Abstract
Objective:
COVID-19, a respiratory infection caused by the novel coronavirus SARS-CoV-2, can cause varying degrees of illness ranging from mild respiratory illness to severe respiratory failure. Systemic sclerosis is a chronic autoimmune disease, with an increased prevalence of infections as compared to the general population. In this study, we compare the clinical outcomes and resource utilization for COVID-19 hospitalizations in patients with and without systemic sclerosis.
Methods:
We used the National Inpatient Sample database, 2020, to study the characteristics, morbidity, mortality, cost, and resource utilization among primary COVID-19 hospitalizations with and without systemic sclerosis.
Results:
There were 1,050,040 patients aged ⩾ 18 years with a diagnosis of COVID-19. Of these, 775 (0.07%) patients had a secondary diagnosis of systemic sclerosis. Although there was no statistically significant difference regarding individual outcomes; in-hospital mortality, vasopressor use, cardiac arrest, acute kidney injury, and disposition to facility were numerically higher in hospitalizations with systemic sclerosis. The composite endpoint of major adverse events was higher in the systemic sclerosis cohort (adjusted odds ratio 1.52, 95% confidence interval: 1.06–2.17, p = 0.022).
Conclusion:
COVID-19 patients with systemic sclerosis had worse outcomes (i.e. higher composite endpoint of major adverse events) than those without systemic sclerosis. Further studies are needed to establish a better understanding of the relationship between COVID-19 and systemic sclerosis.
Introduction
COVID-19, a respiratory infection caused by the novel coronavirus SARS-CoV-2, was declared a pandemic in 2020. While the majority of patients tend to have mild respiratory illness, a minority of patients require hospitalization and mechanical ventilation. 1 Systemic sclerosis (SSc) is a rare chronic autoimmune disease characterized by inflammation and fibrosis of the skin and internal organs. SSc patients have high inpatient needs, and previous literature has shown a greater risk of hospitalization in incident cases of SSc than in age- and sex-matched controls. 2 Infections are more common among SSc patients and contribute to morbidity and excess mortality. 3 The risk factors associated with infections in SSc patients include esophageal and pulmonary involvement, severe Raynaud’s phenomenon or calcinosis, and the use of specific treatments for the management of the disease. 4 Pneumonia has been described in patients who have SSc with pulmonary involvement, particularly in those who have interstitial lung disease, 5 suggesting that pulmonary fibrosis may be a predisposing factor for pulmonary infections, and consequently, worse outcomes in these patients. 6
The outcomes of SSc patients hospitalized with COVID-19 have not been well characterized. Thus, the objective of this study was to compare the clinical outcomes and resource utilization for the treatment of COVID-19 in patients with and without SSc.
Methods
Study population
Using the National Inpatient Sample (NIS) database from 2020, we conducted a retrospective cohort study of admissions in patients 18 years or older with a principal discharge diagnosis of COVID-19 (using the ICD-10-CM codes U07.1) with and without a secondary diagnosis of SSc (using the ICD-10-CM codes M34xx).
Outcomes
Our primary outcomes were in-hospital mortality and the need for invasive mechanical ventilation (IMV). Our secondary outcomes included pressor use, acute kidney injury (AKI), AKI requiring dialysis, cardiac arrest, major adverse events (composite of in-hospital mortality, IMV, pressor use, AKI, AKI requiring dialysis and cardiac arrest), transfer to Skilled Nursing Facility (SNF), Intermediate Care Facility (ICF) or any other facility, hospital length of stay (LOS), and mean hospital costs.
Statistical analysis
For each hospitalization, baseline demographic characteristics, hospital characteristics, and clinically relevant co-morbidities were identified using Elixhauser comorbidities and ICD-9-CM and ICD-10-CM codes. The chi-square test was used for univariate analyses in between-group comparisons for categorical variables and weighted simple linear regression for continuous variables. Weighted logistic and linear regression for categorical and continuous outcomes respectively, adjusted for age, sex, race, and comorbidities listed in Table 1, and hospital characteristics, were performed to determine the association between SSc and predefined clinical outcomes in the principal COVID-19 hospitalizations. We performed subgroup analyses for our primary outcomes according to the age of the patients (age 18–64 vs age 65 and greater). All statistical analyses were performed using Stata 16.1 (StataCorp, College Station, TX, USA), considering survey design complexity by incorporating sampling weights, primary sampling units, and strata. A p-value < 0.05 was considered statistically significant.
