Abstract
Sensorineural hearing loss due to ototoxic drugs remains as a conflict as the treatment option with aminoglycosides. Ototoxic mouse model was produced with the administration of ototoxic drugs aminoglycoside kanamycin and loop-diuretic furosemide, thus validation of auditory function of the mouse model is needed to determine the efficacy of the drugs. Kanamycin sulfate 550 mg/kg (VWR life sciences, PA, USA) and furosemide 130 mg/kg (Lasix, Handok, Korea) were administered through subcutaneous and intraperitoneal injection respectively. Auditory brainstem response and distortion otoacoustic emission tests were performed on days 3,5,7,10,14 post administration of the ototoxic drug. Thresholds in response to the stimulus given in the auditory brainstem recordings and distortion otoacoustic emission tests were obtained. The hearing threshold shift to high stimulus intensity was observed post administration of the ototoxic drug. Latency of the ABR peak waves were recorded and analyzed, latency delay was observed as hearing threshold increases. These findings will further support in the application of this animal model in various studies regarding ototoxic hearing loss.
Keywords
Introduction
It has long been known that the major irreversible toxicity of aminoglycosides is ototoxicity. 1,2 Aminoglycosides have variable cochleotoxicity and vestibulotoxicity. 3 Streptomycin and gentamicin are primarily vestibulotoxic, whereas amikacin, neomycin, dihydrostreptomycin, and kanamycin are primarily cochleotoxic. We demonstrated, in our previous study, “one-shot” injection of a combination of kanamycin and furosemide in the mouse model of ototoxicity could be a novel technique to induce local inner ear injury. 4 This mouse model has demonstrated hearing loss, as the hearing threshold was recorded 70 dB in the auditory brainstem response (ABR) test with click stimulus within 7 days of injection.
Auditory-evoked potentials originating from the brainstem in mice are widely used due to similarity to those in humans, however different strains of mice have unique responses to ototoxic drugs. 5 Standardization of stimulation and recording parameters in auditory functional tests, including the auditory brainstem response (ABR) and distortion product otoacoustic emission (DPOAE), has not been achieved in many species of mice, including the mouse model with ototoxic drugs. 6
In this study, a combination of an aminoglycoside, kanamycin, and a loop diuretic, furosemide was used to induce ototoxic hearing loss through a single-dose regimen in C57BL/6J strain mouse, which is frequently used as a model in auditory research because of its susceptibility to aging and ototoxicity. 7 This study aims to establish a database on the ototoxic hearing loss pattern in this mouse model through the ABR and DPOAE samples collected.
Materials and method
Animals
Twenty male C57BL/6J mice were subjected to ABR under anesthesia. During the recording time, the mice were 5 to 7 weeks old, weighing between 25 and 30 grams; they were given free access to food and water. The recordings were performed in the animal laboratory of Yonsei University Wonju College of Medicine in Wonju, Korea in accordance with the policies of the institutional animal care and use committee (YWC-180703-1).
Ototoxic drug
Kanamycin sulfate 550 mg/kg (VWR life sciences, PA, USA) and furosemide 130 mg/kg (Lasix, Handok, Korea) were administered via subcutaneous and intraperitoneal injection, respectively. 4 The mouse in the control group received saline through subcutaneous injection followed by intraperitoneal injection.
ABR procedure
Prior to ABR recordings, the mice were anesthetized with 100 mg/kg ketamine (Yuhan, Seoul, Korea) and 10 mg/kg xylazine (Rompun, Bayer, Ansan, Korea) by intraperitoneal injection. The anesthetized mice were tested in a sound attenuating chamber with a built-in Faraday cage. An isothermal pad was used to maintain the body temperature of the test subject. TDT RZ6/BioSigRZ system (Tucker Davis Technologies, Alachua, FL, USA) was used for stimulus generation, data management, and ABR collection.
Twelve-millimeter-long, gauge 27 subdermal needle electrodes (27GA 13 mm, Rochester Electro-Medical, USA) were used to record the ABR (Figure 1(a)). One channel was recorded: active electrodes were placed in the vertex; reference electrode was placed axial to the pinnae, which is the same side as the stimulus delivery; and ground electrode was placed on the contralateral side. The electrodes were connected with the low impedance headstage (RA4LI, TDT) that interfaces with the TDT amplifier.

