Abstract
Objective:
Systemic sclerosis is an autoimmune disease characterized by fibrosis of the skin and internal organs including the lung. Mucosal-associated invariant T cells are innate-like T lymphocytes able to produce various cytokines and cytotoxic mediators such as granzyme B. A large body of evidence supports a role of mucosal-associated invariant T cells in autoimmune disease but more recent reports suggest also a potential role in fibrotic conditions. Therefore, we herein addressed the question as whether mucosal-associated invariant T cells may have an altered profile in systemic sclerosis.
Methods:
Mucosal-associated invariant T cell frequency was analyzed by flow cytometry, using fresh peripheral blood from 74 consecutive systemic sclerosis patients who were compared to 44 healthy donors. In addition, in-depth mucosal-associated invariant T cell phenotype and function were analyzed in unselected 29 women with systemic sclerosis who were compared to 23 healthy women donors.
Results:
Proportion of circulating mucosal-associated invariant T cells was significantly reduced by 68% in systemic sclerosis compared to healthy donors (0.78% in systemic sclerosis vs 2.5%, p < 0.0001). Within systemic sclerosis subsets, mucosal-associated invariant T cells were reduced in patients with interstitial lung disease (systemic sclerosis-interstitial lung disease) (0.56% vs 0.96% in patients without interstitial lung disease, p = 0.04). Moreover, in systemic sclerosis patients, mucosal-associated invariant T cells displayed an activated phenotype indicated by markedly increased CD69+ mucosal-associated invariant T cell frequency (20% mucosal-associated invariant T cell CD69+ compared to 9.4% in healthy donors, p = 0.0014). Interestingly, mucosal-associated invariant T cells from systemic sclerosis-interstitial lung disease patients had a more pronounced altered phenotype compared to systemic sclerosis without interstitial lung disease with a correlation between mucosal-associated invariant T cells expressing CCR6+ and mucosal-associated invariant T cell frequency (r = 0.8, p = 0.006).
Conclusion:
Circulating mucosal-associated invariant T cells were reduced and exhibited an activated phenotype in systemic sclerosis patients. This peripheral mucosal-associated invariant T cell deficiency may be related to enhanced apoptosis and/or homing in inflamed tissue, particularly in systemic sclerosis-interstitial lung disease patients.
Introduction
Systemic sclerosis (SSc) is an orphan connective tissue disease with high morbidity and mortality. 1 The pathogenesis of this disease remains elusive but is likely to involve environmental events in a genetically susceptible individual, resulting in vascular alterations, immunity dysregulation, and fibrosis of the skin and internal organs. At early stages, it is thought that in response to microvascular impairment, the immune system is activated, implying both innate and adaptive immunity. 2 As shown by their infiltration in fibrotic tissues, T cells appear to play a prominent role. T CD4+ cells could be primarily involved and several line of evidence support that they have an activated profile and produce high amount of Th2 cytokines, such as interleukin (IL)-4 and IL-13, subsequently stimulating fibroblast proliferation. 3 Although the role of IL-17 in SSc has not been definitely established, 4 the increased levels of IL-17 in sera together with the accumulation of IL-17+ cells in skin 5 suggest a role of IL-17 in SSc. Moreover, granzyme B, mainly produced by cytotoxic T cells, is suspected to participate in SSc pathogenesis by cleaving autoantigens targeted by autoantibodies. 6 Innate immunity is also important in SSc pathogenesis, with a key role of monocytes, macrophages, and dendritic cells in addition to innate lymphoid cells. Finally, at the injury site, microvascular damage and immune cells recruitment result in a progressive fibrosis characterized by the presence of α-smooth muscle actin-positive, apoptosis-resistant myofibroblasts that secrete transforming growth factor (TGF)-β and other profibrotic mediators. 2
Mucosal-associated invariant T (MAIT) cells are innate-like T cells that account for about 5% of peripheral T cells and are abundant in human blood, liver, and mucosal tissues. They express an invariant T-cell receptor (TCR) alpha chain which is Vα7.2-Jα33 and Vα19-Jα33 in humans and mice, respectively. 7 These cells are restricted to major histocompatibility complex (MHC) class I-related (MR1) molecule which presents vitamin B2-derived metabolites derived from bacteria and yeast. 8 Once activated, either by TCR or by cytokines, 9 MAIT cells produce numerous pro-inflammatory cytokines (interferon (IFN)-γ, tumor necrosis factor (TNF)-α, IL-17) and cytotoxic factors such as granzyme B.10,11 Compelling evidences support a role of MAIT cells in autoimmune diseases,12,13 as well as in fibrotic diseases such as in liver fibrosis. 14
A single study investigated circulating MAIT cells in SSc and suggested a decrease in circulating MAIT cells compared to healthy controls, without any correlation with clinical or biological features. 15 This latter study enrolled 27 SSc patients and only restrictedly analyzed circulating MAIT cell proportion.
In the present study, we addressed the question as whether MAIT cells may have an altered profile in SSc using a much larger population of consecutive patients.
Methods
Healthy adult donors and SSc patients
Peripheral blood samples were collected from 44 healthy controls (healthy blood donors) and 74 consecutive patients with SSc from rheumatology department of Cochin hospital. SSc patients were classified according to classification criteria of the American College of Rheumatology and European League Against Rheumatism. 16
For all SSc patients, clinical data have been collected from medical records, including age, gender, disease duration, skin subtype (diffuse or limited), digital ulcers (past or current), joint synovitis, and current treatment (Table 1). Interstitial lung disease (ILD) was identified on the basis of a high-resolution computed tomographic scan and was assessed with the forced vital capacity (FVC) and the diffusing capacity for carbon monoxide (DLCO). Pre-capillary pulmonary arterial hypertension was defined as a mean pulmonary arterial pressure above 25 mmHg with a pulmonary arterial wedge pressure lower than 15 mmHg on right heart catheterization. The extent of skin fibrosis was assessed with the modified Rodnan skin score (mRSS), 17 the disease severity with the Medsger score, 18 and the disease activity with the Valentini 2016 score. 19 We also collected biological data, including the immunological status (presence of anti-centromere antibodies, anti-Scl70 antibodies, and/or anti-RNA polymerase III antibodies) and systemic inflammation markers (C-reactive protein, erythrocyte sedimentation rate).
