Abstract
Background:
Deep brain stimulation is explored as a new intervention for treatment-resistant substance use dependence. A candidate brain region is the nucleus accumbens, due to its involvement in reward and motivation. This study aimed to explore effects of NAcore and NAshell deep brain stimulation on aspects of heroin taking and seeking in a self-administration model for rats.
Methods:
NAcore and NAshell deep brain stimulation was applied during 25 or 100 µg/kg/infusion heroin self-administration on an FR4 schedule of reinforcement and during cue- and heroin-induced reinstatement. In a separate group, effects of NAcore deep brain stimulation on heroin self-administration on a progressive ratio schedule and the first extinction session were examined.
Results:
NAcore and NAshell deep brain stimulation did not alter heroin self-administration on an FR4 schedule. NAcore deep brain stimulation decreased cue – but not drug-induced reinstatement of heroin seeking, whereas NAshell deep brain stimulation did not affect reinstatement responding. In the second experiment, NAcore deep brain stimulation reduced responding during a progressive ratio schedule of heroin reinforcement. Finally, deep brain stimulation facilitated extinction from day 1 throughout the course of extinction learning.
Conclusion:
Taken together, the differential effects of NAcore and NAshell deep brain stimulation on heroin taking and seeking are in line with the distinct functional roles of these sub-regions therein. Conditioned cues have been shown to be very powerful stimuli for the persistence of addiction and relapse to drug use. Therefore, the present findings that NAcore deep brain stimulation decreases motivation for heroin taking and cue-conditioned behaviour and facilitates extinction learning are very promising, supporting the positive findings from clinical case studies.
Introduction
Substance use disorder (SUD) is recognised as a chronic brain disorder. It is characterised by compulsive and continued drug consumption regardless of the knowledge of negative consequences to the subjects’ health status, their environment and society and high rates of relapses into drug use. Even after 5 years of abstinence, a large proportion of the substance abusers relapse, despite currently available treatments (Flynn et al., 2003; Hser et al., 2001). For this reason, development of new therapies is highly needed.
A potential new therapeutic intervention for SUD is deep brain stimulation (DBS). DBS consists of selective electric stimulation of specific brain areas with implanted electrodes. Over the past years, DBS has shown promising results, not only for neurological disorders such as Parkinson’s disease (Benabid, 2003) but also for psychiatric disorders such as depression (Mayberg et al., 2005), obsessive-compulsive disorder (Denys et al., 2010), anorexia nervosa (Oudijn et al., 2013) and, more recently, SUD (Kuhn et al., 2011, 2014; Luigjes et al., 2012).
A candidate brain target for the application of DBS for SUD is the nucleus accumbens (NA; Luigjes et al., 2012; Muller et al., 2013). The NA is part of the mesolimbic system and is a heterogeneous structure, consisting of two anatomically and functionally distinct sub-regions, the core region (NAcore) and shell region (NAshell; Groenewegen et al., 1999). Both sub-regions have been demonstrated to play important but distinct roles in reward and motivation. In this regard, the NAshell is primarily involved in the rewarding effects of drugs (Sellings and Clarke, 2003). For instance, early studies have shown increased dopamine release in the NAshell, and not in the NAcore, in response to intravenous (IV) administration of different drugs of abuse, including heroin (Pontieri et al., 1995; Tanda et al., 1997). In contrast, the NAcore has been shown to be primarily involved in mediating conditioned behaviour in response to cues predicting motivationally relevant events such as a reward (Ambroggi et al., 2011; Corbit and Balleine, 2011; Jones et al., 2010; Theberge et al., 2010).
Recent studies have reported a reduction in consumption in patients treated with NA DBS for their alcohol abuse (Kuhn et al., 2011) or even complete abstinence of alcohol (Muller et al., 2009) and opioids (Kuhn et al., 2014; Valencia-Alfonso et al., 2012; Zhou et al., 2011). These clinical observations are paralleled by rodent studies that have shown beneficial effects of NAcore and NAshell DBS for different drugs of abuse. For instance, several studies have demonstrated a reduction in alcohol preference by acute DBS in both NAcore and NAshell (Henderson et al., 2010; Knapp et al., 2009; Wilden et al., 2014) as well as a reduction in the expression of alcohol sensitisation following NAcore DBS (Nona et al., 2015). Furthermore, NAshell DBS reduced cue-induced and cocaine priming–induced reinstatement of drug seeking (Guercio et al., 2015; Vassoler et al., 2008), whereas NAcore DBS was found to be ineffective (Vassoler et al., 2013). With respect to opiates, chronic unilateral NAcore and NAshell DBS have been shown to reduce conditioned place preference (CPP) for morphine (Liu et al., 2008; Ma et al., 2013; Yan et al., 2013). Similarly, bilateral and unilateral NAcore DBS during abstinence after acquiring heroin self-administration reduced cue-induced and heroin priming–induced reinstatement of drug seeking (Guo et al., 2013). Collectively, these studies suggest that NA DBS decreases both opiate and psychostimulant seeking, albeit with differential involvement of the NA sub-regions.
In contrast to direct comparisons of NAcore/NAshell DBS on alcohol (Henderson et al., 2010; Knapp et al., 2009) and cocaine self-administration (Vassoler et al., 2013), the effects of NA DBS on heroin taking and -seeking have not been established yet. This article studied the effects of acute NAshell and NAcore DBS on different aspects of heroin taking and -seeking in an operant rodent model of heroin self-administration.
