Abstract
Keywords
Introduction
Systemic sclerosis (SSc) is an autoimmune rheumatic disorder characterized by immune dysregulation of both the innate and adaptive immune responses, vascular dysfunction, and cutaneous and visceral fibrosis. It is hypothesized that an environmental trigger, in genetically predisposed patients, induces molecular and cellular alterations in target cells, which in addition to an undefined endothelial injury leads to transmigration of inflammatory cells into the tissues. Early T- and B-cell inflammatory infiltrates in the skin and other involved visceral organs lead to autoantibody, cytokine, and growth factor production, which leads to increased collagen and extracellular matrix (ECM) deposition in the involved tissues.1 –3
Vascular dysfunction is likely initiated by defects in the endothelial cells themselves, tissue hypoxia which leads to the production of reactive oxygen species, or an injury from inflammatory cells producing local cytokines. The dysfunctional endothelial cells express an increase in adhesion molecules that leads to an increased transmigration of inflammatory cells through the endothelial cell lining and into the underlying vessel walls and tissues. Damaged endothelial cells exacerbate vasospasm; mediate proliferation and contraction of smooth muscle cells in the vessel wall; enhance procoagulant activity and reduce fibrinolysis leading to intravascular microthrombi; and enhance the release of chemotactic and adhesion factors activating local inflammation.4,5 The endothelial damage plays a central role in the pathophysiology of the SSc vasculopathy which leads to the clinical picture of Raynaud’s.
The vasculopathy seen in SSc more commonly involves the microvascular circulation, which causes the clinical symptoms of Raynaud’s and can lead to the symmetrical pattern of digital ulcerations. On more rare occasions, the macrovascular circulation can also be affected by the same process, and this again may present in the same pattern but results in asymmetrical symptoms and tissue injury.4 –10
For this study, we examined patients with SSc and “medication refractory Raynaud’s” (patients who were on standard of care therapy for sustained vasospasm and continued to suffer from severe, unremitting pain, and/or chronic ulcerations) likely related to macrovascular involvement of the medium-sized arteries and digital arteries of the hand. We identified patients whose symptoms were treated with microsurgical revascularization surgery at our institution and described the patient demographics, surgical procedures, and long-term outcomes.
Methods
Following institutional review board approval, patients at the University of Pennsylvania between 2009 and 2022 were identified based on ICD-9 and ICD-10 diagnosis codes for SSc, Raynaud’s, and vascular grafting. The patients included in the study were required to fulfill the ACR/EULAR 2013 classification criteria for SSc. 11 Patients who proceeded with the vascular intervention were medication refractory to more than one class of agents used for Raynaud’s and that included patients who have used such agents as calcium channel blockers (CCBs), phosphodiesterase-5 inhibitors (PDE-5i), endothelin receptor antagonists (ERAs), and antiplatelet therapy. We undertook a retrospective chart review and collected demographic data as well as preoperative and postoperative patient-reported outcomes, such as clinical symptoms, clinical exam findings, and imaging results. Pre-surgical clinical symptoms included numbness, pain, ulcers, infections, necrosis, and previous amputations. In regard to the surgical procedure, we reported on which vessel was reconstructed with bypass grafting (ulnar or radial artery), origin of vascular graft, and whether sympathectomy or other concomitant surgery was performed. After the procedure, we collected data on Doppler assessment and strength of blood flow through the graft. We reviewed all postsurgical visits with the vascular surgery and the rheumatology teams and documented subjective patient symptoms and objective physician findings related to Raynaud’s symptoms, pain, ulcers, and occlusion and need for additional surgeries.
Results
Demographics
Our search, during the years of 2009–2022, identified 36 patients, of which 10 patients, 12 hands (with no prior revascularization) met the inclusion criteria described above. Eight out of the 10 patients were female, and the average age of all patients at surgery was 52.3 ± 17.7 years of age (range 20–74 years old), and the average time since the first Raynaud’s symptoms was 37.9 ± 27.2 months (range 5–110 months). All patients had a positive antinuclear antibody, and 7 of 10 had an anticentromere pattern, 5 of 10 were former smokers, 4 of 10 had high blood pressure, and 4 of 10 had elevated lipids. Nine of the 10 patients had limited cutaneous SSc, compared to only one patient with diffuse cutaneous SSc (Table 1).
