Abstract
Over the past decade, opioid use has been at the forefront of a public health crisis throughout the United States. In response to the tremendous negative societal, personal, and economic impacts that the growing opioid crisis has caused, several governmental agencies began to respond. These efforts include declaration of a nationwide public health emergency, increased public health surveillance of the epidemic, research support for pain and addiction, and increased access to overdose-reversing drugs such as naloxone. Naloxone access, in particular, has become a priority. In the United States, pharmacists have had the opportunity to play a crucial role in promoting access to naloxone. Since initial approval by the Food and Drug Administration (FDA) in 1971 as an antidote to opioid agonist overdose, naloxone access has evolved significantly. Today many states have authorized standing orders for naloxone, allowing it to be dispensed by pharmacists without a patient-specific prescription, and all 50 states and the District of Columbia allow medical providers to prescribe take-home naloxone to at-risk patients. While the opioid epidemic itself remains a contentious topic of political, ethical, and medical debate, it is widely acknowledged that mitigation strategies that could lessen morbidity and mortality are essential. Improved access to naloxone is one such strategy which remains at the forefront during this public health crisis.
Keywords
Over the past decade, opioid use has been at the forefront of a public health crisis throughout the United States (US). The US Centers for Disease Control and Prevention (CDC) reported opioid overdose deaths increased from 4030 to 16,917 between 1999 and 2011. 1 This increase was accompanied by a corresponding uptick in opioid prescribing. In fact, increases in opioid-prescribing paralleled increases in morbidity, mortality, opioid-related overdose death rates, and substance-use-disorder-treatment admissions from 2006 to 2012. 2 In response to the tremendous negative societal, personal, and economic impacts that this growing public health crisis (also called the “opioid crisis” or “opioid epidemic”) caused, several governmental agencies began to respond.
In 2016, the CDC developed and published a controversial set of guidelines on the prescribing of opioids for chronic pain, which, in part, included limits of morphine-equivalent daily doses (MEDD) to ⩽90 mg/day for anyone on long-term opioids. 3 Such a cap could be more harmful than good given the widespread lack of understanding for the variation in potency, receptor-binding, affinity, physical tolerance, and pharmacokinetic properties among different opioids. Furthermore, there are inconsistencies among MEDD calculators which limit reliable conversion from one opioid to another. 4 In addition to concerns related to MEDD, the conclusions drawn in the CDC guidelines are largely based on poor quality evidence and, it is noted that no pharmacists were included in the author panel.5,6 Perhaps, most concerning is the fact that these misguided CDC recommendations have now begun to be incorporated into state-specific laws and insurance policies.
