Abstract
According to the standard theory of memory consolidation, recent memories are stored in the hippocampus before their transfer to cortical modules, a process called systemic consolidation. The ventral midline thalamus (reuniens and rhomboid nuclei, ReRh) takes part in this transfer as its lesion disrupts systemic consolidation of spatial and contextual fear memories. Here, we wondered whether ReRh lesions would also affect the systemic consolidation of another type of memory, namely an olfaction-based social memory. To address this question we focused on social transmission of food preference. Adult Long-Evans rats were subjected to N-methyl-
Keywords
Introduction
To persist, declarative-like memories must undergo systemic consolidation (Frankland and Bontempi, 2005), a process through which a recent memory located in the hippocampus is progressively transferred to the prefrontal cortex (PFC). In rodents, spatial (Lopez et al., 2012; Loureiro et al., 2012) and contextual fear (Frankland et al., 2004) has been shown to consolidate at systems level. This consolidation requires an information exchange between hippocampal and cortical networks. Whereas information transmission from the hippocampus to PFC can be monosynaptic, the transmission in the opposite direction requires a relay (Jay and Witter, 1991). From a neuroanatomical point of view, the ventral midline thalamus (i.e. reuniens and rhomboid (ReRh) nuclei) is in a prime position to ensure such bidirectional information exchange (rev Cassel et al., 2013; rev Dolleman-Van-Der Weel et al., 2019; Varela et al., 2014; Vertes et al., 2006). Using a classical water maze task in rats, we discovered that fibre-sparing lesions of the ReRh nuclei had no effect on task acquisition and recent spatial memory retrieval (5 days post-acquisition) but hindered remote memory retrieval (25 days post-acquisition). Furthermore, ReRh lesions prevented the post-learning persistence of new dendritic spines in the hippocampus and the delayed spinogenesis in the medial PFC (mPFC; Klein et al., 2019). These behavioural and morphological observations are compatible with a lesion-induced disruption of systemic consolidation. More recently, we found that ReRh lesions hindered systemic consolidation of a contextual fear memory (Quet et al., 2020). We therefore wondered whether ReRh lesions would also affect the systemic consolidation of another type of memory, namely social memory, which might be hippocampus-independent (Burton et al., 2000; Thapa et al., 2014). To try to answer this question, we used the social transmission of food preference (STFP) task (Galef and Wigmore, 1983; Van Der Kooij and Sandi, 2012). STFP occurs while an observer rat interacts with a conspecific demonstrator, which has just eaten a new flavoured food. Subsequently, the observer has to choose between two unknown foods, one of which previously presented by the demonstrator. An observer rat eating more of the latter food shows STFP. Manipulations such as cholinergic denervation of—or muscarinic blockade in—the PFC alter STFP performance (Berger-Sweeney et al., 2000; Boix-Trelis et al., 2007). Post-acquisition lesions of the hippocampus reduce STFP performance if the lesion is performed 1 day after the presentation of flavoured food, which is not the case if the lesion is performed 21 days after it (Clark et al., 2002; Ross and Eichenbaum, 2006). Likewise, pre-acquisition hippocampal lesions have no effect on information encoding and short-term memory, but alter retention after a 1 day delay (Bunsey and Eichenbaum, 1995). Lesburguères et al. (2011) showed that both the hippocampus and orbitofrontal cortex (OFC) were necessary in the early post-acquisition phase of STFP (0–12 days), not in the late post-acquisition phase (15–27 days): when the hippocampus was inactivated repeatedly during the late phase, rats remembered the task. Studies using c-Fos expression as a functional marker also support a time-limited role of the hippocampus in the acquisition and recall of STFP memory (Countryman et al., 2005; Smith et al., 2007). However, some authors have claimed that the hippocampus is not involved in STFP. For instance, Burton et al. (2000) failed to observe any deficit in the learning or retention of STFP after combined damage to the hippocampus and subiculum, a lesion producing deficits in a one-trial spatial memory task. Therefore, should STFP rely on the hippocampus, one may expect a disruption of its systemic consolidation after ReRh lesions. If not, ReRh lesions would perhaps not affect this type of consolidation and not interfere with social memory persistence.
