Abstract
Background:
With the expanding indications and thus broader use of immune checkpoint inhibitors, clinicians are faced with a new kind of immune-related adverse events. Because of their immune modulating effects, immune checkpoint inhibitors have the potential to worsen autoimmunity in general. Paraneoplastic syndromes can be caused by tumor-induced autoimmune mechanisms. The use of immune-activating substances such as checkpoint inhibitors might lead to exacerbation of paraneoplastic syndromes causing premature discontinuation of the immunotherapy.
Case presentation:
We report on a 64-year-old patient with metastasized renal cell carcinoma who developed acute kidney failure after cytoreductive nephrectomy. Work-up revealed a paraneoplastic syndrome that caused tubulointerstitial nephritis (TIN). Glucocorticoid therapy successfully reversed the acute kidney injury. However, adjuvant therapy with Nivolumab provoked a flare-up of the paraneoplastic syndrome on two occasions, eventually leading to a treatment discontinuation.
Conclusions:
Many cases of Nivolumab-induced TIN have been described lately. However, our case demonstrates therapy failure due to a flare-up of a pre-existing paraneoplastic syndrome of the renal cancer. Against this background, it can only be speculated that some of the TIN cases discussed in prior literature might also have been flare-ups of subclinical autoimmunity.
Keywords
Introduction
Paraneoplastic syndromes are observed up to 40% in renal cell carcinoma and are caused by cytokine- or hormone-like biologically active agents provoking constitutional symptoms (i.e. fever, weight loss, anemia, or fatigue) or specific derangements, such as hypercalcemia, polycythemia, hypertension, and liver dysfunction. 1 The exact mechanism remains unclear, but tumor-induced activation of the immune system leading to autoimmunity has been described. 2
Anti-tumor therapy is the only causative and effective treatment option for paraneoplastic syndromes. The recent development of immune checkpoint inhibitors (CPI) offers new perspectives for the treatment of metastatic renal cell carcinoma (mRCC), and CPI such as nivolumab, ipilimumab, and pembrolizumab are approved in this situation. 3 While the use of CPI has significantly improved overall survival, the activation of the immune system bears the risk of causing immune-related adverse events. The impact of CPI-induced immune system activation on paraneoplastic syndromes is not clear yet, which is problematic, given this cancer’s high autoimmune potential.
Case report
A 64-year-old white male presented with new onset of left-sided flank pain radiating to the inguinal and testicular region. He had never experienced this pain before, but had suffered from fatigue, loss of appetite, and weight loss over the recent weeks. He was afebrile, denied night sweats, macrohematuria, or dysuria. He did not take any regular medication. Physical examination was without pathological findings. Laboratory findings showed an elevated C-reactive protein (76.6 mg/l) with normal leukocyte count as well as a normocytic anemia (hemoglobin: 108 g/l), mild thrombocytosis (359,000/l), mild hyponatremia (131 mmol/l), and hypercalcemia (2.59 mmol/l). The creatinine level was 0.99 mg/dl. His on past history regarding use of potential nephrotoxic drugs was negative. A CT scan showed a left kidney tumor of 11 cm × 12 cm in diameter infiltrating the renal fascia with regional nodal, ipsilateral adrenal and multiple pulmonary metastases (Figure 1(a)). According to IMDC risk classification the patient had poor risk mRCC. A cytoreductive left nephrectomy in line with the interdisciplinary tumor board recommendation was performed (Figure 1(b)). The immediate postoperative course was uneventful, and the patient was discharged on the fifth postoperative day (postoperative creatinine: 1.35 mg/dl, eGFR: 55 ml/min). Histological work-up revealed a clear cell renal cell carcinoma (pT3a R1).
