Abstract
Objectives:
Systemic sclerosis is a rare autoimmune connective tissue disease characterized by progressive skin and organ fibrosis, and diffuse fibro-proliferative vascular modifications. Roughly 2%–10% of systemic sclerosis cases develop during juvenile years (under 18 years of age); however, there are limited data on the incidence and prevalence of juvenile systemic sclerosis. This analysis assessed the incidence and prevalence of juvenile systemic sclerosis in the United States, overall and by age group.
Methods:
Juvenile systemic sclerosis patients with ⩾2 medical diagnostic claims for systemic sclerosis within a 1-year period were identified from the Optum de-identified Clinformatics® Data Mart Database between 2007 and 2021, using International Classification of Diseases diagnostic codes. Incidence and prevalence rates were estimated in the overall population and in a subgroup of patients with at least one filled prescription or infusion of methotrexate, mycophenolate mofetil or cyclophosphamide. Estimates were sex- and/or age-adjusted according to the 2020 US census.
Results:
Forty-eight incident and 103 prevalent cases were identified. Overall, the age- and sex-adjusted incidence and prevalence of juvenile systemic sclerosis were 2.4 per million person-years and 12.0 per million, respectively. When stratified by age group, incidence and prevalence rates increased with age in the overall and subgroup analyses.
Conclusion:
In this analysis, incidence and prevalence rates of juvenile systemic sclerosis increased with age. Increasing incidence and prevalence were observed from 10 years old and from 6 years old, respectively. While the overall data are broadly consistent with previous reports, these data provide new information on the estimated incidence and prevalence of juvenile systemic sclerosis by age group.
Plain language summary
Systemic sclerosis (SSc) is an autoimmune disease, which means the immune system attacks healthy body tissue by mistake. SSc causes thickening/scarring of tissue (known as fibrosis) in the skin, lungs and other organs, as well as changes in blood vessels. Around the world, SSc is rare; however, the impact SSc has on patients with the disease is high. While most patients with SSc are diagnosed in their adult years, there are a small number of patients diagnosed before the age of 18 years (juvenile SSc [jSSc]).
Little is known regarding the number of patients diagnosed with jSSc and even less is known about the number of patients by age group. Here we assessed the incidence (number of newly diagnosed patients over a given time period) and prevalence (number of new and existing cases at a given time point) of jSSc.
In this analysis, patients with jSSc were identified from a database of medical insurance claims between 2007 and 2021 in the United States. The incidence and prevalence of jSSc overall, and in patients who had been prescribed medicines to suppress their immune system that are commonly used to treat SSc, were assessed.
Cases were analysed overall and by age group (0–<6, 6–<10, 10–<12 and 12–<18 year olds). There were very few cases of jSSc overall (48 cases in the incident group and 103 in the prevalent group). In each group, the number of jSSc cases increased with age, with most cases found in patients aged 12–<18 years (34 in the incident group and 79 in the prevalent group). These findings were similar in the subgroup analysis of patients who had been prescribed medicines to suppress their immune system.
This analysis highlights the rarity of jSSc and provides more comprehensive information for healthcare professionals about which age groups are most likely to have the disease.
Introduction
Systemic sclerosis (SSc) is a rare1,2 autoimmune connective tissue disease, 3 characterized by progressive skin and organ fibrosis, and diffuse fibro-proliferative vascular modifications.4,5 Globally, the incidence and prevalence of SSc are relatively low, estimated at 86.4 per million person-years (95% confidence interval (CI) 17.8, 235.7) and 188.7 per million (95% CI 15.5, 252.8), respectively; 3 however, the disease burden is high. 6 SSc is commonly diagnosed in patients between 30 and 60 years old; however, it can occur in children (juvenile SSc (jSSc)).7,8
It is estimated that between 2% and 10% of all patients with SSc develop the disease during their juvenile years.1,2,9 –13 In patients with jSSc, previous studies suggest that the median (interquartile range [IQR]) age at onset of Raynaud’s phenomenon (RP), one of the earliest symptoms of SSc, 14 is around 10.4 years (7.3–12.9), with the median (IQR) age at onset of first non-Raynaud’s symptoms being around 10.9 years (7.4–13.2). 15
It is important for paediatricians to recognize, diagnose and treat SSc as early as possible as the 5-year mortality rate of patients with jSSc is approximately 7.5%. 16 jSSc can lead to complications later in life, including pulmonary arterial hypertension; 9 however, studies have demonstrated that adults who develop SSc during their juvenile years have better survival rates than those who develop SSc as adults,9,17 highlighting the importance of early recognition and intervention. This could potentially be due to survival bias, since patients with jSSc included in these studies may have been adults at the time of data collection; further research is needed to assess this fully.