General characteristics of SSc patients.

SSc: systemic sclerosis; SD: standard deviation; IQR: interquartile range; ILD: interstitial lung disease; HRCT: high-resolution computed tomography; DLCO: diffusing capacity for carbon monoxide; FVC: forced vital capacity; ACA: anti-centromere antibodies; sPAP: systolic pulmonary arterial pressure; CRP: C-reactive protein; MRSS: modified Rodnan skin score.
Values are median with IQR or mean with SD, frequencies with percentages.
Our bio-collection was approved by the Ethics Committee (Comité de Protection des Personnes (CPP) Ile-de-France) (Am3968-3-2307) and all participants provided written informed consent before sampling.
Peripheral blood mononuclear cell preparation and flow cytometry analysis
After isolation of peripheral blood mononuclear cells (PBMCs) from fresh blood samples using Ficoll-Paque (Leucosep Dutsher), PBMCs were stained in Facs Buffer (phosphate-buffered saline (PBS) containing 5% fetal calf serum (FCS) and 0.1% sodium azide) with the following fluorochrome-conjugated anti-human monoclonal antibodies (mAbs): anti-CD3 APC (SK7), CD4 BV711 (OKT4), TCR Vα7.2 BV421 (3C10), CD161 BV605 (HP-3G10) from BioLegend and CD8 APC-Cy7 (SK1). This labeling allowing the identification and quantification of MAIT cells was performed for all patients (44 healthy donors and 74 SSc patients).
For 23 healthy donors and 29 SSc patients, we also studied MAIT cell phenotype and function. Along with labeling for identification, we added a surface staining, an intranuclear staining for B-cell lymphoma 2 (Bcl-2) and Ki67 detection and an intra-cellular staining for detection of cytokines and granzyme B (GrB), as previously reported by Nel et al. 20 The fluorochrome-conjugated anti-human monoclonal antibodies used are detailed in Supplementary Table 1.
Samples were acquired on a BD Biosciences LSR-Fortessa cytometer. Results were analyzed with FlowJo analysis software V10.1.
Statistical analyses
The results are expressed as mean with standard deviation, median with range, and numbers with frequencies, according to variables distribution determined by Shapiro–Wilk test. Mann–Whitney test was used to compare two groups. Correlations were performed using Spearman’s test.
For multivariate analysis, we performed a linear regression, with MAIT cell frequency as the dependent variable (variable to explain) and immunosuppressive medication and ILD as the covariates (explanatory variables). Since the distribution of the residuals does not follow a normal distribution, we calculated confidence intervals and p-values by bootstrapping (1000 iterations).
The analyses were performed using GAPHPAD PRISM 8.0 (GraphPad Software, San Diego, CA, USA). Correlograms and circular bar plot were produced with R studio software. All p-values are two-sided, and p-values less than 0.05 were considered to be statistically significant.
Results
Study population
Most of the healthy donors were female (93%) and the mean age was 45.6 years. Among the 74 SSc patients (85% were female, mean age ± SD: 58 ± 13.8 years), the median (interquartile range (IQR)) disease duration was 9 (3–14) years, 43% had the diffuse cutaneous SSc, and 34% had an ILD. Twenty-six percent of SSc patients received glucocorticoids and 31% conventional synthetic immunosuppressive medication (Azathioprine, Methotrexate, Mycophenolate mofetil, Tacrolimus, or Leflunomide). Only seven patients were treated with biological therapies. Details are shown in Table 1.
Circulating MAIT cell frequency is reduced in SSc patients
In human blood, MAIT cells are divided into two main subsets, CD8+ and CD8− CD4− (double negative, DN), usually representing 80% and 15% of total MAIT cells, respectively. 21 Thus, MAIT cells were identified, such as TCR Vα7.2+ and CD161++ cells, among CD8+ and double-negative T cells after exclusion of iNKT cells and γδ T cells (Supplementary Figure 2A).