Materials and methods
Subjects
All experimental animals were used in compliance with the rules of the European Community Council Directive (2010/63/EU) and the French Agriculture Ministry. All approaches have been validated by the ethical committee of the University of Strasbourg (CREMEAS—authorisations #5822-2016062214582106).
We used 95 adult, Long–Evans, male rats (Janvier Labs, Le Genest-Saint-Isle, France) weighing 250 g at their arrival at the laboratory (7–8 weeks old). They were housed 2 per cage in quiet facilities, under a 12-h light/dark cycle (light on at 7:00 a.m.) with food and water ad libitum, controlled temperature (23°C), and a hygrometry of about 55%. Before any experimental manipulation (surgery or behavioural training), rats were individually handled for 2 min/day over five consecutive days. We used 17 rats to measure the spontaneous preference for thyme- and cumin-flavoured foods and 78 rats for the STFP experiment (12 demonstrators and 42 observers, after removing rats with misplaced or insufficient lesions: 1-day groups, nSham = 10, nReRh = 12; 25-day groups, nSham = 10, nReRh = 10). The sample size was justified by the fact that the effects of a single interaction between observers and demonstrators on diet choice are robust and long-lasting (Galef et al., 1984; Galef and Wiskin, 2003). In addition, in previous studies on STFP, groups of 5 to 12 rats were used (e.g. Gold et al., 2011; Hsu et al., 2018; Lesburguères et al., 2011; Wooden et al., 2014). Finally, it is to note that these rats were the same as in Quet et al. (2020), except for those in the preliminary experiment used to measure the spontaneous preference for thyme- versus cumin-flavoured foods.
Lesion surgery
For permanent fibre-sparing excitotoxic lesions of the ReRh, subjects (mean body weight = 300 g ± 15 g) were anaesthetised with sodium pentobarbital (50 mg/kg, i.p.) and secured in a stereotactic frame (incisor bar: −3 mm). As previously described (Cholvin et al., 2013; Klein et al., 2019; Loureiro et al., 2012), neurotoxic lesions targeting the ventral midline thalamus (ReRh) were made using slow microinfusions (over 5 min) of 0.12 M N-methyl-
Spontaneous preference for thyme- and cumin-flavoured food
The aim of this experiment was to verify the natural preference of rats for thyme over cumin, as previously shown by Lesburguères et al. (2011) in order to use this biased thyme–cumin pair to induce a socially acquired food preference. Indeed, interaction with a demonstrator that has eaten cumin-flavoured food can reverse this innate thyme preference so that observers subsequently choose cumin over thyme. This protocol offers the advantage of decreasing chance level below 50% during the test and then optimising the possibility to detect changes in memory performance (Bessières et al., 2017).
The day before the experiment, rats (n = 17) were food-deprived from 30 min before light extinction (6:30 p.m.) to the acquisition or test session on the next day. The preference test (between 10 a.m. and 2 p.m.) was run over 30 min, during which rats had access to two cups: one containing cumin (0.5%)-flavoured powdered food and the other one containing thyme (0.75%)-flavoured powdered food. The total consumption of each flavoured food was measured. This test was repeated 2 days later in the same rats.
STFP
The classical three-step experimental design (Figure 1(a)) was similar to previously published protocols (Bunsey and Eichenbaum, 1995; Burton et al., 2000; Lesburguères et al., 2011; Ross et al., 2005), although many variants can be found in the literature (rev Bessières et al., 2017). Briefly, demonstrator rats (food-deprived and previously habituated to eat standard (non-flavoured) powdered food) were allowed a time-limited access to one cup filled with the chosen flavoured powdered food. Then, the demonstrator was placed in contact with the observer to enable collection of olfactory information from the demonstrator’s breath during social interactions. After a delay (days to weeks), the observer rat was provided with 2 cups, one containing a novel food, and the other with the food the demonstrator rat had consumed before interacting with the observer rat. At the end of the food preference test (30 min), cups were removed, weighed, and the quantity of demonstrated food versus total food eaten allowed to assess olfactory memory performance (e.g. Lesburguères et al., 2011).
