Abstract
A patient with advanced breast cancer developed acute kidney injury and generalized renal tubular dysfunction manifested as Fanconi syndrome and distal renal tubular acidosis after treatment with palbociclib, a cyclin-dependent kinases 4 and 6 (CDK4/6) inhibitor. A kidney biopsy showed acute tubular injury characterized by dilated tubular lumens, flattened epithelium, and loss of brush borders, with no significant interstitial fibrosis. This case illustrates the importance of close and long-term follow-up of kidney function and electrolyte levels after the initiation of a CDK4/6 inhibitor.
Keywords
Background
Palbociclib, a CDK4/6 inhibitor, prevents cancer growth by blocking the transition from the G1 to the S phase of the cell cycle. Palbociclib has been shown to prolong progression-free and overall survival and is considered the standard care for patients with human epidermal growth factor receptor 2 (HER2)-negative metastatic breast cancers, usually in combination with endocrine therapy. 1 Although uncommon, renal adverse events from palbociclib such as acute kidney injury (AKI), hypokalemia, and hypophosphatemia have been described. 2 Here we present a rare description of nephrotoxicity associated with treatment with palbociclib for breast cancer.
Case description
A 68-year-old woman was admitted to this hospital because of a severe electrolyte imbalance, metabolic acidosis, and kidney function impairment.
Twelve years earlier, the patient suffered from an estrogen receptor (ER) negative, progesterone receptor (PR) positive, and HER2-positive invasive ductal carcinoma of the left breast. She underwent breast-conserving surgery and adjuvant chemotherapy consisting of four cycles of cyclophosphamide, epirubicin, and fluorouracil, followed by four cycles of docetaxel. Thereafter, she had received trastuzumab for 1 year and subsequently endocrine therapy with an aromatase inhibitor for 5 years. The patient remained stable until two and a half years before the current admission, when breast cancer metastasis to lungs and mediastinal and hilar lymph nodes was noted by computed tomography during a routine follow-up visit. Positron electron tomography scan also demonstrated an increase in 18F-FDG uptake in the above-mentioned areas and the fourth lumbar vertebra, suggesting bone metastasis. Palbociclib, a CDK4/6 inhibitor, was then administered in a dose of 125 mg/day for 3 weeks followed by 1 week off, along with fulvestrant. An estimated glomerular filtration rate (eGFR) calculated by the MDRD equation at that time was 76.28 mL/min/1.73 m2. One and a half years before the current admission, treatment with monthly zoledronic acid (ZA) 4 mg was initiated due to progressive bone metastases. Approximately 2 months before the current admission, fatigue and poor appetite developed. One week before the current admission, she presented to the cardiology clinic of this hospital with general weakness and dyspnea on exertion which had developed and worsened during the previous month. The high-sensitivity troponin I level was 34.3 pg/mL (reference range, 0–35). A chest radiography revealed a normal cardiac silhouette. An ECG showed normal sinus rhythm with nonspecific ST-segment changes. U-waves were seen. Because of renal function abnormalities, she was referred to the nephrology department of this hospital and was admitted for further evaluation.
On examination, the temperature was 36.0°C, the heart rate 65 beats per minute, the blood pressure 100/51 mm Hg, the respiratory rate 20 breaths per minute, and the oxygen saturation 99% while the patient was breathing ambient air. She had anemic conjunctivae and decreased muscle power in her lower extremities. The remainder of the physical examination was normal. Medications included palbociclib, fulvestrant, ZA, and rosuvastatin. She took no over-the-counter remedies or supplements. She had lost more than 10 kg of weight in the preceding year. There was no fever, skin rash, joint pain, or spinal cord injuries.
Laboratory test results are shown in Table 1. Her serum level of creatinine was elevated (2.4 mg/dL). Urine chemistries revealed 4+ glucose, 2+ protein, and elevated urine amino acids (18/21). The pH was 6.5. Microscopic examination of the urinary sediment revealed 3–5 red cells per high-power field. Other blood tests revealed hypokalemia (2.3 mmol/L), hypophosphatemia (1.3 mg/dL), hypouricemia (2.2 mg/dL), and metabolic acidosis (HCO3− 14.5 mmol/L). The total protein:creatinine ratio in a spot urine sample was 2.91 mg/mg, which was nearly three-fold that of urinary albumin excretion (1.07 mg/mg). The finding of glycosuria in the absence of hyperglycemia, severe generalized aminoaciduria, increased fractional excretion of phosphate and uric acid in the presence of hypophosphatemia and hypouricemia, metabolic acidosis, and predominantly nonalbumin proteinuria was consistent with a proximal tubular dysfunction and a diagnosis of Fanconi syndrome. 3
Biochemical parameters at the time of presentation.