There are limited data published on the epidemiology of jSSc.5,10,18,19 A 2018 study by Beukelman et al. 10 using International Classification of Diseases (ICD)-9 codes and administrative claims data estimated the prevalence of jSSc in the United States (2010–2014) to be three per one million children under 16 years of age. A physician survey in the United Kingdom and Ireland conducted between 2005 and 2007 estimated a yearly incidence of 0.27 per one million children. 18 To our knowledge, there is only one epidemiological study reporting age-stratified prevalence estimates of jSSc. Fernández-Ávila et al. 19 reported prevalence rates in Colombia of 25, 21 and 29 per million people in children aged 0–4 years, 5–9 years and 10–14 years, respectively, increasing to 69 per million people in children aged 15–19 years. To date, no study has assessed both the incidence and prevalence of jSSc stratified by age. In order to better understand the epidemiology of jSSc and whether this differs by age group, we conducted age-stratified analyses of incidence and prevalence using medical claims data in the United States.
Methods
Patient population
Individuals younger than 18 years with ⩾ 2 medical diagnostic claims for SSc within a 1-year period between 2007 and 2021 were identified from the Optum de-identified Clinformatics® Data Mart (CDM) Database. Optum’s CDM population database includes data on over 65 million commercially insured people in the United States and represents a geographically diverse population. The CDM contains data on medical encounters from all healthcare sites (hospital, inpatient, outpatient, emergency room, physician’s office) for all types of provided services, including specialty, preventive and office-based care.
Patients were selected if they had a diagnosis code of 710.1 (Systemic sclerosis) or 517.2 (Lung involvement in systemic sclerosis) using the ICD-9-clinical modification (CM), or a diagnosis code of M34 (Systemic sclerosis) or M34.x (subcategories of Systemic sclerosis) for ICD-10-CM. Use of ICD-10 began on 1 October 2015.
Analyses and adjustments
Incidence and prevalence estimates were age- and/or sex-adjusted to the US population using 2020 US census data. 20 Prevalent cases were identified between 2007 and 2021. Incident cases were identified between 2008 and 2021 to allow a 1-year lookback period to rule out any previous SSc diagnosis. Incidence was calculated as the number of patients diagnosed with SSc during the follow-up period out of the total number of person-years. Person-years were calculated as follows: if the patient had an SSc diagnosis during the follow-up period, they contributed person-years from the index date to the first SSc diagnosis date; however, if the patient had no SSc diagnosis during the follow-up period, they contributed person-years from the index date to the end of the follow-up period. Prevalence was calculated as the number of patients who had an SSc diagnosis at baseline or during the follow-up period out of the total number of patients in the cohort. The observation period was from 2007 to 2021. Incidence and prevalence were estimated overall and by developmental age group (0–<6 years, 6–<10 years, 10–<12 years and 12–<18 years). Age categories were selected based on standard age cut-offs (6 years and 12 years) among children, as well as the median age of symptom onset reported in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry (10.3 years). 6
A subgroup analysis was conducted in patients with at least one filled prescription or infusion of methotrexate, mycophenolate mofetil or cyclophosphamide before the age of 18. The overall estimates were age- and sex-adjusted, while the estimates by age category were only sex-adjusted. A sensitivity analysis was performed in patients who had at least one SSc diagnostic claim (rather than ⩾ 2 claims) during the follow-up period.