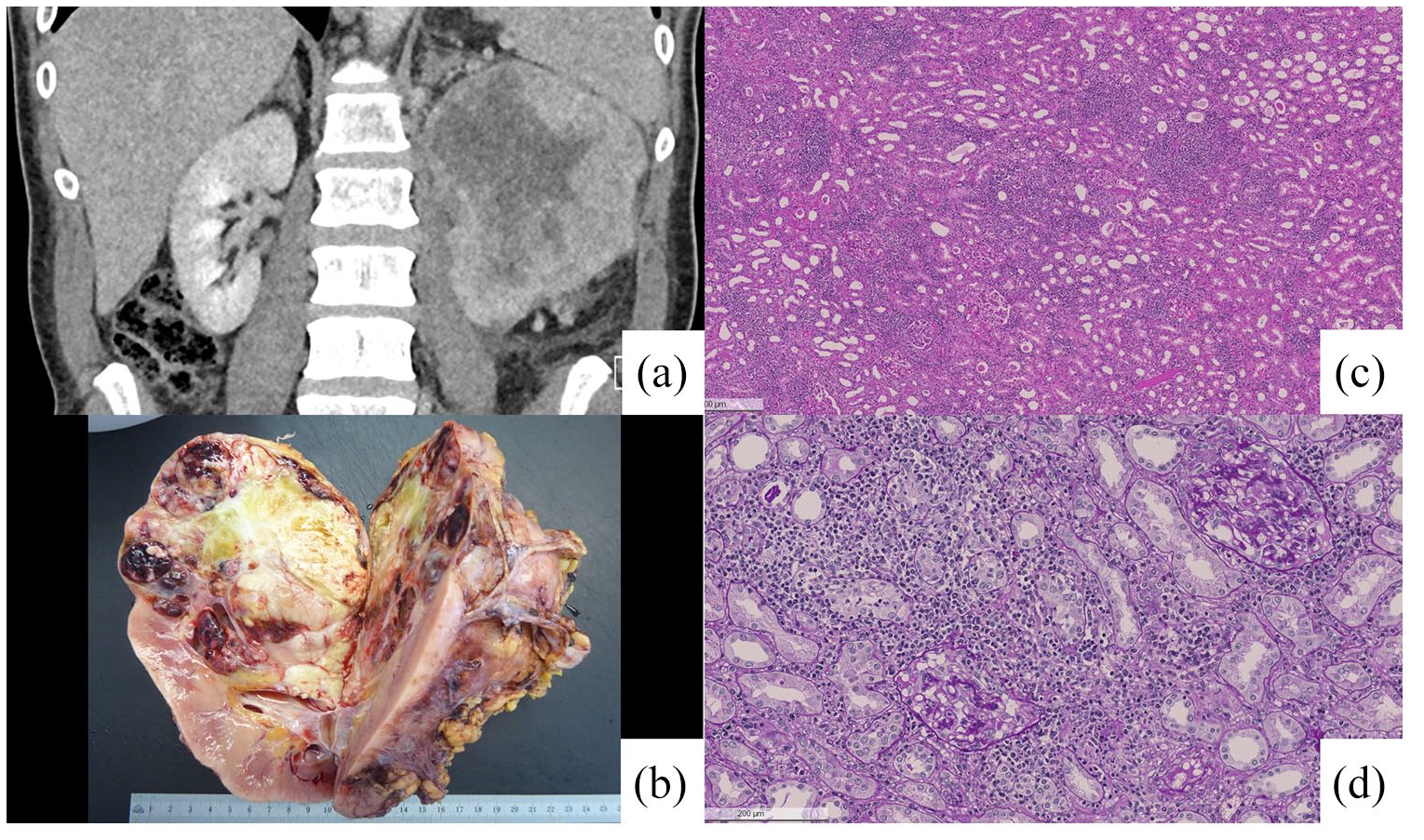
(a) Coronal view of the CT-scan with venous contrast upon initial presentation showing the renal tumor (9 cm × 11 cm × 12 cm) of the upper pole of the left kidney with infiltration of the gerota fascia and ipsilateral adrenal metastasis, (b) Macroscopic examination of the nephrectomy specimen showing an inhomogenous tumor of the upper pole next to the renal pelvis with large areas of necrosis, (c) Kidney tissue with tubulointerstitial nephritis and infiltrates mostly consisting of lymphocytes and plasma cells (H&E stain), and (d) Detail of tubulointerstitial nephritis with interstitial infiltrates and tubulitis (periodic acid-Schiff stain).
