Abstract
Tyrosine kinase inhibitors (TKIs) have improved outcomes for Philadelphia chromosome positive acute lymphoblastic leukemia (ALL) and chronic myeloid leukemia (CML). There has been reports of dasatinib induced proteinuria or nephrotic syndrome. We present a case of a 14-year-old male with high-risk B- ALL who developed nephrotic syndrome due to dasatinib. Proteinuria and nephrotic syndrome should be considered as related adverse effects from dasatinib. This is the first reported case of dasatinib induced nephrotic range proteinuria in a patient with high-risk B-ALL with hyper-eosinophilia.
Keywords
Background
Tyrosine kinase inhibitors have improved outcomes for Philadelphia chromosome positive acute lymphoblastic leukemia (ALL) and chronic myeloid leukemia (CML). Tyrosine kinase inhibitors (TKIs) have several notable adverse effects which can range in severity. Dasatinib, a more potent TKI, may increase the risk of bleeding, regardless of platelet count, and may also cause pleural effusions. 1 Case reports in patients with CML have demonstrated proteinuria and nephrotic syndrome.2–4 Nephrotic syndrome is defined as proteinuria which can be urine protein/creatinine ratbibrio ⩾200 mg/mmol or 3+ protein (⩾300 mg/dL) on urine dipstick, hypoalbuminemia, presence of edema and hyperlipidemia.5,6 Mechanistically, dasatinib’s inhibition of the vascular endothelial growth factor (VEGF) pathway is considered to be the main mechanism promoting the development of proteinuria. 4 Here we report a case of a 14-year-old male with high-risk B-ALL and hypereosinophilia related to his translocation t(5:14)/IGH:IL3 who was treated with dasatinib and subsequently developed nephrotic syndrome likely related to the use of dasatinib.
Case presentation
A 14-year-old male from Nigeria was diagnosed with high-risk B-cell ALL. His rare presentation included hypereosinophilia related to t(5:14); IGH-IL3. Induction therapy included vincristine (1.5 mg/m2), daunorubicin (25 mg/m2), pegaspargase (2500 Int’l Units/m2), and dexamethasone (5 mg/m2) along with intrathecal (IT) cytarabine and methotrexate. Imatinib (400 mg) was added to the induction protocol (DAY15) as his translocation was in close proximity to a neighboring PDGFR beta mutation at 5q31-33. 7 Despite the augmentation he continued to have 13% blasts in his bone marrow. Owing to the need of a stronger TKI, he was switched from imatinib to dasatinib (100 mg) during consolidation.
Gross hematuria developed 9 days after dasatinib treatment along with hypertension requiring antihypertensive therapy. At that time, it was attributed to dasatinib considering he had normal platelet count. It resolved After 14 days (see Figure 1 for trend) of holding dasatinib therapy, a notable improvement in microscopic hematuria was observed. Dasatinib was resumed after 3 weeks for an additional 11 days. Gross hematuria developed again with progressive proteinuria during this second course of dasatinib. His proteinuria (see FFigure 1 for trend) was accompanied with a peak urine protein-to-creatinine ratio (UPC) of 2.41, consistent with nephrotic-range proteinuria. He had serum albumin ranging between 3.0 and 3.5 g/dL in addition with 7%–8% weight gain. “The patient serum creatinine ranged 0.5–0.65 mg/dL. His cystatin C ranged between 0.51–0.62 mg/L.”