Methods
Animals
In total, N = 48 male Wistar rats were obtained from Harlan CPB (Horst, The Netherlands). At the start of the experiments, the rats weighed between 260 and 290 g. The rats were housed in Macrolon cages (42.5 cm × 26.6 cm × 18.5 cm) in pairs until the implantation of an IV catheter and DBS electrodes. They were kept under a reversed light–dark cycle (lights on from 7 p.m. until 7 a.m.) at controlled room temperature (21 ± 2°C) and relative humidity of 60 ± 15%. All experiments were carried out during the dark phase of the light–dark cycle. Water and standard chow food were available ad libitum in the home cage during the entire experiment. All experiments were approved by the Animal Care Committee of the VU University and VU University Medical Center Amsterdam.
Surgery
Prior to surgery, rats received subcutaneous (s.c.) injections of the analgesic Ketofen (5 mg/kg, 1%) and the antibiotic Baytril (8.3 mg/kg, 2.5%). Rats were then implanted with an IV silicone catheter in the right jugular vein, under isoflurane gas anaesthesia (±2%). Following catheter implantation, DBS electrodes (Plastics One, Roanoke, VA, USA) were bilaterally implanted using the following coordinates relative to bregma (Paxinos and Watson, 1998): NAcore 2.3 mm anteroposterior, 7.4 mm dorsoventral, 2.7 mm mediolateral under an angle of 8° relative to the midline sagittal plane of the skull; NAshell 2.4 mm anteroposterior, 8.4 mm dorsoventral, 2.6 mm mediolateral under an angle of 12° relative to the midline sagittal plane of the skull (Pattij et al., 2007). Catheter pedestal and DBS electrodes were anchored to the skull with six stainless steel screws and dental acrylic cement. The experiments started following 1 week of recovery. During self-administration, IV catheters were daily flushed with 0.05 mL sterile saline solution containing 0.25 mg/mL heparin and 0.08 mg/mL gentamicin.
Heroin self-administration
Apparatus
Self-administration of heroin was performed in operant chambers (Med Associates Inc., St. Albans, USA) in sound-attenuating ventilated cubicles. One wall of the operant chamber was equipped with two nose poke holes. On the opposite wall was a house light situated that was on during the sessions.
Acquisition
The rats were trained to self-administer heroin (diacetylmorphine-HCl, dissolved in 0.9% sterile saline; Slotervaart Hospital, Amsterdam, The Netherlands) by making a nose poke into the active hole. Each trial was signalled by a stimulus light located above the active hole, which was extinguished when a heroin infusion was delivered. Acquisition training started with a fixed-ratio-1 (FR1) schedule of reinforcement, meaning that every nose poke resulted in an infusion of heroin (100 µg/kg/infusion) over a time period of 2 s. Heroin infusions were accompanied by a yellow stimulus light in the active nose poke hole combined with a 2-s tone signal (70 dB, 2900 Hz). Responses in the inactive hole were registered, but had no programmed consequences. Each heroin infusion was followed by a time-out period of 15 s, during which any nose poke was without consequences. Responses in the active and inactive nose poke hole were monitored during availability of the drug and during the time-out period. After 10 days, the rats had acquired a stable response on FR1 and the FR schedule of reinforcement was increased to FR2 (3 days) and FR4 (3 days), meaning that every second or fourth nose poke in the active hole was reinforced. The training consisted of 2.5 h daily sessions which took place on weekdays (Monday–Friday).
Progressive ratio
Following the acquisition phase, the rats were switched to a progressive ratio (PR) schedule of reinforcement, to assess motivation for heroin taking. The number of active nose pokes required for one heroin infusion was progressively increased within one session according to the equation 5 ×
Extinction
Responding for heroin was extinguished in daily sessions of 60 min (Monday–Friday). The rats were placed in the operant chamber with solely the house light turned on. Nose pokes in the previously active hole did not result in the delivery of heroin or presentation of the heroin-associated cues. Responses in the previously active and inactive nose poke hole were registered.
Cue- and drug-induced reinstatement
Following extinction, a cue-induced reinstatement test was performed for 60 min, to model relapse induced by heroin-associated stimuli. The conditions were the same as during an FR4 session in the acquisition phase, with the exception that the rats did not receive heroin. Every fourth active lever press was accompanied by the cues. Following 4 days of abstinence, a drug-induced reinstatement test was performed by injecting 0.25 mg/kg heroin s.c. (De Vries et al., 1998) 15 min prior to a 60-min reinstatement test. Responses in the previously active and inactive nose poke hole were registered.
DBS
DBS was performed with a digital stimulator (DS8000; World Precision Instruments, Sarasota, FL, USA) and stimulus isolator (DLS100; World Precision Instruments), using biphasic square pulses with 60 µs pulse width, 200 µs zero time and 130 Hz frequency. Stimulation intensities varied across experiments between 0 (=sham stimulation), 35, 75 and 100 µA. These stimulation parameters are comparable to previously reported work on DBS in rats (Guo et al., 2013; Knapp et al., 2009; Levy et al., 2007; Rouaud et al., 2010; Tan et al., 2010; Vassoler et al., 2008). Stimulation was always applied in the operant cage, started 5 min before actual testing and lasted for the duration of the entire session.