Baseline characteristics of included patients.
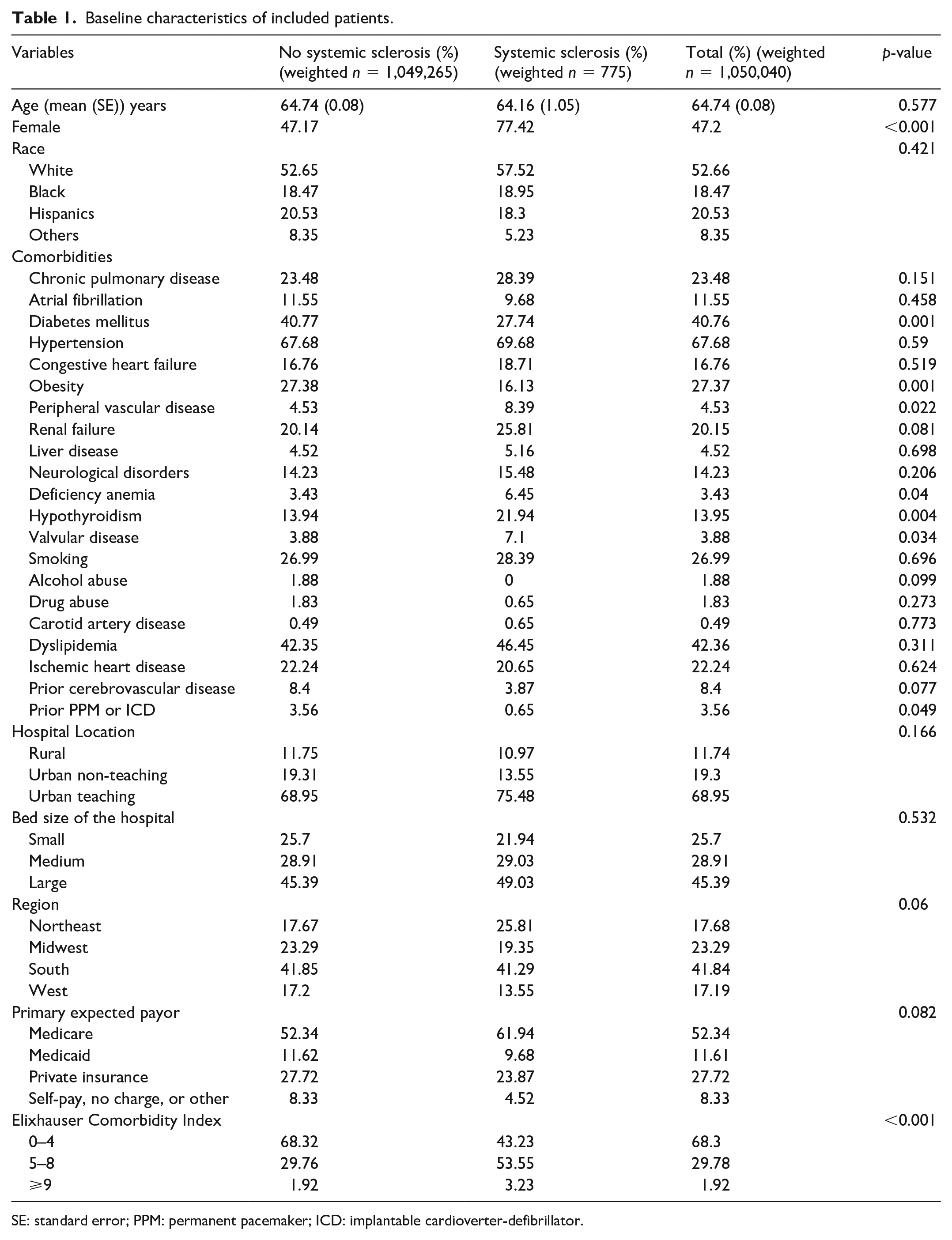
SE: standard error; PPM: permanent pacemaker; ICD: implantable cardioverter-defibrillator.
