Abstract
Scleroderma renal crisis is a rare complication of systemic sclerosis characterized by a rapid decline in kidney function due to acute renal vascular injury. Recently, activating autoantibodies targeting the angiotensin II type 1 receptor and the endothelin-1 type A receptor have been implicated in the pathophysiology of scleroderma renal crisis by sensitizing the angiotensin II type 1 receptor and endothelin-1 type A receptor in renal resistance arteries to their natural ligands. Here, we describe a cohort of 10 patients with scleroderma renal crisis refractory to standard treatment, including blockade of the renin-angiotensin system. Multimodal therapy was initiated, targeting at the removal of anti-angiotensin II type 1 receptor and anti-endothelin-1 type A receptor autoantibodies by plasma exchange and the reduction of vasoconstrictive activity. Further treatment options included angiotensin II type 1 receptor and endothelin-1 type A receptor blockade, iloprost, intravenous immunoglobulins, and immunosuppression. Six patients were hypertensive. On kidney biopsy, concentric intimal sclerosis was present in all patients, whereas acute vascular injury was evident in eight. Levels of anti-angiotensin II type 1 receptor and anti-endothelin-1 type A receptor autoantibodies were significantly reduced by multimodal treatment. Kidney function improved in three patients with histological signs of severe acute renal vascular damage. This report demonstrates that intensive multimodal therapy taking account of potentially pathogenic anti-angiotensin II type 1 receptor and anti-endothelin-1 type A receptor autoantibodies in concert with other vasodilatory interventions provides a salvage option for patients with refractory scleroderma renal crisis.
Introduction
Scleroderma renal crisis (SRC) is a rare but severe vascular complication in patients with systemic sclerosis (SSc). A recent meta-analysis revealed a prevalence of 4.9% for SRC. 1 Activation of the renin-angiotensin-aldosterone system (RAAS) in response to reduced renal cortical blood flow after damage of small intrarenal arteries is a pathophysiological hallmark of SRC. 2 In addition, expression of endothelin-1 (ET-1) and its receptors is upregulated,3,4 resulting in excessive vasoconstrictive activity.
Cases present with acute kidney injury as indicated by declining renal function and acute as well as chronic vascular lesions in small arteries on kidney biopsy. 5 About 90% develop hypertension, 2 often severe with serious end-organ damage such as thrombotic microangiopathy (TMA) 6 that can also be found in renal biopsies. 5 Despite some improvements over the last decades, mostly due to the introduction of angiotensin-converting enzyme inhibitors (ACEI), the clinical outcome is still unsatisfactory with 31% dying within the first year and 35% requiring long-term renal replacement therapy. 1
A rather novel concept links autoimmunity to vascular damage by agonistic autoantibodies (Abs) targeting the angiotensin II type 1 receptor (AT1R) and the ET-1 type A receptor (ETAR). In SSc patients, high anti-AT1R Abs and anti-ETAR Abs levels indicate an increased risk for SRC and mortality. 7 Mechanistically, anti-AT1R and anti-ETAR Abs not only activate endothelial cells 8 but also elicit contraction in renal resistance arteries. 9 Importantly, we found evidence that Abs targeting the AT1R and the ETAR hypersensitize the receptors to their natural ligands angiotensin II (Ang II) and ET-1. 9 This might facilitate the excessive vasoconstriction found in SRC and enhance the vicious cycle of vascular injury, reduced renal blood supply, and RAAS activation.
With this case series, we aimed to document the effects of a broad multimodal therapeutic approach that also addresses anti-AT1R and anti-ETAR Abs.
Case description
We report the course of 10 patients with SRC who presented or were referred to our clinic between January 2006 and February 2018 and were followed until May 2021. The study was approved by the local ethics committee (EA1/013/705), and all patients provided written informed consent to participate in a scientific study. SRC was defined as an otherwise unexplained rapid decline of renal function (increase in serum creatinine ⩾ 50%) in a patient with SSc. All patients had already been treated with ACEI or AT1R blockers (AT1RB) without sufficient clinical response, that is, significant improvement of kidney function and resolution of hypertension, if present. Patient characteristics are summarized in Table 1 and are reported on single patient level in Supplementary Table.