Experimental design
Experiment 1: NAcore and NAshell DBS during heroin self-administration and reinstatement
One group equipped with NAcore DBS electrodes (n = 16) and one group with NAshell DBS electrodes (n = 16) were trained to self-administer heroin as described above. Upon stable FR4 responding, rats were habituated to the stimulation procedure during six additional sessions on an FR4 schedule. To this end, rats were connected to the DBS cables during the session without current delivery. Rats were then tested in a randomised within-subject design on 0, 35, 75 and 100 µA during FR4 responding. Sessions with stimulation were always alternated with one session without stimulation. Subsequently, rats were trained for 6 days on FR4, receiving 25 µg/kg/infusion heroin to increase responding, and tested in a randomised design on 0 and 100 µA. After that, responding was extinguished during 20 sessions as described above. Then each rat was tested twice for cue-induced reinstatement in a randomised order with 0 or 100 µA. Reinstatement tests were separated for 48 h, and the rats remained in their home cage between tests. Finally, each rat was tested twice for drug-induced reinstatement in a randomised order with 0 or 100 µA. Again both tests were separated by 48 h and the rats remained in their home cage between tests.
Experiment 2: NAcore DBS during PR and first day extinction
In Experiment 1, NAcore DBS decreased cue-induced reinstatement, without any effects of NAshell DBS on aspects of heroin taking and -seeking. To further explore the effects of NAcore DBS on different aspects of heroin taking and -seeking, in Experiment 2 the effects of NAcore DBS on PR and first day extinction were explored. For this purpose, a new group of n = 16 rats was trained to self-administer heroin as described above. Upon stable FR4 responding, rats were trained on a PR schedule of reinforcement for 7 days and 7 subsequent days with connection to DBS cables for habituation to the stimulation procedure. Following habituation, rats were tested during PR responding in a randomised within-subject design with stimulation intensities of 0, 35, 75 and 100 µA. Sessions with stimulation were always followed by one session without stimulation. After testing on PR, rats were re-trained on FR4 for three sessions after which daily extinction training started. During the first extinction session, half of the rats was stimulated with 0 µA and the other half with 100 µA. Extinction learning was continued for 14 sessions to monitor extinction behaviour in both groups.
Statistical analyses
All data are presented as means ± standard errors of the mean and were analysed using IBM SPSS Statistics 20.0 (IBM Corporation, Armonk, NY, USA). Behavioural data were analysed using repeated measures analysis of variance (ANOVA) with DBS intensity as within-subjects factor, or paired Student’s t-tests. In the PR experiments, a square root transformation was performed on the number of active responses and a log transformation on the final ratio reached, the latter since these values were derived from an exponential function (Richardson and Roberts, 1996). In case of violation of homogeneity, tested with Mauchly’s test of sphericity, corrected degrees of freedom and resulting more conservative
Results
Histology and exclusion of rats
As indicated in Figure 1, most DBS electrodes were placed in the NAcore and NAshell at the level of 2.20 mm rostral to bregma. The majority of the NAcore DBS electrodes placements were dorsomedially located, whereas most of the NAshell DBS electrodes were ventromedially located. Three rats had only one electrode each in the NAcore or NAshell and one rat had one electrode at the border of 2.70 mm rostral to bregma. Since their behavioural response was not different from the rest of the group, they were included in the analyses. In Experiment 1, one rat from the NAcore group was excluded from all analyses because of an infection around the tip of the electrode. Of 16 rats with DBS electrodes aimed at the NAshell, one rat was excluded due to misplaced electrodes. Other exclusion criteria were IV catheter failure and loss of the IV catheter or DBS electrodes. These rats were included in the analyses until catheter failure or loss of electrodes occurred, and this is described in the specific experiment.