We first studied MAIT cell proportion in SSc patients compared to healthy controls (Figure 1(a) and (b)). Circulating MAIT cell frequency was significantly reduced in SSc compared to healthy donors (median (IQR): 0.78% (0.26–1.8) in SSc vs 2.5% (1.4–4.4) in healthy donors, p < 0.0001, Figure 1(c)).
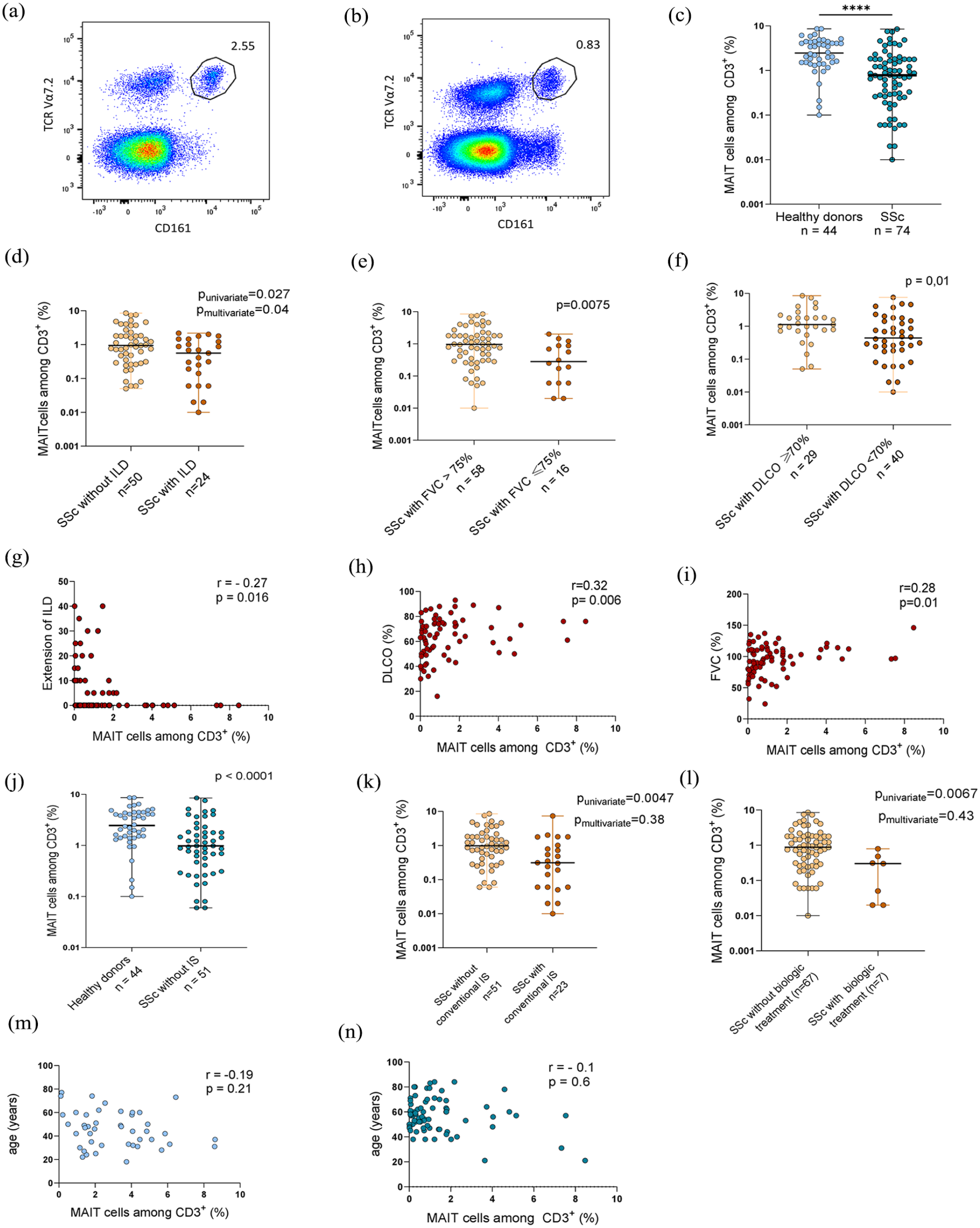
MAIT cell frequency in SSc patients (n = 74) and healthy controls (n = 44) (a–c). MAIT cell frequency according to lung involvement, current immunosuppressive therapy, and age (d–m). (a) Pseudocolor plot representation of cells identified as MAIT cells (Vα7.2+ and CD161++) in a healthy donor. (b) Pseudocolor plot representation of cells identified as MAIT cells (Vα7.2+ and CD161++) in a SSc patient. (c) Proportion of circulating MAIT cells among CD3+ cells in SSc and controls. (d) Proportion of circulating MAIT cells among CD3+ cells in SSc-ILD (n = 24) compared to SSc without ILD (n = 50). (e) Proportion of circulating MAIT cells among CD3+ cells in SSc with FVC ⩽75% (n = 16) compared to SSc with FVC > 75% (n = 58). (f) Proportion of circulating MAIT cells among CD3+ cells in SSc with DLCO <70% (n = 40) compared to SSc with DLCO ⩾ 70% (n = 29). (g) Correlation between MAIT cell frequency and the extension of ILD (%) in SSc patients (n = 74). (h) Correlation between MAIT cell frequency and DLCO % of the predicted value in SSc patients (n = 73). (i) Correlation between MAIT cell frequency and FVC % of the predicted value in SSc patients (n = 74). (j) Proportion of circulating MAIT cells among CD3+ cells in SSc without immunosuppressive therapy (n = 51) compared to healthy donors (n = 44). (k) Proportion of circulating MAIT cells among CD3+ cells in SSc patients with conventional synthetic immunosuppressive therapy (n = 23) compared to SSc without conventional synthetic immunosuppressive therapy (n = 51). (l) Proportion of circulating MAIT cells among CD3+ cells in SSc patients with biologic treatment (n = 7) compared to SSc without biologic treatment (n = 67). (m) Correlation between MAIT cell frequency and the age (years) in healthy donors (n = 44). (n) Correlation between MAIT cell frequency and the age (years) in SSc patients (n = 74).
We also compared the frequency of different MAIT subpopulation (CD4+, CD8+, and double-negative MAIT cells) for 29 SSc patients and 23 healthy donors. In SSc patients, circulating MAIT cell deficiency affected the three subsets of MAIT cells: 64% reduction of CD8+ MAIT cells in SSc patients compared to healthy donors (0.54% in SSc vs 1.5%, p = 0.0005, Supplementary Figure 1A), 58% reduction of CD4− CD8− MAIT cells in SSc patients compared to healthy donors (0.11% in SSc vs 0.27%, p = 0.05, Supplementary Figure 1B), and 42% reduction of CD4+ MAIT cells in SSc patients compared to healthy donors (0.038% in SSc vs 0.065%, p = 0.0032, Supplementary Figure 1C).
Relationships between circulating MAIT cells and organ involvement
We next investigated whether MAIT cell frequency was associated with clinical variables in SSc patients. After adjusting on immunosuppressive therapy, MAIT cell frequency was still reduced in patients with interstitial lung disease (SSc-ILD) (median (IQR): 0.56% (0.1–1.34) vs 0.94% (0.3–2.1) as compared to SSc patients without ILD, p = 0.04, Figure 1(d)), with a negative correlation between MAIT cell frequency and the extent of ILD (r = 0.27, p = 0.016, Figure 1(g)). Interestingly, MAIT cell deficiency was even lower in patients with severe ILD and reduced FVC (median (IQR): 0.28% (0.06–0.88) in patients with FVC < 75% vs 0.96% (0.31–1.82) in patients with FVC ⩾ 75%, p = 0.0075, Figure 1(e)) or reduced DLCO (median (IQR): 0.44% (0.18–1.45) in patients with DLCO < 70% vs 1.1% (0.72–2) in patients with DLCO ⩾ 70%, p = 0.01, Figure 1(f)). We found a correlation between MAIT cell proportion and % DLCO (r = 0.32, p = 0.006, Figure 1(h)) and between MAIT cell proportion and % FVC (r = 0.28, p = 0.01, Figure 1(i)).