Patient characteristics, histological findings, treatment, and outcome data of patients with refractory SRC.
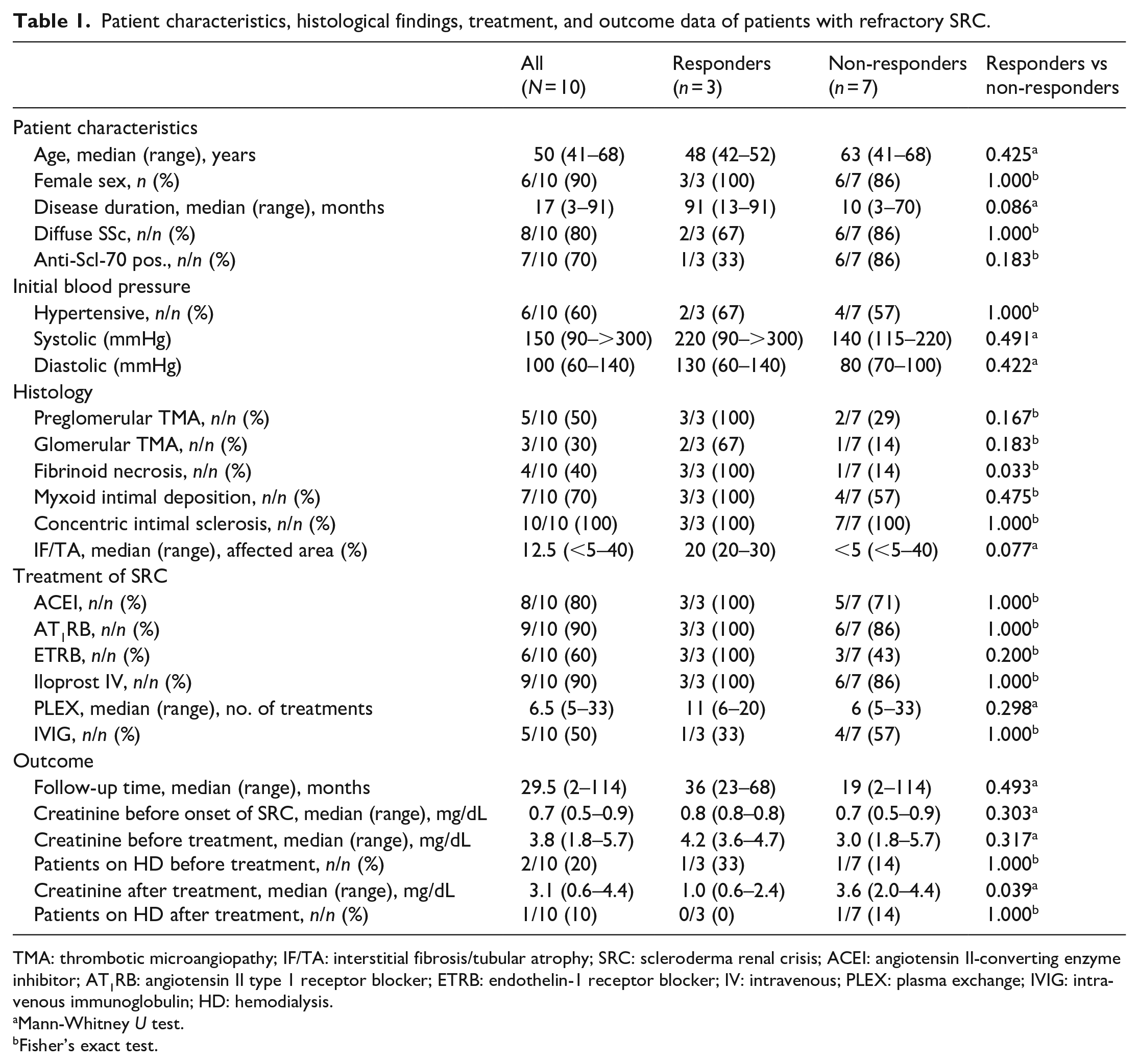
TMA: thrombotic microangiopathy; IF/TA: interstitial fibrosis/tubular atrophy; SRC: scleroderma renal crisis; ACEI: angiotensin II-converting enzyme inhibitor; AT1RB: angiotensin II type 1 receptor blocker; ETRB: endothelin-1 receptor blocker; IV: intravenous; PLEX: plasma exchange; IVIG: intravenous immunoglobulin; HD: hemodialysis.