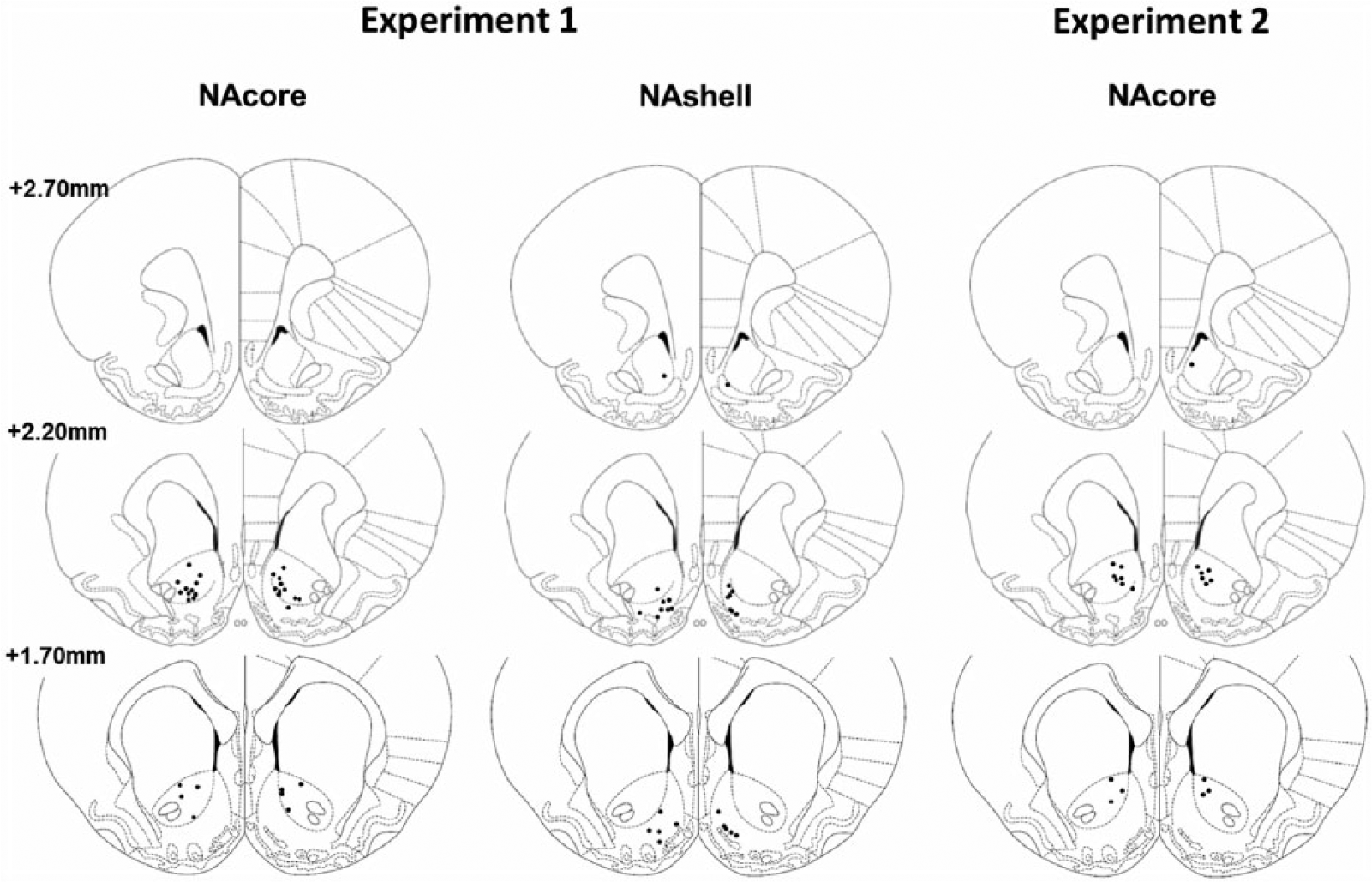
Verification of DBS electrodes’ placement in either the NAcore (Experiments 1 and 2) or NAshell (Experiment 1) at the level of 2.70, 2.20 and 1.70 mm rostral to bregma. Rats with placements of DBS electrodes at 2.70 mm rostral to bregma in the NAcore group were excluded from analysis.
In Experiment 2, 3 of the 16 rats died unexpectedly during surgery, 2 rats had misplaced electrodes, 1 rat lost his DBS electrodes during acquisition training and 1 rat had a clogged IV catheter. These rats were excluded from all analyses, resulting in n = 9.
Experiment 1
Acquisition of heroin self-administration
Apart from one rat with a misplaced electrode in both groups, four rats lost their electrodes during acquisition training and one rat suffered from IV catheter failure, resulting in n = 10 in NAcore group. In the NAshell group, three rats had a clogged IV catheter and two rats lost their electrodes, resulting in n = 10. All rats readily acquired heroin self-administration on an FR1 schedule and learned to discriminate between active and inactive nose poke holes (session F(10,180) = 42.92, ε = 0.62,
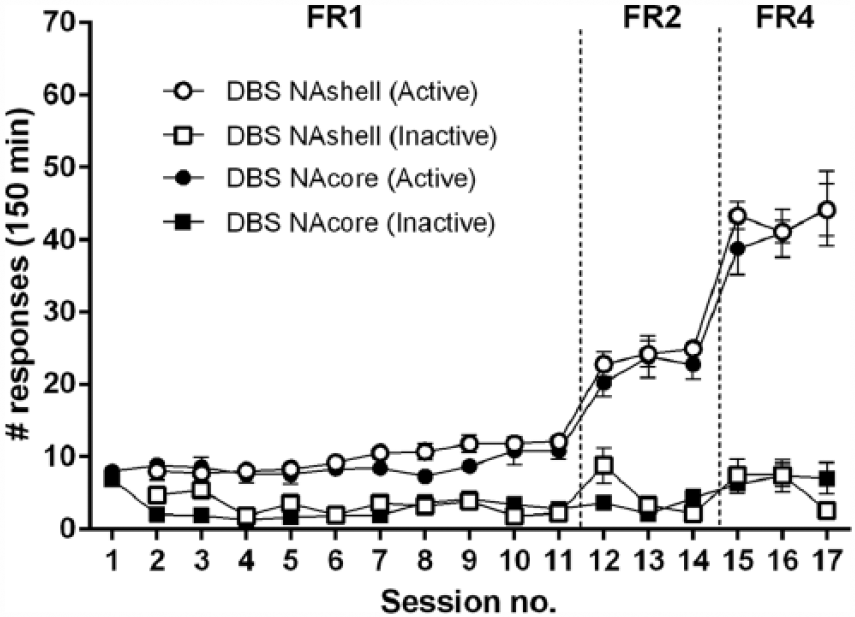
Number of active (circles) and inactive (squares) responses during acquisition of heroin self-administration for NAcore (black symbols; n = 10) and NAshell (white symbols; n = 10) DBS implanted rats (Experiment 1).
