Abstract
Objective:
Sex and gender are of growing scientific interest in disease onset and course. While sex differences have been shown to exist in systemic sclerosis, there is a paucity of data on gender. Our objective was to examine the association between occupation, a gender-related role and outcomes in systemic sclerosis.
Methods:
An occupation score ranging from 0 to 100, with lower scores representing occupations traditionally held by men and higher scores traditionally held by women, was constructed using the National Occupational Classification 2016 and data from Statistics Canada. Subjects in the Canadian Scleroderma Research Group registry were assigned an occupation score based on self-reported occupation. Multivariate models, adjusted for sex, age, smoking and education were used to estimate the independent effect of occupation score on systemic sclerosis outcomes.
Results:
We included 1104 subjects, of which 961 were females (87%) and 143 (13%) males. There were differences between females versus males: disease duration (9.9 vs 7.6 years, p = 0.002), diffuse disease (35% vs 54%, p < 0.001), interstitial lung disease (28% vs 37%, p = 0.021) and pulmonary hypertension (10% vs 4%, p = 0.033), but not pain, response to treatment and mortality. The median occupation scores differed between females and males (84.3 (interquartile range 56.8, 89.4) vs 24.9 (4.3, 54.1), p < 0.001). The Spearman correlation between sex and occupation score was 0.44, indicating a weak correlation. In adjusted analyses, occupation score was not an independent predictor of disease subset (diffuse vs limited), interstitial lung disease, pulmonary hypertension, pain, response to treatment or mortality.
Conclusion:
We did not find independent associations between an occupation score, a gender-related role and outcomes in systemic sclerosis. These results should be interpreted with caution as occupation may be a poor measure of gender. Future research using a validated measure of gender will be needed to generate robust data on the effect of gender in systemic sclerosis.
Introduction
Systemic sclerosis (SSc) has a strong female predominance, with a female-to-male ratio reported as high as 9:1.1–6 This suggests that sex-related factors contribute to the aetiopathogenesis of SSc. Indeed, considerable research exploring the role of sex in SSc has been published.7–9 Although many of those studies reported gender differences, most were in fact conflating sex and gender and were in fact focusing on sex-related variables. Sex and gender, although often used interchangeably, refer to different constructs. Sex is considered a biological variable, defined by chromosomes and reflected physiologically by sex hormones, gonads, external genitalia and internal reproductive organs.10–12 Gender is defined by the World Health Organization as the ‘socially constructed roles, behaviours, activities, and attributes that a given society considers appropriate for men and women’. 13 There is emerging evidence that, in addition to sex, gender can also influence disease onset and progression in a number of diseases. 14 To our knowledge, there has been very little research on gender differences, in the strict sense, in SSc.
Although there is an increasing interest in the role of gender in health and disease, there are few validated measures thereof. Some have proposed using occupation as a measure of gender,15–17 in part because of varying occupational exposures associated with occupation. 18
We undertook this exploratory study to investigate the role of gender in SSc using occupation as a surrogate marker of gender. We hypothesized that occupation would be associated with SSc outcomes independently of sex.
Methods
Data were extracted from the Canadian Scleroderma Research Group (GSRG) registry. Patients in this multi-centre registry have a diagnosis of SSc made by an expert rheumatologist, are ⩾ 18 years of age, fluent in English or French, and likely to be compliant with study procedures and visits. Patients are seen at baseline and annually thereafter. Standardized sociodemographic, history, physical examination and laboratory data are collected at each study visit.
Sex and other sociodemographic information including age, education, smoking status and occupation are self-reported by patients. Clinical data, recorded by expert rheumatologists, include disease duration from the onset of the first non-Raynaud’s disease manifestation. Skin involvement is assessed using the modified Rodnan skin score (mRSS), a widely used clinical assessment where the examining rheumatologist records the degree of skin thickening ranging from 0 (no involvement) to 3 (severe thickening) in 17 areas (total score range 0–51). Limited cutaneous disease (lcSSc) was defined as skin involvement distal to the elbows and knees with or without facial involvement; diffuse cutaneous disease (dcSSc) was defined as skin involvement proximal to the elbows and knees and/or of the trunk.