Mann-Whitney U test.
Fisher’s exact test.
On presentation, three patients had severe hypertension and three patients were moderately hypertensive. Three patients were normotensive and one hypotensive (Table 1). Of the patients with hypertension, patient 1 initially presented to a peripheral hospital with blurred vision. Bilateral papillary edema was diagnosed and high-dose intravenous glucocorticoids were administered. Few hours later, the patient developed malignant hypertension (>300 mmHg systolic) and was transferred to the intensive care unit (ICU) of our department.
Renal biopsy revealed concentric intimal sclerosis in all patients. Four patients had very severe acute vascular injury as indicated by fibrinoid necrosis. Glomerular and preglomerular TMA, signs of acute endothelial injury, were found in five and three patients, respectively (Table 1 and Supplementary Table).
Levels of anti-AT1R and anti-ETAR Abs in patient serum were measured with a solid-phase sandwich enzyme-linked immunosorbent assay (ELISA) (CellTrend GmbH, Luckenwalde, Germany). 8 Median levels were 22.1 U/mL for anti-AT1R Abs and 21.0 U/mL for anti-ETAR Abs (Figure 1(a) and (b)). The previously reported relative risk for SRC in SSc patients was 11.6 for anti-AT1R Abs with a cut-off value of 15.8 U/mL and 8.0 for anti-ETAR Abs (cut-off value 16.5 U/mL). 7
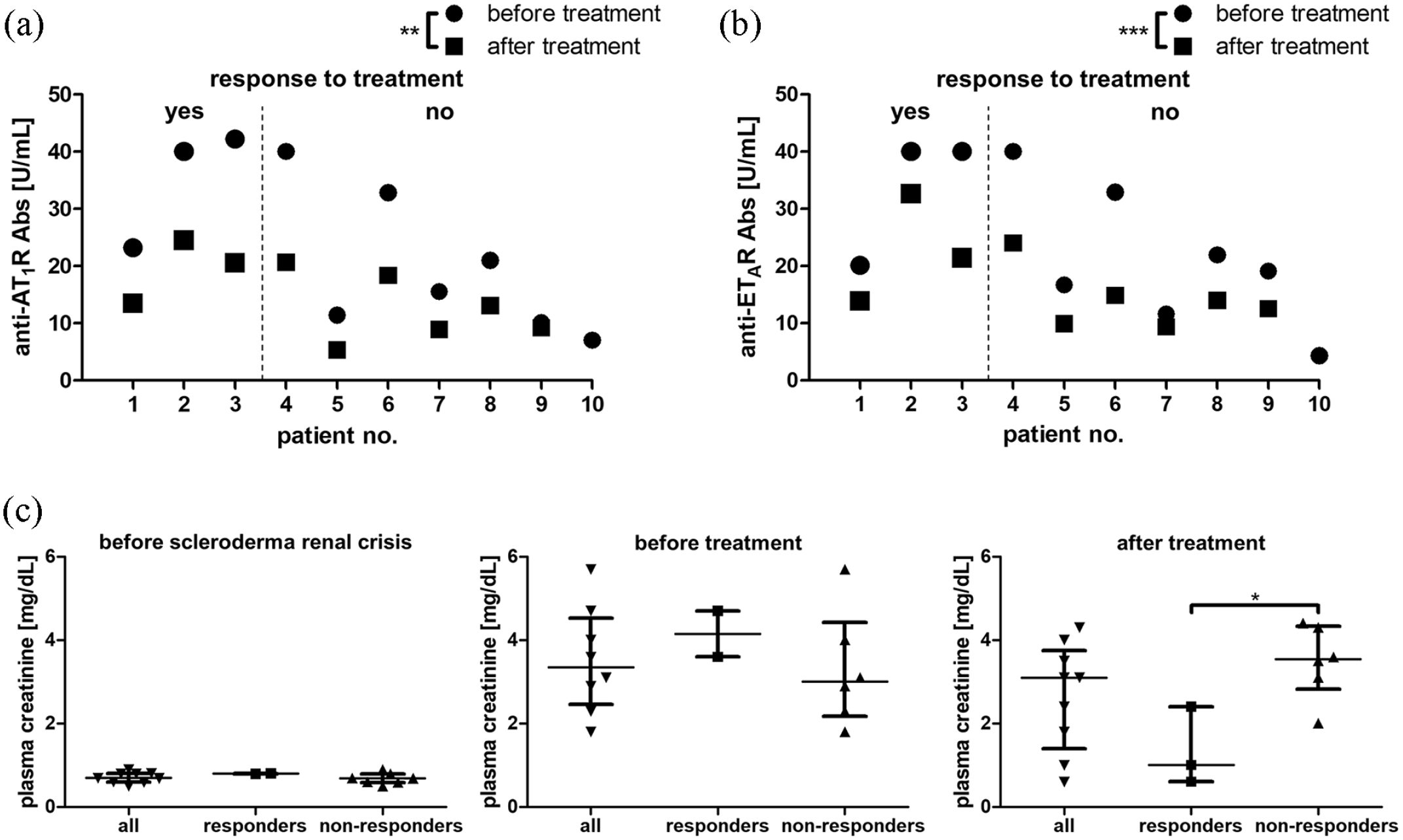
Autoantibody levels and kidney function in response to multimodal targeted therapy for scleroderma renal crisis (SRC). Levels of autoantibodies directed against (a) the angiotensin II type 1 receptor (anti-AT1R Abs) and (b) the endothelin-1 type A receptor (anti-ETAR Abs) in patients with SRC refractory to treatment