Ethics
Institutional Review Board approval is not required for publications citing Optum data. Optum data only contains de-identified health information as described by the Health Insurance Portability and Accountability Act (HIPPA) Privacy Rule. No direct identifiers of individuals, employers, households or providers are included; as such, the study is exempt from patient consent requirements.
Results
Patient demographics
Of the patients with ⩾ 2 claims, we identified 48 incident cases and 103 prevalent cases, which were included in this analysis (Table 1). In both the incident and prevalent cohorts, the majority of patients were female (68.8% and 71.8%, respectively). When looking at ethnicity, the majority were Caucasian (68.8% and 60.2%, respectively). Similar demographics were seen in patients receiving immunosuppressants before the age of 18 years (Table 1). In the sensitivity analysis of patients with one or more recorded medical diagnostic claim for SSc, 231 incident cases and 365 prevalent cases were identified. In the incident cohort (n = 48 cases), at the time of jSSc diagnosis, patients also reported infection, asthma, gastroesophageal reflux, respiratory muscle weakness, depression, malignancy (specifically unifocal Langerhans-cell histiocytosis) and cardiac involvement. The most commonly reported comorbidities were infection (n = 7; 14.6%) and asthma (n = 5; 10.4%).
Patient demographics.
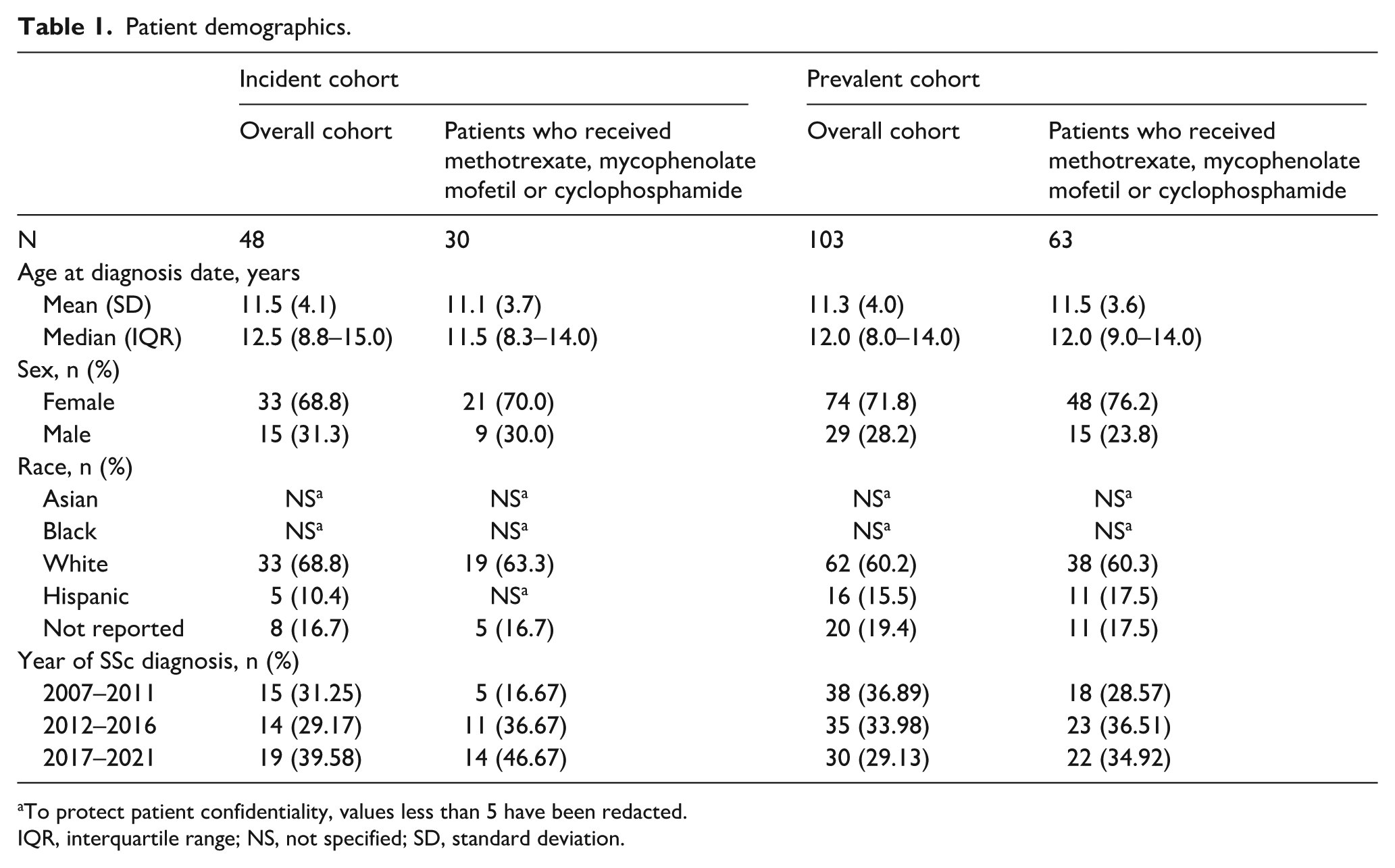
To protect patient confidentiality, values less than 5 have been redacted.
IQR, interquartile range; NS, not specified; SD, standard deviation.
Incidence of jSSc increases with age
The adjusted incidence of jSSc in the overall cohort was 2.4 per million person-years (95% CI 1.68, 3.07). Incidence was observed to increase with age, from 1.2 (95% CI 0.02, 2.27) per million person-years in patients aged 0–<6 years to 4.3 (95% CI 2.88, 5.79) in those aged 12–<18 years (Table 2). A trend of increasing incidence was observed in patients from 10 years old. Differences in incidence were statistically significant when comparing patients aged 12–<18 years with patient groups aged under 10 years (0–<6 years and 6–<10 years).
jSSc age- and sex-adjusted incidence per million person-years.
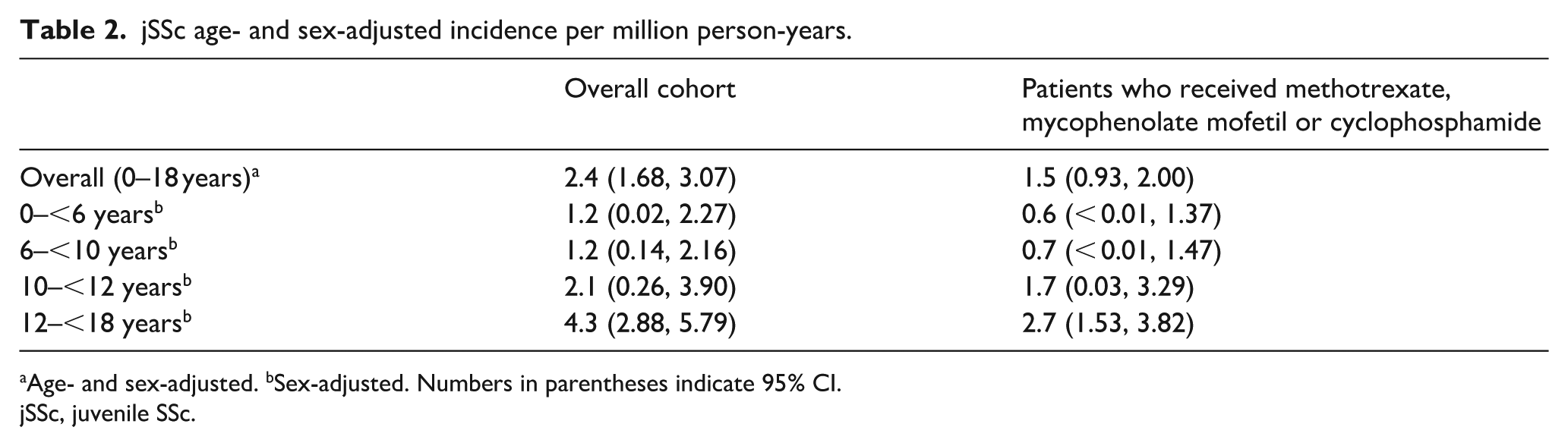
Age- and sex-adjusted. bSex-adjusted. Numbers in parentheses indicate 95% CI.
jSSc, juvenile SSc.