The CDC has not been the only US agency to respond to the “opioid epidemic.” In 2017, the acting Secretary, Dr Eric Hargan, of the US Department of Health and Human Services (HHS) declared a nationwide public health emergency regarding the opioid crisis and initiated a 5-point strategy to combat the crisis.7,8 These included improving access to treatment and recovery services, promoting use of overdose-reversing drugs (including naloxone), increasing public health surveillance of the epidemic, providing support for research on pain and addiction, and advancing better practices for pain management. 8
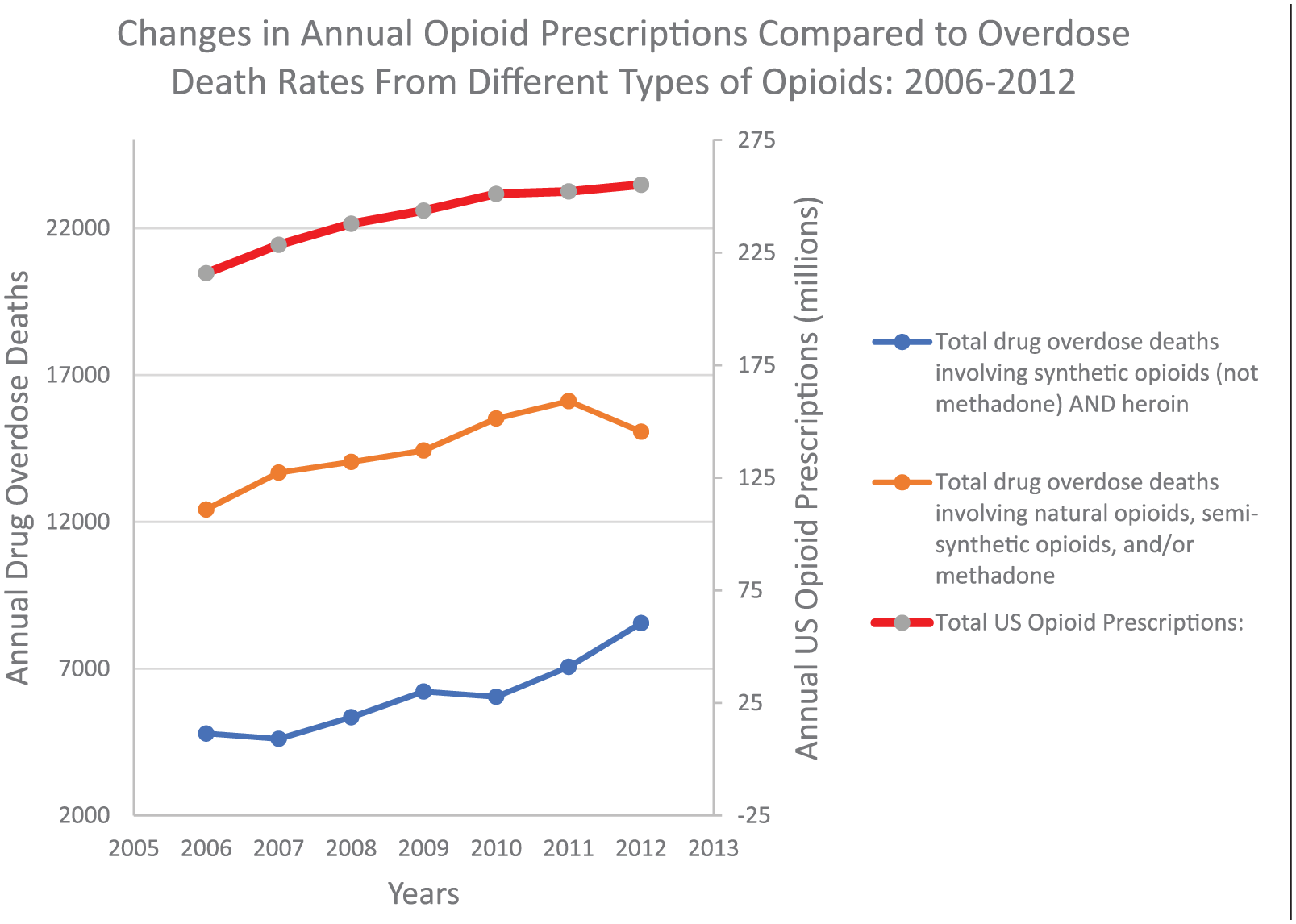
Evaluation of more recent data trends has shown overall reductions in national opioid prescribing rates from 2012 to 2017, which is noted to have begun prior to publication of the CDC guidelines, and, notwithstanding, an overall leveling off of overdose deaths involving natural opioids, semi-synthetic opioids, and methadone (all combined representing the majority of prescription opioids), as shown in Figure 1.9–11 However, despite the efforts of the Department of HHS, overdose death rates involving any opioid continue to rise, with significant increases of deaths involving non-methadone synthetic opioids (mostly from illicit fentanyl analogs) and heroin, as shown in Figure 2.9–11 In response, Food and Drug Administration (FDA) commissioner Dr Scott Gottlieb has made several statements about increasing access to naloxone including outlining ways to reduce cost of different naloxone formulations, and expedited naloxone generic availability.12,13

In the United States, pharmacists have also had the opportunity to play a crucial role in promoting access to naloxone. Since initial approval by the FDA in 1971 as an antidote to opioid agonist overdose, naloxone access has evolved significantly. Initially, naloxone was limited to injection formulation and administration by healthcare personnel only. Today, however, many states have authorized standing orders for naloxone allowing it to be dispensed by pharmacists without a patient-specific prescription. 14 Even in those without such legislation, pharmacists are still in a unique position to not only properly educate patients about naloxone but also identify patients who may need naloxone and assist patients with obtaining prescriptions for it, as well. Education is a crucial piece of naloxone dispensing, particularly with the rise in overdoses associated with illicit fentanyl and derivatives. 15
As illicit fentanyl analogs become more ubiquitous, it has become clear that higher and/or repeated doses of naloxone are needed to reverse more potent opioids. 16 In fact, it is for this reason that Kaléo recently changed the strength of their auto-injector from 0.4 to 2 mg per dose. Pharmacists can further engage in facilitating increased access to naloxone by utilizing risk stratification tools such as the risk index for serious prescription opioid induced respiratory depression or overdose (RIOSORD) to determine which patients are most likely to benefit from naloxone. 17 Currently, no standardized guidelines exist stating which patients should and should not receive naloxone. Therefore, clinical judgment and risk stratification tools drive the discussion.