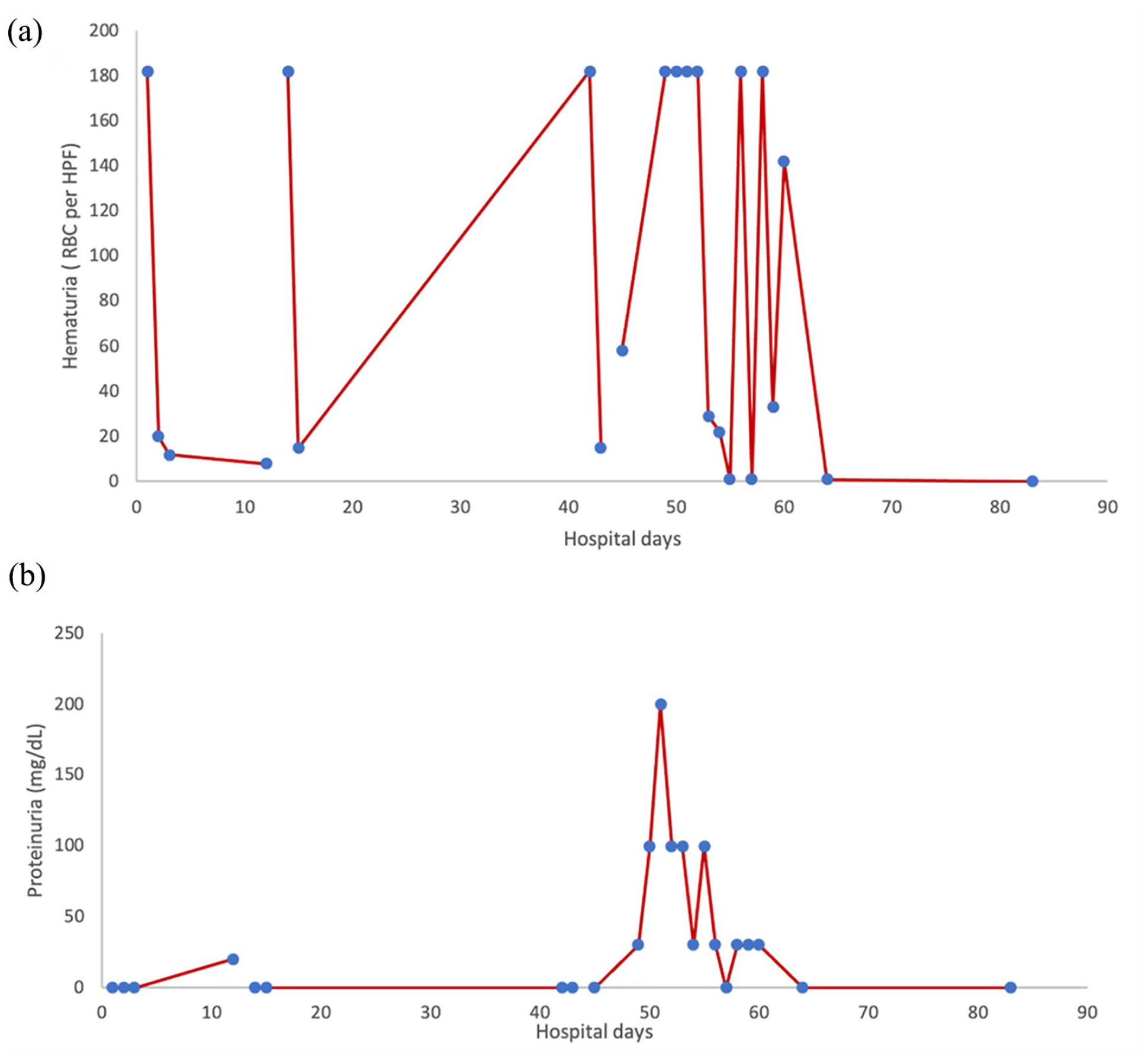
(a) Plot showing range of red blood cells in the urine during TKI therapy. (b) Plot showing range of protein in the urine during TKI therapy.
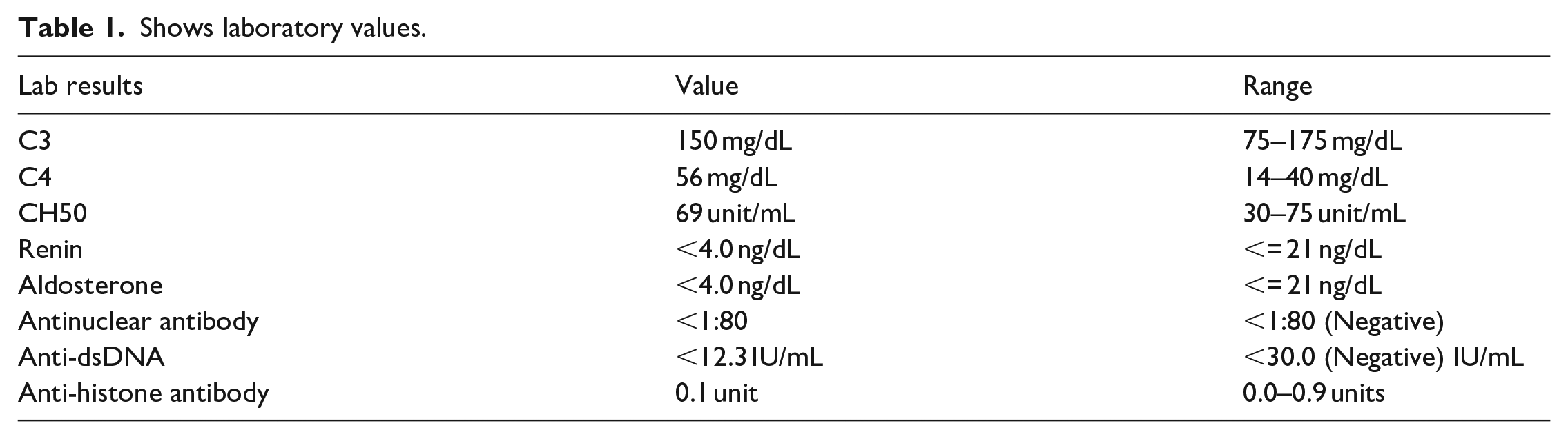
With the development of hypertension, hematuria, and significant proteinuria, further evaluation was sought which included additional reassuring labs (see Table 1) and a kidney biopsy (see Figure 2). A renal ultrasound showed a size discrepancy right kidney (7 cm vs 8.7 cm); however, both were within 2 z-score for his height and weight. His renal biopsy (Figure 2) revealed no gross abnormalities seen