Abstract
We report the case of a 63-year-old man with severe hypercalcemia, acute kidney injury, and concomitant diagnosis of germinal center diffuse large B-cell lymphoma (nHL). Hypercalcemia is a common finding in patients with cancer. Multiple mechanisms can lead to hypercalcemia of malignancy, and different malignancies are often associated with a specific mechanism of hypercalcemia. Parathyroid hormone-related protein (PTHrP) and excessive production of 1,25-dihydroxyvitamin D can independently (and rarely, concomitantly) lead to development of hypercalcemia in patients with a variety of malignancies. Literature review of hypercalcemia related with PTHrP and 1,25-dihydroxyvitamin D co-secretion shows that most reported cases are associated with solid tumors and responded to tumor directed therapy. Our case of nHL was associated with increased PTHrP and 1,25-dihydroxyvitamin D making this an unusual presentation.
Background
Hypercalcemia is a common finding in patients with cancer. Multiple mechanisms can lead to hypercalcemia of malignancy, and different malignancies are often associated with a specific mechanism of hypercalcemia. The etiology of hypercalcemia in non-Hodgkin lymphoma (NHL) has been most often attributed to either elevated parathyroid hormone-related protein (PTHrP) or 1,25-dihydroxyvitamin D serum levels. 1
Here, we present an unusual case of a patient with a nHL and hypercalcemia of malignancy in the setting of simultaneous elevations in PTHrP and 1,25-dihydroxyvitamin D levels.
Case description
The patient is a 63-year-old male who presented with constitutional symptoms.
His past medical history was significant for hyperuricemia, benign prostatic hyperplasia, chronic hepatitis C (treated 10-years earlier with sofosbuvir and ribavirin), and H. pylori infection (treated the year before). The patient was on vitamin D (10,000 UI/day) supplementation and allopurinol.
He was referred to an hematology outpatient clinic due to night sweats, unexplained significant weight loss, right sided paresthesia, abdominal pain, generalized muscle weakness, and polyuria over the last 6 months. Physical exam was remarkable for mild dehydration, unaltered mental status, and normal blood pressure.