When naloxone was first approved by the FDA, it was approved for intravenous (IV) administration only. This presented a challenge for rapid reversal in those with poor IV access (a commonality among IV drug abusers) or for those with significant delays in achieving IV access, although intramuscular administration was an option. Subsequently, a mucosal atomizer was used off-label (by attaching the atomizer to the syringe of naloxone) to create “make-shift-intranasal” naloxone kits at a dose of 0.5 mg/0.5 mL. Unfortunately, the reliability to deliver consistent doses of this medication was questionable at best, given difficulty in accurately attaching the device itself and intensive training involved and lack of compliance data to support use. 18
Since that time, two additional naloxone products have been approved in the United States. In April, 2014, Evzio® was approved as a handheld, single-use auto-injector designed to deliver 0.4 mg of naloxone intramuscularly. 19 This product has auditory prompts for emergency responder to follow. Recently, the available concentration was changed to 2 mg/0.4 mL in part to compensate for high potency illicit fentanyl analogs requiring higher naloxone doses. In November, 2015, Narcan® was approved as a naloxone nasal spray, offering significant improvements over the off-label naloxone syringe attached to a mucosal atomizer previously used for emergency overdose reversal. 20 Narcan is designed to deliver 4 mg of naloxone intranasally, although about one-tenth is absorbed transmucosally, which makes this an approximate overall equivalent dose to the original Evzio formulation. In 2017, a new Narcan formulation containing 2 mg/0.4 mL was approved by the FDA for chronic opioid users at high risk of severe opioid withdrawal. At time of publication, commercial availability of the lesser concentrated dose is still lacking.
The FDA has continued to make efforts to increase naloxone access. Most recently, an unprecedented step was taken to transition naloxone availability from prescription-only status to over-the-counter (OTC), allowing consumers to purchase the medication devoid of counseling or interaction with a healthcare provider. 13 To this end, the FDA, for the first time, has developed and tested a drug facts label (DFL) to support development of an OTC naloxone device. 13 They also conducted the CONFER study to assess consumer comprehension of the DFL, which was designed to support the use of naloxone in the OTC setting. 21 The CONFER study concluded that the DFL was acceptable as written with adjustments to be made specific to each delivery system marketed. The DFL performed well in facilitating comprehension of the steps required to evaluate an overdose victim and administer naloxone. The only shortcoming identified was in comprehension of the need to call emergency services immediately. Despite this, the DFL has been deemed adequate as written. 18
All 50 states and the District of Columbia currently allow medical providers to prescribe take-home naloxone to patients at risk of an opioid overdose. 14 As of 1 July 2017, all 50 states plus the District of Columbia also had additional naloxone access laws allowing for expansion of the distribution of naloxone beyond the traditional prescription format. 22 Importantly, only six states have laws that provide civil and criminal immunity for prescribers, dispensers, lay administrators, and lay distribution and possession. 23
The majority of these expansion laws broaden naloxone access through the use of third-party prescriptions and non-patient-specific prescriptions. A third-party prescription is one that is issued to an individual who is not at risk of overdose, intended for use on someone else. 14 Prescribers can write these prescriptions for family members, friends, or professionals who work with at-risk individuals. Third-party prescription of naloxone is permitted in all states except for Kansas and Minnesota. 23 Some states have extra requirements such as training or receipt of instructional materials before third parties can acquire naloxone. 14