In the incidence cohort, 30 (62.5%) patients received immunosuppressants before the age of 18 years (Table 3). In this subgroup, the adjusted incidence was 1.5 per million person-years (95% CI 0.93, 2.00). A similar trend was seen with incidence increasing from 0.6 (95% CI < 0.01, 1.37) per million person-years in patients aged 0–<6 years to 2.7 (95% CI 1.53, 3.82) in patients aged 12–<18 years (Table 2).
jSSc incident cases.

jSSc, juvenile SSc.
The number of incident cases increased with age in both the overall cohort (4 in patients aged 0–<6 years, up to 34 in those aged 12–<18 years) and in the subgroup of patients who received immunosuppressants (2 in patients aged 0–<6 years, up to 21 in those aged 12–<18 years) (Table 3).
In a sub-analysis of these data, when considering the change in diagnostic coding from ICD-9 (2007 to September 2015) to ICD-10 (October 2015 to 2021), there was no significant change in incidence of jSSc per million person-years.
Prevalence of jSSc increases with age
The adjusted prevalence of jSSc in the overall cohort was 12.0 (95% CI 9.71, 14.36) per million (Table 4). Prevalence was observed to increase with age, from 1.7 (0.35, 3.10) in patients aged 0–<6 years to 26.8 (20.88, 32.70) in patients aged 12–<18 years. A trend of increasing prevalence was observed in patients from 6 years old. Differences in prevalence were statistically significant when comparing patients aged 12–<18 years with patient groups aged under 12 years (0–<6 years, 6–<10 years and 10–<12 years).
jSSc age- and sex-adjusted prevalence per million.
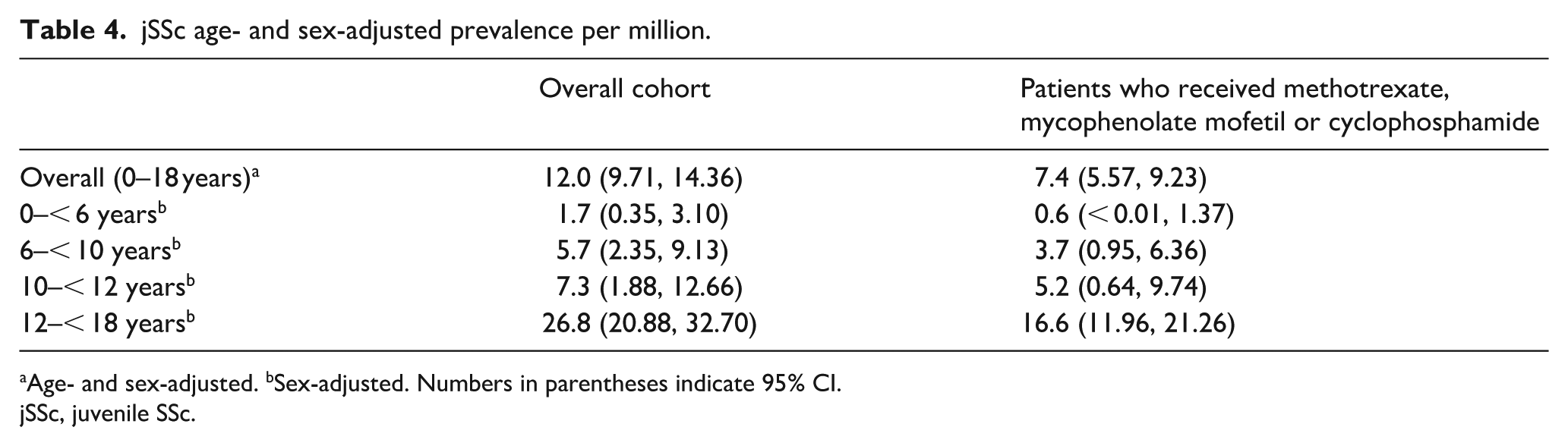
Age- and sex-adjusted. bSex-adjusted. Numbers in parentheses indicate 95% CI.