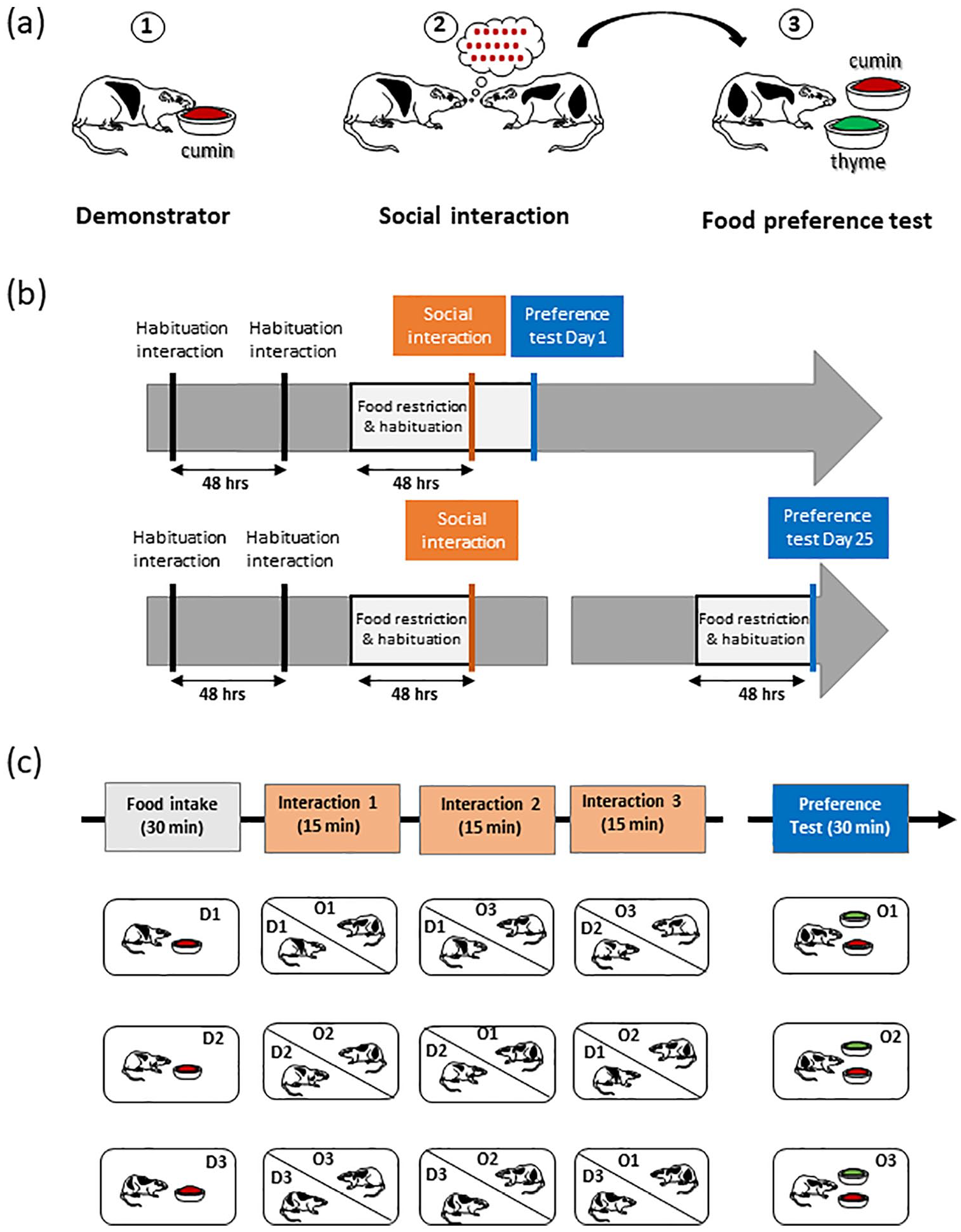
Design of the experiments. (a) General principle of STFP with the classical three-step procedure:
Habituation and food restriction
Despite the fact that the STFP paradigm is a non-aversive procedure with a high degree of ethological relevance, it calls for great vigilance on some aspects. As a social task, it requires motivation to interact with a rat congener without any social stress. Thus, all observer rats (O) were habituated to interact twice (2 days apart) with their future demonstrator rats (D) in an interaction cage (5 min/interaction) in the testing room. This cage is divided into two compartments (Gold et al., 2011), separated by a transparent plastic wall with drilled holes. The holes were large enough to allow rats to insert their nose, thereby facilitating olfactory exchanges of information but avoiding occurrence of aggressive behaviour.