NAcore DBS and NAshell DBS during FR4 schedule of reinforcement
None of the amplitudes of DBS applied to NAcore or NAshell affected responding for heroin on the regular dose of 100 µg/kg/infusion (active responses: NAcore DBS F(3,27) = 1.50, n.s., NAshell DBS F(3,27) = 1.22, n.s.; inactive responses: NAcore DBS F(3,27) = 1.38, n.s., NAshell DBS F(3,27) = 0.44, ε = 0.78, n.s.; rewards: NAcore DBS F(3, 27) = 1.47, n.s., NAshell DBS F(3, 27) = 1.27, n.s.; Figure 3(a) and (c)). Similarly, 100 µA DBS did not influence responding for heroin on a lower dose of 25 µg/kg/infusion in both NAcore and NAshell stimulated rats (active responses: NAcore DBS F(1,6) = 3.08, n.s., NAshell DBS F(1,7) = 1.83, n.s.; inactive responses: NAcore DBS F(1,6) = 0.52, n.s., NAshell DBS F(1,7) = 2.72, n.s.; rewards: NAcore DBS F(1, 6) = 3.07, n.s.; F(1, 7) = 1.78, n.s.; Figure 3(b) and (d)). During test days of 25 µg/kg/infusion, three additional rats were removed from the analyses, due to loss of DBS electrodes, catheter failure and technical issues during test days, resulting in n = 7. In addition, two rats of the NAshell group did not receive heroin properly during test days, resulting in n = 8 for this group.

DBS during FR4 schedule of reinforcement did not affect the number of active and inactive responses and rewards. (a
NAcore DBS and NAshell DBS during cue-induced and drug-induced reinstatement
To explore the effects of NAcore and NAshell DBS on reinstatement of heroin seeking, rats were tested for cue-induced and drug-induced reinstatement. Specifically, NAcore DBS was able to decrease reinstatement responding when conditioned cues were presented (0 µA DBS versus 100 µA DBS t(7) = 2.44,
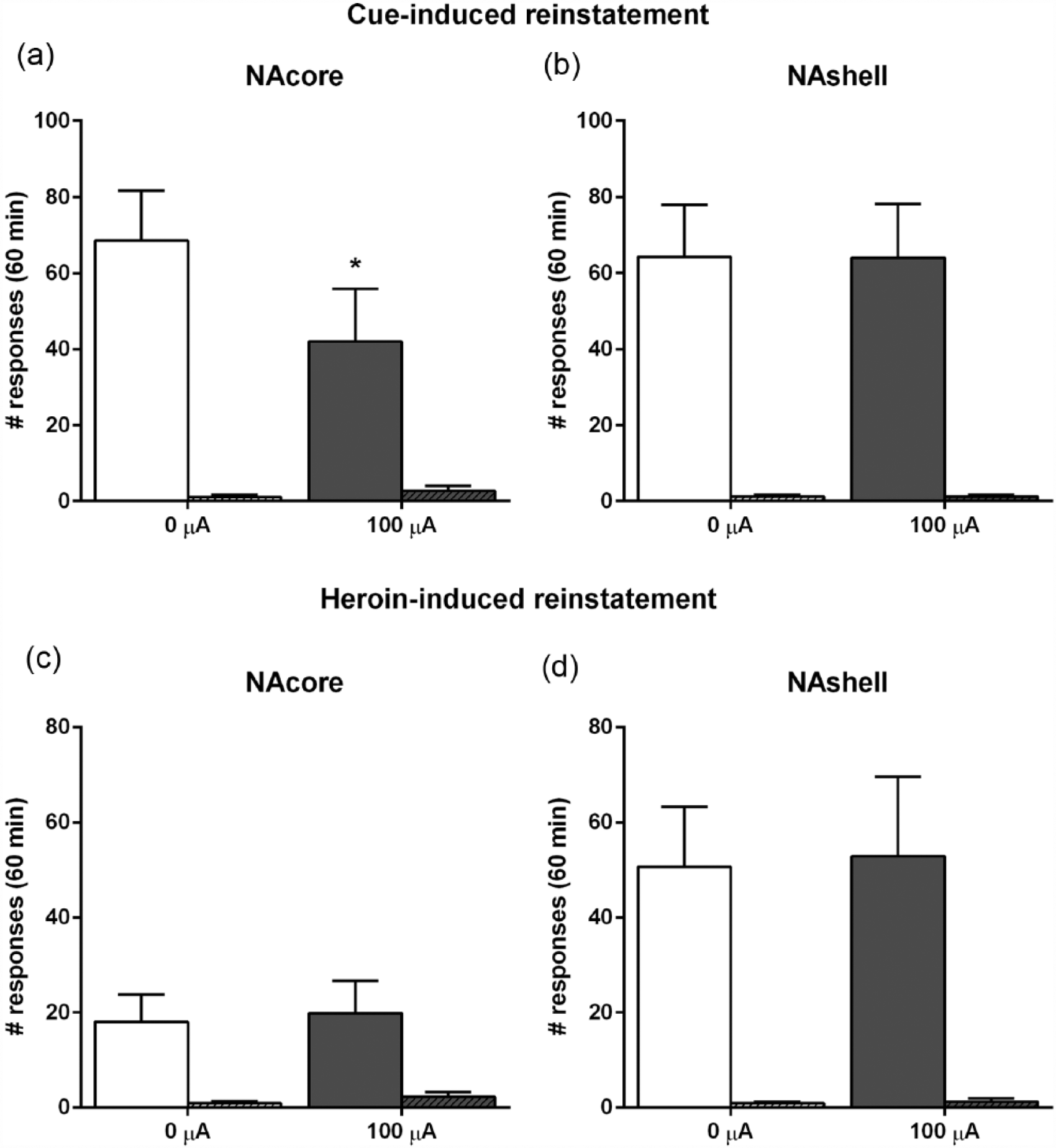
Number of responses in the active and inactive (hatched bars) holes following exposure to heroin-associated cues when stimulated in the (a) NAcore (n = 8) and (b) Nashell (n = 8) and following heroin priming for (c) NAcore DBS (n = 8) and (d) NAshell DBS (n = 8).