jSSc, juvenile SSc.
In the prevalence cohort, 63 (61.2%) patients received immunosuppressants before the age of 18 years (Table 5). In this subgroup, the adjusted prevalence was 7.4 (95% CI 5.57, 9.23) per million. A similar trend was seen with prevalence increasing with age group from 0.6 (95% CI <0.01, 1.37) per million in patients aged 0–<6 years to 16.6 (95% CI 11.96, 21.26) per million in patients aged 12–<18 years.
jSSc prevalent cases.

jSSc, juvenile SSc.
The number of prevalent cases increased with age in both analyses. In the overall cohort, there were 6 cases in the 0–<6 years group increasing to 79 cases in the 12–<18 years group, while in patients who received immunosuppressants, there were 2 cases in the 0–<6 years group and 49 in the 12–<18 years group (Table 5).
In a sub-analysis of these data, when considering the change in diagnostic coding from ICD-9 (2007 to September 2015) to ICD-10 (October 2015 to 2021), there was no significant change in prevalence of jSSc per million people.
Age of diagnosis was similar for incident and prevalent cases of jSSc
In the overall population, the mean age of diagnosis was 11.5 years (standard deviation (SD) 4.1) and 11.3 years (SD 4.0) for the incident and prevalent cohorts, respectively (Table 1). In the subgroup of patients who received immunosuppressants, the mean age of diagnosis was 11.1 (SD 3.7) and 11.5 (SD 3.6) years for the incident and prevalent cohorts, respectively.
The median age of diagnosis was 12.5 years (interquartile range (IQR) 8.8–15.0) and 12.0 years (IQR 8.0–14.0) for incident and prevalent cohorts, respectively. The median age of diagnosis was similar to the overall population in the subgroup of patients who received immunosuppressants: 11.5 years (IQR 8.3–14.0) and 12.0 years (IQR 9.0–14.0) for the incident and prevalent cohorts, respectively.
In the incident cohort, there were 33 females and 15 males; the mean age of diagnosis was 11.1 (SD 4.0) and 11.2 (SD 4.4) for the females and males, respectively. In the prevalent cohort, there were 74 females and 29 males; the mean age of diagnosis was 11.0 (SD 3.8) and 10.7 (SD 4.3) for the females and males, respectively.
Discussion
Limited data have been published regarding the estimated incidence and prevalence of jSSc. The current analysis adds important new data on the epidemiology of jSSc in the United States and is, to our knowledge, the first database study to estimate both incidence and prevalence by age group.
The overall age- and sex-adjusted incidence and prevalence estimates of jSSc were 2.4 per million person-years and 12.0 per million in children under 18 years of age, respectively. When assessed by age group, both incidence and prevalence increased with age, with the greatest incidence and prevalence observed in the 12–<18-year-old group. The adjusted incidence of jSSc increased from 1.2 per million person-years in patients aged 0–<6 years to 4.3 per million person-years in patients aged 12–<18 years, with the adjusted prevalence increasing from 1.7 per million in patients aged 0–<6 years to 26.8 per million in patients aged 12–<18 years. Similar results were seen in the subgroup analysis of patients who had been prescribed immunosuppressants. In the overall incident population, the mean age at diagnosis was 11.5 years and the median age was 12.5 years. In the subgroup of patients who received immunosuppressants, the mean and median ages at diagnosis were similar to the overall incident population (11.1 and 11.5 years, respectively). The higher incidence and prevalence of jSSc in children aged 12–<18 years suggests that enrichment for this age group in future paediatric clinical trials in SSc may be warranted. In a sub-analysis of these data, when moving from ICD-9 to ICD-10, there was no significant change in jSSc incidence or prevalence.