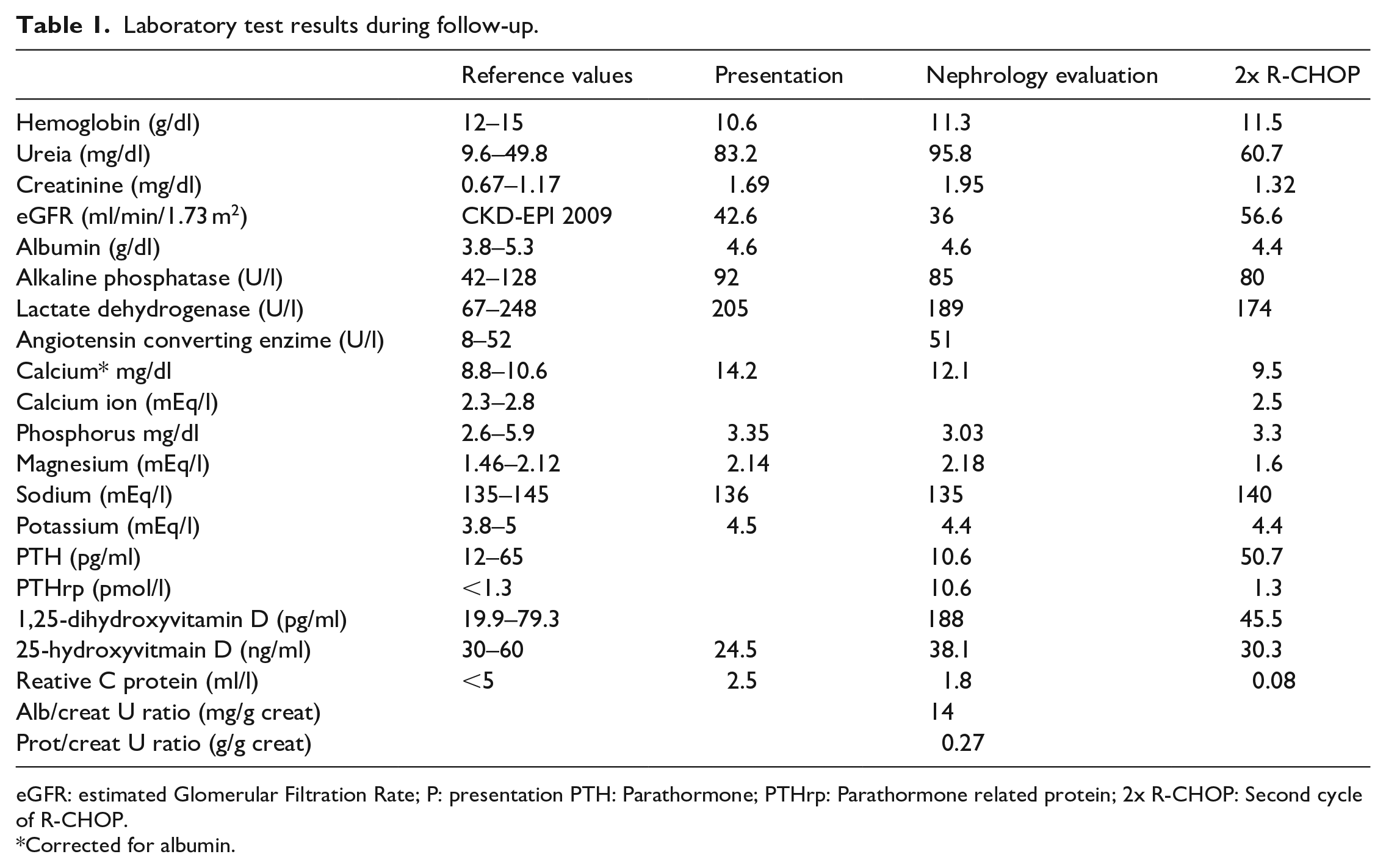
Blood work-up showed mild anemia, severe hypercalcemia (14.2 mg/dl), and stage 1 (KDIGO) acute kidney injury (AKI) with serum creatinine of 1.69 mg/dl, and normal urinalysis. An abdominal ultrasound was performed revealing splenomegaly and renal ultrasound unveiled normal sized kidneys without obstruction.
Despite reinforced hydration and vitamin D suspension, AKI and moderate hypercalcemia persisted. A nephrology consultation was requested at this time. PTH levels were appropriately low and vitamin D and phosphorus levels were normal (Table 1). There was no M-spike in serum protein electrophoresis, immunofixation and skeletal surveys were normal.
Laboratory test results during follow-up.
eGFR: estimated Glomerular Filtration Rate; P: presentation PTH: Parathormone; PTHrp: Parathormone related protein; 2x R-CHOP: Second cycle of R-CHOP.
Corrected for albumin.
The patient was acutely started on fluid therapy and prednisolone 20 mg/day, with partial favorable response (Ca 12.1 mg/dl). No bisphosphonates were given.
CT-scan showed a solid heterogeneous splenic mass, with 14 cm and heterogeneous contrast enhancing. A splenic biopsy was performed which yielded the diagnosis of germinal center diffuse large B cell lymphoma (nHL).
Upon hematologic diagnosis (22 days later), chemotherapy with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) was initiated. At this time, serum creatinine and calcium levels presented further improvement, and calcium levels normalized with a second cycle of chemotherapy (Ca 9.5 mg/dl).
PTHrP and 1,25-dihydroxyvitamin D levels (collected prior to chemotherapy institution, with results only available afterward), were clearly above the normal range [10.6 pmol/l (normal value <1.3) and 188 pg/ml (normal value: 19.9–79.3), respectively]. Both normalized with chemotherapy institution.
Discussion
Our case highlights the importance of a thorough diagnostic work-up of hypercalcemia in patients with underlying malignancy for successful treatment of this challenging condition.