Experiment setting. (a) Auditory brainstem response test settings; electrodes were inserted as follows: active electrode (red) at the vertex, reference electrode (black) at the ipsilateral ear to the stimulus, and ground electrode (green) at the contralateral ear. (b) Distortion product otoacoustic emissions test settings; pure tone stimulus from two separate sound sources were given, and the responses were recorded.
Acoustic stimuli were generated by auditory processor (RZ6, Tucker Davis Technologies, Alachua, FL, USA). The stimulus signal and signal response data were acquired by automated processing through BioSigRZ software installed on the PC. Stimuli were delivered in a closed field setting by a magnetic speaker (MF1, TDT, Alachua, FL, USA) with a PVC tubing and a conical cap inserted into the subject’s ear.
DPOAE procedure
Mice were anesthetized prior to the recordings; stimuli were generated using a etymotic research microphone (ER10B+) connected to a pair of MF1 microphones inserted into the subject’s ear canal (Figure 1(b)).
ABR and DPOAE recording
ABR and DPOAE were recorded from the bilateral ears, 1 day prior to administration (Day-1) of the ototoxic drug and on days 3, 5, 7, 10, 14 post administration of the ototoxic drug. Prior to ABR and DPOAE recordings, mice were anesthetized with a mixture of ketamine 100 mg/kg and 10 mg/kg xylazine. The stimuli were given at a maximum of 90 dB to a minimum of 10 dB for clicks and 8, 16, 20, 26, 32 kHz for tone bursts. Ten decibel steps reducing the SPL to obtain the auditory thresholds. Using a pair of MF1 microphones and an etymotic research microphone, ABR recordings were measured, followed by measurement of DPOAE. The stimuli were generated using TDT software.
Statistical analyses
Statistical analyses were processed using GraphPad Prism software. Data obtained from the peak detection were expressed as the mean ± standard error of the mean (SEM) and values from different time points were compared using repeated measures two-way ANOVA. In all tests, the mean had p-value of ≤0.05.
Results
Changes of auditory thresholds in the mouse model of ototoxicity