Patient demographics.

F, female; M, male; NA, not assessed; N, no; Y, yes; APLs, antiphospholipid antibodies; HTN, hypertension; DM, diabetes mellitus; HLD, hyperlipidemia; SSc, systemic sclerosis; mo, months; L, left; R, right; Meds, medications; CCB, calcium channel blocker; PDE, 5i, phosphodiesterace 5 inhibitor; ASA, aspirin; PPI, proton pump inhibitor; HCQ, hydroxychloroquine; MMF, mycophenolate mofetil; ERA, endothelin receptor antagonist; SSRI, selective serotonin reuptake inhibitor; MTX, methotrexate; AZA, azathioprine.
Pre-surgical symptoms and imaging findings
Pre-surgical imaging revealed occlusion of the ulnar artery in seven hands, the radial artery in two hands, and one hand with both radial and ulnar occlusion. In one hand, there was a discontinuous ulnar artery, while another hand had multiple discontinuous digital arteries and the palmar arch. In addition, diminutive vessels were described in palmar arches of four hands, in the digital arteries of four hands, in the ulnar artery of five hands and the radial artery of three hands. In four hands, collateral vessels were observed (Table 2). All 12 hands at presentation had medication refractory Raynaud’s pain, and digital ulcers were seen in 11 of 12 hands. Three patients had history of digital infections, and three different patients suffered from tissue necrosis. One patient had a previous digital amputation. In seven hands, there were documented complaints of digital numbness (Table 3).
Findings of pre-surgical vascular imaging obtained from magnetic resonance angiography and computed tomography angiography.

R1, right thumb; R4, right ring finger; R5, right little finger; L5, left little finger.
CTA of wrist only, hand not imaged.
Pre-surgical symptoms and postsurgical long-term follow-up symptoms.

Y, yes; N, no; Mo, months; DMA, digital minimi artery
Surgical intervention
Surgical grafting was performed on the ulnar artery in 11 hands and radial artery in one hand. Grafts were obtained from the following regions: eight saphenous vein, two cephalic vein, and two veins from the upper extremity. In 9 of 12 hands, there were concomitant sympathectomies performed. Additional procedures were performed in three hands: one carpal tunnel release, one ulnar nerve neuroplasty at the elbow, and one ulnar nerve release in Guyon’s canal. There were no significant surgical complications reported with any of the cases. Intraoperative Doppler ultrasound indicated excellent pulsatile blood flow through the graft and digits prior to closing in all cases.
Postsurgical medical treatment
In 6 of 12 hands, aspirin (ASA) was increased from preoperative 81 mg/day to 325 mg/day after the operation, for varying durations. Four hands were continued on ASA 81 mg/daily, and one hand was initiated on ASA 325 mg two times a day. Two hands received enoxaparin for 4 weeks postoperatively, three hands were on heparin drip perioperatively, and one hand was placed on clopidogrel. Three hands were treated with tadalafil postoperatively, six hands on sildenafil, two hands on ERAs, and seven on CCBs (Table 4).
Surgical intervention and postsurgical medical management.