Consistent with the current study, an analysis by Beukelman et al. 10 estimated the overall prevalence of jSSc in the United States to be three per million; however, prevalence was not assessed by age group. A study by Fernández-Ávila et al. 19 reported jSSc prevalence rates by age group in Colombia. In patients aged 0–4 years, 5–9 years and 10–14 years, prevalence rates were higher than those observed in the current analysis (25, 21 and 29 per million, respectively), increasing to 69 per million in individuals aged 15–19 years. 19 Fernández-Ávila et al. analysed data from the Colombian health system, which included a high proportion of the total population. The study included patients aged 19 years and younger with a shorter observation period (January 2012 to December 2016) than our study. Furthermore, the case ascertainment strategy differed from that used in the current study, and therefore a direct comparison may not be appropriate.
Prevalence rates from the subgroup analysis of patients with at least one filled prescription or infusion claim for methotrexate, mycophenolate mofetil or cyclophosphamide before 18 years of age are slightly higher than previously published data from Beukelman et al. 10 (7.4 in the current analysis vs 3 per million people in the Beukelman analysis), although the case ascertainment strategies between the two analyses differed. Similar to the current study, Beukelman et al. 10 used the ICD-9 code 710.1 (Systemic sclerosis) to identify patients, but to increase their specificity for SSc, Beukelman et al. excluded any patient with a diagnosis code of localized scleroderma (ICD-9 701.0).
There are differences between the current analysis and the Beukelman et al. 10 analysis. First, Beukelman et al. used Truven MarketScan®, an employer-based commercial insurance claims database (including inpatient and outpatient claims, outpatient prescription claims, clinical utilization records and healthcare expenditures) whereas the current analysis used data from Optum’s CDM population database (which contains data from a single insurance company including commercial and Medicare Advantage plans covering 50 states in the United States). While Optum CDM does include Medicare Advantage plans, all patients included in the current analysis were covered by commercial healthcare insurance. In Beukelman et al., the observation period in the analysis was shorter than the current study (4 years (2010–2014) vs 14 years (2007–2021)). In addition, Beukelman et al. only included patients who had ⩾ 1 medical claim for jSSc, while the current analysis included patients with ⩾ 2 medical claims for SSc on different dates within a 1-year period. Furthermore, Beukelman et al. only included children up to 16 years of age, while the current study included children up to 18 years of age. This could partly explain the increased rate seen in the current study versus Beukelman et al., as cases of SSc are known to increase with age.
Registry studies such as the CARRA 6 and the jSSc inception cohort, 15 which focused on patients who began manifesting symptoms prior to the age of 18 years, provide useful information on the clinical characteristics of individuals with jSSc. The median age at disease onset in the CARRA cohort was 10.3 years (IQR 6.3–13.1), and the median time from disease onset to diagnosis was 0.8 years (IQR 0.14–1.9). 6 In the inception cohort, Foeldvari et al. 15 reported the median (IQR) age at onset of RP to be 10.4 years (7.3–12.9) and the median (IQR) age at first non-Raynaud’s symptoms to be 10.9 years (7.4–13.2), which suggest a slightly earlier onset than the mean and median estimates of jSSc diagnosis in our study (11.5 and 12.5 years, respectively). However, since there was no information available for the time period prior to patient enrolment in the Optum database, it is possible that age at diagnosis of jSSc in our study was overestimated (‘left censoring’), for example, if patients had delayed entry into the database post-diagnosis due to a lack of insurance coverage, different insurance provider or an interruption in enrolment. Furthermore, the report by Foeldvari et al. 15 was limited to patients who developed their first non-Raynaud’s symptoms before the age of 16 years, which will have had an impact on the age of disease onset. In the current analysis, comorbidities were assessed in the incident cohort, with infections and asthma being the most commonly reported.