As an appetitive task, STFP requires diet restriction to elicit adequate motivation for food. Considering that food restriction may induce anxiety (Heiderstadt et al., 2000; Toth and Gardiner, 2000), we used a moderate and time-limited food restriction paradigm before the interaction and the test (Bessières et al., 2017). Regular food pellets were removed from the cages 3 days before the social interaction and preference tests, with food access during two daily sessions: (1) at noon, rats had free access to plain powdered food in a small cup over a 30-min period in the test cage, for habituation to powdered food and (2) at 6:00 p.m., regular food pellets were provided over a 30-min period in the home cage.
STFP
The demonstrator rats were placed in the testing room in an individual cage and had access to a food cup filled with cumin-scented powdered food for 30 min. Observer rats were then presented with a rotation of three demonstrator rats for 15 min each and a total of 45 min of social interactions (Figure 1(c); Boix-Trelis et al., 2007; Ross et al., 2005; Wooden et al., 2014). After these rotations, for the 1-day delay group, the food restriction diet was prolonged for 1 additional day. For the 25-day delay group, all rats returned to their home cage where they received a standard food diet. On Day 22, that is, 3 days before the preference test, rats were food-restricted using the same procedure than before the social interactions.
Food preference tests
On the test day (Day 1 or Day 25), observer rats were placed in individual cages in the testing room. They had free access to two pre-weighed food cups, one containing the thyme (0.75%)-powdered food (new food) and one containing the cumin (0.5%)-powdered food (demonstrated food). The position of the demonstrated food (left or right) in the cage was counterbalanced. The test lasted 30 min, and then, cups were removed, weighed, and an olfactory memory score was computed with the following formula: ((amount of familiar food eaten)/(amount of total food eaten)) × 100 (Bessières et al., 2017; Lesburguères et al., 2011).
Histological verifications
Perfusion and tissue sectioning
All rats were subjected to a lethal dose of pentobarbital (200 mg/kg, i.p.) and perfused transcardially with a cold (4°C) 4% paraformaldehyde (PFA) solution. Brains were removed and transferred to a 20% sucrose solution for 72 h at 4°C before being snap frozen (isopentane, −40°C) and stored at −80°C. Serial coronal sections (40 µm) were cut throughout the midline thalamus using a cryostat (Microm HM560; Thermo Scientific) to assess the lesion placement and extent.
Immunohistochemistry
To characterise the ReRh lesions, immunolabeling of the NeuN protein was performed on free-floating brain sections evenly distributed along the entire rostro-caudal extent of the ReRh. As previously described (e.g. Klein et al., 2019), NeuN immunochemistry was performed using a mouse NeuN antibody (1:2000, ref MAB377; Millipore) as primary antibody and a biotinylated anti-mouse horse antibody (1:500; Vector Laboratories) as secondary antibody (see Loureiro et al., 2012). Briefly, sections were rinsed three times during 10 min in a PBS merthiolate buffer before being soaked for 1 h in 5% normal donkey serum in PBS containing 0.5% Triton X-100. The sections were then transferred into the primary anti-NeuN antibody solution and kept there for 18 h at room temperature. Then, they were soaked in a buffer solution containing the secondary antibody. Staining was performed with the avidin–biotin peroxidase method (Vectastain ABC kit; Vector Laboratories) coupled with diaminobenzidine.
Lesion analysis
Lesions were drawn on relevant plates of the rat brain atlas (Paxinos and Watson, 2007) and replicated on electronic copies of the atlas. Automated pixel counts of the thalamic nuclei in the target regions were used to estimate lesion extent, including in regions adjacent to the ReRh (Loureiro et al., 2012). According to Groenewegen and Witter (2004), the Re nucleus is bordered by the so-called perireuniens (pRe). As the Re, the pRe has connections with limbic structures, particularly with the mPFC (Hoover and Vertes, 2012). Acceptable lesions were defined as having >50% damage to the ventral midline thalamus (Re, Rh and peri-Re nuclei combined) with at least 15% damage to each of the nuclei in order to prevent the inclusion of too asymmetric lesion (Loureiro et al., 2012; Quet et al., 2020).