Waveforms acquired from the ABR recordings of all subjects were similar. Figure 2 illustrates the typical waveforms of ABR recorded from two different types of stimuli—click and 20 kHz tone burst—in a mouse with normal hearing before the injection of the ototoxic drugs. Upon click stimulation, the maximum stimulation intensity was at 90 dB SPL with 10 dB reducing steps to reach 10 dB stimulation. Five distinct positive peaks (I-V) of ABR waves were identified within 7 msec of initiation of stimulus; Peak II showed highest amplitude level and Peak V remained identifiable with low stimulus intensities. For tone burst stimulation, Peak III had the highest amplitude and Peak V remained identifiable with low stimulus intensity. In ABR recordings of both, click and tone burst stimuli, the peaks shifted to the right, which indicates that the latencies of the peak waves were delayed with decreasing stimulus delivery.
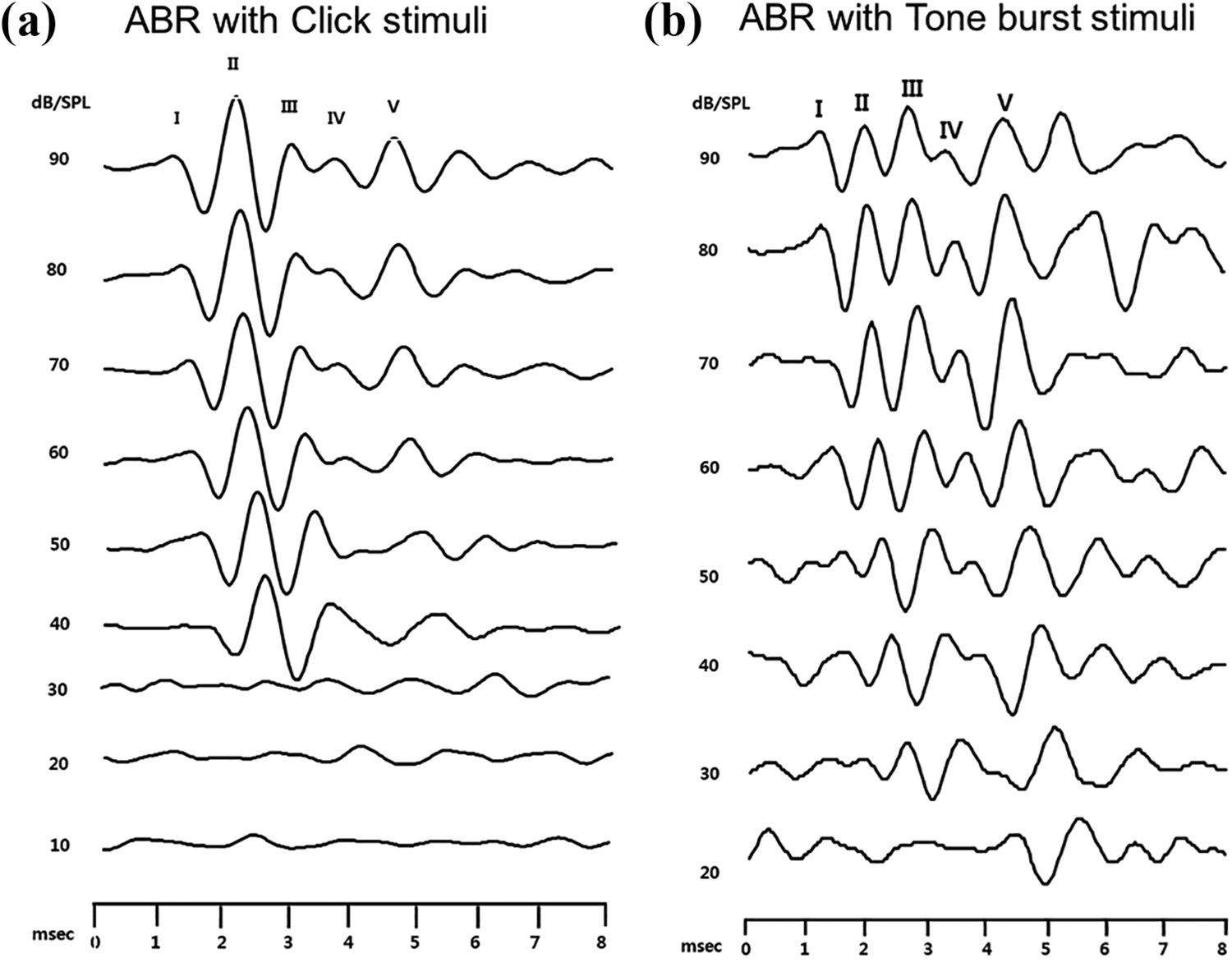
ABR recordings according to click (a) and tone burst 20 kHz (b) in a C57BL/6J mouse with normal hearing prior to ototoxic drug administration. Five peaks after the initiation of acoustic stimulus labeled with Roman numbers, maximum stimuli intensity was at 90 dB SPL.
Figure 3 showed the changes of auditory thresholds in click ABR, 20 kHz tone burst ABR, and DPOAE after administration of the ototoxic drugs up to 2 weeks. The mice model exhibited profound hearing loss over 70 dB thresholds within 7 days of the injection. In tone burst ABR, the greater hearing loss was observed significantly on day 3 at high frequencies (20 kHz and 32 kHz) than at low frequency (8 kHz). After 5 days of hearing loss, there was no difference in frequency specificity. This phenomenon was displayed in DPOAE test. In the 32 kHz, the acute changes of auditory hearing thresholds were shown from the initial days (days 1 and 3) after the injection. The frequency specificity was also present on day 3, and if the injury by ototoxic drugs was severe after 5 days, the hair cells appeared to be damaged from basal turn to apical turn.