In the CSRG, the presence of ILD is determined using a published clinical decision rule. 19 This algorithm considers ILD to be present if a high-resolution computed tomography (HRCT) scan of the lung was interpreted by an experienced radiologist as showing ILD or, in the case where no HRCT is available, if either a chest X-ray was reported as showing increased interstitial markings (not thought to be due to congestive heart failure) or fibrosis, and/or if a study physician reported the presence of typical ‘velcro-like crackles’ on physical examination. Pulmonary hypertension (PH) is defined as SPAP >⩾45 mmHg measured by TTE (an estimate that correlates strongly with the values reported in right-sided heart catheter studies). 20
Medication history is recorded by a study physician at each visit. Response to treatment is measured in subjects with dcSSc using the CRISS score, which is a provisional composite response index in dcSSc approved by the American College of Rheumatology. It includes disease areas that are susceptible to improvement, including skin involvement based on the modified Rodnan skin score (mRSS), lung involvement based on the forced vital capacity (FVC), patient and physician global assessments of disease and function based on the Health Assessment Questionnaire Disability Index (HAQ). 21
Patient-reported outcomes include pain severity, assessed with an 11-point numerical rating scale (NRS) ranging from 0 to 10 (where 0 = no pain and 10 = very severe pain) in the last week.
Autoantibody analyses were performed on sera collected at baseline study visit by Mitogen Diagnostics Laboratory, University of Calgary. Anti-centromere (CENP-A and CENP-B; ACA), anti-topoisomerase 1 (ATA) and anti-RNA polymerase III (RP11 and RP155; ARNAP) autoantibodies were detected by Euroline systemic sclerosis profile line immunoassay (Euroimmun GmbH, Luebeck, Germany) according to manufacturer’s instructions. Autoantibodies were reported as absent (negative, equivocal and low titres) and present (moderate and high titres).
Mortality is recorded by research staff at the time of a missed study visit.
In the absence of a validated measure of gender in the CSRG data collection protocol, occupation was used as a surrogate measure. We constructed the occupation score using the National Occupational Classification (NOC) 2016 and data collected by Statistics Canada. 22 Statistics Canada collects population-level occupational data using 47 NOC categories by sex. Some occupations such as elementary school teaching and nursing are more common in women, while others such as police officers and construction trades are more common in men. We calculated the proportion of women in each of the 47 specific occupations in the general population. The possible range was from 0% to 100%. The lower the proportion, the more traditionally held by men an occupation was considered, and the higher the proportion, the more traditionally held by women. Then, each patient was assigned an occupation score ranging from 0-100 based on their self-reported occupation. For example, a female patient working in a predominantly male occupation was assigned a female sex but a masculine occupation score, and a male patient working in a predominantly female occupation was assigned a male sex but a feminine occupation score.
We examined the associations between occupation and various SSc manifestations including disease subset (diffuse vs limited), ILD and pulmonary hypertension, response to treatment measured with the CRISS, patient-reported pain and mortality. Multivariate logistic and linear regression and Cox proportional hazards analyses adjusting for sex, age, smoking, education and disease duration were used to estimate the independent effect of occupation score on SSc outcomes. p Values ≤ 0.05 were considered statistically significant. Given the exploratory nature of the study, no corrections for multiple testing were done.
Results
As of December 2020, the Canadian Scleroderma Research Group’s pan-national cohort included 1698 subjects, of which 1104 had reported an occupation. Of those, 143 were males and 961 were females. Mean age was 53.9 ± 11.4 years, and 54% reported having received post-secondary education. More males (79.9%) were smokers compared to females (55.4%). The mean disease duration was 9.6 (±9.0) years (Table 1).
Baseline characteristics of the study cohort by sex (mean ± SD or n (%)).