Statistical analyses
To compare the size of the lesion in the ReRh nuclei and adjacent regions between recent (Day 1) and remote (Day 25) time-points, we used one-way analysis of variance (ANOVA) with Delay as the factor. For the spontaneous cumin versus thyme preference, data were analysed using one-way or two-way analyses of variance (ANOVAs) with Food (thyme, cumin) and Delay (Day 1, Day 2) as factors (the latter as a repeated measure). For the STFP experiment, data analyses used a comparison of the means to a standard (i.e. 36.4%, the value corresponding to the spontaneous percentage of preference for cumin vs thyme). Analysis of scores during the test used a two-way ANOVA with factors Group (Sham, Lesion) and Delay (recent, remote). These analyses were completed, when appropriate, with multiple comparisons using the Newman–Keuls multiple range test. Values of p < 0.05 were considered significant.
Results
Spontaneous food preference for thyme- and cumin-flavoured food
The two-way ANOVA of the quantity of cumin versus thyme eaten showed significant Food (F(1, 32) = 10.76, p = 0.0025) and Day (F(1, 32) = 4.68, p = 0.037) effects, but no interaction between the two factors (F(1, 32) = 0.04, p = 0.83). The Food effect was due to a significantly higher quantity of thyme-flavoured over cumin-flavoured food intake at both testing days (p < 0.05, Figure 2(c)). The significant Day effect was due to rats eating more food (both types) on Day 2 versus Day 1 (p < 0.05), as also illustrated in Figure 2(a). Concerning the percentage of preference for each type of flavoured food, data are illustrated in Figure 2(b). The two-way ANOVA showed a significant Food effect (F(1, 32) = 10.00, p = 0.0034), no Day effect (F(1, 32) = 0.00, p = 1.00), and no interaction between the two factors (F(1, 32) = 0.21, p = 0.64).

Spontaneous preference for thyme- versus cumin-flavoured powdered food. (a) Total food eaten (in g) during the two 30-min test sessions, 2 days apart. (b) Percentage of preference for the cumin- versus thyme-flavoured food. The dotted line represents 50%. (c) Quantity of cumin- versus thyme-flavoured food eaten (in g) on each testing day. Values are mean ± SEM of 17 rats. *p < 0.05 versus cumin.
Lesion location and extent
Serial sections throughout the thalamus were stained with NeuN to visualise the lesion placement and extent. After removal of rats with misplaced or too small lesions (n = 12 rats/delay lesioned group), final sample sizes were as follows: 1-day groups, nSham = 10, nReRh = 12; 25-day groups, nSham = 10, nReRh = 10. No significant difference in the extent of the ReRh lesion was found between 1 and 25 days in the Re (F(1, 20) = 2.56, p = 0.125), the pRe right (F(1, 20) = 1.79, p = 0.20) and the pRe left (F(1, 20) = 0.22, p = 0.64). For the Rh, the lesion size was significantly smaller at the 25-day delay (F(1, 20) = 7.90, = 0.011), and for the whole ReRh area, the lesion size tended to be smaller at the 25-day delay, as compared to the 1-day delay (F(1, 20) = 4.06, p = 0.058). In the 12 ReRh rats at the 1-day delay, there was a mean of 92.5% and 77.9% damage to the Re and Rh, respectively, with pRe showing 67.3% and 86.3% damage (left and right, respectively, Table 1). At the 25-day delay, in the 10 ReRh rats, there was a mean of 86.4% and 54.5% damage to the Re and Rh, respectively, with pRe showing 62.6% and 78.5% damage (left and right, respectively; see Table 1). Panels a–a″ in Figure 3 show the smallest and largest lesions at three anteroposterior levels, and panels c and d and c′ and –d′ in the same figure illustrate two typical examples of ventral midline thalamic lesions.
Quantification of the ReRh lesion at both delays for rats that were included in the final behavioural analysis.

SEM: standard error of the mean.
Values are mean ± SEM of the percentage of the lesion surface as compared to the total surface of each subregion (see section ‘Methods’ for the quantification protocol). Values correspond to the average lesion extent on frontal sections at three anteriority Bregma levels (i.e. −1.20 to −1.92 mm, −2.16 to −2.52 mm, −2.64 to −3.36 mm, according to Paxinos and Watson, 2007).
p < 0.05, statistically significant difference from Day 1.