MAIT cell deficiency did not correlate with disease activity and severity scores, SSc cutaneous subtype (diffuse or limited), symptomatic gastroesophageal reflux, systemic inflammation, and the immunological status (Table 2). If MAIT cells were decreased in patients receiving conventional synthetic immunosuppressive medication and patients receiving biologic treatment in univariate analysis (p = 0.0047 and p = 0.0067, respectively), it is important to highlight that MAIT cell deficiency persisted in SSc patients after exclusion of patients receiving immunosuppressive treatment (median (IQR): 0.98% (0.41–1.8) in SSc versus 2.5% (1.4–4.4) in healthy donors, p < 0.0001 Moreover, circulating MAIT cells appeared to be similar in treated and untreated patients by conventional synthetic immunosuppressive medication (p = 0.38, Figure 1(k)) or biologic treatment (p = 0.43, Figure 1(l)) after adjustment on the presence of ILD. This highlights the influence of ILD but not the immunosuppressive therapy on MAIT cell counts.
Proportion of MAIT cells in SSc patients according to clinical feature of the disease.
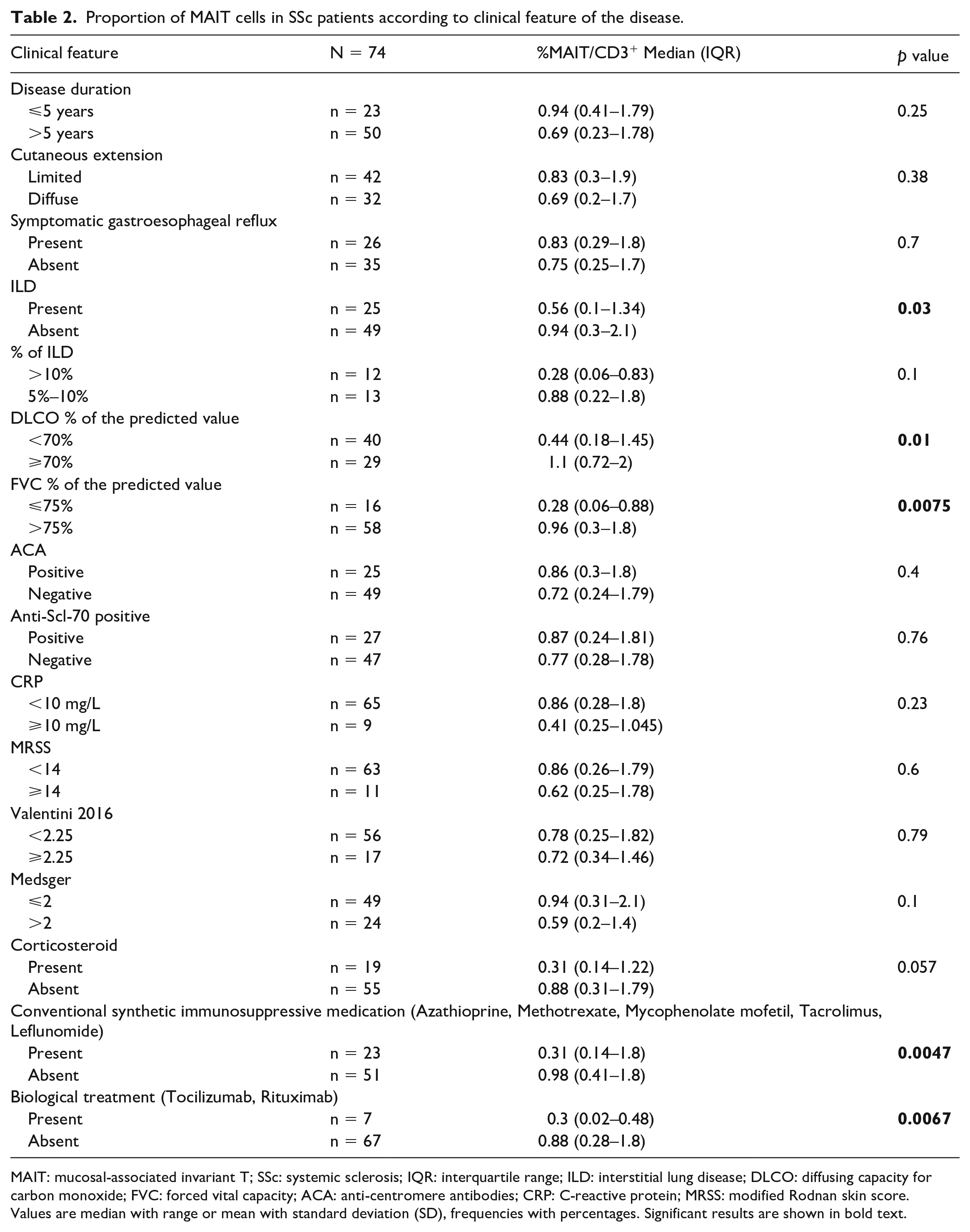
MAIT: mucosal-associated invariant T; SSc: systemic sclerosis; IQR: interquartile range; ILD: interstitial lung disease; DLCO: diffusing capacity for carbon monoxide; FVC: forced vital capacity; ACA: anti-centromere antibodies; CRP: C-reactive protein; MRSS: modified Rodnan skin score.
Values are median with range or mean with standard deviation (SD), frequencies with percentages. Significant results are shown in bold text.
Finally, MAIT cell frequency did not correlate with the age, both in controls (r = −0.19, p = 0.21, Figure 1(m)) and SSc patients (r = −0.1, p = 0.6, Figure 1(n)).
Circulating MAIT cell phenotype is altered in SSc patients
We next analyzed the phenotype of circulating MAIT cells in the participants with SSc and healthy donors (Supplementary Figures 2 and 4). In SSc patients, MAIT cells displayed a strongly activated phenotype indicated by markedly increased CD69+ MAIT cell frequency compared to healthy donors (20% MAIT cell CD69+ compared to 9.4% in HD, p = 0.0014, Figure 2(a) and (b)). We also observed a trend for an increase in proliferation of MAIT cells in SSc patients with an increase in Ki67+ MAIT cells (0.97% Ki67+ MAIT cells compared to 0.74% in healthy donors, p = 0.13, Figure 2(a) and (c)).