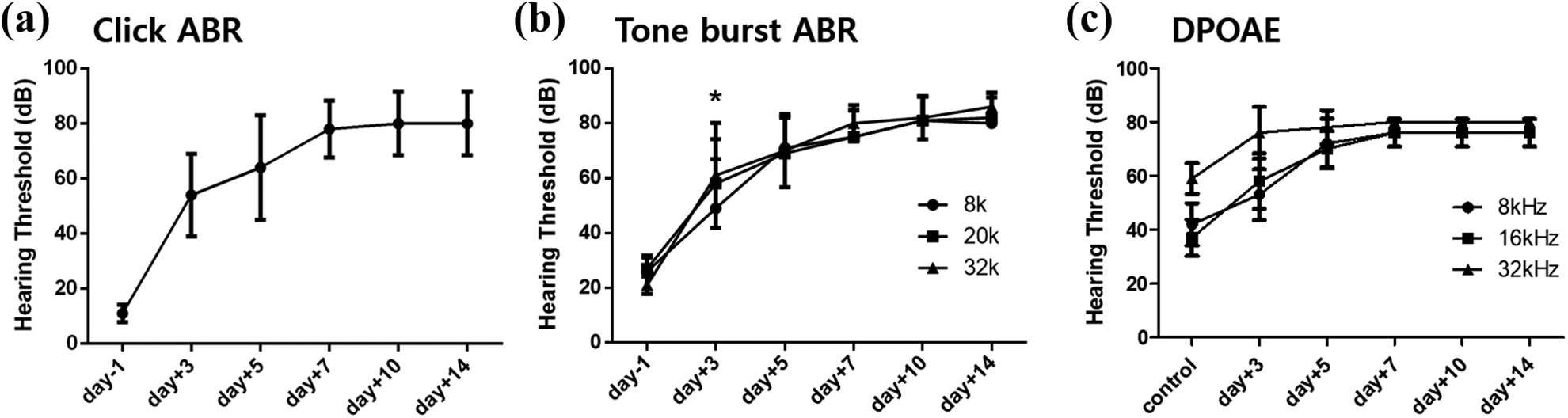
Changes in the hearing threshold of the C57BL/6J mouse pre and post administration of the ototoxic drug in response to click, tone burst, and distortion product otoacoustic emissions. Decrease in the hearing threshold could be observed on day 3 post administration; complete hearing loss noted on day 7 and onward, similar hearing loss pattern could be observed in the three different types of recordings performed.
Changes of latency in peak waves in the mouse model of ototoxicity
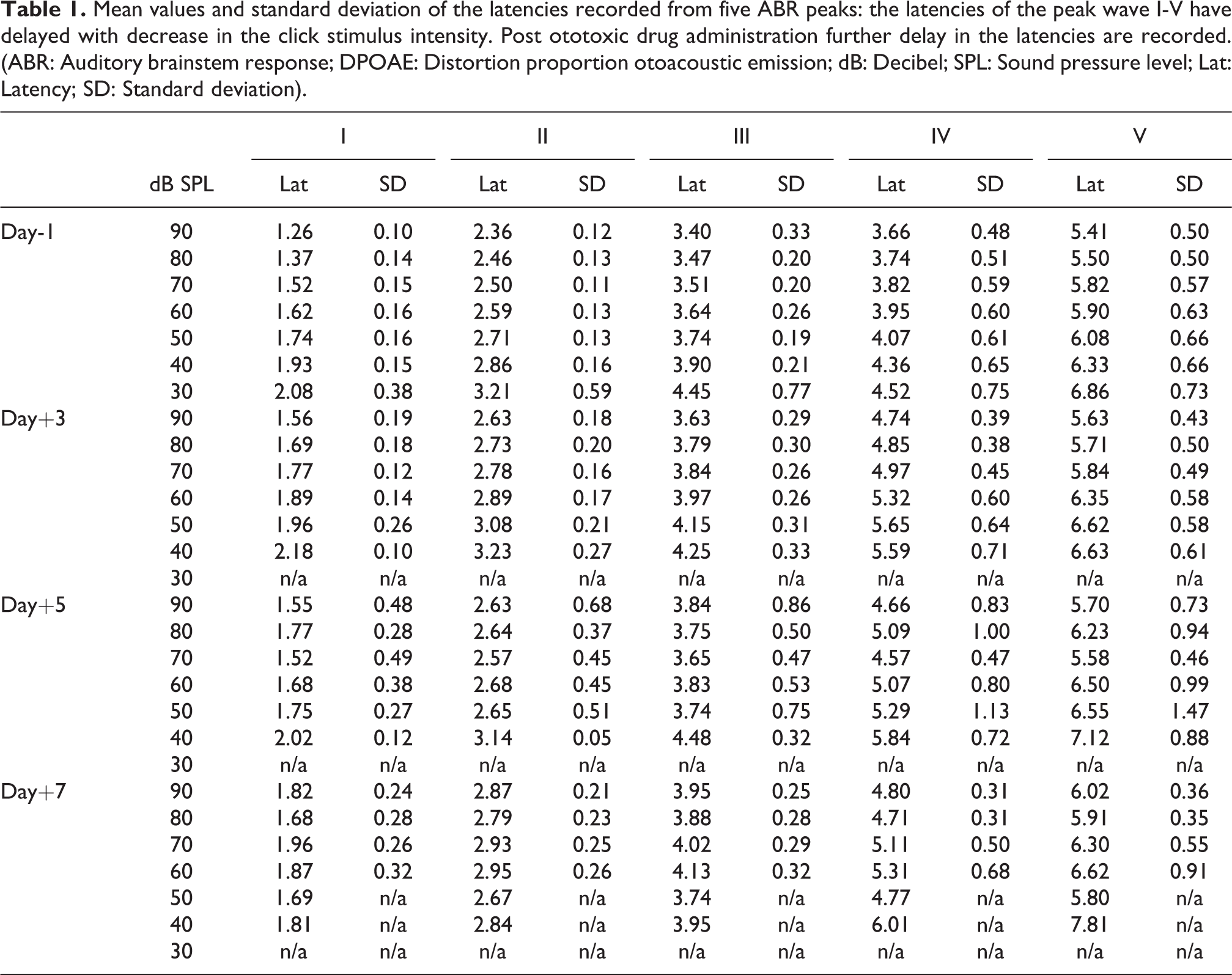
The latencies on each peak wave (I-V) in click ABR were recorded in the subjects pre and post administration of the ototoxic drug kanamycin and furosemide. The latency of peak waves I-V from the day prior to the administration of the ototoxic drug and on days 3, 5, 7 were recorded (Table 1). Distinct loss of hearing was observed on day 5, on which the hearing threshold was at 60dB. The latencies of the peak waves were analyzed in comparison with the normal latency recorded on days prior to administration of the ototoxic drug (Figure 4). (InsertFigure4) The latencies of peak wave I to V increased when decreased click stimulus was given, and the latency of the ABR wave peaks recorded showed delayed latency of the peak waves on day 5 post administration of the drug compared with the same time point in mice with normal hearing. We compared the amplitudes and latencies of peaks II and V (Figure 5) and noted that the latencies on wave peak II and V increased with significance on day 5 post administration of the ototoxic drug; moreover, there were no identifiable wave peaks when low intensity stimulus of 30 dB and below was given. (InsertFigure5)
Mean values and standard deviation of the latencies recorded from five ABR peaks: the latencies of the peak wave I-V have delayed with decrease in the click stimulus intensity. Post ototoxic drug administration further delay in the latencies are recorded. (ABR: Auditory brainstem response; DPOAE: Distortion proportion otoacoustic emission; dB: Decibel; SPL: Sound pressure level; Lat: Latency; SD: Standard deviation).