Approximately 90% of patients with hypercalcemia have either hyperparathyroidism or cancer. 2
When the nephrology consultation was requested, hypercalcemia confirmation (calcium corrected for albumin), measurement of ionized calcium levels and primary hyperparathyroidism work-up was performed. Patient’s low parathyroid hormone level made the diagnosis of hyperparathyroidism unlikely.
Elevated 1,25-dihydroxyvitamin D could point to vitamin D intoxication. So, the next step was to evaluate the patient’s 25-hydroxyvitamin D levels. The measurement was below the normal range. Even so over the counter vitamin D consumption was suspended on initial evaluation.
In short, our patient had an elevated corrected calcium level, low PTH level, and presented with B-symptoms. Hypercalcemia in patients with hematologic malignancies has been well reported in the literature and occurs in up to 20 to 30% of cancer patients.3,4
The next approach was to consider other mechanisms that would explain the hypercalcemia, like excessive bone resorption or lytic metastases. Serum protein electrophoresis, immunofixation and skeletal surveys were normal, so multiple myeloma and osteoclastic metastasis of solid tumors were excluded.
Our patient presented symptomatic hypercalcemia in association with both raised PTHrP and 1,25-dihydroxyvitamin D consistent with hypercalcemia of malignancy. These can independently lead to development of hypercalcemia in a variety of malignancies, particularly in nHL.
PTHrP shares the same N-terminal end as PTH (the first 13 amino acids are almost identical), binds to the same PTH-1 receptor, and as a result, mimics most of its actions, particularly increasing bone turnover, releasing calcium from stores, stimulating distal tubular calcium reabsorption, and inhibiting proximal tubular phosphate absorption. Structural divergence after the first 13 amino acids of the molecule accounts for their immunologic distinctiveness, with different effects on 1,25-dihydroxyvitamin D. Serum 1,25-dihydroxyvitamin D levels are usually suppressed. PTHrP secretion is commonly seen in various solid tumors (e.g. cancer of the head/neck/lung/kidney) but can also be seen in hematological malignancies such as nHL.1,5
The mechanism by which 1,25-dihydroxyvitamin D is produced in malignancy could be related to 1,25-dihydroxyvitamin D secretion by the cancer cells.
1,25-dihydroxyvitamin D will increase calcium and phosphorus intestinal absorption, leading to negative feedback on the normal parathyroid glands and decreased PTH levels. This is the cause of many cases of hypercalcemia in Hodgkin lymphoma and approximately one-third of cases in non-Hodgkin lymphoma.1,6 It should also be noted that 1,25-dihydroxyvitamin D induced hypercalcemia is also seen in association with chronic granulomatous diseases, such as sarcoidosis and tuberculosis. 7
In patients with HM associated with production of PTHrP, 1,25-dihydroxyvitamin D concentrations are typically normal or suppressed possibly due to direct suppression of 1-α- hydroxylase activity and/or PTH by serum calcium. 8
Likewise, when HM is associated with 1,25-dihydroxyvitamin D, PTHrP is typically suppressed.
The simultaneous production of both by a nHL makes this case unusual.
Histologic confirmation with 1-alfa-hydroxylase and 1,25-dihydroxyvitamin D staining of the spleen biopsy would have been of interest. PTHrP and 1,25-dihydroxyvitamin D normalization with R-CHOP treatment supports that both were produced by the tumor.
