Abstract
Systemic sclerosis is an autoimmune disease characterized by fibrosis and small vessel vasculopathy, which affects various organ systems, such as the heart. Takotsubo cardiomyopathy is a transient cardiomyopathy in reaction to an emotional or physical trigger. There may be clinical and pathogenetic overlap between Takotsubo cardiomyopathy and primary systemic sclerosis heart disease, and some patients with systemic sclerosis have been diagnosed with recurrent Takotsubo cardiomyopathy. Our large systemic sclerosis clinical cohort was reviewed to identify cases diagnosed with Takotsubo cardiomyopathy. The clinical features, laboratory and imaging results were reviewed and evaluated to perform a comparison between cases. We identified five patients with systemic sclerosis, all female (age 68.6 ± 5.7 years), who were diagnosed with Takotsubo cardiomyopathy. Two of these patients had recurrent episodes: one case with a history of multiple episodes and the other with one recurrence. Typical features included repolarization abnormalities on the electrocardiogram and transient left ventricular dysfunction observed using echocardiography or cardiac magnetic resonance imaging. Our findings build upon previous reports and observations that systemic sclerosis may cause Takotsubo cardiomyopathy. To our knowledge, this is the largest case series of Takotsubo syndrome in patients with systemic sclerosis. This association may provide novel insights into the aetiopathogenesis of Takotsubo cardiomyopathy as part of primary systemic sclerosis heart involvement.
Introduction
Systemic sclerosis (SSc) is an autoimmune rheumatic disease with an estimated UK prevalence of approximately 1 in 10,000, although varying prevalence has been reported between 7 and 489 per million. 1 Pathologically, SSc is characterized by fibrosis of various organ systems and small vessel vasculopathy. 1 Organ manifestations in SSc most commonly include skin, lungs, gastrointestinal tract, and cardiovascular system. Although cardiac involvement, when present, is associated with significant mortality, primary cardiac involvement in SSc is often unrecognized, 2 and the precise pathological mechanisms are incompletely understood. One challenge is that the typical age and demographic of SSc mean that other cardiac comorbidities may be present.
Cardiac manifestations include asymptomatic conduction abnormalities or low-grade inflammation, which can eventually evolve into fulminant heart failure and myocarditis. Autopsy studies have reported inflammatory, fibrotic as well as vascular myocardial abnormalities in 80% of SSc patients. 3 The diagnosis of primary cardiac disease, however, remains challenging as clinical findings of cardiac abnormalities in the early stage of disease are uncommon and, if present, very subtle and non-specific. 2 The diagnostic workup is further hampered due to the lack of criteria for distinguishing between primary cardiac involvement and other more common causes of cardiomyopathy such as hypertension and ischaemia. 2 Furthermore, another cardiac manifestation which appears to be underreported is transient heart disease, which may clinically resemble Takotsubo cardiomyopathy (TTS). 4
TTS or stress-induced cardiomyopathy is a transient cardiomyopathy in reaction to an emotional or physical trigger. The pathophysiology of TTS is not completely elucidated. It has been linked to sympathetic nervous system activation and a catecholamine surge. 4 However, recent investigation proposed an additional inflammatory pathway which might be relevant in the context of autoimmune rheumatic diseases.3,4
Although TTS has traditionally been regarded as a benign, transient condition, it has recently been reported that acute mortality may be significant and patients can experience persistent symptoms, even though left ventricular function is completely normalized. 5 In addition, approximately 1% to 4% of patients will experience a recurrent episode of TTS, most commonly within 5 years after the first event. 4 There are no known risk factors to identify which patients will develop a recurrence, although triggering events are likely to recur with increased age. Furthermore, as the pathophysiological pathways are incompletely understood, there are no established strategies for preventing recurrences of TTS, although there might be a protective value of angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB) therapy initiated after a TTS episode. 5
In this case series, we present five patients, with a history of SSc, who were diagnosed with recurrent TTS and use these to anchor a comprehensive consideration of possible pathogenetic and clinical links between these diagnoses.