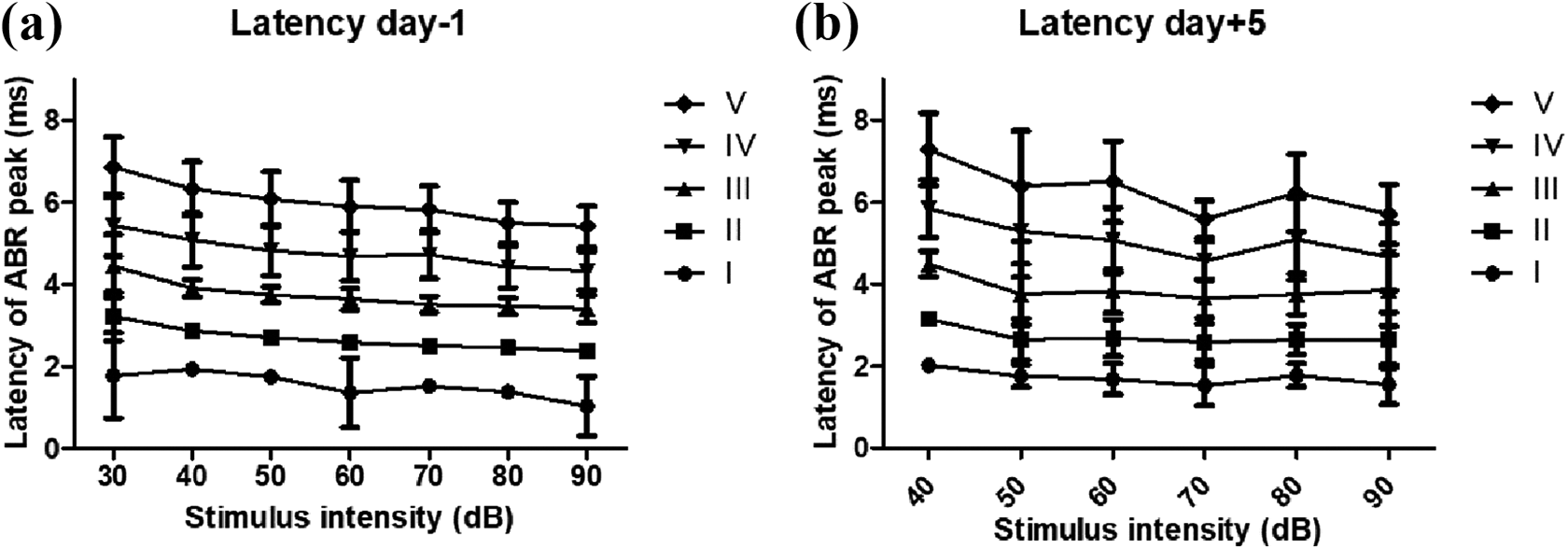
Latency of peak in ABR click stimulus; 1-day prior to administration of kanamycin/furosemide regimen and 5-day post administration of drug. Increased peak intensities were observed in ABR recordings 5 days’ post administration of ototoxic drugs.
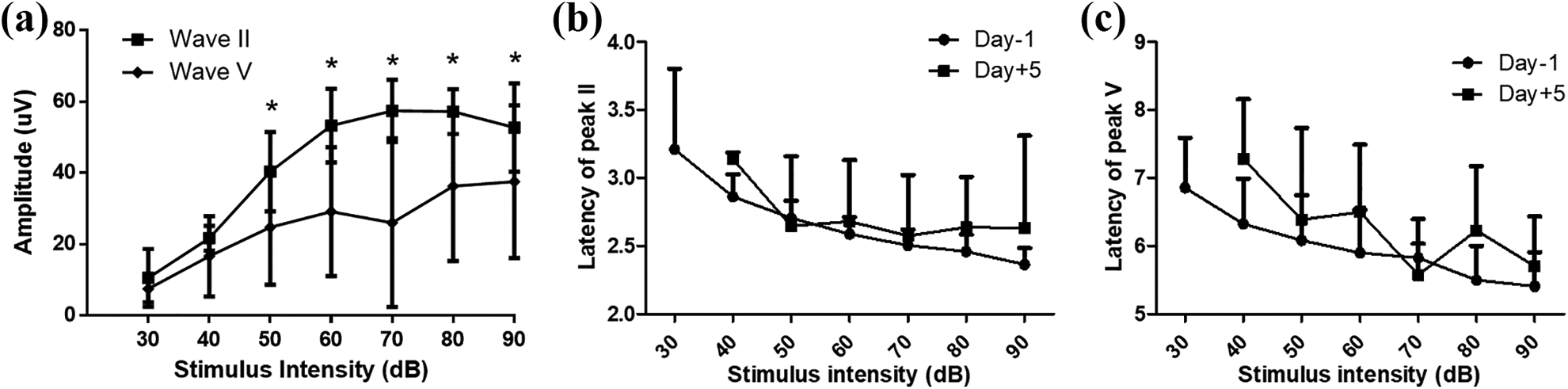
Latencies of two ABR peaks II and V. In comparison to the latencies recorded 1-day prior to drug treatment, the latencies of the ABR peaks were delayed twofold post administration of the ototoxic drugs.
