Abstract
Introduction:
Digital ulcers (DUs) stand out as one of the most prevalent and clinically meaningful manifestations of systemic sclerosis (SSc) and are associated with significant morbidity. While systemic (pharmacological) therapy is currently established as the ‘standard of care’, effective local ulcer management remains crucial for all cases of DUs. This is particularly true for patients who cannot tolerate systemic treatments or in the case of refractory SSc-DUs. On this background, there is a pressing demand for the formulation of evidence-based guidelines to assist clinicians and patients in navigating the local treatment options for DUs.
Methods:
A steering committee of international experts was established by the World Scleorderma Foundation (WSF) Digital Ulcer (DU) ad hoc committee. Two systematic literature reviews on local non-surgical and surgical treatments for the management of SSc-DUs were performed to inform the development of local treatment recommendations for SSc-DUs. Consensus methodology was used to develop the final treatment recommendations.
Results:
Six overarching treatment principles and eight local treatment recommendations (five non-surgical and three surgical) were agreed upon for the management of SSc-DU. Among topical non-surgical options, botulin toxin can be conditionally recommended for refractory and/or severe DUs. Among surgical treatments, autologous adipose tissue grafting might be recommended for DU healing when combined with background systemic treatments.
Conclusion:
These recommendations are specifically tailored to guide treatment decisions concerning both local and non-pharmacological approaches to managing SSc-related DUs. Our work has highlighted a notable quality gap in comparison to systemic treatments, underscoring the scarcity of high-quality studies concerning this topic.
Keywords
Introduction
Systemic sclerosis (SSc) is a rare complex systemic autoimmune disease characterized by vasculopathy, fibrosis of the skin and internal organs, and abnormal immune system activation. 1 In SSc, digital ulcers (DUs) are a severe common complication of vasculopathy, often occurring early in the disease course, affecting up to 50% of patients.2,3 DUs can cause substantial pain, disability, including work disability, with a considerable impact on patients’ quality of life. 4 Moreover, DUs are associated with a worse prognosis and are also a significant cause of morbidity for SSc patients.5,6
While local wound care is a fundamental aspect of managing DUs, specific recommendations for SSc patients are currently lacking.7,8 In clinical practice, the use of the ‘TIME’ (tissue management, infection and inflammation, moisture balance, and wound edge and epidermal advancement) methodology has been developed for the local management of generic wounds and can be systematically applied to identify crucial factors for optimal wound (ulcer) bed management. 9 However, to date, in the absence of specific recommendations for the local management of SSc-DUs, the use of systemic (pharmacological) therapies is currently considered the ‘standard of care’. Therefore, there is a high clinical need for developing local strategies specifically for SSc-DU management for two main purposes. First, this management approach can synergize with systemic treatments for refractory DUs; in addition, local treatment may be sufficient in patients with sporadic DUs or DUs without complications. Second, local management may be the only treatment strategy available for patients who do not tolerate systemic pharmacological therapies.
Against this background and recognizing the urgent need for guidance on the local management of SSc-DUs, the aim of the World Scleroderma Foundation DU ad hoc committee was to formulate practical recommendations, supported by current clinical evidence and expert opinion. These recommendations may provide a pragmatic and standardized approach to the local management of SSc-DU.
Methods
Research participants
A project steering committee of internationally recognized experts in the field of SSc (MH, YAS, CPD, OD, TF, DEF, DK, TK, MK, MM-C and JP) was established to determine the overarching research goals and select research questions to support the recommendations. A dedicated local treatment working group (CC, YAS, DG and PM) was established to perform two systematic literature reviews (SLRs) on the local non-surgical and surgical treatments for SSc-DU and was responsible for the summary and presentation of results to the steering committee. Methodological guidance and development of the final search strategy for the SLRs was provided by an expert methodologist (AA) and senior medical librarian (JWS). There was no external involvement of third parties in the process (including financial support) of developing these recommendations. The overarching principles were agreed upon among all members of the steering committee on the basis of the available literature on the management of DUs in SSc patients. 7
Systematic literature reviews
The steering committee reached consensus that the literature search informing local DU treatment guidelines should consider the following questions:
What is the role of DU assessment in the approach to local management?