The rate of patients treated with immunosuppressive agents in our analysis (~60%) was lower than the inception cohort (~90%). 21 This may be due to the nature of the data sources and also how cases were ascertained in these studies. The inception cohort included a set of pre-defined clinical inclusion criteria, 21 while the current study included all individuals younger than 18 years with ⩾ 2 medical diagnostic claims for SSc within a 1-year period between 2007 and 2021 from the Optum CDM database. In addition, sites participating in registry studies may be expert centres with more advanced patients, potentially explaining the difference in immunosuppressive agent prescription between these cohorts. While the inception cohort is aligned with the current analysis as both include patients up to the age of 18 years, there may be differences between the two patient populations. The inception cohort included patients with non-RP symptoms before the age of 16 years, whereas the current analysis included patients younger than 18 years with ⩾ 2 medical diagnostic claims for SSc within a 1-year period between 2007 and 2021. In addition, the current analysis focused on US claims regardless of disease severity, while the inception cohort may have included patients with more advanced SSc requiring specialist input, resulting in a higher percentage treated with immunosuppressant therapies.
A strength of the current analysis is that the database includes over 65 million patients with a wide geographical representation and mixture of demographics. While we did not use a validated algorithm, our analysis potentially has higher specificity than previous analyses, as it uses more stringent eligibility criteria by only including patients with two or more medical claims within a 1-year period, thus reducing the chance of misdiagnoses within the analysis pool.
This analysis has several limitations inherent to the nature of claims data. The CDM database only includes insured patients and thus may limit the generalizability of the results to the overall SSc population in the United States. Furthermore, patient information may be incomplete if patients moved to care settings outside of the CDM database or if they switched to an insurance provider not included in the CDM database. As this analysis required at least 1 year of continuous enrolment, patients with major interruptions in insurance coverage may have been excluded from the study. Prevalence may be a more reliable factor than incidence due to misclassification of newly diagnosed patients. The use of medical claims within a 1-year period encompasses the median time from disease onset to diagnosis as identified in the CARRA registry (0.8 years). 6 By using a 1-year time period, the majority of new onset cases would therefore be diagnosed during this time period. When using claims data, it should be noted that diagnosis codes may be driven by reimbursement considerations rather than clinical diagnosis and are subject to possible coding errors.22,23 Furthermore, the absence of a claim does not guarantee that a patient did not have the disease during this period. We cannot be certain if a medical diagnostic claim was made in an attempt to rule out a disease or condition; as such, we acknowledge this as a potential limitation. Finally, due to the impact of COVID-19, there is the potential for missing data as patients may not have been able to visit the doctor.
Conclusion
Analysis of medical claims data from US individuals under 18 years of age demonstrated that both the incidence and prevalence of jSSc increased with age. Analysis by developmental age group demonstrated increasing incidence from 10 years old and increasing prevalence from 6 years old. Results were consistent in a subgroup of patients treated with immunosuppressants.
Footnotes
Acknowledgements
The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). The authors did not receive payment related to the development of the manuscript. Paul Todd, PhD, of Nucleus Global, an Inizio company, provided writing, editorial support and formatting assistance, which was contracted and funded by Boehringer Ingelheim. Boehringer Ingelheim was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations.
Data availability
We assessed claims data from the Optum Clinformatics Data Mart Database, which is a database of commercially and non-commercially insured populations in the United States. All patient data were anonymized; therefore, informed consent was not necessary. Restrictions apply to the availability of these data because of a contract between Optum and Boehringer Ingelheim, and data are therefore unavailable to the public. For inquiries on the data set analysed in this study, please contact Optum (![]() ).
).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: All authors are employees of Boehringer Ingelheim Pharmaceuticals Inc, Ridgefield, CT, USA.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported and funded by Boehringer Ingelheim International GmbH.