Discussion
Of the various animal models, the mouse model is preferred due to the similarities in the microstructures of the hearing organ of a mouse to those of the hearing organ in humans; moreover, it is an economical model for the experiment. 8 The auditory function tests such as ABR and DPOAE have become a useful and practical procedure for the determination of hearing levels in animals. 9 The ABR patterns of mice typically consist of five vertical positive waves. 10 In Wave I, voltage arises from the cochlea and/or compound action potential of the auditory nerve. Waves II to V reflect the evoked activity at ascending generators in the auditory midbrain and are known to originate from cochlear nuclei, contralateral superior olivary complex, lateral lemniscus, and contralateral lateral inferior colliculus, respectively. 6 However, the values of ABR parameters differ based on the strain of the mouse. 11 Millions of mice are produced annually at the Jackson Laboratory. The Neuroscience Mutagenesis Facility at the Jackson Laboratory has undertaken a large scale auditory screening project. Zhou et al. reported auditory brainstem responses in 10 inbred strains of mice. 11,12 Scimemi et al. reported the normative data reported in C57BL/6J mouse, which can be used as a reference for further investigations on murine models of hearing loss. 6 DPOAE should serve as a useful tool to study the function of outer hair cells (OHCs) on the cochlea. Parham et al. reported the values of DPOAEs recorded in young and aging C57BL/6J mice. 13 In mice, cochlear pathology progresses from base to apex, owing to which DPOAE changes are first seen in the high-frequency region of the cochlea. 14 Although normative data in mice auditory functional tests were published, there are no data for hearing patterns of the pathologic mouse model, especially the mouse model of ototoxicity. In the previous study, we suggested a mouse model established with a “one-shot” injection of a combination of ototoxic drugs, kanamycin and furosemide; in this study, we have arrived at a database to establish the ototoxic hearing loss pattern in this mouse model through ABR and DPOAE tests.
The threshold, amplitude, and latency analysis of the ABR provides information on the peripheral hearing status and the integrity of brainstem pathways. A click stimulus covering a wide frequency band is a commonly used stimulus to evaluate ABR. The click stimulus sound and the 4 kHz stimulus sound among the tone burst stimulus were compared in the mouse; the waveforms were similar but showed differences in latency. 15 Thus, we have applied click stimuli in our study to evaluate the changes in latency. The latency in the C57BL/6J mouse is similar to that of other mice in the previous study 11,16 and can be considered a baseline to evaluate the rate of transmission of auditory signals modeled on mice or the function of the central nervous system. The amplitude of waves I and V in C57BL/6J mouse increased monotonically with increasing intensity, which is similar to what is commonly used in evoked potential studies. 11 Burkard et al. reported that the slope of latency–intensity functions of waves I and V were ∼8 to 9 μs/dB in gerbils but ∼13 to 16 μs/dB in rats when examined under click stimulation conditions. 12 In this study, the slope of latency–intensity function of wave II ranged from 2.36 to 3.21 μs/dB whereas that of wave V ranged from 5.41 to 6.86 μs/dB. When considering wave I-V peak latency, the peak latency of waves decreased with increasing click intensity stimulus.
The ototoxicity of aminoglycoside antibiotics has been well established in mice experiments. Furosemide and other loop diuretics have well-known synergistic effects with aminoglycoside antibiotics when the two drugs are administered together and cause profound hearing loss. 17,18 Upon combined treatment with each drug, i.e. after administration of an aminoglycoside antibiotic followed by a loop diuretic, complete OHC loss with IHC damage has been observed in our previous study. 4 This mouse model could be further used in the area of ear science research. The utilization of this mouse model to validate standard values by auditory function tests like ABR and DPOAE could be helpful to researchers in a similar field of study.
Conclusion
Establishment and validation of the hearing loss pattern in a mouse model of ototoxicity is much needed for researchers to determine auditory function in mice. Thus, these findings could support research that utilizes a one-shot mouse model.
Footnotes
Author contributions
YJA: conception and design, collection and assembly of data, data analysis and interpretation, manuscript writing; JSC: data analysis and interpretation; DHK: collection and assembly of data; TB: collection and assembly of data; YJS; administrative support, collection and assembly of data, data analysis and interpretation, manuscript writing, final approval of manuscript.
AuthorAvailability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Animal studies were performed in accordance with the policies of the institutional animal care and use committee (YWC-180703-1).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (No. NRF-2020R1A2C1009789) and by National Information Society Agency(NIA) funded by the Ministry of Science, ICT.