What is the efficacy of local treatment for SSc-DU?
What is the safety of local treatment for SSc-DU?
What local treatment protocols, including debridement, are being used?
What is the role for combining local with systemic (pharmacological) treatment for DU?
What are the financial costs of DU in SSc? Specifically, are there cost-savings associated with local treatment for DU?
Two dedicated SLRs,10,11 undertaken with the project methodologist and librarian (AA, JWS), were performed according to the patient, intervention, comparison, outcome (PICO) model. PubMed, MEDLINE (OVID), Embase (OVID), Web of Science, Cochrane Library, Emcare (OVID) and Academic Search Premier databases were searched from inception to August 2022. Study titles and abstracts were screened, and then relevant data were extracted after full-text review by two study authors (CC, YAS), supervised by the working group leaders (DG, PM). The risk-of-bias (RoB) assessment was performed independently by two authors (CC, YAS). For randomized trials (RTs), the Cochrane RoB tool 12 was used, whereas the ROBINS-I 13 was applied to observational (OBS) cohort studies. All disagreements were resolved by consensus.
Recommendations
A summary of evidence and outcomes was developed by CC and YAS. Using these results, a set of draft treatment recommendations was prepared. The draft recommendations were presented to the steering committee and discussed at an online consensus meeting. A final draft of systemic treatment recommendations, that incorporated the feedback from the consensus meeting, was distributed and discussed via email for final approval from the steering committee members. All recommendations were supported unanimously by the steering committee. As most of the studies would focus on DUs of ischemic nature10,11 our set of recommendations will apply mostly for this specific subset of DUs; however, we cannot rule out that some of these local treatments might be beneficial also for DUs of different natures.
Results
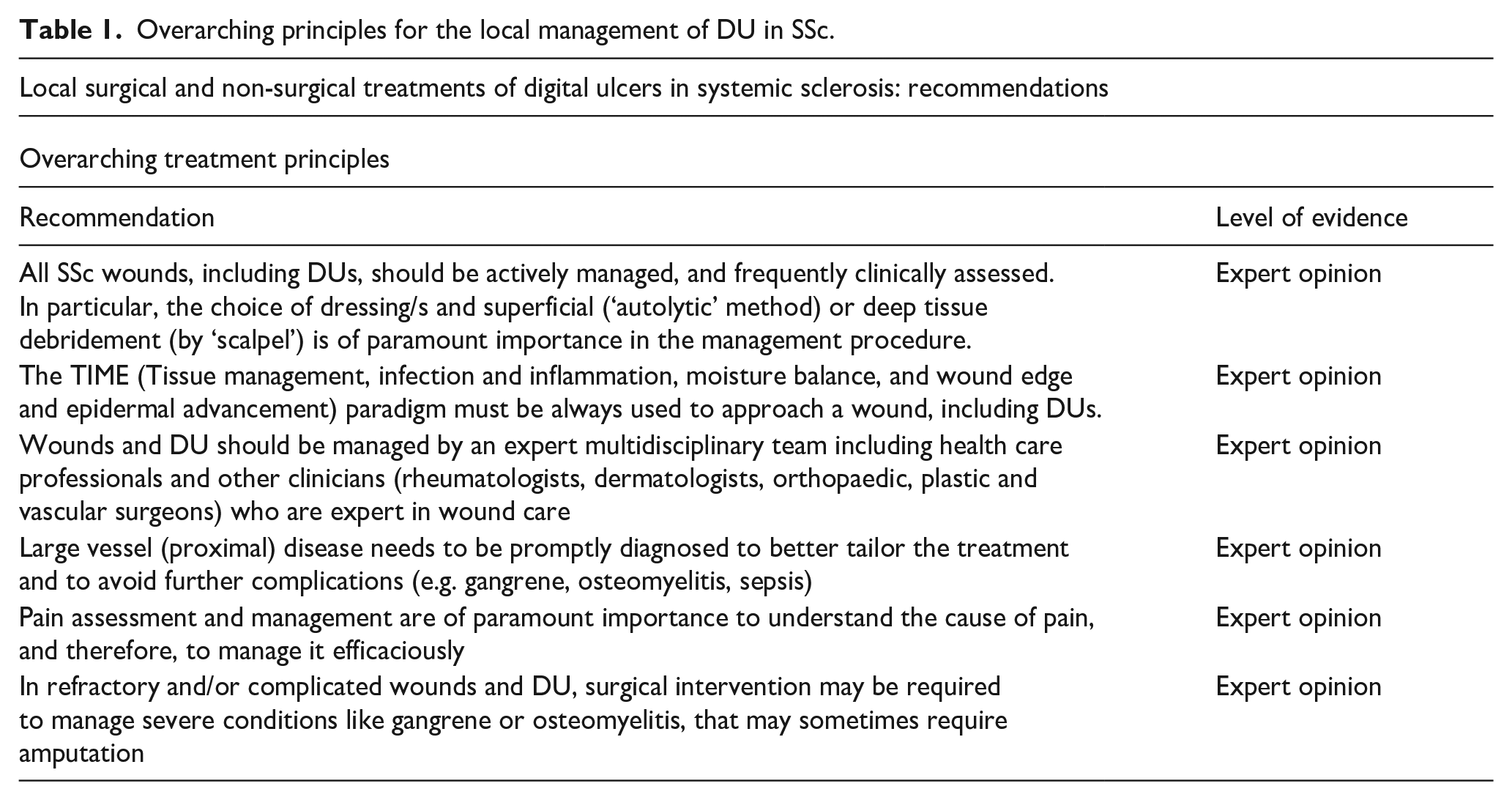