Evaluation of the placement and extent of ReRh lesions. (a-a′-a″). Schematic representation of the smallest (black) and largest (grey) lesion extent of the ventral midline thalamus (Re and Rh) in coronal sections taken at three anteroposterior levels from Bregma (according to Paxinos and Watson, 2007). (b–d) Photomicrographs showing typical examples of NeuN-immunostained brain sections from a Sham rat (b) and from two rats with ReRh lesions (c and d); scale bar, 1 mm. (b′–d′) Photomicrographs showing larger magnification of the region including the Re and Rh as shown in (b–d), respectively; scale bar, 250 µm.
Concerning other thalamic nuclei, data showed mostly unilateral damage localised along the canula track. These unilateral damage encroached onto the submedius nucleus (Day 1, 57.5%; Day 25, 59.9%), the centromedian/paracentral nuclei (Day 1, 18.7%: Day 25, 13.1%), the paratenial/mediodorsal nuclei (Day 1, 28.9%; Day 25, 25.4%), and the anteromedian nucleus (Day 1, 36.9%; Day 25, 30.1%). At the anterior and posterior levels of the canula track, these thalamic nuclei were not affected. All of these areas showed no damage on the contralateral injection side, except the submedius nucleus (Day 1, 13.5%; Day 25, 21.7%). We found no significant Delay effect (Day 1, Day 25) on the lesion extent in these thalamic nuclei (F(1, 20) = 0.0442 to 1.157, p > 0.1 for all).
STFP: retrieval performance
Panel a in Figure 4 shows that the ReRh lesion did not affect food motivation. The ANOVA of the total food consumed showed no significant Group (F(1, 38) = 0.035, p = 0.85) or Delay (1 day, 25 days, F(1, 38) = 1.75, p = 0.19) effects, and there was no interaction between the two factors (F(1, 38) = 2.14, p = 0.15). Sham rats ate between 2.7 and 4.0 g of powdered food in line with previous data (Burton et al., 2000; Lesburguères et al., 2011; Ross and Eichenbaum, 2006).

Recent and remote memory performance after ReRh lesions in rats. (a) Total food eaten (in g) during the 30-min test session either 1 day (recent) or 25 days (remote) after interaction with the demonstrators. (b) Percentage of cumin-flavoured food preference. The dotted line represents the innate cumin preference (i.e. 36.4%). (c) Quantity of cumin-flavoured food eaten (in g) during each testing day. Values are mean ± standard error of the mean (SEM) of 10 or 12 rats/group. *p < 0.05 versus innate cumin preference.
Panels b and c show that the previously demonstrated cumin-flavoured food was preferred by Sham rats, as it amounted to 89% of the total food intake one day (recent memory), a value significantly above the standard of 36.4% (innate preference; p < 0.001). This preference for the cumin-flavoured food persisted after the long delay (remote memory), as it amounted to 69% preference in Sham rats (vs 36.4%, p < 0.001). The excitotoxic ReRh lesion had no effect on the STFP, whatever the post-interaction delay. Indeed, lesioned rats showed a strong preference for cumin-flavoured food (recent, 79%; remote, 73%, p < 0.001 and p < 0.05, respectively, vs 36.4%). The two-way ANOVA showed that lesioned rats did not differ from Sham rats: there were no significant Group (F(1, 38) = 0.15, p = 0.70) or Delay (F(1, 38) = .2.68, p = 0.11) effects, and no significant interaction between the two factors (F(1, 38) = 0.78, p = 0.38).
Our data clearly show that ReRh nuclei lesions did not affect retrieval of recent and remote associative olfactory memory, thereby indicating that systemic consolidation of this memory worked normally. This is at variance with our previous data showing a role for the ReRh nuclei in persistence of spatial and contextual fear memories (Loureiro et al., 2012; Quet et al., 2020, respectively).
Discussion
Building upon evidence showing that the ReRh nuclei were essential to systemic consolidation of spatial (Ali et al., 2017; Klein et al., 2019; Loureiro et al., 2012) and contextual fear memories (Quet et al., 2020; Vetere et al., 2017), the current study addressed the possibility that these nuclei also contribute to the persistence of a social memory. The paradigm used was that of STFP, a memory test of high ethological value (Galef and Wigmore, 1983; Van Der Kooij and Sandi, 2012). As for spatial and contextual fear memory (Loureiro et al., 2012; Quet et al., 2020), the lesion did not hinder acquisition and recent recall of the olfactory information. Unlike what happened for spatial and contextual fear memories, however, we found no evidence for an implication of the ReRh nuclei in the systemic consolidation of STFP memory.