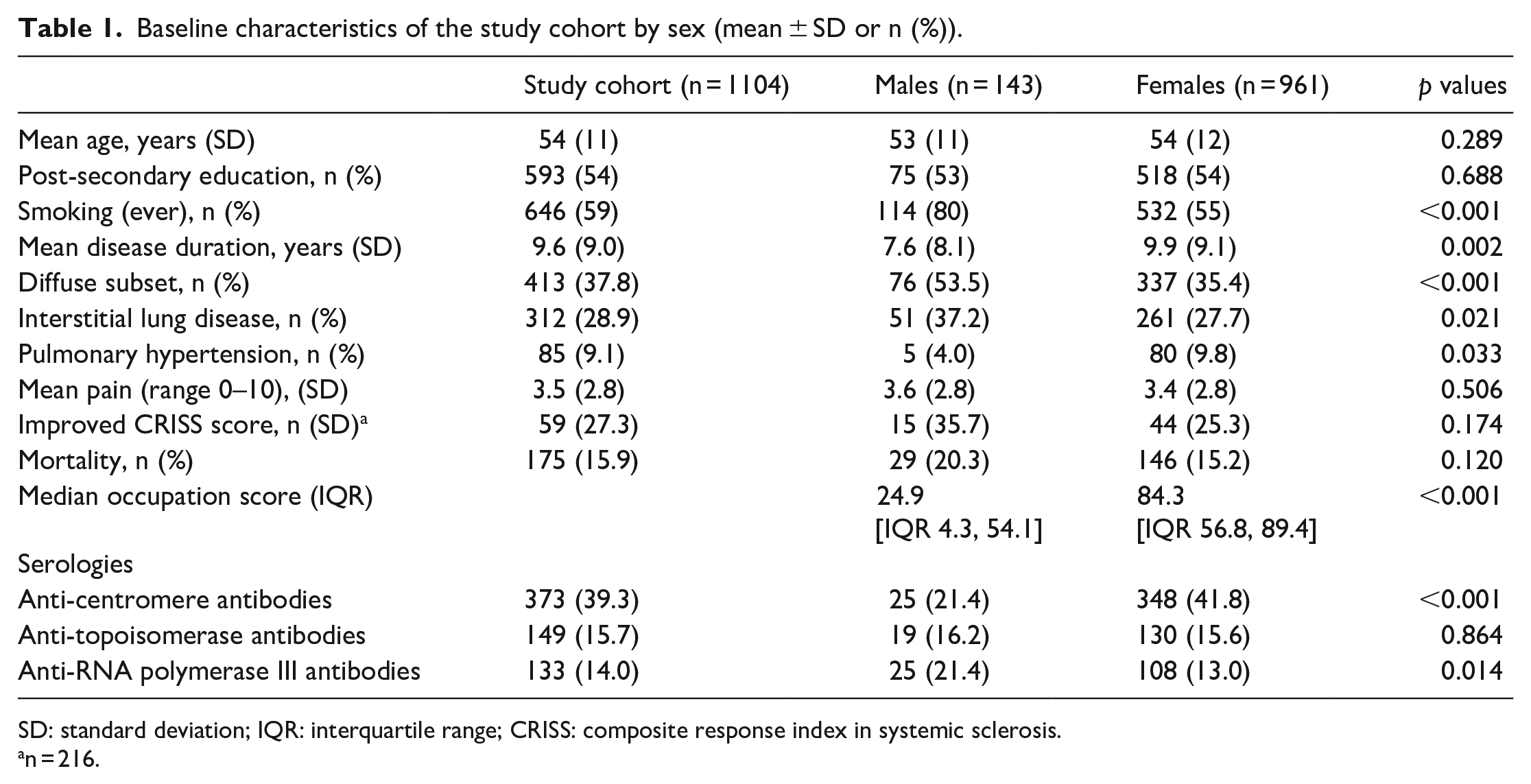
SD: standard deviation; IQR: interquartile range; CRISS: composite response index in systemic sclerosis.
n = 216.