On the 14th postoperative day the patient presented to the urological outpatient clinic with an acute kidney failure (creatinine: 3.85 mg/dl). After exclusion of a postrenal cause, the patient was transferred to internal medicine for further work-up. An urea excretion fraction of 65% made a prerenal cause improbable, and renal perfusion abnormalities were ruled out through duplex sonography. The past and present history regarding use of potential nephrotoxic drugs was negative. He only took paracetamol postoperatively. Sterile leukocyturia and low molecular weight proteinuria implied TIN. It was decided against a biopsy of the single remaining kidney. However, further histological work-up of the nephrectomy specimen demonstrated an acute to subacute TIN with interstitial infiltration of mostly lymphocytes and plasma cells, macrophages, a few neutrophils and eosinophils, tubulitis with lymphocytes in tubular epithelium, edema and focal beginning interstitial fibrosis. No granulomas were found (Figure 1(c) and (d)). Kidney function improved quickly under high dose that is, methylprednisolone therapy (125 mg/day). The patient was discharged on oral prednisolone with swift tapering to 10 mg per day making the patient suitable for an immunotherapy.
Rapidly after administration of a single dose of the PD1-inhibitor nivolumab (240 mg) creatinine levels rose again indicating a flare-up of TIN and supporting the postulated autoimmune paraneoplastic syndrome (Figure 2). Prednisolone treatment was increased again (100 mg per day), and the renal function improved. Thereafter, treatment with nivolumab was switched to sunitinib. Unfortunately, the last restaging CT scan after the second cycle of sunitinib showed progressive disease. Consequently, the therapy was changed to cabozantinib. Despite clinical improvement imaging revealed further rapid local disease progression with extension into the abdominal wall. To prevent abdominal wall perforation, nivolumab was readministered. Two weeks later the patient was readmitted with a recurrence of acute renal insufficiency. Unfortunately, the patient’s condition deteriorated further, and he died due to the very extensive and progressive metastatic tumor only 5 months after the initial presentation.
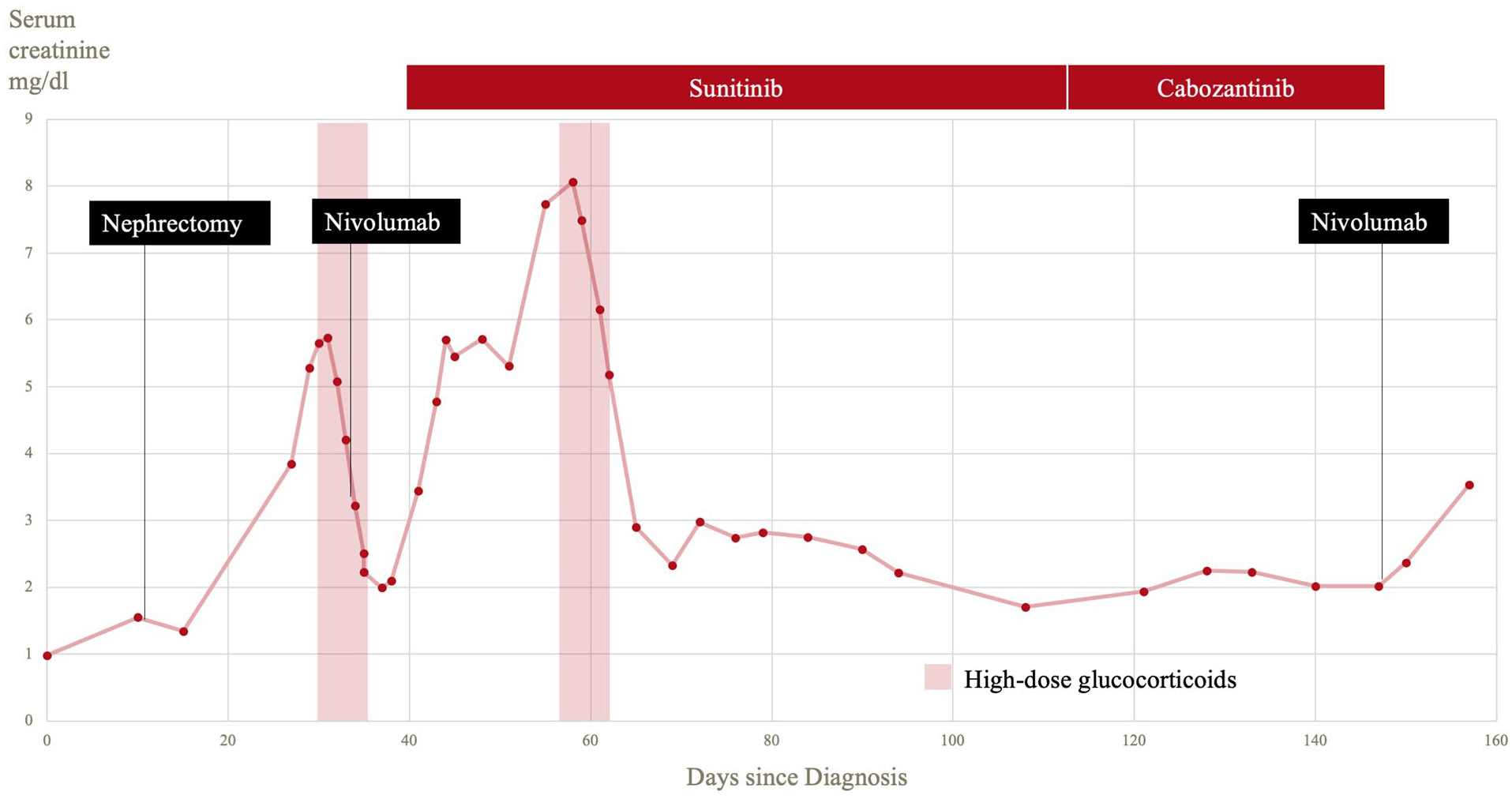
Plot of the renal function during the course of the disease. Day 0 marks the initial presentation in the emergency room. The two episodes of acute renal failure have been treated with high dose glucocorticoids. The initial dosages were 125 mg respectively 100 mg (for 3 days) and were gradually reduced.