Dopamine innervation of dorsal hippocampus from ventral tegmental area (VTA) has been associated with STFP performance in mice (Matta et al., 2017). Based on lesion or pharmacological inactivation, STFP formation and retrieval, even at a recent time point, may not depend on the hippocampus (Alvarez et al., 2001; Burton et al., 2000; Thapa et al., 2014). Thus, our data showing that ReRh lesions were without any effect on the consolidation of an STFP task are compatible with the non-implication of the hippocampus in such a type of memory. However, in other studies based on lesion, immediate early gene imaging, and reversible inactivation/receptor inhibition approaches, STFP formation and recall at recent time-points (i.e. 1 to 7 days) seem to rely on the hippocampus, as well as on several neocortical structures, including the orbitofrontal, piriform and entorhinal cortices (Bunsey and Eichenbaum, 1995; Lesburguères et al., 2011; Ross and Eichenbaum, 2006). The hippocampus is involved in the information consolidation process, not in long-term storage of the cue (Bunsey and Eichenbaum, 1995). It is also involved in recent recall of the cue, as Ross and Eichenbaum (2006) observed that hippocampal lesions made 1 day after the demonstrator–observer interaction disrupted subsequent recall of the olfactory cue (30-day post-interaction). When the lesion was made 21 days after the interaction, recall was normal. In the same study, recent recall of the cue activated the ventral subiculum shortly after acquisition (immediate or 1 day), a response which decreased with longer delays (2 days or 21 days), whereas in later retrieval tests, the regions showing augmented activity were the orbitofrontal, piriform and entorhinal cortices, among other olfactory projection areas. Lesburguères et al. (2011) confirmed the increased activity in the dorsal and ventral hippocampus during recent (day 7) memory recall, and in the OFC after remote (from day 15) memory recall. Lesburguères et al. (2011) also showed that upon information encoding, OFC neurons undergo a tagging process, which might serve as a matrix for hippocampus-driven cortical rewiring supporting enduring memory. Recently, Loureiro et al. (2019) found STFP to depend on projections from the piriform cortex—one of the olfactory projection areas to which the study by Ross and Eichenbaum had pointed—to the neurons of the mPFC (prelimbic and infralimbic regions), which project onto the nucleus accumbens. Thus, STFP seems to involve several distributed and interdependent structures of the brain.
Because the hippocampus seems necessary for early consolidation of the olfactory cue and cortical modules for its retrieval at a remote time point, we hypothesised that the olfactory cue memory might undergo a ReRh nuclei-dependent systemic consolidation. Our data show that the cue memory may last for at least 25 days, which is in line with Lesburguères et al. (2011). However, we found no evidence for a contribution of the ReRh nuclei to systemic consolidation of STFP. One possible reason for this could be related to physical characteristics of the lesions. As in our former studies (Ali et al., 2017; Klein et al., 2019; Loureiro et al., 2012), we used fibre-sparing NMDA lesions, which could have been less extended in the current study, therefore less efficient, but it was not the case. Furthermore, these lesions prevented systemic consolidation of a contextual fear memory (Quet et al., 2020). Another possible reason could be differences in the lesion extent in one particular nucleus, namely the rhomboid nucleus. Indeed, the Rh nucleus was damaged to an average extent of about 80% in the recent memory group versus about 54% in the remote memory group, most probably reflecting a sampling fluctuation. It is of note, however, that in all of our studies, the extent of the Rh lesions varied between about 35% (Ali et al., 2017; Loureiro et al., 2012) and 42% (Klein et al., 2019). Therefore, it is relatively unlikely that the difference in lesion extent accounts for the absence of remote memory deficits. Yet another—very theoretical—explanation could be that systemic consolidation of an olfactory cue is slower than for other types of items (e.g. a location or a context) and therefore requires more than 25 days for being constructed in cortical modules. This possibility is discarded by data from Ross and Eichenbaum (2006), which showed that as soon as 21 days after training, retrieval of the olfactory cue had become resistant to hippocampal lesions. Likewise, Lesburguères et al. (2011) clearly showed the engagement of the OFC and the disengagement of the hippocampus 15 days after demonstrator–observer interaction.