In contrast, NAcore DBS during drug-induced reinstatement did not alter responding in the previously active hole (0 µA DBS versus 100 µA DBS t(7) = −0.19, n.s.) and inactive hole (0 µA DBS versus 100 µA DBS t(7) = −0.23, n.s.). Likewise, NAshell DBS did not change drug-induced reinstatement (active: 0 µA DBS versus 100 µA DBS t(7) = −0.12, n.s.; inactive 0 µA DBS versus 100 µA DBS t(7) = −0.63, n.s.; Figure 4(c) and (d)).
Experiment 2
Acquisition of heroin self-administration
All rats readily acquired heroin self-administration on an FR1 schedule and learned to discriminate between active and inactive nose poke holes (session F(10,80) = 2.542,

Number of active (circles) and inactive (squares) responses during acquisition of heroin self-administration for NAcore (n = 9) DBS implanted rats (Experiment 2).
NAcore DBS during PR schedule of reinforcement
Upon stable baseline responding on an FR4 schedule, rats were trained and tested on a PR schedule with three different DBS intensities. Repeated measures ANOVA on the number of active responses revealed a significant effect of DBS treatment (F(3,24) = 3.25,
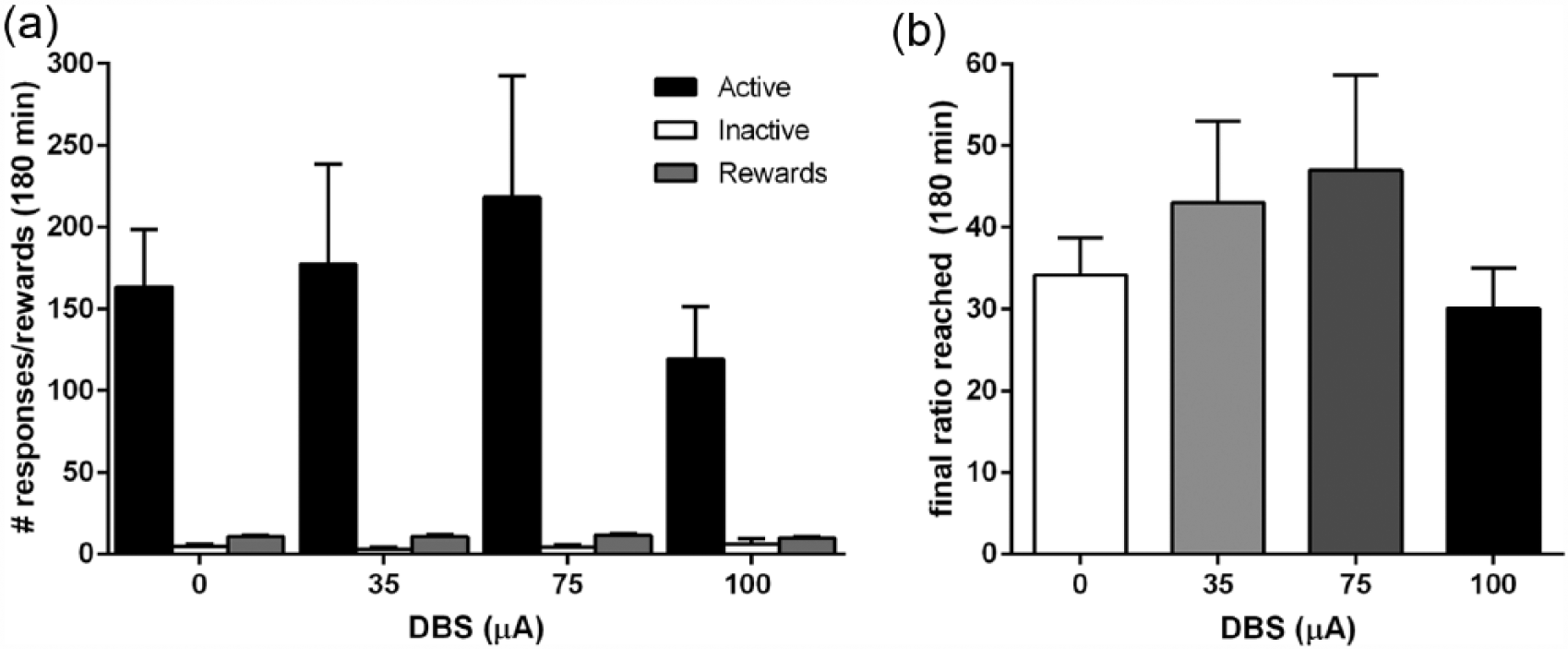
(a) NAcore DBS effects on number of active and inactive responses and obtained rewards and (b) final ratio reached on a progressive ratio schedule of reinforcement (n = 9).