Discussion
With the growing indications and prescriptions of the highly effective immune checkpoint inhibitors, clinicians are faced with a wide variation of novel adverse events. Those immune-related adverse events differ significantly from the adverse effects seen with conventional chemotherapeutics in terms of appearance and management. 4 While most immune-related adverse events remain mild, up to 10% of the patients can develop severe to life-threatening conditions. Rash, colitis and hepatitis have been found most commonly. 5 Drug trials have shown that 2% of the reported side effects caused by immune checkpoint inhibitors were acute kidney injury (AKI). Acute TIN was the most frequent cause for AKI, but also glomerular involvement and electrolyte disturbances were observed. 6 In the recent published literature it is discussed that acute TIN after CPI may portend a higher likelihood of response through antigenic overlap between tumor cells and renal tubular cells. 7
Non-infectious TIN is usually described as being caused by drug hypersensitivity, autoimmune diseases or neoplasm. Drugs mostly associated with TIN are proton pump inhibitors, non-steroidal anti-inflammatory drugs, certain antibiotics, anticonvulsants, and diuretics. 8
Observations of mutual influences of cancers and the kidney function were well known prior to the development of immune checkpoint inhibitors. 9 The first case reports date back almost 100 years ago.10,11 Looking for immune-mediated nephropathies in renal cell carcinoma patients, Magyarlaki et al. 12 examined nephrectomy specimens histologically. Coincidently, approximately one third of those specimens showed TIN. It is unfortunately unclear, if these patients were exposed to well-known triggers of TIN because the corresponding patient characteristics were only specified as asymptomatic. Recent published studies also analyzed non-neoplastic renal tissue in kidney cancer patients, while some studies did not report any tubulointerstitial nephritis.13,14 Salvatore et al. 15 and Bijol et al. 16 (Bijol) could also demonstrate tubulo interstitial nephritis in nephrectomy specimen for renal cell carcinoma. In retrospective, the TIN in our case was present before the nephrectomy, since it was histologically demonstrated in the nephrectomy specimen. In the history of our patient prior to the nephrectomy, no potential trigger for TIN apart from the cancer itself could be found.