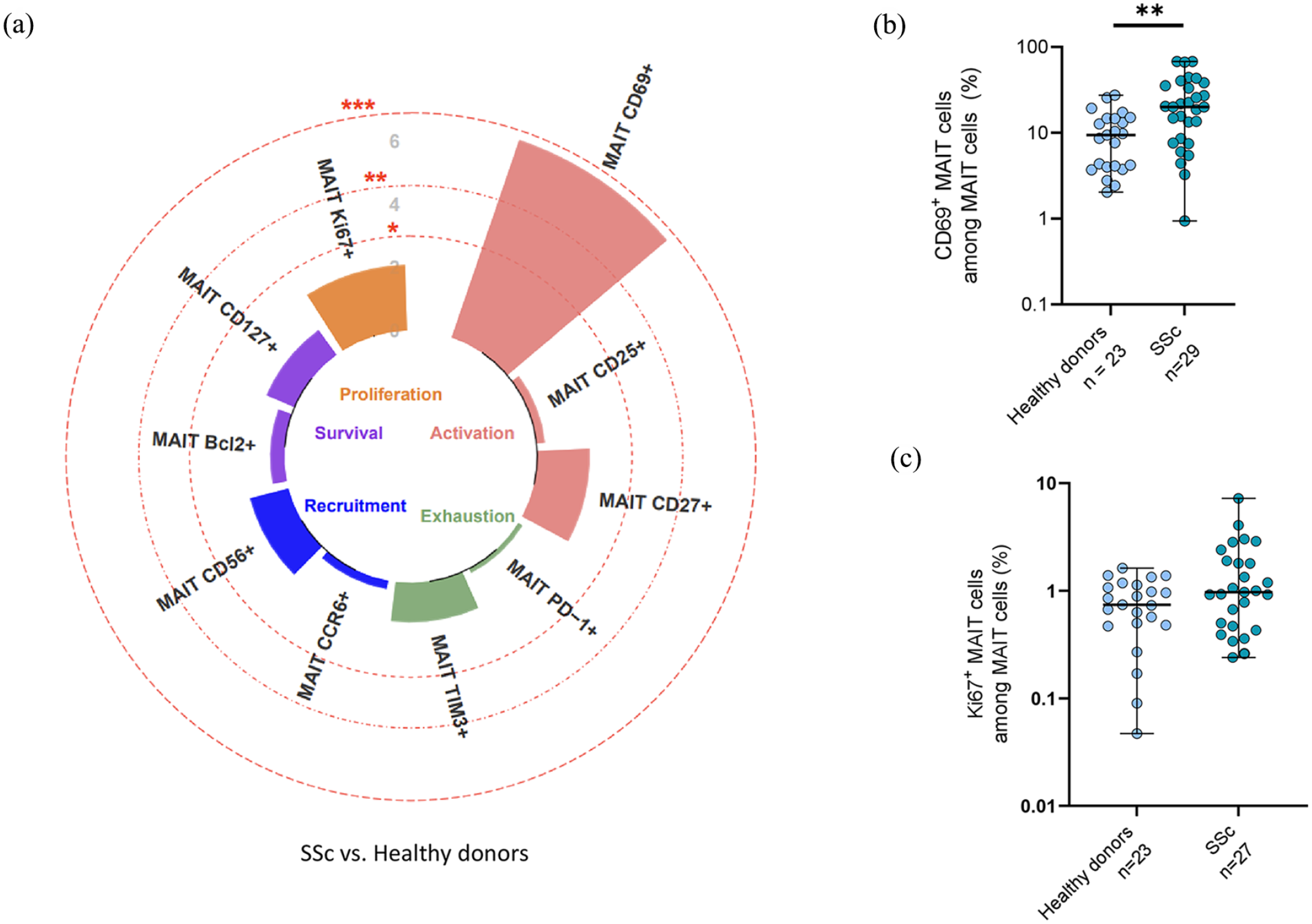
Phenotype of MAIT cells in healthy donors (n = 23) and SSc patients (n = 29). The phenotype analysis included the study of activation (CD69, CD25, CD27), exhaustion (PD1, Tim3), recruitment (CD56, CCR6), survival (Bcl2, CD127), and proliferation (Ki67). (a) Circular bar plot represents negative loge p values of MAIT cell phenotype between healthy donors (n = 23) and systemic sclerosis (n = 29). (b) Proportion of MAIT cells expressing CD69 among MAIT cells in healthy donors (n = 23) and SSc patients (n = 29). (c) Proportion of MAIT cells expressing Ki67 among MAIT cells in healthy donors (n = 23) and SSc patients (n = 27).
We then performed correlograms with all these MAIT cell variables (Figure 3(a) and (b)). In SSc patients (Figure 3(b)), we identified a correlation between MAIT cell frequency and survival markers (CD127) (r = 0.4, p = 0.02, Figure 3(e)), and between MAIT cell frequency and frequencies of MAIT cells expressing exhaustion marker (PD-1) (r = −0.4, p = 0.035, Figure 3(f)). These correlations were absent in healthy donors (Figure 3(a)). We also found, only in SSc patients, a correlation between frequencies of MAIT cells expressing adhesion/activation molecule (CD56) and activation marker (CD69) (r = −0.7, p < 0.0001, Figure 3(g)). Altogether, our results reveal MAIT cell alterations in SSc patients compared to healthy donors.