Case description
Our large SSc clinical cohort, consisting of over 2700 patients, was reviewed to select cases diagnosed with TTS. A total of five cases of TTS were identified, and subsequently their clinical features were evaluated in order to enable a comparison between the cases (Table 1). All patients provided informed written consent form for publication.
Patient demographics.
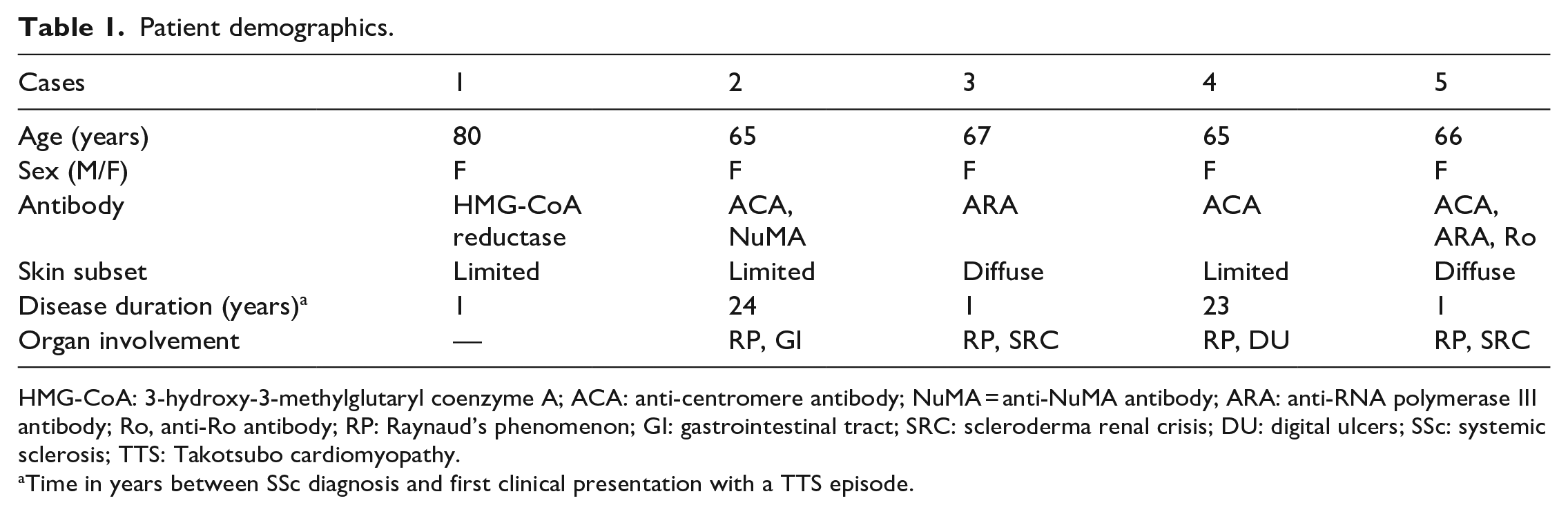
HMG-CoA: 3-hydroxy-3-methylglutaryl coenzyme A; ACA: anti-centromere antibody; NuMA = anti-NuMA antibody; ARA: anti-RNA polymerase III antibody; Ro, anti-Ro antibody; RP: Raynaud’s phenomenon; GI: gastrointestinal tract; SRC: scleroderma renal crisis; DU: digital ulcers; SSc: systemic sclerosis; TTS: Takotsubo cardiomyopathy.
Time in years between SSc diagnosis and first clinical presentation with a TTS episode.
Case 1
A 76-year-old woman with a history of limited cutaneous SSc overlap with myositis was diagnosed with nine separate episodes of TTS in 2016 and 2017. Antibody screening performed revealed an HMG-CoA (3-hydroxy-3-methylglutaryl coenzyme A) reductase antibody. Subsequently, in 2020, she reported central chest pain which was diagnosed as another recurrence of TTS. The echocardiogram during that episode showed severe left ventricular dysfunction and a subsequent cardiac magnetic resonance imaging (MRI) noted microvascular ischaemia and mid-wall non-ischaemic fibrosis suggesting prior inflammation-mediated changes.