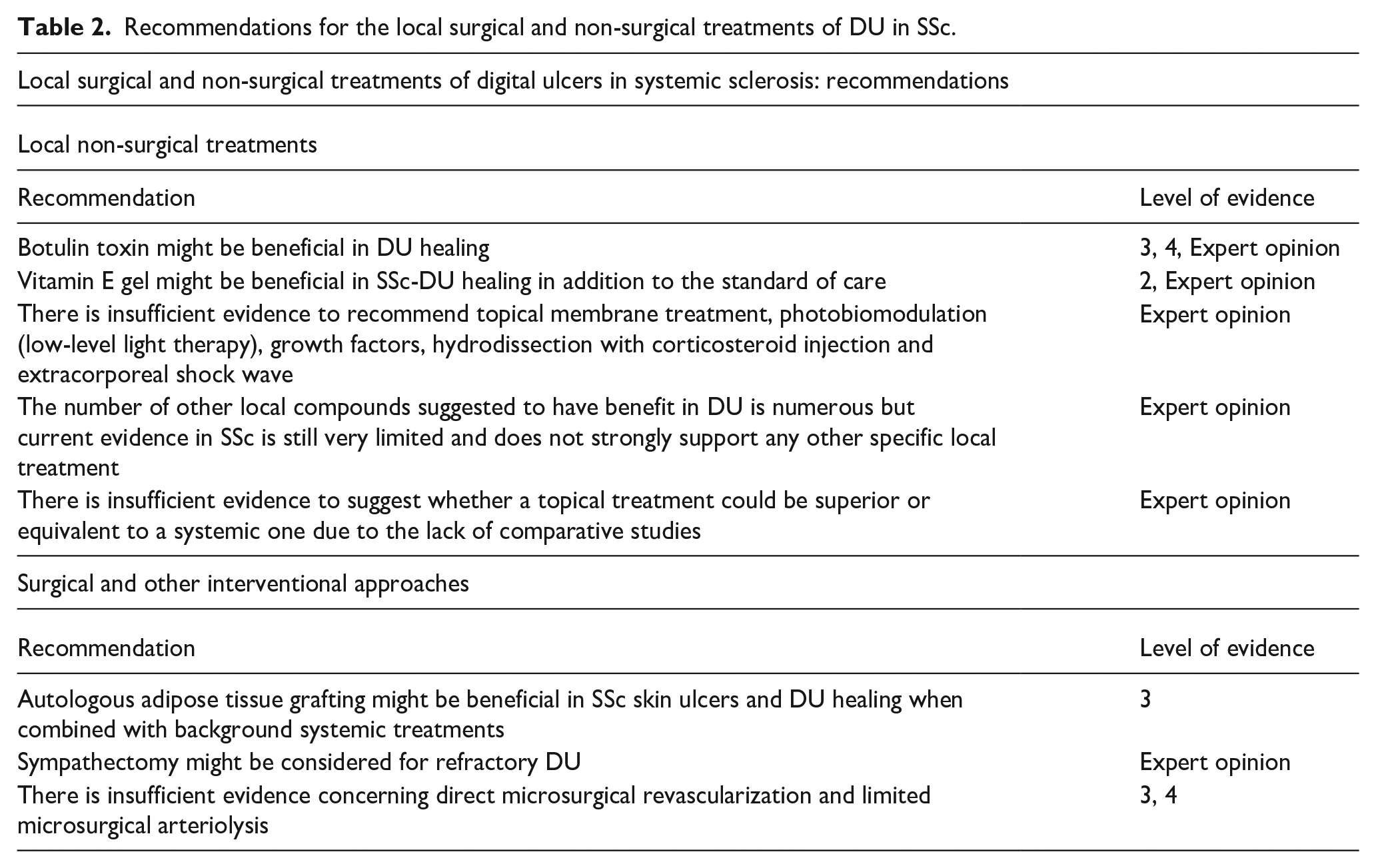
This study yielded six overarching treatment principles (Table 1) and eight treatment recommendations (Table 2) for the local surgical (three recommendations) and non-surgical management (five recommendations) of SSc-DU (see Figure 1). The final recommendations were grouped together as ‘local non-surgical treatments’ and ‘surgical treatments and other approaches’ for SSc-DU.
Overarching principles for the local management of DU in SSc.
Recommendations for the local surgical and non-surgical treatments of DU in SSc.
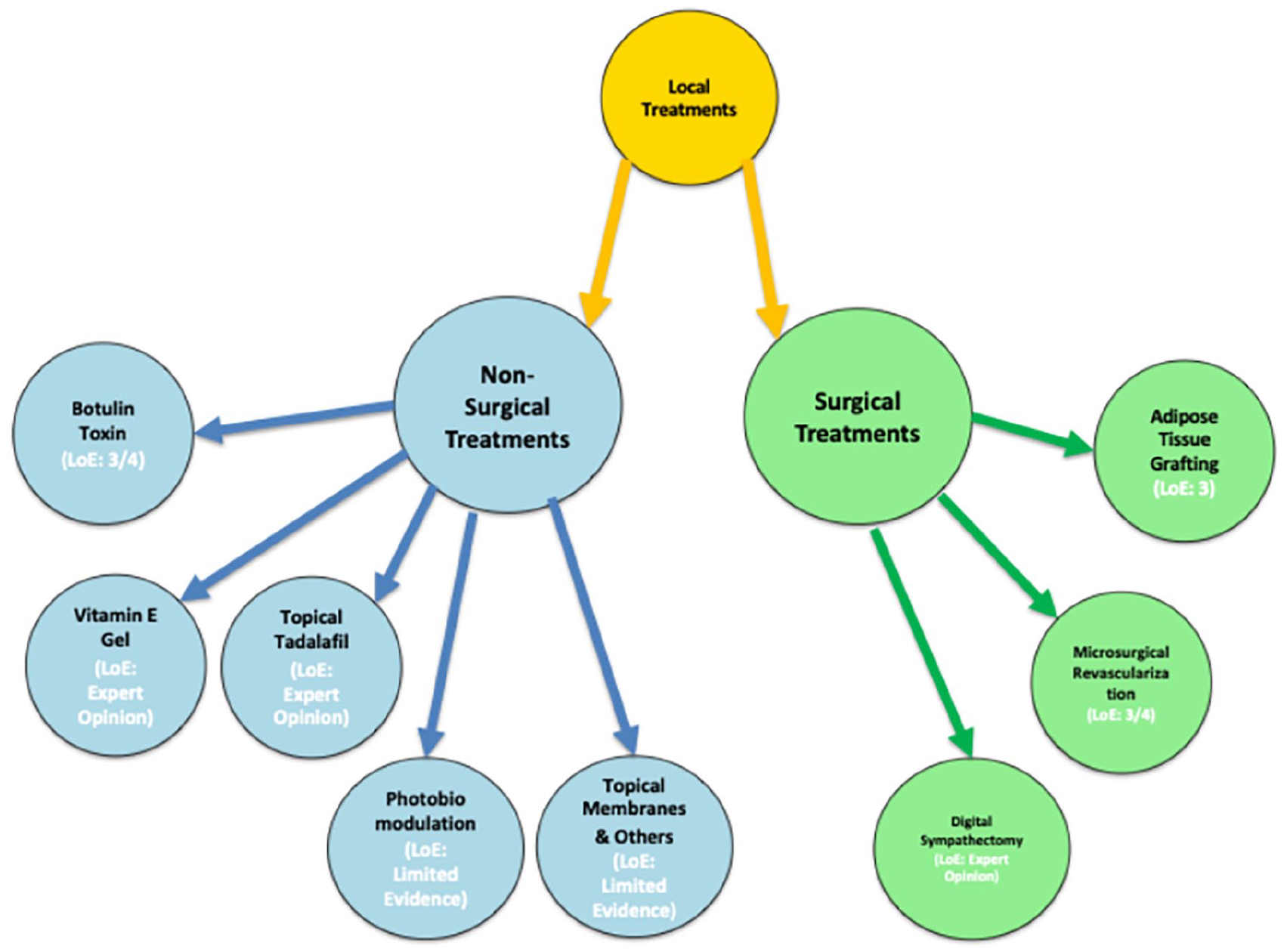
Network diagram recapitulating surgical and non-surgical treatment recommendations.
Local non-surgical treatments
Botulin toxin might be beneficial in DU healing (level of evidence (LoE) 3/4, expert opinion):
The use of botulin (‘botulinum’) toxin for SSc-DUs was reported in five observational studies.14–18 The use of botulin toxin ‘A’ was most commonly described. The injection procedure was associated with a good degree of efficacy regardless of its modality (as single-finger or whole-hand injection). The doses injected were highly variable (see Supplementary File 1). Overall, the procedure was well tolerated, and the most common side effect was transitory hand weakness. Although there are no placebo-controlled studies, there is strong expert opinion which considers the use of botulin toxin currently as a reasonable approach for refractory and/or severe DUs, particularly in the case of a threatened (i.e. critically ischaemic digit). Nonetheless, the optimal type of botulin toxin (e.g. A or B) for SSc-associated digital vasculopathy and the type of approach (full hand or single finger) have yet to be defined. It is also notable that the only higher evidence level multicenter, randomized, double-blind, placebo-controlled, parallel-group phase III trial in patients with systemic sclerosis with Raynaud’s phenomenon (SSc-RP) could not show beneficial effects. 19 Therefore, controlled studies are required to confirm our expert opinion in the setting of DU healing.