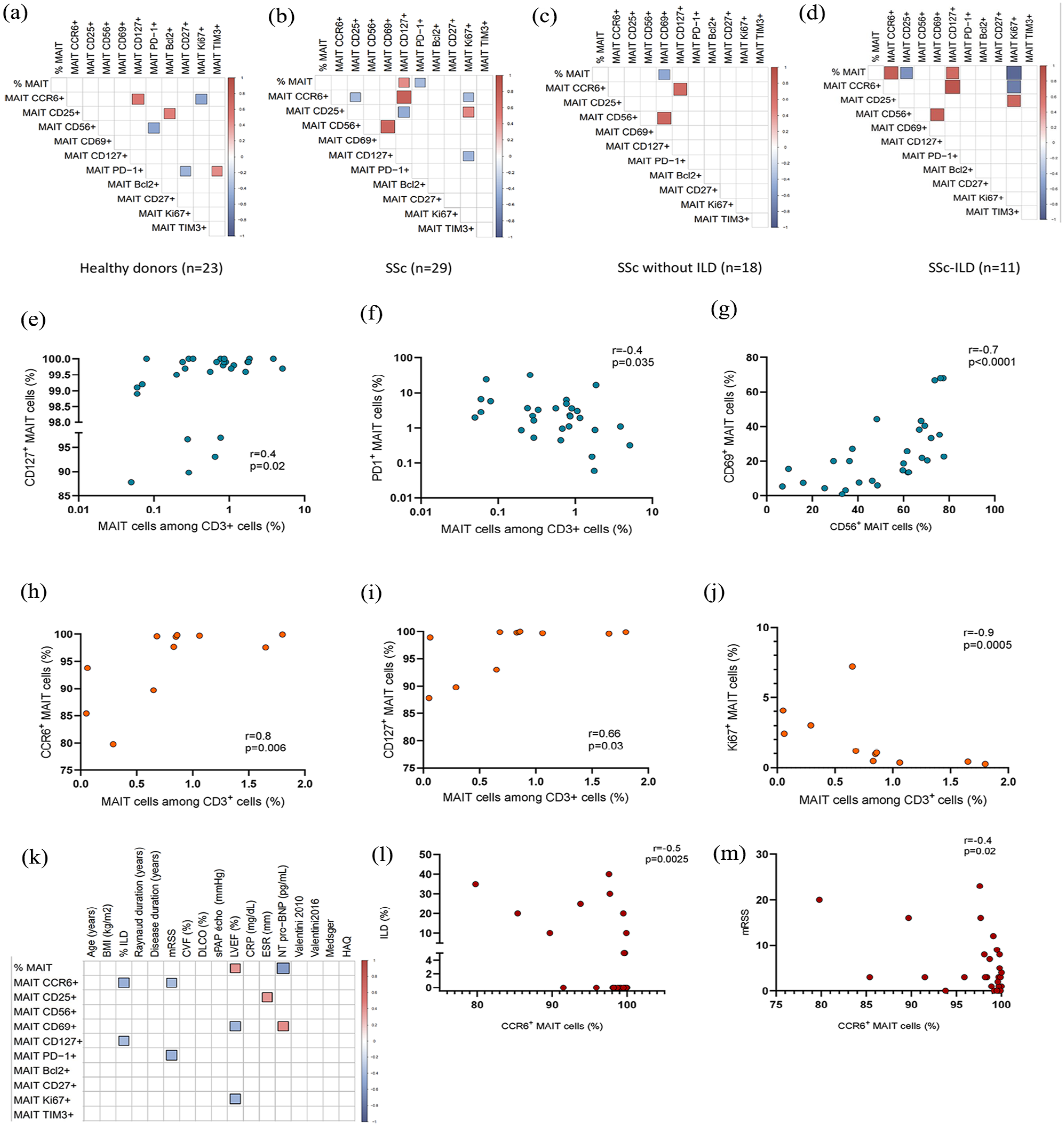
Correlation between circulating MAIT cell frequency and phenotypes. (a–d) Correlograms of circulating MAIT cell frequency and marker expression (a) in healthy donors (n = 23), (b) in SSc patients (n = 29), (c) in SSc without ILD (n = 18), and (d) in SSc-ILD (n = 11). (e–h) Correlation in SSc patients (n = 29) between MAIT cells among CD3+ cells and CD127+ MAIT cells (e) and PD1+ MAIT cells (f), between CD56+ MAIT cells and CD69+ MAIT cells (g). (i–j) Correlation in SSc-ILD patients (n = 11) between MAIT cells among CD3+ cells and CCR6+ MAIT cells (h), CD127+ MAIT cells (i), and Ki67+ MAIT cells (j). (k) Correlograms of circulating MAIT cell frequency, phenotypes, and clinical variables in SSc patients (n = 29). (l) Correlation between frequency of MAIT cells expressing CCR6 and % extension of ILD in SSc patients (n = 29). (m) Correlation between frequency of MAIT cells expressing CCR6 and mRSS in SSc patients (n = 29).
Finally, correlograms of circulating MAIT cell phenotypes and clinical variables (Figure 3(k)) exhibited negative correlations between frequency of MAIT cells expressing CCR6 and % of ILD (r = −0.5, p = 0.003, Figure 3(l)) and mRSS score (r = −0.4, p = 0.02, Figure 3(m)).
Alteration of MAIT cell phenotype is more pronounced in SSc-ILD
As MAIT cell frequency was decreased in the sub-group of SSc patients with ILD (Figure 1(d)), and also because MAIT cells expressing CCR6 were negatively correlated with ILD extent (Figure 3(l)), we focused on MAIT cells phenotype in SSc-ILD patients.
We observed positive correlations between MAIT cell frequency and MAIT cell frequency expressing CCR6 (tissue recruitment molecule) (r = 0.8, p = 0.006, Figure 3(h)) and CD127 (survival marker) (r = 0.66, p = 0.03, Figure 3(i)). Moreover, Ki67+ MAIT cells negatively correlated with MAIT cell frequency (r = −0.9, p = 0.0005, Figure 3(j)). Theses correlations were lost in SSc patients without ILD (Figure 3(c)).
Modification of MAIT cell function in SSc-ILD
Although no difference in the production of cytokines by MAIT cells was found in SSc patients compared to healthy controls (Supplementary Figures 3 and 5), the profile appeared different in cells from patients with SSc-ILD compared to SSc without ILD. We analyzed the ability of MAIT cells to produce Th1 cytokines, IL-17, and GrB. Although not reaching statistical significance, we observed a 30% increase in MAIT cells producing none of the effector molecules (27.8% in SSc-ILD compared to 19.4% in SSc without ILD, Figure 4(b)–(d)), with decrease of monofunctional and bifunctional subsets in SSc-ILD compared to SSc without ILD (Figure 4(d)). Furthermore, the main secreted population which are the IFN-γ+ TNF-α+ MAIT cells had a trend to be reduced in SSc-ILD (median (IQR): 38.5% (14.5–47.5) compared to 47.5% (29.6–53.7) in SSc without ILD, p = 0.08, Figure 4(e)).