Case 2
A 65-year-old woman with a history of limited cutaneous SSc, positive anti-centromere antibody (ACA) and positive NuMA presented with severe abdominal pain. A volvulus was diagnosed which was concluded to be gastrointestinal involvement of the SSc. During hospital admission, her electrocardiogram (ECG) showed abnormalities matching with an infero-lateral non q-wave myocardial infarction. The troponin was elevated to 1100 ng/L (reference range: 0–14 ng/L). There was hypokinesia of the inferior segments on the echocardiogram. A cardiac stress MRI was performed which showed regional wall motion abnormalities consistent with TTS, moderate left ventricle (LV) dysfunction and elevated T1-values in the mid-ventricular and apical segments of the LV (Figure 1, top row). Considering absence of ischaemia on the cardiac stress MRI, an angiogram was not performed. Patient was treated with an ACEi and betablockade. Three years later, despite continuation of this therapy, this patient experienced another TTS episode (Figure 1, bottom row).
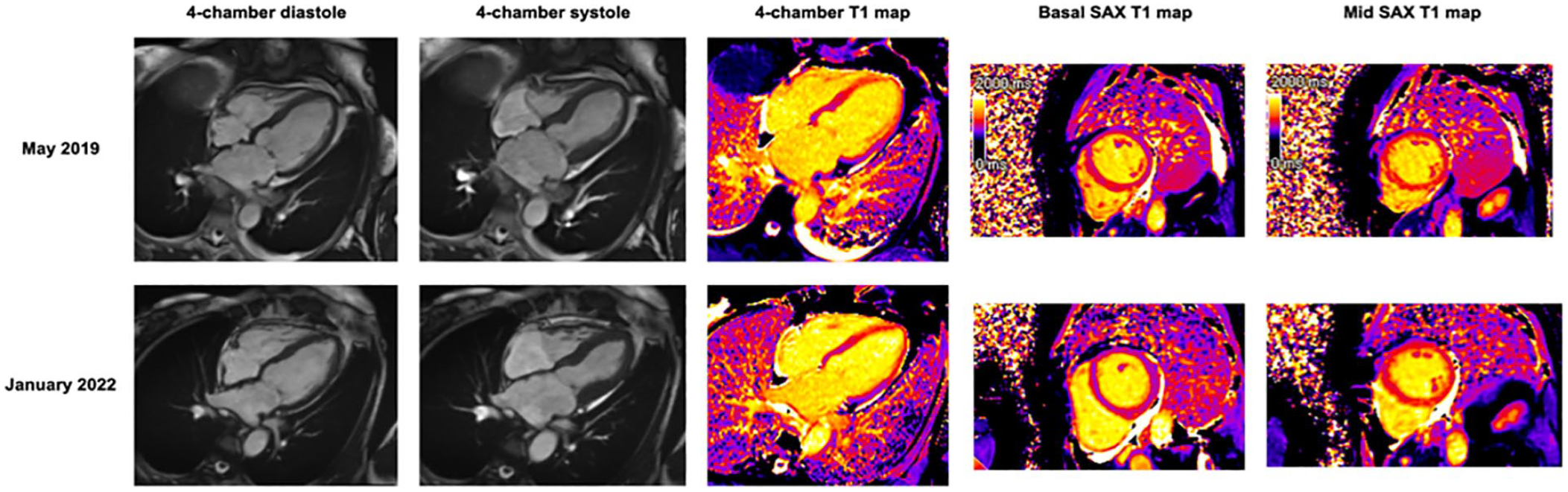
Cardiac MRI findings of a case with recurrent Takotsubo cardiomyopathy. The four-chamber SSFP images in diastole and systole depicting apical ballooning. T1 mapping performed in four-chamber and short-axis orientation demonstrating an increase in extracellular volume in the mid till apical segments of the left ventricle (high T1), consistent with edema.