NAcore DBS during first day of extinction
To assess NAcore DBS effects on the first day of extinction learning, half of the rats were stimulated with 100 µA during the first day of extinction. An independent t-test on the number of active responses revealed a significant difference between stimulated and non-stimulated rats (t(9) = 2.97,

NAcore DBS during first day of extinction. (a) Total number of active and inactive (hatched bars) responses and (b) within-session time-course of number of active responses for stimulated (100 µA, DBS n = 6) and non-stimulated (0 µA, DBS n = 5) rats. (c) Extinction of heroin seeking in stimulated and non-stimulated rats; stimulation was applied during extinction session 1 only (grey bar).
Discussion
This study aimed at exploring the effects of NAcore and NAshell DBS on different aspects of heroin taking and -seeking behaviour. Previous studies have reported positive effects of NAcore DBS on opioid seeking (Guo et al., 2013; Liu et al., 2008; Ma et al., 2013; Yan et al., 2013). However, these studies have solely reported the effects of long-term stimulation either during the CPP conditioning phase (Liu et al., 2008; Yan et al., 2013) or during multiple days of extinction or abstinence (Guo et al., 2013; Ma et al., 2013) prior to assessment of relapse sensitivity. To our knowledge, this is the first study that reports direct effects of NAcore and NAshell DBS on heroin taking and -seeking, providing the opportunity to assess the immediate effect of DBS on specific components of addictive behaviour.
The results show that heroin seeking is selectively attenuated by NAcore DBS and is unaffected by NAshell DBS. More precisely, NAcore DBS facilitated extinction learning on the first extinction day and reduced drug-seeking responses during a cue-induced reinstatement test and attenuated responding during a PR schedule of heroin reinforcement. These findings appear not secondary to changes in motor behaviour since (1) responding in the inactive hole was not impaired throughout the experiments, (2) responding for heroin on an FR4 ratio was not affected by DBS and (3) rats were still able to adapt responding to a change in heroin dose. The latter observations suggest that NA DBS does not influence the direct pharmacological effects of heroin. This study substantiates the clinical efficacy of DBS for heroin dependence (Valencia-Alfonso et al., 2012).
Differential role of NAcore and NAshell in expression of heroin-seeking behaviour
The lack of effect of DBS on heroin intake suggests that NAcore and NAshell are not critical for heroin self-administration, which is in line with lesion and local inactivation studies showing that neither region appears essential in mediating the rewarding effect of heroin (Alderson et al., 2001; Hutcheson et al., 2001; Schlosburg et al., 2013; Walker et al., 2000). On the other hand, the present data indicate a clear distinction between NAcore and NAshell DBS in modulating reinstatement of heroin seeking, which is in line with previous pharmacological intervention studies. For example, intracranial administration of the dopamine antagonist SCH23390 or gamma-aminobutyric acid (GABA) agonists baclofen and muscimol decreased discrete cue-induced reinstatement of heroin seeking when infused into the NAcore, but not NAshell (Bossert et al., 2007; Rogers et al., 2008). The NAshell has, however, been suggested to play a role in context-induced reinstatement of heroin seeking (Bossert et al., 2012), a measure that was not determined in this study. Collectively, these studies indicate that the NAshell does not seem to play a role in the expression of heroin-seeking behaviour in response to discrete cues or drug priming. This corresponds with the present results, in which a presumed DBS-mediated inactivation of the NAshell did not alter responding for heroin or reinstatement to heroin seeking, despite the potential reduction in neuronal firing in close proximity of the DBS electrodes (Hu et al., 2011).
The NAcore has been shown to be primarily involved in mediating learned behaviour in response to cues predicting motivationally relevant events (Ambroggi et al., 2011; Corbit and Balleine, 2011; Jones et al., 2010; Theberge et al., 2010), such as the cue and light stimuli related to the incentive effects of heroin in the self-administration paradigm. Heroin-associated cues are shown to play an important role in the maintenance of drug use and relapse (Carter and Tiffany, 1999; Preller et al., 2013), also after a long period of abstinence (Preller et al., 2013). In line with this, it has been reported that cue-reactivity as measured with intracranial electroencephalography (EEG) recordings was related to NA DBS effectiveness for heroin dependence at the same contact points of the DBS electrodes (Valencia-Alfonso et al., 2012). The clinical efficacy of DBS in heroin use might thus be attributed to successful interference with cue-conditioned behaviour.