Institutional Review Board approval
Since NIS contains publicly available de-identified data, Institutional Review Board (IRB) approval was not sought.
Results
Out of a total of 1,050,040 weighted principal COVID-19 hospitalizations, 775 (0.07%) patients had coexisting SSc. COVID-19 patients with SSc were more likely to be female and had a higher prevalence of peripheral vascular disease, deficiency anemia, hypothyroidism, and valvular heart disease and a lower prevalence of diabetes, obesity, and prior permanent pacemaker or implantable cardioverter-defibrillator compared to COVID-19 patients without SSc (Table 1). The SSc cohort had a higher Elixhauser Comorbidity Index. In addition, SSc patients with COVID-19 had a higher burden of interstitial lung disease (17.4%) than patients without COVID-19 (1.13%).
There were no statistically significant differences between the two groups with regard to any individual outcomes but some outcomes including in-hospital mortality, pressor use, cardiac arrest, AKI, and disposition to facility (SNF, ICF, or any other facility), were slightly numerically higher in the SSc cohort (Table 2). The composite endpoint of major adverse events was higher in the SSc cohort (adjusted odds ratio [aOR] 1.52, 95% confidence interval [CI]: 1.06–2.17, p = 0.022; Table 2). In our subgroup analyses, there was a trend toward increased in-hospital mortality in 18 to 64 years old COVID-19 patients with SSc (aOR 2.05, 95% CI: 0.97–4.33, p = 0.06), but no other interactions between age and primary outcomes were found (Table 3).
Results for each outcome.
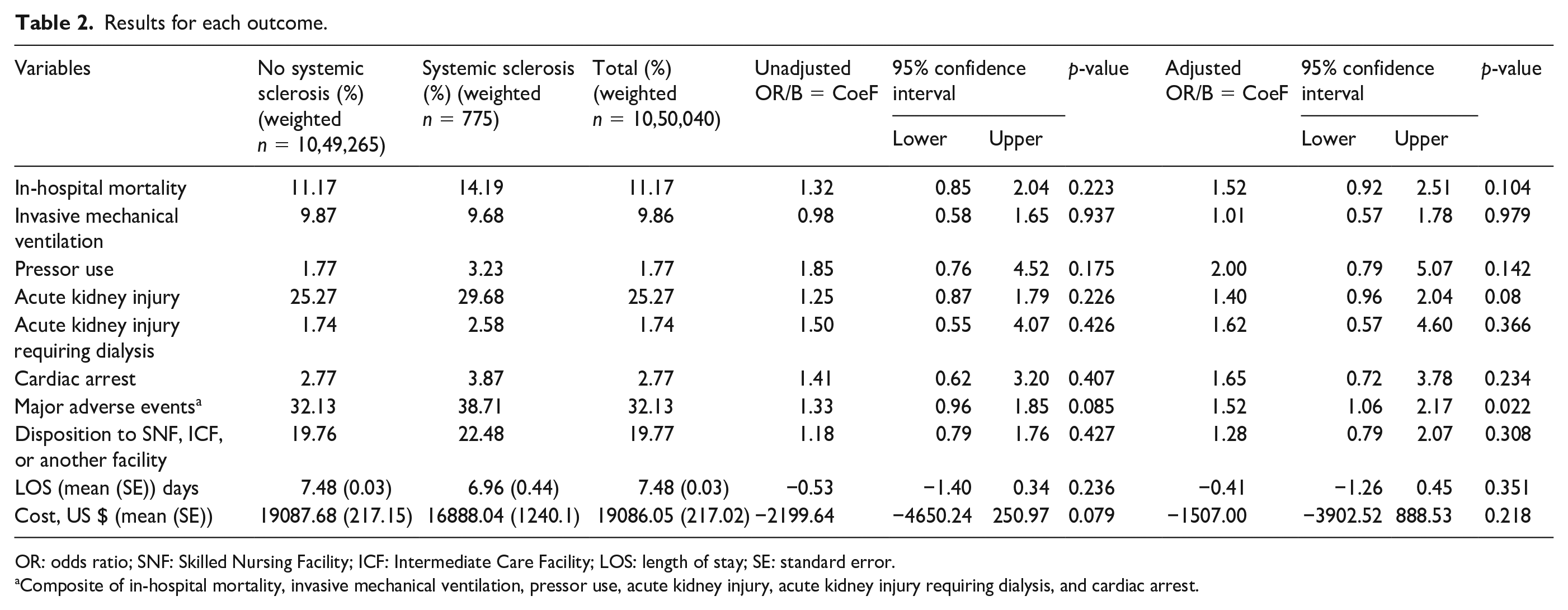
OR: odds ratio; SNF: Skilled Nursing Facility; ICF: Intermediate Care Facility; LOS: length of stay; SE: standard error.