Whether or not kidney biopsy should be obtained prior to corticosteroid therapy is subject to current debate. 17 Seeking biopsy can prevent unnecessary empirical corticosteroid therapy or potentially ill-advised discontinuation of immune checkpoint inhibitors which could ultimately lead to inferior outcome. 18 However, some studies showed no clear benefit of a prior kidney biopsy in comparison to empirical corticosteroid therapy in cases with a clinical high suspicion for TIN. Consequently, biopsy should only be pursued if there are enough reasons to suggest that the result would make a change of management probable and should carefully be evaluated in patients with a solitary kidney. 18
During the postoperative phase the TIN flared up in our patient for unknown reasons and lead to AKI, which was successfully managed with glucocorticoids. It is possible, that the surgery as an inflammatory stimulus itself was responsible for the flare of the pre-existing TIN. After improvement of the AKI one single dose of the PD-1 inhibitor nivolumab exacerbated the preexisting paraneoplastic TIN and could again be successfully treated with glucocorticoids. The fact that this reaction occurred very rapidly after a single dose of nivolumab points to a recall reaction in the context of hypothesized pre-existing autoimmunity. The same reaction occurred as nivolumab was used as a salvage therapy later in the course of the disease, which is in line with recent literature: a case cohort study showed that three of four patients who were re-treated with an immune checkpoint inhibitor experienced a flare-up of the AKI. 7
In the past, TIN has been found during histological analysis of nephrectomy specimens with renal cell carcinoma. However, to the best of our knowledge this is the first case of clinically relevant paraneoplastic TIN causing acute renal failure. TIN has to be noted as another possible autoimmune disease which can be caused by a renal cell carcinoma. Our case also shows the potential harm using immune checkpoint inhibitors in a patient suffering from autoimmunity beforehand. Hence, we conclude that it is essential to check patients for autoimmune conditions before the use of an immune checkpoint inhibitor, since severe aggravations may occur, potentially causing organ failure. These risks have to be carefully evaluated and weighed against the potential benefits of the treatment. Especially a potential re-challenge has to be thoroughly assessed.
Footnotes
Acknowledgements
The authors wish to thank Ulrike Gruber Mösenbacher, Department of Pathology, Cantonal Hospital Graubuenden, Chur, Switzerland, for the histological diagnosis of clear cell renal cell carcinoma and recording the macroscopic figure.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support from Cantonal Hospital Graubuenden.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Guarantor
Thomas Fehr, Department of Internal Medicine, Cantonal Hospital Graubuenden, Loestrasse 170, CH-7000 Chur, Switzerland. Tel. +41 81 256 63 05
E-mail address:
Authors’ contributions
T.P. Scherer and K. Saba drafted the manuscript. T.P. Scherer designed the figures. R. Strebel, R. Cathomas, A. Gaspert and T. Fehr were the responsible physicians and gave critical feedback regarding their specialities. A. Gaspert performed the histological analysis. Th. Fehr supervised the case report. All authors have read and approved of the final manuscript.