with angiotensin II-converting enzyme inhibitors (ACEI) or AT1R blockers (AT1RB) before (circles) and after (squares) multimodal therapy. **p < 0.01, ***p < 0.001 for comparison of all patients before and after treatment. (c) Plasma creatinine levels before onset of SRC, during refractory SRC before initiation of multimodal therapy, and after intensive treatment. Values are shown for the entire cohort (all; n = 10) as well as separately for patients with a response to treatment (responders; n = 3) and those without (non-responders; n = 7). Patients on dialysis or lost to follow-up were excluded at the respective time points. *p < 0.05.
Since standard treatment had not been successful in these patients at high risk of permanent renal failure and severe complications related to hypertension, additional interventions based on pathophysiological considerations were started. Vasoconstriction was addressed by AT1RB and/or ACEI, ET-1 receptor blockade (ETRB), and the vasodilatory prostacyclin analogue iloprost. Plasma exchange (PLEX) against fresh frozen plasma was initiated to reduce levels of anti-AT1R and anti-ETAR Abs. Five patients received intravenous immunoglobulins for immunomodulation after PLEX. To limit de novo production of autoantibodies, immunosuppressive treatment was continued or initiated, preferentially employing potent antibody-suppressing agents (Supplementary Table). Treatment modalities were individually adjusted depending on each patient’s needs (blood pressure, tolerability, response to treatment, previous immunosuppression, cell count, etc.) at the attending physician’s discretion. Dialysis treatment was initiated and stopped as deemed individually necessary.