2. Vitamin E gel might be beneficial in SSc-DU healing in addition to the standard of care (LoE Expert opinion):
The efficacy and safety of vitamin E gel were tested in a single study where a topical gel was added to the local standard of care. 20 Experts have significant concerns about generalizability as this is based on one study only with a small sample size.
3. There is insufficient evidence to recommend topical membrane treatment, photobiomodulation (low-level light therapy), growth factors, hydrodissection with corticosteroid injection and extracorporeal shock wave (LoE Expert opinion):
The experts consider that these procedures are of interest as they may aid DU healing. However, given the methodological limitations and small sample sizes of the studies,21–25 experts strongly suggest that further rigorous investigation is required in order to reach sufficient evidence to support their use (Expert opinion).
4. The number of other local compounds suggested to have benefit in DU is numerous, but the current evidence in SSc is still very limited and does not strongly support any other specific local treatment (LoE Expert opinion):
There is a strong expert opinion favouring the local treatment of wounds and DUs through application of topical tadalafil (e.g. to avoid systemic vasodilation). 26 Therefore, studies on the local vasoactive treatment of SSc-DUs are missing and are therefore highly warranted.
5. There is insufficient evidence to suggest whether a topical treatment could be superior or equivalent to a systemic one due to the lack of comparative studies (LoE Expert opinion):
There is no strong evidence that a local treatment could be beneficial on its own; therefore, a combined approach including systemic and local treatment, taking into account the possible side effects and the specific drug interactions, should be advised. On this background, studies comparing the use of a systemic treatment versus a local treatment are warranted to support the use of local treatment/s as an alternative to systemic therapies.
Surgical and other approaches
Autologous adipose tissue (AT) grafting might be beneficial in SSc skin ulcers and DU healing when combined with background systemic treatments (LoE 3, Expert opinion):
Autologous AT grafting was investigated in seven studies, of which one randomized controlled trial (RCT), four cohort prospective studies and two case series.27–33 Two main cell types were extracted after isolating AT: adipose tissue used as a whole (ATDC) and stromal vascular fraction (SVF) separation and injection, and both were evaluated (see Supplementary File 1). Different techniques of AT handling, separation of centrifuged layers, site of injection and isolation of SVF were evaluated in the studies. In the only RCT, 31 the DU healing was superior in the group of patients treated with AT compared to those receiving a sham procedure. The experts agree that autologous tissue grafting might be beneficial, but further research is required to define the optimal technique for AT preparation (and/or separation), the site of injection, the appropriate dosage and the time interval between grafting intervals and better understand mechanisms of action. The current use of the technique strongly depends on local expertise and experience; however, this warrants further rigorous investigation.
2. Digital sympathectomy might be considered for refractory DU (LoE Expert opinion):
Several papers have reported the results of digital sympathectomy for SSc skin ulcers and DU, although currently there is not sufficient evidence-based literature to support this procedure.34–36 However, expert opinion addresses the possibility to use selective digital sympathectomy only in cases with severe skin ulcers and DU as well as in the case of threatened digits. Local expertise and the specific kind of techniques used can vary, and cases should be discussed individually in a multidisciplinary team.
3. There is insufficient evidence concerning direct microsurgical revascularization and limited microsurgical arteriolysis (LoE 3/4, Expert opinion):
Direct microsurgical revascularization (radial-to-common digital artery bypass graft) and limited microsurgical arteriolysis (adventitial stripping) were only evaluated in small case series,37,38 as such, the experts cannot advise their use for SSc-DU.
Discussion
In SSc patients, DUs are a prevalent and early disease manifestation contributing significantly to morbidity and negatively impacting patients’ quality of life. 39 The timely and accurate management of DUs is crucial not only to manage tissue inflammation, pain and infection but also to prevent irreversible tissue damage and disability. Over the years, there have been significant international collaborative efforts to identify pharmacological strategies that could be effective both for DU healing and prevention, 40 but it is globally accepted that local treatment strategies are also pivotal in DU management. This is particularly important for patients who do not tolerate systemic medications or whose DUs are refractory to systemic treatments. This is why there is a pressing need to define which non-surgical and surgical treatments are useful and should be considered in the management of DUs. Nonetheless, the range of potential local treatments is large and varies significantly according to local expertise.