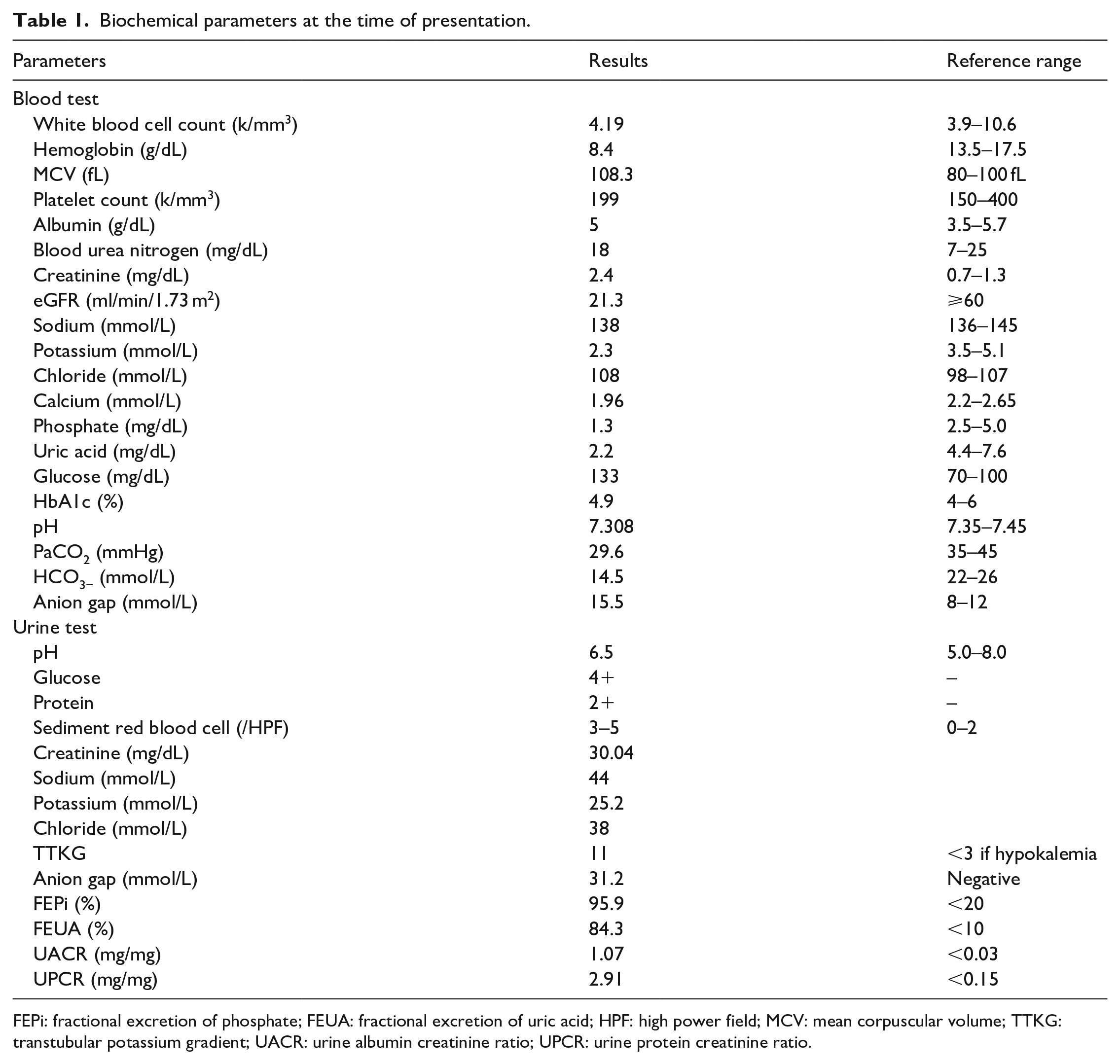
FEPi: fractional excretion of phosphate; FEUA: fractional excretion of uric acid; HPF: high power field; MCV: mean corpuscular volume; TTKG: transtubular potassium gradient; UACR: urine albumin creatinine ratio; UPCR: urine protein creatinine ratio.
Although her oral intake had decreased recently due to poor appetite, this alone was unlikely to explain her significant hypokalemia. A urinary potassium:creatinine ratio of 9.5 (reference range, <2.5) or transtubular potassium gradient (TTKG) of 11 (reference range, <3) in a patient with hypokalemia points to abnormal renal potassium loss. The patient was found to have a mixed normal anion gap (AG) and high AG metabolic acidosis (∆AG/∆HCO3− ratio <1; ∆AG = 15.5 − 12 = 3.5, ∆HCO3− = 24 − 14.5 = 9.5). She did not have diarrhea. The combination of renal potassium loss in association of normal AG metabolic acidosis is strongly supportive of the diagnosis of renal tubular acidosis (RTA), coincident with high AG metabolic acidosis due to underlying chronic kidney disease (CKD). Patients with proximal RTA often have hypokalemia as potassium binds bicarbonate in urine. However, this is usually mild unless urinary acidification by the distal renal tubule is also impaired, resulting in more urinary potassium loss. In this patient, severe hypokalemia raised the possibility of the presence of a distal RTA.