on light microscopy including segmental sclerosis nor immune complex deposition on immunofluorescence; however, the glomerular basement membranes/podocytes were effaced and indicative of minimal change disease. 8 His proteinuria and hematuria resolved after stopping Dasatinib. One month post discontinuation his urinalysis was without hematuria and proteinuria and his UPC was <0.2 (normal < 0.21). Given the constellation and timing of symptoms, the most likely cause is nephrotic-range proteinuria secondary to his use of dasatinib.
Shows laboratory values.
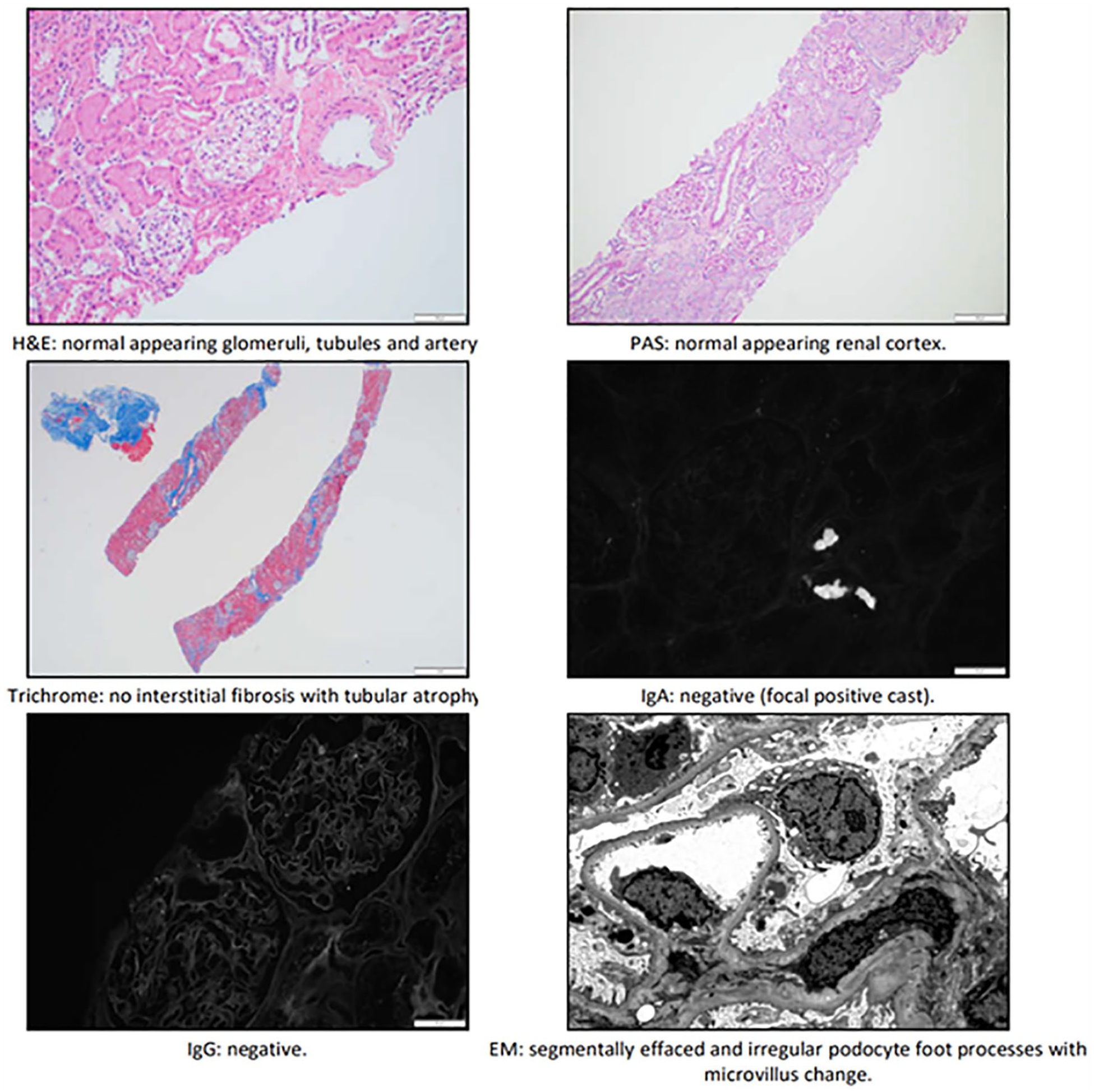
Kidney biopsy specimen.