Case 3
A 67-year-old woman with a history of diffuse cutaneous SSc and positive anti-RNA-polymerase III antibody presented with a scleroderma renal crisis (SRC), for which she required dialysis. Subsequently, she developed symptoms of acute left ventricular failure. ECG demonstrated sinus rhythm with a frequency of 110 beats per minute; left cardiac axis; normal p-waves; normal conduction times; slight ST-elevation in V1, V2 and ST-depression in I, AVL, V5 and V6. No P-R segment depression was found. She was diagnosed as having a possible ST-segment elevation myocardial infarction (STEMI) of the left anterior descending artery (LAD). Echocardiogram showed a reduced left ventricular function with dyskinesia in the apical segments and hyperdynamic basal anterior and inferior wall. The troponin was elevated to 110 ng/L (reference range: 0–14 ng/L) and brain natriuretic peptide (BNP) levels were 22,800 ng/L (reference range: 0–400 ng/L). Cardiac MRI demonstrated preserved global biventricular systolic function, but impaired biventricular longitudinal function. Native myocardial T1 was markedly elevated with mildly elevated T2 (the latter suggesting a component of oedema). Although coronary artery disease was not excluded using a coronary angiogram, the combination of typical regional wall motion abnormalities, relatively low troponin value and an increased T2 value on cardiac magnetic resonance imaging (CMR) was considered suggestive for TTS. An echocardiogram was repeated after 1 month and demonstrated normalized left ventricular function (LVF; EF: 55%–60%), further supporting the TTS diagnosis.
Case 4