Composite of in-hospital mortality, invasive mechanical ventilation, pressor use, acute kidney injury, acute kidney injury requiring dialysis, and cardiac arrest.
Subgroup analysis for primary outcomes according to age.
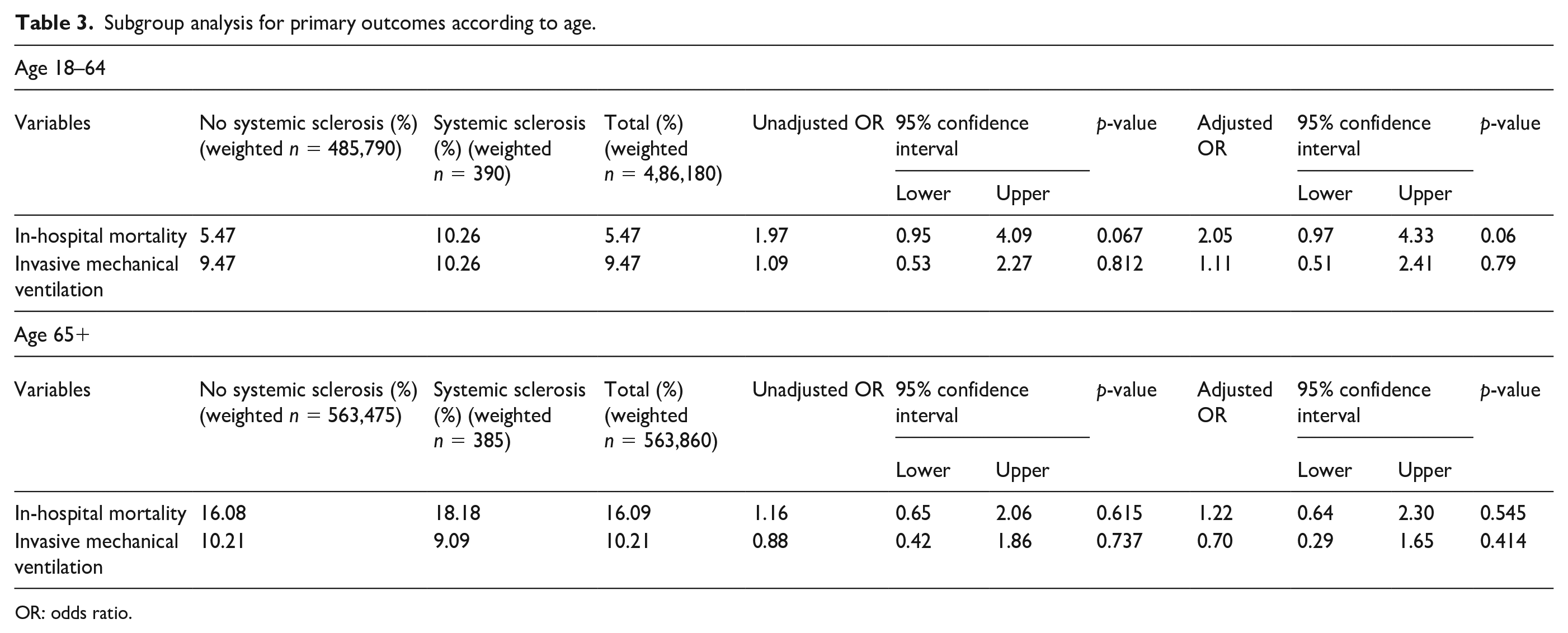
OR: odds ratio.