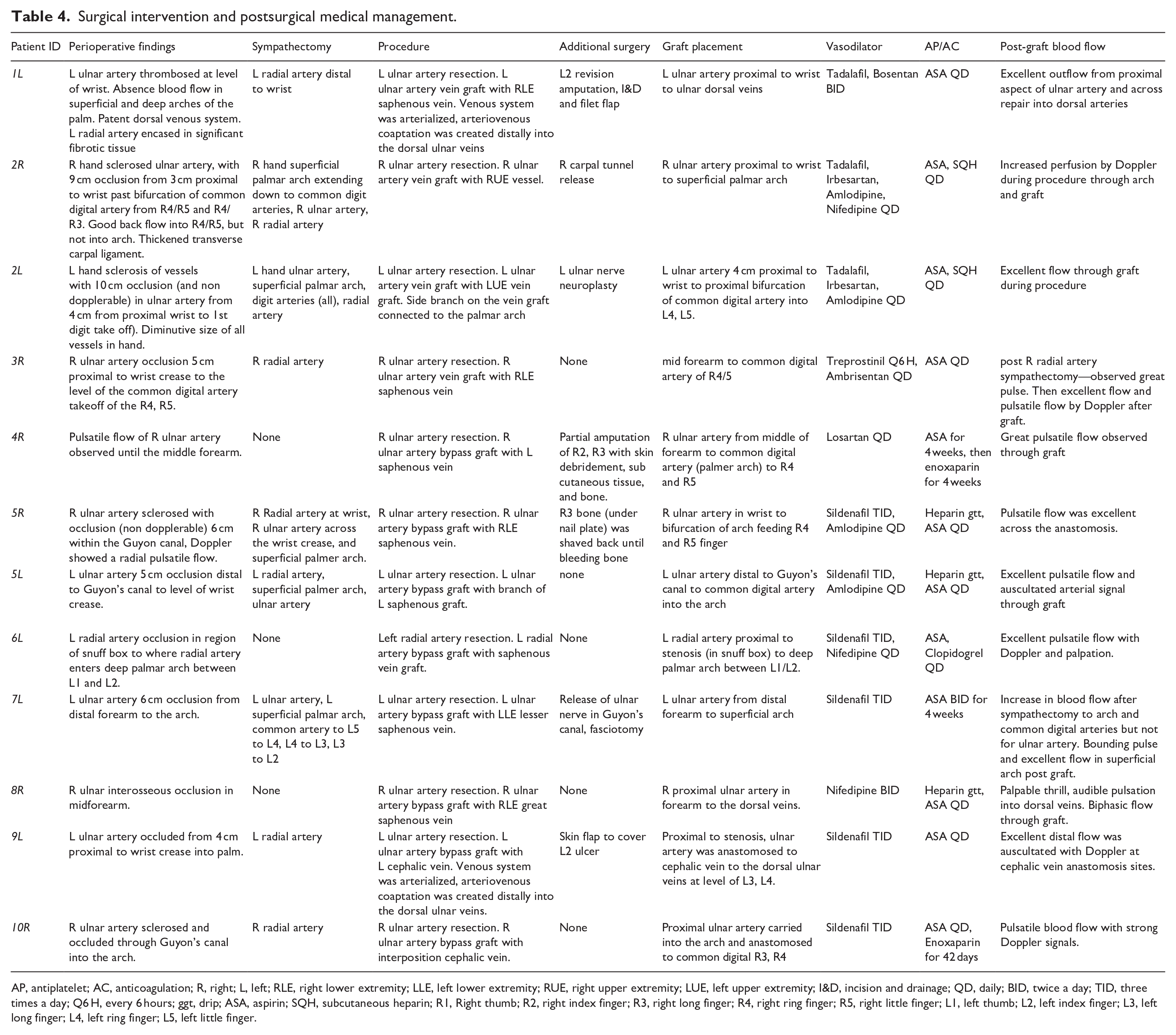
AP, antiplatelet; AC, anticoagulation; R, right; L, left; RLE, right lower extremity; LLE, left lower extremity; RUE, right upper extremity; LUE, left upper extremity; I&D, incision and drainage; QD, daily; BID, twice a day; TID, three times a day; Q6 H, every 6 hours; ggt, drip; ASA, aspirin; SQH, subcutaneous heparin; R1, Right thumb; R2, right index finger; R3, right long finger; R4, right ring finger; R5, right little finger; L1, left thumb; L2, left index finger; L3, left long finger; L4, left ring finger; L5, left little finger.
Long-term symptom and physical exam follow-up
The average time of follow-up after the surgical procedure was 45.4 ± 38.7 months (range 8–120 months). Postsurgical follow-up with Doppler was available in 11 of 12 hands approximately 1–2 months postoperatively. In seven hands, the signal was strong through the graft, but in four hands the signal was subjectively decreased downstream of the graft. Postsurgical Raynaud’s symptoms were recorded in eight hands: two hands were documented as “active,” two hands were documented as “active in winter only,” and four hands were documented as “improved” or “minor symptoms in cold only.” The two hands with “active” Raynaud’s also had active pain in hands and fingers. When patients were asked about pain relief, 10 hands were documented as “improved” or “pain only in the cold.” In three hands, ulcers continued to develop. One of these hands had a finger amputation several years after grafting and has remained ulcer free. One patient had chronic ulcers in the setting of medication noncompliance. A final patient had ongoing chronic ulcers, followed by an occlusion requiring revascularization. At the final follow-up visit, the other 11/12 hands had no clinical or radiographic signs of an occluded graft (Table 3). Three patients died within the first 2 years after revascularization. All other patients were still being followed to date.