Mechanism of DBS
Interestingly, in previous work, NAcore DBS applied prior to reinstatement tests was found to attenuate both cue-induced and heroin priming–induced reinstatement (Guo et al., 2013). Two important methodological differences between the former study and this study are the duration and timing of DBS application. In the study of Guo et al. (2013), DBS was applied during the extinction phase and terminated before reinstatement testing, whereas in this study DBS was only applied during the reinstatement test. A possible explanation is that only repeated or long-term NAcore DBS is effective in decreasing heroin priming–induced reinstatement as opposed to a single 1-h stimulation. In fact, long-term NAshell DBS seems to be effective in reducing morphine seeking (Ma et al., 2013) and alcohol taking (Wilden et al., 2014), contradicting the null effects of acute NAshell DBS in this study.
A particular point of note is that local accumbal inactivation does not seem to translate directly to all effects obtained with DBS. In this regard, for instance, a decrease in heroin priming–induced reinstatement by both NAcore and NAshell inactivation by intracranial baclofen–muscimol infusion or lesions has been reported (Rogers et al., 2008; Wang et al., 2002). Emerging evidence suggests that DBS does not only alter regional activity but also induces global changes in neuronal network activity, and therefore should not be directly compared to local intracranial drug manipulations or lesions. Evidence for this view comes from extensive research on subthalamic nucleus DBS as a treatment for Parkinson’s disease, which shows antidromic activation and neurotransmitter release in structures connected to the subthalamic nucleus following DBS (Deniau et al., 2010), such as the globus pallidus (Benazzouz et al., 1995) and striatum (Gubellini et al., 2006). A similar network influence is suggested for NA DBS (Holtzheimer and Mayberg, 2011), with particular focus on the frontostriatal network. Indeed, acute NAcore DBS has been shown to upregulate expression of BDNF, Fos and pERK (Do-Monte et al., 2013; Rodriguez-Romaguera et al., 2012) and to increase dopamine and serotonin release in the medial prefrontal cortex (mPFC; Van Dijk et al., 2013), without altering local monoamine release (Van Dijk et al., 2011). In further support, increased blood oxygen level–dependent
Although speculative, potential antidromic DBS-induced inactivation of the PFC and amygdala might explain the decreased responding to motivationally relevant cues. The PFC and amygdala, together with the NAcore region, are well known to be strongly involved in cue-conditioned responding for heroin (Daglish et al., 2001; Fanous et al., 2013). Glutamate release from the PFC to the NAcore appears critical for cue-induced and drug-induced reinstatement to heroin seeking (LaLumiere and Kalivas, 2008; Shen et al., 2014). Also, glutamatergic efferent projections from the amygdala to the NAcore are involved in Pavlovian responding to cue-conditioned and heroin-primed reinstatement of heroin seeking (Fuchs and See, 2002; Jones et al., 2010; Rogers et al., 2008; See et al., 2003). In line with these findings, morphine-induced CPP has been shown to increase glutamate levels in the NA, which were reduced by NAcore DBS (Yan et al., 2013). Additionally, glutamate levels in the ventral pallidum (VP) and the ventral tegmental area (VTA) were reduced, whereas GABA levels in NA, VP and VTA were increased by NAcore DBS, thereby reversing the effects of morphine CPP on glutamate and GABA release (Yan et al., 2013). The projections from the NA to the VP are important for motor output, thus stimulation of the NAcore potentially leads to a reduced output to downstream brain areas.
Interestingly, facilitated extinction of heroin seeking by NAcore DBS is consistent with a recent finding that NAcore and not NAshell DBS is able to decrease extinction of fear memories (Rodriguez-Romaguera et al., 2012). This is in support of the hypothesis that extinction of drug-related memories and fear-related memories comprises a similar neural circuitry with overlapping pathways in the mPFC (Peters et al., 2009). In addition, this adds to the increased evidence that the therapeutic effect of NAcore DBS is (partly) due to modulation of mPFC activity.
The present findings extend previous work in a CPP (Liu et al., 2008) and a self-administration paradigm with opioids (Guo et al., 2013) and two bottle choice paradigm (Knapp et al., 2009), alcohol sensitisation (Nona et al., 2015) and alcohol consumption after a period of abstinence (Henderson et al., 2010), confirming that NAcore DBS is effective in decreasing drug seeking. However, the results contrast those of Vassoler et al. (2013) who report that NAshell but not NAcore DBS attenuates cocaine-primed reinstatement. This discrepancy could be explained by the involvement of partly distinct circuits thought to be involved in cocaine and opioid dependence (Badiani et al., 2011). Reinstatement of heroin seeking seems to entail a wider variety of brain regions compared to cocaine seeking (Rogers et al., 2008) and might therefore be differentially influenced by DBS.
Conclusion
In conclusion, the present results build on previous work pinpointing NAcore DBS as a promising intervention for treatment-resistant heroin dependence by reducing cue-induced responding for previously motivational relevant events (Valencia-Alfonso et al., 2012). NAcore DBS was found to selectively decrease cue-conditioned responding for heroin, whereas NAshell DBS did not alter heroin taking or -seeking. This is in line with the distinct functional roles of NAshell and NAcore in heroin seeking. Considering the fact that conditioned cues have shown to be very powerful stimuli for the persistence of SUD and relapse to drug use (Carter and Tiffany, 1999), the present findings that NAcore DBS decreases cue-conditioned behaviour are very promising.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was funded by ZonMW grant no. 31160204.