Patients with proximal RTA are able to reclaim filtered bicarbonate fully if the filtered load is low but not when it is normal. 4 Therefore, the urine is acidic (pH <5.3) when serum bicarbonate is below the reabsorptive capacity of the proximal tubule but alkaline (pH ⩾5.3) when it is above this level. In contrast, patients with distal RTA have persistently alkaline urine due to a defect in urinary acidification. Her urine AG was positive (urine Na + K – Cl = 31.2). Her urine pH was 6.5 on admission when her serum bicarbonate was 14.5 mmol/L. These features established a diagnosis of distal RTA and suggested that she could have had a mixed proximal and distal RTA.
Her eGFR was 21.3 mL/min/1.73 m2, which had been normal (>60 mL/min/1.73 m2) before the use of palbociclib. The changes of eGFR from April 2020 over time were shown in Figure 1. What is causing this patient’s AKI on CKD or CKD progression? The ratio of urea nitrogen to creatinine was less than 20 (7.5), the urine sodium level was greater than 20 mmol/L (44 mmol/L), and the fractional excretion of sodium was greater than 1% (2.5%). Renal ultrasound revealed increased cortical echogenicity but no evidence of nephrolithiasis or hydronephrosis. These findings are inconsistent with a prerenal or postrenal cause of AKI but are more suggestive of intrinsic kidney injury. The various causes of intrinsic kidney disease can be grouped into three diagnostic categories: glomerular disease, acute interstitial nephritis, and acute tubular necrosis. The absence of dysmorphic red blood cells in the urine and of eosinophilia are not typical of glomerular disease or with acute interstitial nephritis. In addition, a comprehensive panel for autoimmune profile was negative for antinuclear antibody, anti-GBM antibody, and antineutrophil cytoplasmic antibody. Therefore, it is highly suspicious that the intrinsic kidney injury was secondary to ischemic or toxic tubular insults.
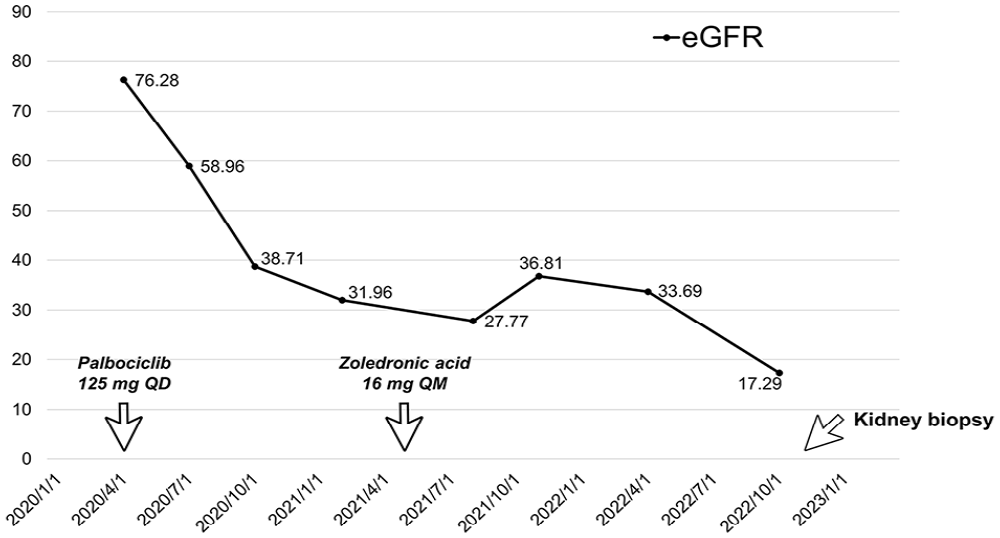
Time course of palbociclib administration and changes of estimated glomerular filtration rate (eGFR).
Percutaneous kidney biopsy was performed. Light mi-croscopy revealed dilated tubular lumens, flattened epithelium, and loss of brush borders, with no significant interstitial fibrosis (Figure 2), which provided a pathologic basis for the patient’s abnormal renal function. Direct immunofluorescence staining was negative for IgG, IgA, IgM, C3, and C1q in the mesangium and the glomerular basement membrane. No electron-dense deposits were observed on electron microscopy. These findings are compatible with acute tubular injury. In this case, the temporal association between starting palbociclib and the onset of kidney disease suggests that this drug was the most likely cause of nephrotoxicity. The dose of palbociclib was reduced to 75 mg/day. Her eGFR did not decline furth-er over the following months. The patient was treated supportively with oral potassium, phosphate, and alkali supplementation and was doing well, although her serum electrolytes remained suboptimal despite regular replacement of the deficient substances.