Discussion
Surgical intervention in patients with Raynaud’s with underlying connective tissue disease, specifically SSc, has gained momentum in the last decade. The benefits of sympathectomy have a long history dating to 1980s when Adrian Flatt 12 described periarterial sympathectomy in patients with refractory vasospastic diseases suffering from connective tissue disease, trauma, and frostbite. His method involved the removal of several millimeters of adventitia from digital arteries, the outer layer of the arterial wall that includes autonomic nerves which play a central role in blood vessel tone, in two women with connective tissue disease resulting in a permanent 1–2°F temperature increase, and alleviation of pain. 12 Today, the sympathectomy has evolved to include reverse interposition venous grafting in those with complete occlusion of medium-sized hand/wrist arteries. In this case venous grafts from other areas of the body are used to bypass these occluded medium-sized arterial vessels. Among SSc–Raynaud’s patients, surgical interventions to alleviate symptoms are often reserved for cases where there is both microvascular and macrovascular disease. Unfortunately, there are limited studies examining when patients with RP should be referred for arterial venous grafting and the long-term results of the intervention.
In this study, we set out to track the long-term effects of venous grafting in SSc patients with cold-exposure digit color changes refractory to medical therapy. We reviewed all notes in the electronic medical record system starting with the first signs of hand and/or digit pain and tracked the patient’s progression until their last note in the chart. The average follow-up time was 45.4 ± 38.7 months (range 8–120 months).
Similar to previous studies, we found the majority of medium-sized vessel occlusions in our SSc–Raynaud’s population occurred in the ulnar artery.7 –10,13,14 Our population of patients were predominantly patients with limited cutaneous systemic sclerosis (LcSSc) who had a positive anticentromere antibody with an average disease duration from the initial Raynaud’s symptoms of 37.9 ± 27.2 months. When examining patients’ subjective reports of long-term postsurgical outcomes, 9 of 12 hands had subjective improvement in pain and Raynaud’s color change. The objective clinical outcome indicated that 7 of 12 hands had no additional ulcer occurrences, and three hands had only one more event of noncomplicated ulcer. In the hands with ongoing ulcerations, one ulcerated hand was in the setting of poor medication compliance, one in the setting of a repeat occlusion and finally one patient who had chronic ulcerations on one finger that resolved upon amputation.
A more recent retrospective study of 46 patients (58 hands) who received sympathectomies and arterial vein grafting (33 hands), with follow-up for an average of 3.97 years, found that the patients had 77.4% graft patency, 94.8% sustained ischemic pain resolution, and 78% had prevention of future ulcer development. 15 Of the rheumatic disease patient population studied, only 22 hands were from SSc patients (37.9%). 15 A small subsection of the study population was monitored for long-term outcomes by a phone interview (n = 10, 7 autoimmune rheumatic disease) which indicated that their Raynaud’s severity and frequency, drastically decreased, were ulcer free, and experienced dramatic decreases in pain over an average of 11.6 years post-surgery. 15 Unfortunately, the conclusions from this study are difficult to compare to our study because the results were not documented based on whether patients have SSc and were primarily subjective findings.
One of the largest studies examining the outcomes of surgical interventions of 178 refractory Raynaud’s patients (351 hands) was completed during 2003–2009. 13 The team first created a novel Raynaud’s hand severity classification system (Types 1–5) to guide surgical decision based on pre-surgical magnetic resonance arteriography (MRA) evaluation of the arteries with diminished blood supply i.e. ulnar, radial, palmar arch or digital arteries. 13 Second, based on classification type, they tailored the surgical intervention which was a combination of arterial or venous grafts and digital sympathectomy. 13 Of note, only 47% of patients in the study had an underlying autoimmune rheumatic disease; scleroderma, rheumatoid arthritis, and systemic lupus erythematosus. The study found that surgical intervention improved patient pain and ulcerations in at least 50% of the patients. 13 However, the timeline for data collection was not provided, and the improvement of pain and decrease in ulceration was lumped into a single category. Unfortunately, the Raynaud’s hand severity classification and type of surgical intervention performed were not described.