In response to multimodal treatment, kidney function recovered in patients 2 and 3 and improved in patient 1. Nevertheless, patient 1 had to start hemodialysis after 22 months. Dialysis could be stopped after 6 weeks in patient 2 and kidney function remained stable until loss to follow-up after 68 months. Patient 6 was still on dialysis 20 months after onset of SRC, but diuresis increased gradually from anuria to 1000 mL per day. Patient 7 progressed to renal failure requiring dialysis after 2 months and was lost to follow-up after 4 months. Patient 8 was transiently on hemodialysis between months 7 and 21 and had stable, but highly compromised kidney function thereafter (creatinine 4.2–4.5 mg/dL). Patients 4, 5, 9, and 10 showed no clinically relevant response to treatment with regard to renal function (Supplementary Table; Figure 1(c)). Overall, median dialysis-free survival was 23 months (Figure 2(a)).
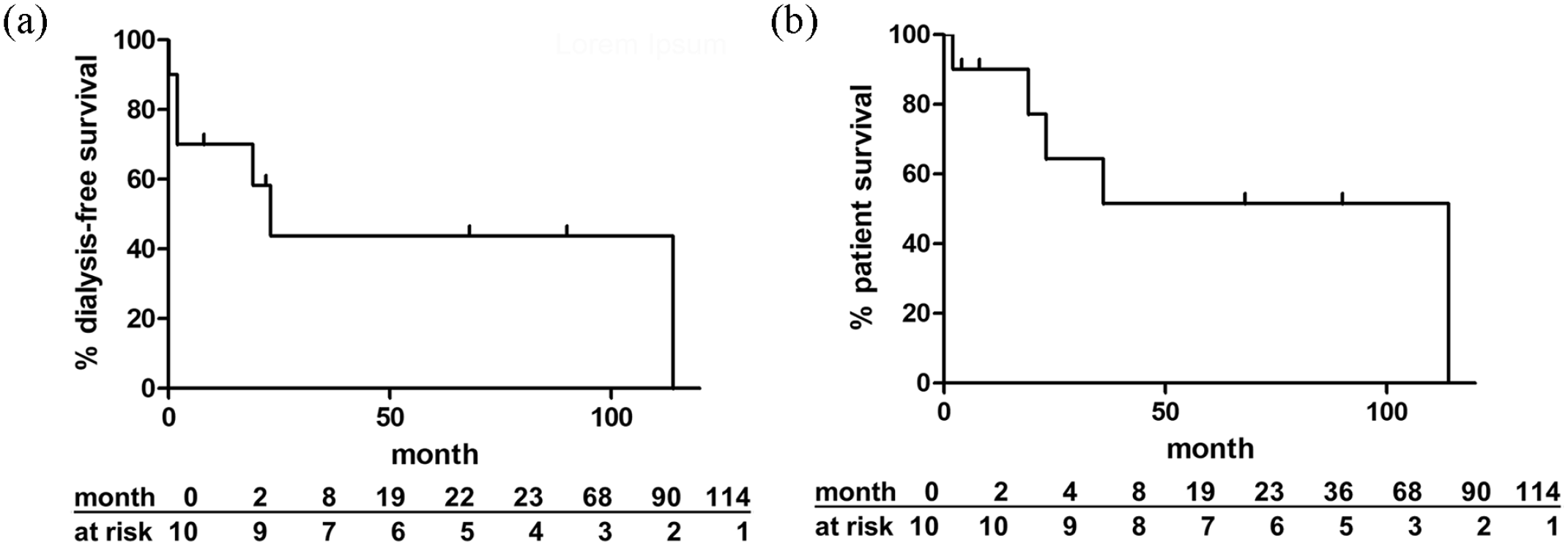
Outcome of patients receiving multimodal targeted therapy for refractory scleroderma renal crisis (SRC). Kaplan–Meier curves for (a) dialysis-free survival and (b) patient survival of all patients in the study cohort with SRC.
Patient 3 died after 23 months despite recovery of renal function because of sepsis. Patient 4 died after 19 months because of renal failure (last creatinine 9.2 mg/dL) and respiratory failure due to lung fibrosis. Patient 6 died after 36 months of an unknown cause. Patient 8 survived for 114 months and died of myocardial infarction. Patient 9 died after 2 months because of pneumonia and heart failure. Median patient survival was 36 months (Figure 2(b)).