A 65-year-old woman with a history of limited cutaneous SSc and positive ACA presented with chest pain. Further assessment showed an elevated troponin of 2911 ng/L (reference range: 0–14 ng/L). ECG demonstrated sinus rhythm with a frequency of 70 beats per minute, normal cardiac axis, normal p-waves with slight PR-segment depression in multiple leads (Figure 2). Small QRS complexes in the extremity leads and QS pattern in leads V1-V2 were observed and compared with previous ECGs. Subsequently, an angiogram was performed, which showed no significant coronary artery disease. Cardiac MRI was refused by the patient due to claustrophobia. Repeated echocardiogram demonstrated complete LVF recovery and supported the diagnosis of TTS.
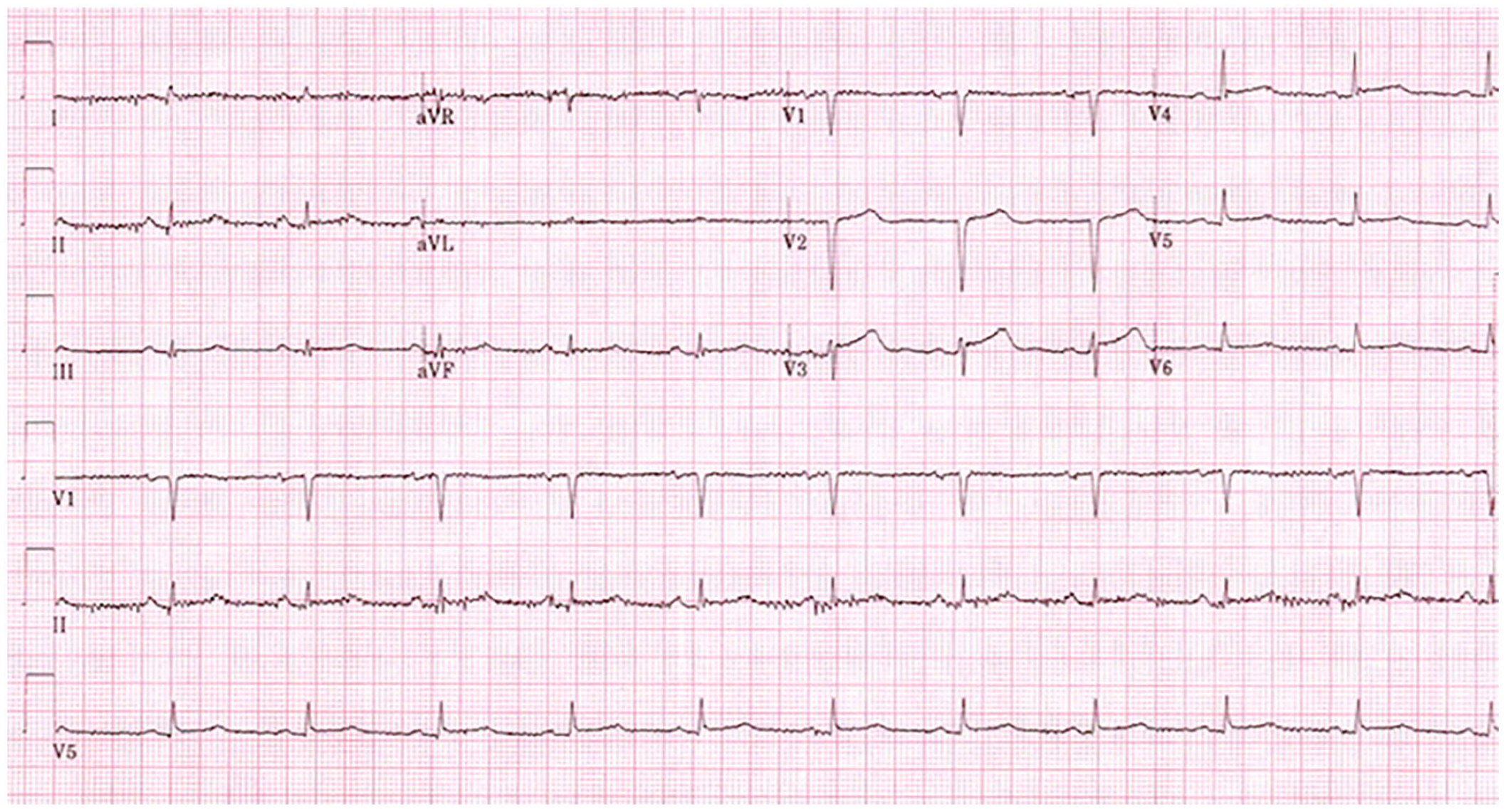
ECG findings of a case with recurrent Takotsubo cardiomyopathy. In patient 4, described above, there were ECG abnormalities, including minor PR-segment depression in multiple leads, small QRS complexes in the extremity leads and QS pattern in leads V1-V2.
Case 5
A 66-year-old woman with a history of Raynaud’s phenomenon (RP) presented with progressive shortness of breath without associated chest pain or infective symptoms. She had significant thickening of her skin, especially over her fingers and forearms. Antibody screening revealed positive ACA and anti-Ro antibodies. She was diagnosed with diffuse cutaneous SSc. An echocardiogram was performed showing left ventricular dysfunction with regional wall motion abnormalities compatible with TTS. Coronary angiogram showed unobstructed coronary arteries. During admission, she developed acute renal impairment, referred to as SRC. She was treated with ACEi, and an echocardiogram performed during follow-up demonstrated complete recovery of biventricular systolic function and normalized wall motion.
Discussion
To our knowledge, this is the largest case series of TTS in patients with SSc, including two recurrent cases. Based on our evaluation and review of these cases, certain learning points have emerged that may inform understanding of both TTS and SSc cardiac involvement.