Finally, systemic consolidation of an STFP-related olfactory cue might just not require ReRh nuclei, because STFP is not hippocampus-dependent. Olfactory cue-related signals generated in the olfactory bulbs are relayed in the piriform cortex to the OFC and mPFC. The mPFC projects onto the nucleus accumbens, whose dopaminergic neurons reach the hippocampus. An implication of this circuit in STFP has been documented in various reports (e.g. Loureiro et al., 2019; Matta et al., 2017; Ross et al., 2005) and damage or dysfunction at any level in this circuit seems to have disruptive consequences on STFP acquisition or recall (Brightwell et al., 2005; Bunsey and Eichenbaum, 1995; Jeon et al., 2007; Loureiro et al., 2019; Peters et al., 2003; Ross and Eichenbaum, 2006; Ross et al., 2005). Therefore, information exchange between cortical and hippocampal modules underlying systemic consolidation could occur in this circuitry, which does not require the ReRh nuclei hub. Our current knowledge of STFP, underlying circuits, and systemic consolidation does not allow us to directly compare the processes and precise networks at play in social, contextual and spatial memorisation. With regard to contextual and spatial memories, the recall of a recent memory is not accompanied by an activation of the mPFC (Frankland et al., 2004; Lopez et al., 2012) and recent memory is not altered by a reversible inactivation of the mPFC (e.g. Cholvin et al., 2013 for spatial memory). Furthermore, regarding contextual fear, it appears that its systemic consolidation is clearly dependent on a functional network involving the mPFC, the hippocampus and the thalamus, including the Re nucleus (Wheeler et al., 2013), and the same could be true for a spatial memory (Ali et al., 2017; Klein et al., 2019; Loureiro et al., 2012). Regarding STFP, it should be noticed that a recent memory of the olfactory cue might depend on both the hippocampus (Ross and Eichenbaum, 2006) and the projection from the piriform to mPFC (Loureiro et al., 2019). This initial co-dependence of a recent memory on the olfactory cue could indicate that the processes involved in the consolidation of the memory are different, at least in part, between STFP and place or context memories. If this were the case, it would mean that the systemic consolidation of STFP does not rely on the engagement of a standardised cortico (network tagging)-hippocampo(recent memory)-cortical(remote memory) circuit.
It is usually considered that the ventral, ventrolateral and dorsolateral regions of the OFC and the hippocampus are not interconnected by direct neuroanatomical connections (Murphy and Deutch, 2018; Sesack et al., 1989). Therefore, direct, bidirectional monosynaptic information exchange between these subdivisions of the OFC and the hippocampus can be excluded from the possible mechanisms accounting for ReRh nuclei-independent systemic consolidation. It is noteworthy, however, that reciprocal connections between the medial OFC and the hippocampus have been described (Jay and Witter, 1991). To what extent these connections could be involved in the systemic consolidation of an STFP memory remains unknown for now. In addition, according to a recent review (Izquierdo, 2017), the medial OFC plays a role in reversal learning, unobservable/uncertain outcome, reinforcer devaluation and pavlovian instrumental transfer, which are all difficult to link to STFP.
Conclusion
Compared to our former results on spatial and contextual memories (Ali et al., 2017; Klein et al., 2019; Loureiro et al., 2012; Quet et al., 2020), our current findings indicate that the ReRh nuclei are most probably not part of a generic, systemic consolidation mechanism processing all kinds of memories in order to make them last. While these nuclei are necessary to the persistence of a spatial or a contextual memory, they do not contribute to that of an olfaction-based social memory.
Footnotes
Acknowledgements
The authors are grateful to O. Bildstein, D. Egesi and G. Edomwony for their excellent assistance in animal care. They also acknowledge E. Panzer and L. Boch for providing appreciable technical help and D. Cochand for correction of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: this work was supported by the Agence Nationale de la Recherche (ANR THALAME, grant 14-CE13-0029-01; coordinator J-C.C.), the University of Strasbourg, the CNRS and the INSERM. The French government and subsequently the Paul Mandel Fund provided a doctoral fellowship of 3 years and 6 months, respectively, to E.Q.
