic nephropathy, hypertensive nephrosclerosis and obesity-related FSGS). Interestingly, beyond six cases of LI as a primary cause of kidney damage, in nine cases LI co-existed with MPGN, acute interstitial nephritis, MN and other findings. 16 In our previous study of pathology spectrum of kidney damage in 36 patients with HL, NHL, CLL, and LPL, only 9 patients presented with AKI or AKI on the top of CKD, and in these particular cases pathology showed mostly LI or acute tubulonecrosis, associated with severe nephrotic syndrome in FSGS. 21
In our current study, we found preponderance of LI: alone in six cases and in combination with AL amyloidosis, IgAN, and FSGS in three cases. These findings are very similar to the above-mentioned data from Mayo clinic. LI was a direct cause of AKI in all these nine cases. We consider PGNMID, AL amyloidosis, MPGN, MN, FSGS, and IgAN, which totally presented in 13 cases, as NHL/CLL-related and HL-related causes of the underlying CKD, as all these variants of kidney damage were described in the NHL/CLL and HL setting.3,6,16,22–29 PGNMID and AL resulted from direct paraprotein deposition, while MPGN, MN, FSGS, and IgAN represent paraneoplastic spectrum.24,25 AKI on the top of CKD in these 13 patients was pre-renal (9 cases), caused by LI (3 cases) and TLS (1 case). We considered both cases of AA amyloidosis as indirectly related to HL and NHL, as they were associated with chronic infectious treatment complications. In the patient with HL, treated with multiple courses of chemotherapy and radiotherapy it was chronic aspergillus lung infection; and in the patient with NHL with the spinal cord involvement, successfully treated with chemotherapy, but with residual lower paraplegia and pelvic disorder, AA amyloidosis was related to the bedsores and chronic urinary tract infection. AA amyloidosis in NHL and HL are also described in the literature.22,30 In both our cases, AKI was pre-renal. We also found unrelated to lymphoma cause of CKD, like in the Mayo clinic cohort there were FSGS, related to obesity, and diabetic nephropathy. In both cases metabolic disorders developed long before lymphoma, and AKI was pre-renal.
AKI in the patients with lymphoma increases probability of loss of kidney function to 67.9% versus 38.8%, and mortality to 14.7% versus 6.8% in patients without AKI, respectively.11,12 In the patients with CLL and AKI mortality rate was 39%. 13 In our study only 14.8% remained on dialysis, and 31.8% of patients died from the complications of the main disease (6.3% on dialysis and 25.35% dialysis-free), which reflects that our cohort constituted of both lymphomas and CLL patients.
Conclusions
AKI in the patients with leukemia and lymphoma is heterogeneous; in our cohort, it was most commonly caused by volume depletion, and less common by tumor lysis syndrome, specific lymphoid infiltration, and urethral compression by lymphatic nodules. Pre-renal AKI was more typical for HL, while LI and TLS were characteristic for CLL and NHL. In a half of cases, AKI developed on the top of CKD, caused by monoclonal gammopathy-related glomerular diseases (AL, PGNMID), paraneoplastic glomerulopathies (IgAN, FSGS, MN, and MPGN), or concomitant kidney diseases (DN). AKI recovery, even though not complete, occurred in 63.8% of cases, and patient’s survival reached 68%. The complexity of AKI causes in lymphomas and leukemia’s, and a substantial number of cases with the combination of two mechanisms, for instance—pre-renal and renal, demands proactive approach and expanded indications for kidney biopsy. Kidney biopsy, if not contraindicated, in the majority of cases may help to decipher AKI cause and guide the treatment strategy.
Study limitations
This is a single-center study and the sample size is relatively small, therefore the generalizability of the results is limited. The analysis of CKD stages was beyond the scope of this study.
Footnotes
Acknowledgements
We thank doctors Vadim Dobrynin, Sergey Loginov, Eugeny Nikitin, Svetlana Minenko, and Eugeny Shutov, for their help in evaluation, diagnosis, and treatment the patients.
Author contributions
Made substantial contributions to conception and design of the study and performed data analysis and interpretation: Elena Zakharova and Ekaterina Stolyarevich. Performed data acquisition, as well as provided technical support: Tatyana Makarova, Eugenia Leonova, and Elena Nikitina.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval/patient’s consent
Not applicable—this is a retrospective non-interventional study, not including any personal data.
Guarantor
EZ.
Trial registration
Not applicable.