Our patient did not achieve remission after consolidation. He subsequently received mini hyper CVD (cyclophosphamide, vincristine, dexamethasone) plus immunotherapy 9 followed by an allogenic stem cell transplant and he remains in remission at the time of this report.
Discussion
Minimal change disease (MCD) is the most common cause of nephrotic syndrome occurring in approximately 70%–90% of nephrotic syndrome. 10 The histological feature of MCD includes extensive fusion or effacement of podocyte processes, similar to the case presented here. 10 It is more commonly diagnosed in children than in adolescents. There are several causes that lead to minimal change disease which includes malignancies, medications, infections, or autoimmune disorders. Treatment consists of salt and fluid restriction in addition to steroids. 11
Since the discovery and usage of imatinib, more TKIs have become available, such as dasatinib, nilotinib, and ponatinib. 12 Adverse events have been variable with each TKI. Dasatinib, used in this case, can increase the risk of bleeding despite platelets count and may lead to gross and microscopic hematuria as with our patient. 12 The patient’s proteinuria, given the timing of onset and resolution along with the dosing of dasatinib, has also been cited elsewhere in the literature. 12 Of note, there is no past medical history of sickle cell trait or disease. B-ALL with hyper-eosinophilia can cause several organ dysfunctions related to eosinophilia deposition, but no eosinophilia or eosinophil deposits were noted on his biopsy. It is important to highlight the patient clinically improved with cessation of his TKI. No steroids were used for his nephrotic-range proteinuria.
The incidence of TKI- related nephrotic syndrome has been evaluated in 256 patients with CML. A total of 67 patients received dasatinib were monitored for 35 months. Six patients developed nephrotic range proteinuria which improved after discontinuation or switching to other TKIs. 13 There are a limited number of cases of nephrotic syndrome reported in pediatric leukemia. One report describes a 5-year-old male who had improvement in proteinuria after stopping dasatinib and he had a kidney biopsy showing similar findings to our patient. 14 Another case report detailed a 9-month-old female with CML who developed nephrotic syndrome, however they opted initially to continue dasatinib and attempt a trial of prednisone. She was able to have partial remission of her symptoms but eventually required cessation of dasatinib with subsequent improvement in her proteinuria 15
The pathogenesis of dasatinib-related nephrotic syndrome or nephrotic-range proteinuria is hypothesized to be related to its effect on podocyte and actin cytoskeleton. An in vitro study did not show those effects while using imatinib or VEGF receptor inhibitor such as vandetanib. 9 Such studies are important for us to understand TKI adverse events and in the future we can find ways to predict which patients will be at higher risk for dasatinib related nephrotic syndrome.
Conclusion
In summary, nephrotic-range proteinuria should be considered as likely related adverse effects from dasatinib. Based on findings in the literature and in this patient, it should be noted that nephrotic syndrome due to dasatinib can be reversible. The timeline for resolution of symptoms varies from days to months. Here we present a unique case of dasatinib-induced nephrotic-range proteinuria in a patient with high-risk B-ALL with hyper-eosinophilia. This is the first reported case to our knowledge.
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
None
Informed consent
Consent was obtained or waived by all participants in this study
Trial registration (where applicable)
NA
Guarantor
David McCall, MD
Contributorship
Case identification and eligibility assessment were done by David McCall. Critical review of the manuscript was done by David McCall and Rita Swinford. Initial manuscript writing was by Shaikha Alqahtani and literature review. Craig Authement was the second author and reviewed clinical history and contributed to discussion. All authors performed reviewed and gave approval of the final manuscript.