Many local treatment strategies lack a sufficient level of evidence to robustly support their use in DU treatment. This report, based on our recent two SLRs focusing on local non-surgical and surgical treatment options for DUs,41,42 gathers the expert opinion and the existing literature to present eight recommendations related to local non-surgical and surgical treatments for DUs in SSc. Of note, while systemic treatments were previously addressed in the European Alliance of Associations for Rheumatology (EULAR) summarized recommendations for SSc management, local treatment recommendations for SSc-related DUs have not yet been included.43,44 This report marks the first development of specific recommendations for the local treatment of DUs in SSc.
In addition to specific local options discussed, and as stated in the overreaching principles, the ad hoc committee recommends the application of the TIME (tissue management, infection and inflammation, moisture balance, and wound edge and epidermal advancement) paradigm to approach SSc-DU. The committee also strongly endorses that SSc-DUs should be managed by an expert multidisciplinary team given their complexity and the potential need of treatment requiring surgical expertise. 45
In our recommendations, two treatment options with the highest level of evidence emerged: botulin toxin among topical non-surgical treatments and autologous AT grafting among surgical treatments. Both these options were provisionally recommended given the existence of multiple studies indicating potential efficacy in the treatment of refractory SSc-DU. Unfortunately, the presence of different treatment protocols for both procedures (i.e. type of toxin used, dose and injection technique for botulin toxin and techniques of AT handling, separation of centrifuged layers, site of injection and isolation of SVF) and the presence of only one RCT for autologous AT grafting limit the strength of these treatment recommendations. Therefore, while both these options could be considered in clinical practice, future studies are mandatory to better understand the details of the procedures and specific indications which are associated with the highest rate of success. Moreover, it should also be kept in mind that the specific expertise of the multidisciplinary team and the equipment needed to implement these treatments play a critical role in deciding which strategy and approach is preferred at an individual level.
For the remaining treatments, no clear recommendations could be made due to the low quality of evidence available and the small number of patients included in studies to date. Moreover, one important point specifically pertaining to local management is the absence of studies comparing the use of systemic treatments with local treatment. Indeed, in most studies, the use of local strategies was implemented in patients already on background systemic therapy (e.g. Phosphodiesterase-5 (PDE5)-inhibitors, endothelin receptor antagonists, prostanoids and calcium channel blockers), making it hard to evaluate the exact role of the topical treatment in the healing process. Therefore, currently, the use of above-mentioned local treatments is largely restricted in patients who are already on systemic treatments. Future SSc-DU research needs to address whether a topical strategy can be implemented as an alternative to systemic pharmacological therapy (see Table 3).
Research agenda: directions for future research.

It has not been possible to perform a health economic assessment of local DU treatments due to insufficient data. Only two studies included data on treatment costs: one topical vitamin E study performed in Italy 19 and one botulin toxin study that compared treatment costs to those of intravenous prostanoids in Iran. 14 Both studies were limited by small sample sizes. Moreover, the local costs associated both with local and systemic treatments (especially in the case of intravenous prostanoid treatment schemes) can vary significantly from country to country, making it hard to generalize about treatment-associated costs on a global scale.