Patient 1 who was initially treated at the ICU because of malignant hypertension responded to PLEX with an impressive improvement of hypertension. Before initiation of PLEX, seven different antihypertensive drugs were needed to achieve blood pressure control. However, already after the first treatment session, all antihypertensives could be stopped. This immediate and strong response of extremely elevated blood pressure levels to PLEX can be explained best by disruption of a functional interaction of anti-AT1R and anti-ETAR Abs with Ang II and ET-1, resulting in a rapid resolution of severe vasoconstriction.
To decipher prognostic factors, we classified three patients as responders and six patients as non-responders based on kidney function (Supplementary Table; Figure 1(c)). The only statistically significant difference between responders and non-responders was the presence of fibrinoid necrosis on biopsy (Table 1). Combined treatment with PLEX significantly reduced anti-AT1R (median levels from 22.1 U/mL to 13.4 U/mL) and anti-ETAR (median levels from 21.0 U/mL to 14.0 U/mL) Abs in all nine patients with available data (Figure 1(a) and (b)). Both responders and non-responders had similar levels of anti-AT1R and anti-ETAR Abs before and after treatment (Figure 1(a) and (b)). There were no significant differences in antibody reduction rates calculated as (antibody level before treatment–antibody level after treatment) / antibody level before treatment (Supplemental Figure).
Conclusion
Although the cohort we studied was highly selected for severe cases, the incidence of chronic renal failure (30%; 3/10 patients) was comparable to that found in unselected cohorts (25%–50%). 10 Of note, follow-up time of one patient who required dialysis was limited to 4 months. Given that kidney function has been observed to recover and dialysis could be stopped even after up to 3 years, 11 it can be speculated that the incidence of end-stage renal disease in our intensively treated patients was even lower with longer follow-up.
Fibrinoid necrosis of the vessel wall indicative of severe acute vascular injury was the only variable significantly associated with a clear improvement in kidney function observed in three patients (Table 1). This is outstanding because such acute changes were associated with worse outcomes in two studies with biopsies from a total of 75 patients with SRC.11,12 We cautiously propose from a pathophysiological perspective based on our and others’ findings the hypothesis that addition of ETRB and PLEX might have crucially contributed to resolution of renal vasculopathy, as virtually all patients in the studies mentioned above were on RAAS blockers but were not treated with ETRB or PLEX. Addition of an ETRB alone might not have been similarly efficient. An open-label study with six SRC patients (five with microangiopathic hemolytic anemia (MAHA)) treated with bosentan for 6 months compared to a historical cohort without ETRB did not show a significant benefit. 13 However, a recent randomized placebo controlled study in 13 patients with chronic kidney disease associated with SSc but without apparent SRC demonstrated an improvement of estimated glomerular filtration rate (eGFR) after 26 and 52 weeks in those six patients who received the ETRB zibotentan for 26 weeks. 14 Thus, continuation of ETRB in the chronic phase might be particularly beneficial. In a retrospective study observing the long-term outcome of SRC, a subgroup of 10 patients with MAHA or ACEI intolerance treated with PLEX had a better outcome than the other 596 patients treated with ACEI alone. 15 Identification of those individuals who would respond to a rather aggressive therapy such as PLEX seems to be crucial. Whether kidney biopsies or less invasive diagnostic tools such as peripheral blood smears for evidence of MAHA or measurement of agonistic anti-AT1R and anti-ETAR Abs provide sufficient diagnostic accuracy requires further research.