In another study of refractory Raynaud’s patients presenting with pain and ulceration who underwent surgical interventions, a different surgical approach was pursued. All patients received pre-surgical MRA, and during surgery all patients received a sympathectomy. After sympathectomy completion, the tourniquet was removed and if blood flow to the digits was adequate no further intervention was pursued. However, if subjectively, the blood flow appeared to be diminished, then an interposition vein graft was placed. The study reported on 33 patients, and of them, eight had bypass surgery, and seven received necrotic tissue debridement. All 33 patients had resolution of ulcers, and subsequently, 0 amputations occurred. The pre-surgical ulceration frequency, postsurgical changes in pain, and the type of arteries grafted were not included in the report. 13
Momeni et al. 16 reported on 17 patients with SSc–Raynaud’s who received combined sympathectomy and a vascular graft which led to 100% healing of digital ulcers with a mean follow-up of 13 months (range 1–54 months).
An interesting systemic literature review on surgical management of digital ulcers and SSc identified 13 significant articles reporting on the decrease of digital ulceration with the following surgical interventions: autologous fat grafting (seven studies), autologous adipose derived stromal vascular fraction grafting (four studies), bone marrow derived cell transplantation (one study), sympathectomy (three studies), and microsurgical revascularization (two studies). 17 All surgical modalities reported improvement in digital ulcerations; however, bone marrow–derived cell transplantation and microsurgical revascularization reported 100% ulcer resolution. 17
One of the challenges of retrospectively analyzing the benefits of surgical intervention in Raynaud’s patients is the definition of success of the surgical intervention. This can be assessed based on physical exam. For example, if negative outcomes are defined as frequent ulcerations or reoccurrence of occlusion, then we can safely say that in our study, the greater majority of hands (9/12) had a decrease or resolution of ulcerations and did not experience repeat occlusions post intervention, and thus were a success.
Success can also be reviewed from the patient perspective. For example, a large multicenter trial of 281 patients analyzed diary recordings of Raynaud’s frequency and Raynaud’s attack duration, impact scales for mood and tension, health assessment questionnaires, and physician assessment scales, found that patients with ulceration had elevated patient determined Raynaud’s Condition Scores, elevated pain scores, disability scores, physician global assessment scores and tension compared to Raynaud’s patients without ulcerations. 18 If surgical intervention can improve these scores for patients by limiting their ulceration frequency, then the surgical intervention can be psychologically beneficial for patients suffering from medication refractory Raynaud’s.
Our case study provides an important observation of 12 SSc–Raynaud’s hands, refractory to pharmacologic therapies, who’s severe Raynaud’s symptom of pain, ulceration frequency and digital color change improved in the majority of hands after vascular grafting and continued to have long-term symptom relief. Treatment of severe SSc–Raynaud’s requires a multifactorial treatment approach that includes pharmacologic and surgical approaches to effectively improve symptoms.
The limitation of this study includes the lack of standardized pre-surgical questionnaires, detailed vascular physical exam, standardized postsurgical and follow-up questionnaires, as well as a standardized postsurgical vascular exam all relating to the retrospective nature of our study. Future prospective studies looking at this same question could use standardized Raynaud’s activity questionnaires to assess the severity of this before and after a peripheral arterial bypass surgery, and a post-bypass questionnaire to better assess the effectiveness of the surgical procedure. Another limitation of our study is a potential placebo effect from the active procedure that can lead to subjective improvement regardless of if the vascular flow has improved by the surgical intervention. The results gathered for this study solely relied on physician documentation and may be limited in regard to patient experiences and complaints.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support from the 2020–21 American College of Rheumatology Medical and Graduate Student Preceptorship Scholarship (Nora Hajnoczky MD)
Statement of ethics and consent
For the following study, IRB approval was obtained through the University of Pennsylvania IRB. IRB PROTOCOL#: 834443. The following approval was exempt.