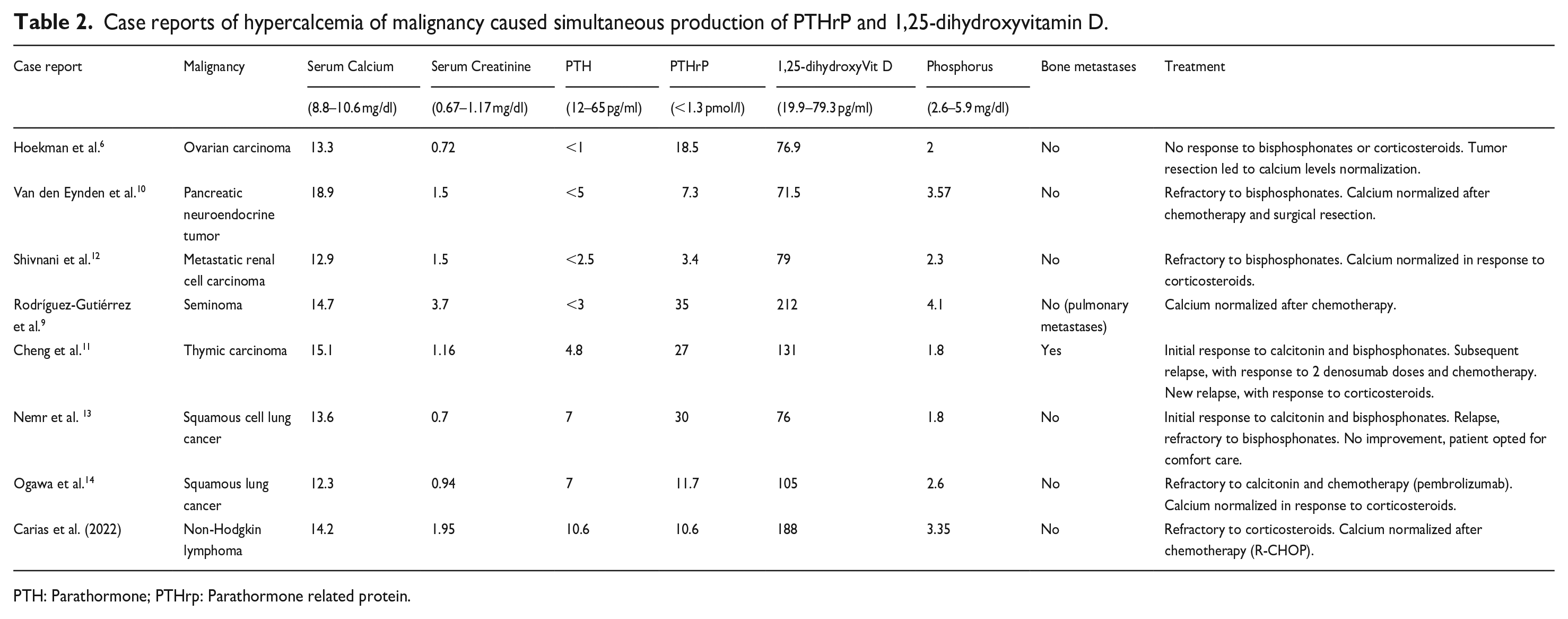
We reviewed the literature for hypercalcemia with PTHrP and 1,25-dihydroxyvitamin D co-secretion and found the following case reports: ovarian carcinoma, 6 seminoma, 9 pancreatic neuroendocrine tumor, 10 thymic carcinoma, 11 metastatic renal cell carcinoma, 12 squamous cell lung cancer, 13 non-small cell lung cancer, 14 which are summarized in Table 2.
Case reports of hypercalcemia of malignancy caused simultaneous production of PTHrP and 1,25-dihydroxyvitamin D.
PTH: Parathormone; PTHrp: Parathormone related protein.
Although the sample was small, precluding solid conclusions, most cases of HM cases related with PTHrP and 1,25-dihydroxyvitamin D co-secretion were associated with solid malignancies. Mean (±standard deviation) of PTHrP and 1,25-dihydroxyvitamin D were 17.9 pmol/l (±11.6) and 117.4 pg/ml (±55.0), respectively, and our case was in accordance with these values. Only one case presented with associated bone metastasis (which might have been a confounding etiologic factor). The response to bisphosphonates was either scarce or temporary.
All patients but one, 12 presented decreased PTHrP and 1,25-dihydroxyvitamin D in parallel to calcium normalization.
In conclusion, HM has been well described in both solid and hematologic malignancies and represents the most common cause of elevated serum calcium in hospitalized patients.
Increased concentrations of plasma PTHrP or 1,25-dihydroxyvitamin D in nHL are already recognized causes of severe hypercalcemia, however, simultaneous production of both by a nHL makes this case unusual.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