Discussion
In this study, SSc patients with COVID-19 had worse outcomes (i.e. higher composite endpoint of major adverse events) compared to COVID-19 patients without SSc. Furthermore, there was a trend toward increased in-hospital mortality in the younger age group. These findings could be attributed to the fact that patients with SSc had more co-morbidities and a higher Elixhauser Comorbidity Index at baseline, and despite adjustment for confounders, some residual confounding is likely to remain. A study in Spain concluded that the higher COVID-19 mortality rates seen in patients with systemic autoimmune diseases were related to the higher burden of comorbidities, secondary to direct organ damage and sequelae of their condition. 7 However, a study conducted on a cohort of SSc patients in Brazil reported that pulmonary arterial hypertension, interstitial lung disease, cardiac involvement, and use of rituximab, but not age, gender, comorbidities or use of corticosteroids, were predictors of worse outcome for COVID-19. 8 Therefore, apart from comorbidities, disease-related factors such as specific organ involvement and treatment history could also play a role in the outcome of SSc patients with COVID-19.
It is important to note that there were no significant differences between SSc patients with COVID-19 versus without regarding the hospital LOS and hospital costs. Although there was a numerically higher number of dispositions to facility (SNF, ICF, or others), this difference was not statistically significant. This is reassuring as the worse outcomes in COVID-19 patients did not translate into an increased healthcare burden. Nevertheless, due to the scarcity of prior literature on this, further studies are needed to confirm these findings.
The pathogenesis of both COVID-19 and SSc has an underlying autoimmune disturbance and the interplay between these two conditions may also contribute to worse COVID-19 outcomes in patients with SSc. An Italian survey study has hypothesized that COVID-19 might amplify the ongoing systemic sclerosis manifestations during the acute phase of viral infection and that later it might also contribute to advanced scleroderma organ damage. 6 In addition, the concurrent use of immunosuppressant therapy for SSc may also make these patients more susceptible to COVID-19 infection. Immunocompromised patients have shown an increased risk of in-hospital mortality related to the Omicron variant of COVID-19. 9
Previously many large population-based or health-system-based studies conducted to date suggest an elevated risk of COVID-19 death in people with rheumatic disease. 10 A recent meta-analysis concluded that patients with rheumatic and musculoskeletal diseases (RMDs) have higher rates of SARS-CoV-2 infection and an increased mortality rate. In concurrence with our study, it reported similar odds of mechanical ventilation in patients with and without an RMD. 11 We leveraged a nationally representative database to provide data on outcomes of SSc patients with COVID-19, therefore, which gives the advantage of a relatively larger sample size than previous small single-center studies.
The main limitation of our analysis is the particular setting examined, namely, hospitalized patients, which does not allow evaluating the actual impact of COVID-19 on the entire population of SSc patients. Moreover, our study is limited by the fact that despite including a large total sample size, only 0.07% of the patients in this study had SSc. Therefore, our results might be underpowered to detect any significant differences between SSc patients and non-SSc patients with COVID-19 in our individual outcomes. Furthermore, despite controlling for many confounding variables, some residual confounding is possible, which could impact the validity of our findings. There is no information available on the pharmacotherapy employed for either SSc or COVID-19 which could have effects on disease prognosis. As the vaccination status of the participants was unavailable, the presentation and outcomes of COVID-19 infection in SSc patients already vaccinated for COVID-19 remain unclear. Additional clinical characteristics like the disease duration of SSc which can impact the inpatient outcomes adversely are not available in the NIS database. Finally, using diagnostic codes to record patient data in database studies also carries a risk of incorrect diagnoses.
In summary, COVID-19 patients with SSc had worse outcomes (i.e. higher composite endpoint of major adverse events) than those without SSc. However, this should be interpreted with caution as the individual outcomes did not differ significantly between the two groups. Further studies are needed to establish a better understanding of the relationship between COVID-19 and SSc, particularly in terms of risk factors, complications, and outcomes.
Footnotes
Acknowledgements
The authors thank Dr Abdul Mannan Khan Minhas for the conducting the statistical analysis and providing an expert evaluation of the results.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
No ethical approval was required for this study.