TTS consists of a wide spectrum of clinical findings, such as transient LV dysfunction, most often observed in reaction to an emotional or physical trigger. This is accompanied by sympathetic nervous system activation and a catecholamine surge which has till date been seen as the explanation for acute myocardial dysfunction. There are various hypotheses about potential mechanistic explanations for the initiation of this clinical entity, including multi-vessel coronary artery spasm caused by endothelial dysfunction, direct cardiomyocyte toxicity and micro-circulatory dysfunction. 4 More recent studies have studied the chronic phase of TTS which seems to be caused by an underlying (persisting) inflammatory reaction.3,4
One particular inflammatory pathway, which has been proposed in the context of autoimmune disease, is the release of cardiac myosins as a result of the catecholamine-medicated damage to the cardiomyocytes. The subsequent inflammatory response, triggered by autoreactive T cells exposed to these cardiac myosins, eventually leads to global myocardial dysfunction. 3 This is a similar process to the Dressler syndrome, which has been observed after myocardial infarctions and cardiac bypass surgery 3 and may cause severe constrictive pericarditis in these situations.
Cardiac RP could also be a potential explanation for TTS in SSc. 6 Peripheral RP is a common feature of SSc, present in approximately 95% of patients. In addition, a visceral form has also been described. Cardiac RP is defined as asymptomatic, cold-induced, transient cardiac ischaemia, resulting in cardiac hypoperfusion and subsequent reversible left ventricular dysfunction, with previous reports visualizing this using positron emission tomography, such as 201-thallium scintigraphy. Echocardiographic studies have also described reversible regional left ventricular wall motion abnormalities, 7 similar to the abnormalities observed in atypical TTS. It is plausible that TTS-like observations in patients with SSc could represent a symptomatic form of cardiac RP.
Multiple studies have suggested that vasodilators, including calcium channel blockers (CCB), have a beneficial effect on myocardial perfusion in SSc and could potentially improve outcome and survival in SSc. This is considered to be due to the effect of CCB on cardiac microcirculation which leads to a reduction in intermittent ischemic episodes and subsequent fibrotic remodelling. 8 This could be a potential therapeutic opportunity for SSc patients presenting with LV dysfunction, including TTS.
SRC is another clinical entity in SSc that can lead to hypertensive emergency and transient left ventricular dysfunction. It is plausible that this may have similarities to TTS in pathogenesis or clinical presentation. The acute renal failure in SRC might function as a trigger to activate the sympathetic nervous system and thereby stimulate catecholamine release. 9 During an SRC, patients could develop a catecholamine-induced vasospasm which might lead to myocardial stunning, similar to TTS. Involvement of innate immune system perturbation with complement activation also underpins both TTS and SRC.
In addition, excess catecholamines play a role in activating/stimulating the renin–angiotensin–aldosterone system (RAAS) 10 and have a negative effect on the already existing hypertension. This results in increased myocardial wall stress and might be an additional pathophysiological pathway leading to cardiac dysfunction in SRC.
ACEi are a recognized therapeutic option for the treatment of SRC which can improve the prognosis of affected patients. 1 Moreover, studies have indicated that ACEi may also have a favourable impact on the recovery of patients with TTS. Furthermore, usage of ACEi has been linked with reduced recurrences. 5 Consequently, it is reasonable to expect a similar beneficial effect in patients with both SRC and TTS or in patients with SSc who experience an isolated episode of TTS, following treatment with ACEi. 8
In general, and outside the context of SSc, TTS is considered to be a self-limiting condition. However, in recent studies, it has been observed that patients can experience persistent symptoms, even after normalization of left ventricular function. As there is a high prevalence of myocardial abnormalities of SSc patients (almost 80%), 3 it could be hypothesized that the inflammatory changes in the heart might contribute to a more chronic (or less reversible) pathology, leading to a more complicated and slower recovery and possibly a higher risk of recurrence.
As demonstrated in this article, there is a wide spectrum of clinical entities involving transient LV dysfunction in SSc patients, which share a common pathophysiological pathway. This could be used to inform future research aimed at understanding these entities using specific clinical and biomarker tests, such as capillaroscopy and cold challenge, and discovering novel therapeutic targets.
Footnotes
Authors’ Note
The Editor/Editorial Board Member of JSRD is an author of this paper; therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Board member had no involvement in the decision-making process.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