Caution should be exercised in interpreting the outcomes of this process, considering the methodological constraints on the data available that affect the robustness of the recommended suggestions. Although our project steering committee benefitted from an expert internal, multidisciplinary membership, we did not include patient representation. However, the patient voice including unmet needs is well known, but formal involvement should be considered in the future design of dedicated studies. 46 Widespread limitations in studies of SSc-DUs include diverse classifications and definitions of ulcers, variations in participants’ baseline characteristics, the absence of standardized outcomes, and a lack of consistent time intervals between systemic treatment initiation and local treatment interventions. These limitations apply unfortunately to the majority of the studies that were examined. Moreover, given the use of topical treatment is usually provided in patients refractory to systemic therapies, studies are biased towards the inclusion of only patients with the most severe and refractory DUs. It is not possible to ascertain the most appropriate timing of implementation of local strategies or whether their earlier use might be more beneficial in the management of DUs.
These recommendations are intended to be complementary to current guidance on the management of SSc (e.g. published by the British Society of Rheumatology and EULAR), by providing a broader clinical overview of treatment approaches outside of the context of published randomized clinical trials.
Conclusion
The proposed recommendations reflect the currently available evidence and international expert opinion pertaining to the local non-surgical and surgical management of SSc-DUs. These recommendations may serve as a general guide to clinicians in the appropriate application of non-pharmacological treatment options for SSc-DUs. They represent the first pragmatic recommendations specifically formulated for the local treatment of SSc-DUs.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983251339821 – Supplemental material for Recommendations for the local management of digital ulcers in systemic sclerosis: A report from the World Scleroderma Foundation (WSF) ‘Ad hoc committee’
Supplemental material, sj-pdf-1-jso-10.1177_23971983251339821 for Recommendations for the local management of digital ulcers in systemic sclerosis: A report from the World Scleroderma Foundation (WSF) ‘Ad hoc committee’ by Corrado Campochiaro, Yossra A Suliman, Dilia Giuggioli, Pia Moinzadeh, Alessia Alunno, Jan W Schoones, Murray Baron, Lorinda Chung, Laura Ross, Nancy Maltez, Begonya Alcacer-Pitarch, Khadija El-Aoufy, Yannick Allanore, Francesco Del Galdo, Christopher P Denton, Oliver Distler, Tracy Frech, Daniel E Furst, Dinesh Khanna, Thomas Krieg, Masataka Kuwana, Marco Matucci-Cerinic, Janet Pope and Michael Hughes in Journal of Scleroderma and Related Disorders
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: MH reports research funding and speaker fees from Janssen, outside of the submitted work. Chair of a Data Safety Monitoring Board: SHED SSc – SHarp dEbridement of Digital ulcers in Systemic Sclerosis: a multi-centre Randomised Controlled Trial feasibility study (REC reference: 21/YH/0278). MK has received consulting fees, speaking fees and/or research grants from Argenx, Asahi Kasei Parma, AstraZeneca, Boehringer Ingelheim, Chugai, GSK, Janssen, Kissei, MBL, Mochida, Ono Pharmaceuticals and Tanabe-Mitsubishi. LC has received consulting fees from Mitsubishi Tanabe, Genentech, Kyverna, IgM Biosciences, Lilly and Janssen. DG has received speaking fees from Boehringer Ingelheim and Janssen. OD has/had a consultancy relationship with and/or has received research funding from and/or has served as a speaker for the following companies in the area of potential treatments for systemic sclerosis and its complications in the last three calendar years: 4P-Pharma, AbbVie, Acceleron, Alcimed, Altavant, Amgen, AnaMar, Argenx, Arxx, AstraZeneca, Blade, Bayer, Boehringer Ingelheim, Cantargia AB, Catalyze Capital, Corbus, CSL Behring, Galderma, Galapagos, Glenmark, Gossamer, Horizon, Janssen, Kymera, Lupin, Medscape, Merck, Miltenyi Biotec, Mitsubishi Tanabe, Nkarta Inc, Novartis, Orion, Prometheus, Redxpharma, Roivant, EMD Serono, Topadur and UCB. Patent issued ‘mir-29 for the treatment of systemic sclerosis’ (US8247389, EP2331143). Co-founder of CITUS AG. None of the other authors report any relevant conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the World Scleroderma Foundation Digital Ulcer ad hoc committee.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