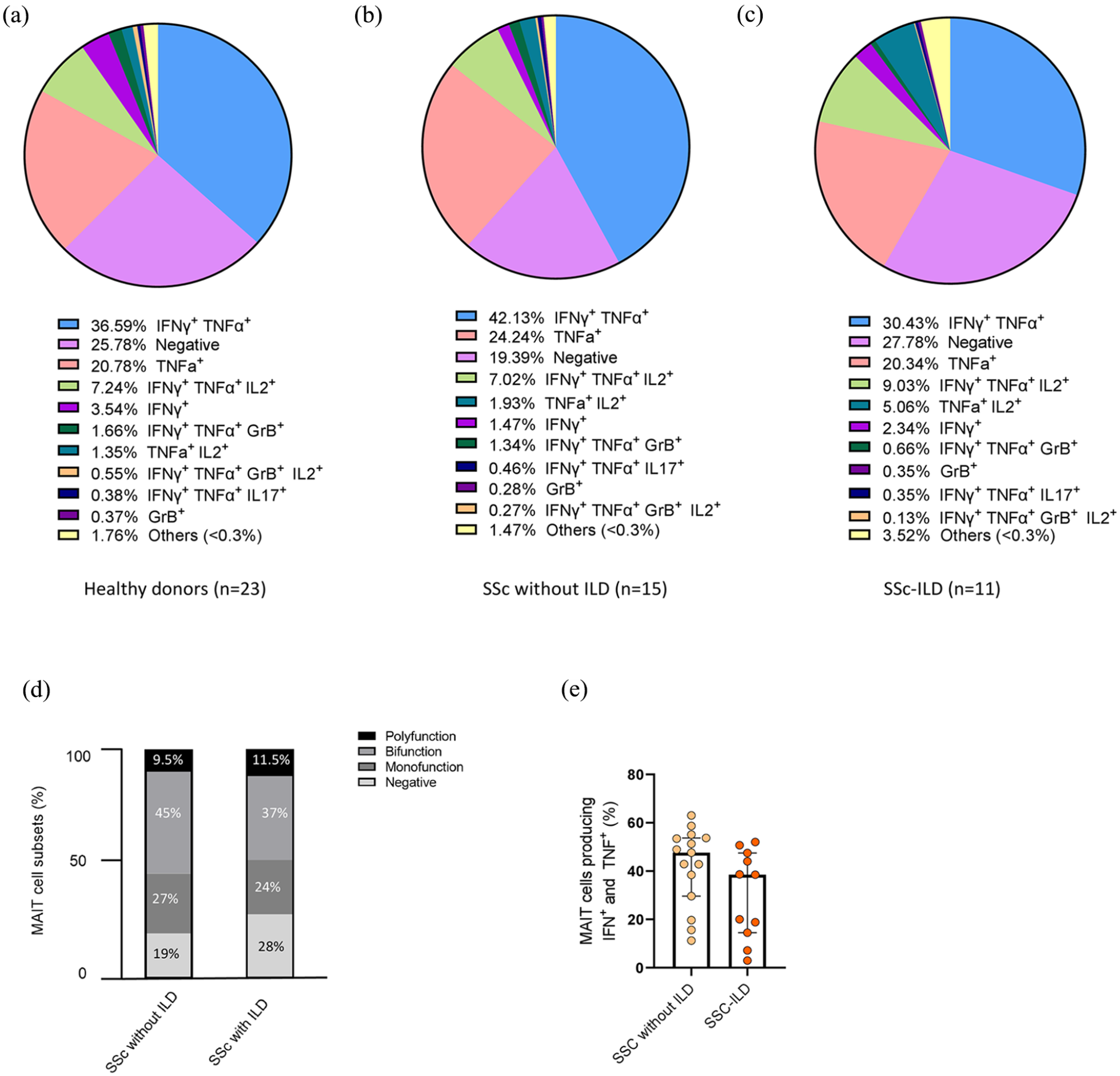
Functional analysis of MAIT cells in healthy donors, SSc without ILD, and SSc-ILD. (a–c) Pie charts showing MAIT cells producing Th1 cytokines, IL-17, and/or GrB, in healthy donors (n = 23) (a), SSc without ILD patients (n = 15) (b), or SSc-ILD (n = 11) (c). (d) Bar plot representative of cumulative Th1 cytokines, IL-17, and GrB, production by MAIT cells, classified as producing none (negative), one (monofunction), two (bifunction), or more than two (polyfunction) of these factors in SSc without ILD (n = 15) and SSc-ILD (n = 11). Percentages represent the mean for each subset. (e) MAIT cells producing IFN-γ and TNF-α frequency in SSc without ILD (n = 15) and SSc-ILD (n = 11).
Discussion
The present study shows that circulating MAIT cells are altered in patients with SSc. Indeed, we herein show (1) a reduced proportion of MAIT cells in SSc, (2) an activated phenotype for the circulating MAIT cells, and (3) a more pronounced altered phenotype in SSc-ILD patients.
In patients with SSc, we demonstrated that circulating MAIT cells were decreased compared to healthy donors. This confirms, in a largest population, the results of Mekinian et al. 15 Altogether, these data are consistent with those in other autoimmune diseases. Indeed, decreased levels of MAIT cells were reported in multiple sclerosis, 22 systemic lupus erythematosus, 23 Sjogren syndrome, 24 rheumatoid arthritis,23,25,26 and type 1 diabetes. 20
Furthermore, MAIT cells of SSc patient appear to display altered cellular homeostasis. Indeed, the positive correlation between frequency of MAIT cells and the frequency of MAIT cells expressing CD127 (survival marker) and the negative correlation between frequency of MAIT cells and MAIT cells expressing PD-1, which were absent in healthy donors, suggest an alteration of MAIT cell homeostasis with a reduced survival in circulating blood. MAIT cell proliferation also appears to be affected, as evidenced by the trend to increase in Ki67+ MAIT cell frequency in SSc patients, suggesting proliferation of MAIT cells to counterbalance their deficit.
MAIT cell localization and their recognition of bacterial ligands might play a role in the interaction between microbiota and the immune system. Microbial infection is key to promote MAIT cell functions but the herein population of patients did not have active infection. However, since dysbiosis has been described in SSc, 27 a perspective might be in the future to investigate the potential influence of dysbiosis on MAIT cell proportion and activation state. As a proxy, detailed gastrointestinal assessment that was not done in our patients could provide indications about the link between gut involvement and MAIT cell biology.