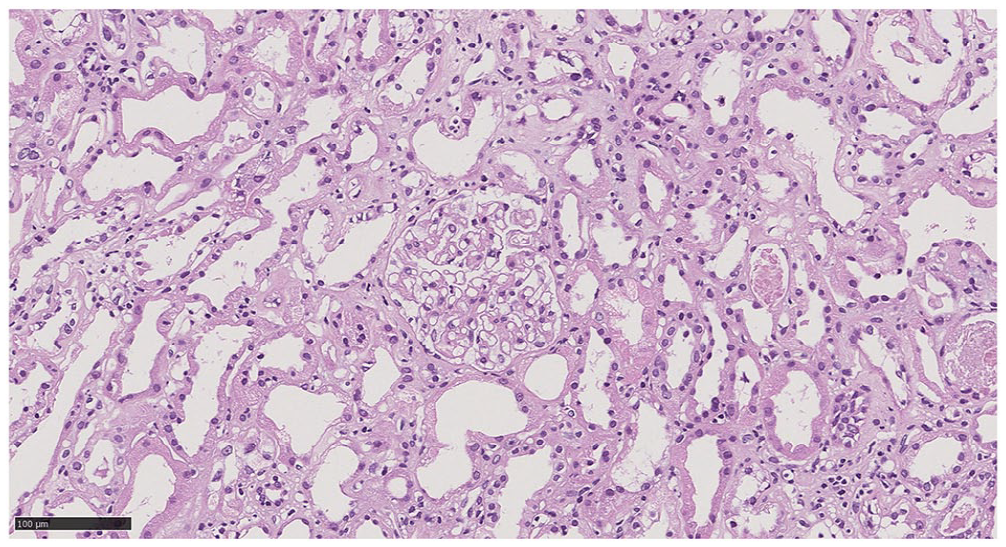
Histopathology of palbociclib-associated acute tubular injury, characterized by dilated tubular lumens, flattened epithelium, and loss of brush borders, scale bar = 100 µm.
Discussion
This 68-year-old woman with advanced breast cancer presented with AKI and generalized renal tubular dysfunction. As there was a significant decrease of eGFR after the administration of palbociclib and evident histopathologic findings on kidney biopsy, her presentation can best be explained by nephrotoxic effects of palbociclib. CDK4/6 inhibitor treatment can be associated with adverse effects such as fatigue, diarrhea, neutropenia, and in rare cases, pneumonitis. 1 However, AKI and combined proximal and distal RTA have not been described previously.
Existing data on nephrotoxicity associated with CDK4/6 inhibitors are limited. Based on the Food and Drug Administration Adverse Event Reporting System, AKI as well as hypokalemia and hypophosphatemia has been reported as a complication following the use of all three agents (palbociclib, abemaciclib, and ribociclib). Recently, Gupta et al. 2 summarized six cases of biopsy-proven AKI that was directly attributed to CDK4/6 inhibitors. The most common histopathologic feature was acute tubular necrosis identical to that of our patient. Given the relative novelty of CDK4/6 inhibitors, the mechanism behind AKI remains to be elucidated, but may be due to the off-target effects of CDK4/6 inhibitors with renal tubule damage from preventing cell-cycle progression. Whether there is a genetic predisposition of certain population resulting in nephrotoxicity or it could be an idiosyncratic reaction requires further studies.
Fanconi syndrome, characterized by urinary loss of glucose, phosphate, uric acid, amino acids, and bicarbonates, usually occurs as a result of proximal renal tubular injury from medications, paraproteins, or heavy metals. 5 Four patients with cancers, who developed AKI and Fanconi syndrome approximately 1 week after treatment with ZA, have recently been reported. 6 Our patient had been receiving ZA for one and a half years before the current presentation. Based on the temporal association and the involvement of the distal renal tubule, Fanconi syndrome secondary to ZA administration seemed less likely in our patient. Nevertheless, high dose ZA infusion for bone metastasis, in the presence of reduced kidney clearance associated with palbociclib, may contribute to the patient’s laboratory abnormalities.
In conclusion, CDK4/6 inhibitors have emerged as promising therapies for advanced breast cancer. Despite that, much remains unknown on nephrotoxicity associated with these drugs. AKI and RTA, although rare, are serious adverse effects following palbociclib administration. Clinicians should be aware of this unusual complication and monitor kidney function and electrolyte levels regularly in patients receiving CDK4/6 inhibitors.
Footnotes
Acknowledgements
NA
Author contributions
SCH conceptualized the study. SYT, CHP, TYL, FPC, and SCH drafted, revised, and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
NA.
Informed consent
The patient granted authorization for unidentified clinical data to be use as research.
Trial registration
NA.
Guarantor
SCH.