Levels of anti-AT1R and anti-ETAR Abs were lowered in all patients but remained above or close to the cut-off value predictive of SRC. 7 Neither absolute autoantibody concentrations before or after treatment nor their reduction rates were significantly different between patients with renal recovery and those without. Even patient 1 with the impressive drop of blood pressure in response to the first PLEX session did not have extremely high autoantibody levels or reduction rates. This apparent lack of correlation of anti-AT1R and anti-ETAR Abs concentrations measured in peripheral blood with clinical manifestations might be related to technical limitations of the detection method. Quantification of antibody levels with an ELISA is influenced by binding affinity and antibody avidity that may vary but cannot be controlled. 16 In addition, Abs directed against G-protein-coupled receptor (GPCR) can stimulate, inhibit, or block receptor-dependent signaling, 16 and differing functionalities have also been shown for anti-AT1R Abs in SSc. 17 While the ELISA is a simple method allowing for easy and fast measurement of antibody levels in general, it is not suited to assess biologic activity of Abs. Since we did detect falling antibody levels with treatment but no correlation with the clinical course, it seems feasible to monitor anti-AT1R and anti-ETAR Abs over time to evaluate the efficacy of treatment with regard to antibody reduction but not as a surrogate for a clinical response. Since the functionality of anti-AT1R Abs as measured by a luminometric bioassay did not correlate with clinical manifestations in SSc patients, 17 even functional assays covering only one target GPCR might not be sufficient to predict the improvement of clinically relevant parameters in response to PLEX. For this purpose, a comprehensive assessment of a whole network of Abs targeting vascular GPCR, including those with vasodilatory properties such as the Ang II type 2 receptor, might be necessary. Moreover, we cannot completely rule out that removal of other mediators such as cytokines by PLEX had contributed to the improved outcome.
An important limitation of our study is the observational character with only a small number of patients and lack of a control group. Furthermore, dual RAAS blockade did not improve clinical endpoints in several cardiorenal studies but was associated with side effects such as acute kidney injury and hypotension. 18 Thus, it is currently not recommended in international guidelines. However, whether or not dual RAAS blockade could offer some benefit in specific clinical situations with very high RAAS activity as found in SRC is not known and needs further investigation.
In conclusion, a multimodal therapeutic approach targeted at intensive blockade of AT1R and ETAR and removal of anti-AT1R and anti-ETAR Abs was associated with improved renal function in three patients with severe acute renal vasculopathy. The relative significance of ETRB and PLEX in refractory SRC needs to be determined. In addition, diagnostic procedures identifying patients who would benefit from these interventions should be explored. Assays evaluating the functional properties of anti-AT1R and anti-ETAR Abs might be particularly useful.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983231168193 – Supplemental material for Intensive receptor blockade and plasma exchange to treat refractory scleroderma renal crisis in patients with agonistic autoantibodies targeting the angiotensin II type 1 and endothelin-1 type A receptors
Supplemental material, sj-pdf-1-jso-10.1177_23971983231168193 for Intensive receptor blockade and plasma exchange to treat refractory scleroderma renal crisis in patients with agonistic autoantibodies targeting the angiotensin II type 1 and endothelin-1 type A receptors by Björn Hegner, Julia Callaghan, Ralf Schindler, Harald Heidecke, Gabriela Riemekasten, Aurélie Philippe and Rusan Catar in Journal of Scleroderma and Related Disorders
Supplemental Material
sj-pdf-2-jso-10.1177_23971983231168193 – Supplemental material for Intensive receptor blockade and plasma exchange to treat refractory scleroderma renal crisis in patients with agonistic autoantibodies targeting the angiotensin II type 1 and endothelin-1 type A receptors
Supplemental material, sj-pdf-2-jso-10.1177_23971983231168193 for Intensive receptor blockade and plasma exchange to treat refractory scleroderma renal crisis in patients with agonistic autoantibodies targeting the angiotensin II type 1 and endothelin-1 type A receptors by Björn Hegner, Julia Callaghan, Ralf Schindler, Harald Heidecke, Gabriela Riemekasten, Aurélie Philippe and Rusan Catar in Journal of Scleroderma and Related Disorders
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bundesministerium für Wirtschaft und Energie (BMWi, Federal Ministry for Economic Affairs and Energy; ZIM project KF2257305AJ1) and the Deutsche Stiftung Sklerodermie (DSS, German Scleroderma Foundation).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