These phenotype modifications (activation and homeostasis alteration) were in line with data obtained in other diseases. Indeed, MAIT cells exhibited an activated profile in liver fibrosis 14 with an upregulation of CD69 and CD25, and in type-1 diabetes 20 with an upregulation of CD25. Moreover, MAIT cells harbored an exhausted phenotype with an increase in PD1 expression in type-1 diabetes 20 and systemic lupus erythematosus. 23 A pro-apoptotic profile was also described in liver fibrosis 14 with a correlation between Bcl2+ MAIT cells and MAIT cell frequency.
The fate of circulating MAIT cells in these diseases, whether they die or migrate to inflamed tissue, remains enigmatic. Of note, an accumulation of MAIT cells, contrasting with circulating MAIT cells deficiency, have been described in salivary glands of Sjogren syndrome, 24 joint of rheumatoid arthritis, 23 and central nervous system of multiple sclerosis, 22 suggesting a migration from peripheral blood to inflammatory sites. Accumulation in liver fibrotic septa, while circulating MAIT cells are reduced in patients with fibrosis liver, is also described. 14 Moreover, Rouxel et al. 28 reported an increased migration of MAIT cells in the inflamed pancreas of NOD (non-obese diabetic) mice during disease progression toward diabetes. Adhesion molecules, integrins, and chemokines appear to play a key role in the migration of MAIT cells in inflamed tissue, 29 where they may contribute to the local inflammation and fibrotic disorders.
In our study, MAIT cell frequency was reduced in patients with ILD (SSc-ILD) even lower in the patients with severe ILD. Moreover, MAIT cells of SSc-ILD display a more pronounced altered phenotype than SSc without ILD, with a trend of functional alteration. Indeed, the analysis of the ability of MAIT cells to produce Th1 cytokines, IL-17 and GrB, revealed a 30% increase in MAIT cells producing none of the effector molecules (negative subset) in SSc-ILD with a trend to decrease the main secreted population, the MAIT cells producing IFN-γ+ TNF-α+. Although not reaching statistical significance, these results, close to those described in long-term type 1 diabetes, 20 suggest functional alteration of MAIT cells in SSc-ILD patients but will require further investigations and in particular in lung tissue.
The deficiency of MAIT cells more pronounced in SSc-ILD, as well as the negative correlation between the extent of ILD and frequency of CCR6+ MAIT cells, suggests a migration of circulating MAIT cells into the lung. Most of the MAIT cells able to migrate thanks to CCR6, a tissue recruitment chemokine, would therefore be within the lung.
Indeed, pulmonary epithelial cells highly expressed CCL20, which is the CCR6 ligand, in an inflammatory environment, 30 which would facilitate MAIT cell migration into the lung. Once in the lung tissue, one hypothesis is a communication between MAIT cells and stromal cells, either by granzyme B or pro-inflammatory cytokines MAIT cell production or by direct interaction, leading to lung cell affection.
The positive correlation between frequency of MAIT cells and frequency of MAIT cells expressing CCR6, and between frequencies of MAIT cells expressing activation marker CD69 and adhesion molecule CD56, reinforces the hypothesis of circulating activated MAIT cells migration into the lung, but will require further investigations. MAIT cell detection could be done by immunostaining or transcriptome analyses on lung parenchyma but unfortunately such lung biomaterial is not available for our patients. However, it could be done in an SSc-ILD mouse model, the transgenic Fra-2 mice. 31
Our study has several limitations. First, the healthy donors were younger than SSc patients, which may be a source of bias as MAIT cell frequency might decrease with age. 32 However, in our population, we did not find any correlation between age and MAIT cell frequency both among SSc patients and healthy donors.
Our sample of consecutive patients reflects routine care where patients have a quite advanced disease and the majority of patients are receiving a specific therapy. Indeed, a larger group of patients at early stages may provide additional insights. A replication cohort would be helpful particularly with numerous patients having ILD but we have studied the largest sample ever investigated and unselected patients reflect the routine practice. We assume to have identified herein stimulating clues that will open the door to several additional projects to clarify the role of MAIT cells in SSc.
To conclude, circulating MAIT cells were reduced and exhibited an activated phenotype in SSc patients compared to healthy controls. This peripheral MAIT cell deficiency may be related to enhanced apoptosis and/or homing in inflamed and fibrosis tissues, in particular the lung in SSc-ILD patients. MAIT cell study in lung tissue and skin, as well as functional studies, is necessary to better understand the implication of MAIT cells in SSc.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983231209807 – Supplemental material for Deficiency and altered phenotype of mucosal-associated invariant T cells in systemic sclerosis
Supplemental material, sj-pdf-1-jso-10.1177_23971983231209807 for Deficiency and altered phenotype of mucosal-associated invariant T cells in systemic sclerosis by Manon Lesturgie-Talarek, Virginie Gonzalez, Lucie Beaudoin, Camelia Frantz, Noémie Sénot, Zouriatou Gouda, Camille Rousseau, Jérôme Avouac, Agnès Lehuen and Yannick Allanore in Journal of Scleroderma and Related Disorders
Supplemental Material
sj-pdf-2-jso-10.1177_23971983231209807 – Supplemental material for Deficiency and altered phenotype of mucosal-associated invariant T cells in systemic sclerosis
Supplemental material, sj-pdf-2-jso-10.1177_23971983231209807 for Deficiency and altered phenotype of mucosal-associated invariant T cells in systemic sclerosis by Manon Lesturgie-Talarek, Virginie Gonzalez, Lucie Beaudoin, Camelia Frantz, Noémie Sénot, Zouriatou Gouda, Camille Rousseau, Jérôme Avouac, Agnès Lehuen and Yannick Allanore in Journal of Scleroderma and Related Disorders
Footnotes
Acknowledgements
We thank all the patients, physicians, and nurses of Cochin hospital (Rheumatology department) who helped with the study. We thank the Cybio facilities of the Cochin Institute. We thank the “Association des Sclerodermiques de France” for funding this study.
Authors’ note
Manon Lesturgie-Talarek and Virginie Gonzalez are shared first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the “Association des Sclerodermiques de France.” The funders were not involved in the study design; the collection, analysis, or interpretation of the data; the writing of this article; or the decision to submit it for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