Non-patient-specific prescriptions allow anyone at risk of an opioid overdose and anyone who may be able to assist in the event of an overdose to receive naloxone. One method of distribution of non-patient-specific prescriptions has been the utilization of standing orders. Through standing orders, prescribers can authorize pharmacies to dispense naloxone without a prescription from a provider. For example, a patient or care-giver may present to a community pharmacy and request naloxone. The pharmacist is then authorized to process an order for that individual using the standing order as the prescription. The pharmacist may also bill the patient’s insurance to request payment for naloxone. This process allows patients and care-givers to procure naloxone with ease by reducing steps involved in obtaining a prescription, as well as allows for reduced cost, ultimately increasing access. 14 The cost of naloxone does, however, remain a concern for both institutions and individuals. Table 1 provides comparative costs for injectable naloxone syringes (intended for use with a nasal atomizer) and the only two FDA naloxone products for take-home naloxone use. 24 On April 19, 2019, after completing an expidited review, the FDA announced approval of the first generic naloxone nasal spray for lay person use. 25 As of publication, it is not yet clear when this product will be available nor what the cost will be. In the same press release, the FDA also announced steps to facilitate in the development of an over the counter naloxone product as well. Currently standing order prescriptions for naloxone are legal in all 50 states except for Idaho, Nevada, and Oregon. 23
Prices of different naloxone products. 24

In addition to utilizing standing orders to authorize pharmacies and pharmacists to dispense naloxone, some states permit individuals in community settings to distribute naloxone through a standing order. These are most often utilized in community-based Overdose Education and Naloxone Distribution (OEND) programs. 14 State laws protect lay-people and professionals who administer and distribute naloxone. Lay distribution and possession of naloxone are not covered by law in 17 out of 50 states. 23
Another method of distribution of non-patient-specific prescriptions is the utilization of protocol orders. Protocol orders have a similar intent and effect as standing orders, but differ in that they are issued by the state board of health or pharmacy licensing board, as opposed to the provider themselves. 14
The third and final method of non-patient-specific prescriptions is collaborative practice agreements. These are formal agreements between individual prescribers and pharmacies or chains within a state that allocate mutually agreed upon provider-type responsibilities to pharmacists (including the ability to essentially prescribe and dispense naloxone to patients). 14 Like the two aforementioned, this also allows for enhanced access to naloxone without the need for direct consultation with the patient’s provider.
As naloxone access has increased, fears have surfaced regarding individuals overdosing deliberately with the knowledge that they can be revived using naloxone or individuals becoming cavalier regarding use of prescription and illicit opioids. Some feared that increased access to naloxone and the institution of good Samaritan laws would actually increase opioid overdose rates. Rees et al. explored this possible correlation in 2017 and found the opposite to be true. They noted a 13% reduction in opioid-related deaths associated with the removal of criminal liability for naloxone possession. Furthermore, their study noted that naloxone access laws led to an initial reduction in opioid-related deaths of 9%–11% and up to a 21% reduction over 2 or more years. 26
Naloxone access throughout the United States in the wake of an opioid crisis has become a priority. Since naloxone is a prescription drug at the time of publication, regulations regarding access from the pharmacist can vary considerably among various states. Although medical providers have become increasingly cognizant of variable opioid-associated respiratory risks, there are no specific guidelines to suggest whether or when a patient is an appropriate candidate for in-home naloxone as part of their opioid regimen. While the ultimate role and utilization of opioids for the management of chronic pain in this era continues to be a simmering boiling pot of contentious debate and discussion, it is generally agreed that mitigation strategies which could lessen morbidity and mortality are essential. As naloxone access has gained popularity among politicians, medical providers, pharmacists, various other healthcare providers, and patient advocacy groups, continual expansion of access will no doubt save lives.
Footnotes
Acknowledgements
This article is the sole work of the authors; stated opinions or assertions do not reflect the opinions of employers or employee affiliates listed. The article was not prepared as part of the authors’ duties as federal employees.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