There were differences in clinical manifestations between females and males. Diffuse disease was more common in males (53.5%) compared to females 35.4% (53.5% vs 35.4%, p < 0.001). Males were more likely to have ILD in comparison to females (37.2% vs 27.7%, p 0.021), whereas PH was more common in females (9.8%) compared to males (9.8% vs 4%, p = 0.033). There was no difference between males and females in pain scores (3.6 ± 2.8 vs 3.4 ± 2.8, p = 0.506), response to treatment (improved CRISS score 25% vs 35%, p = 0.174), or mortality (15% vs 20%, p = 0.120).
The median occupation scores differed between females versus males (84.3 (interquartile range (IQR) 56.8, 89.4) vs 24.9 (4.3, 54.1), p < 0.001). The Spearman correlation between sex and occupation was 0.44, indicating a weak correlation between sex and occupation (Figure 1).
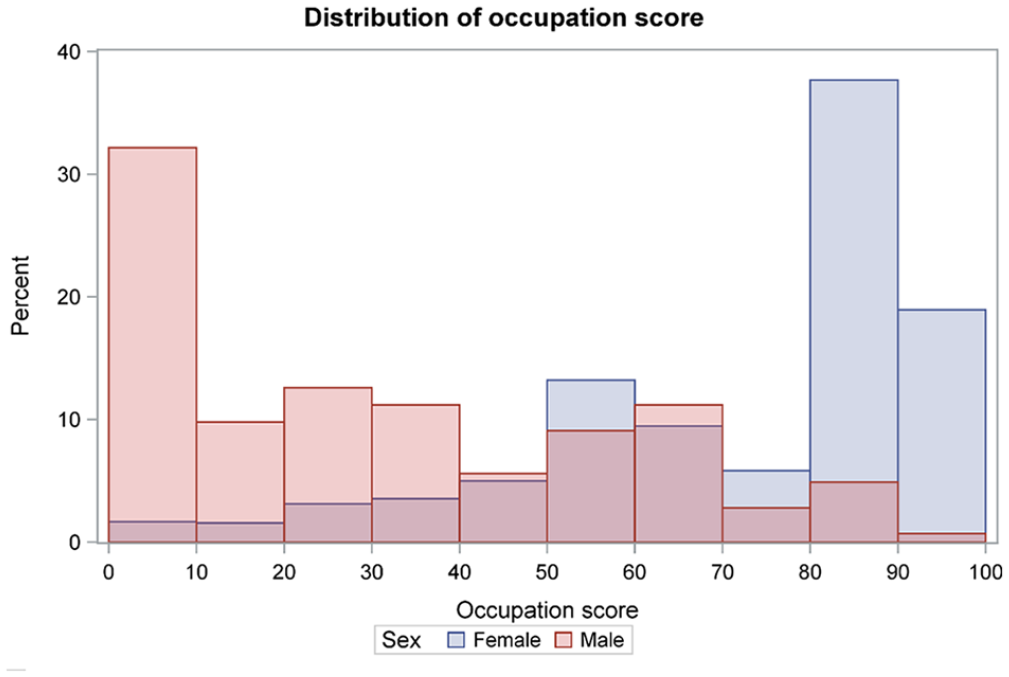
Distribution of occupation scores by sex (n = 1104).
Our results confirm existing data pointing to sex differences in SSc, namely disease subset and ILD. In multivariate models, females were less likely to have dcSSc (odds ratio (OR) 0.44; 95% confidence interval (CI) 0.29, 0.68) or ILD (OR 0.53; 95% CI 0.23, 0.84) compared to males (Table 2). Similarly, the risk of death was lower in women compared to men (hazard ratio (HR) 0.50; 95% CI 0.30, 0.84) (Supplementary Table 1).
Associations between occupation score, sex and SSc manifestations (OR and 95% CI).
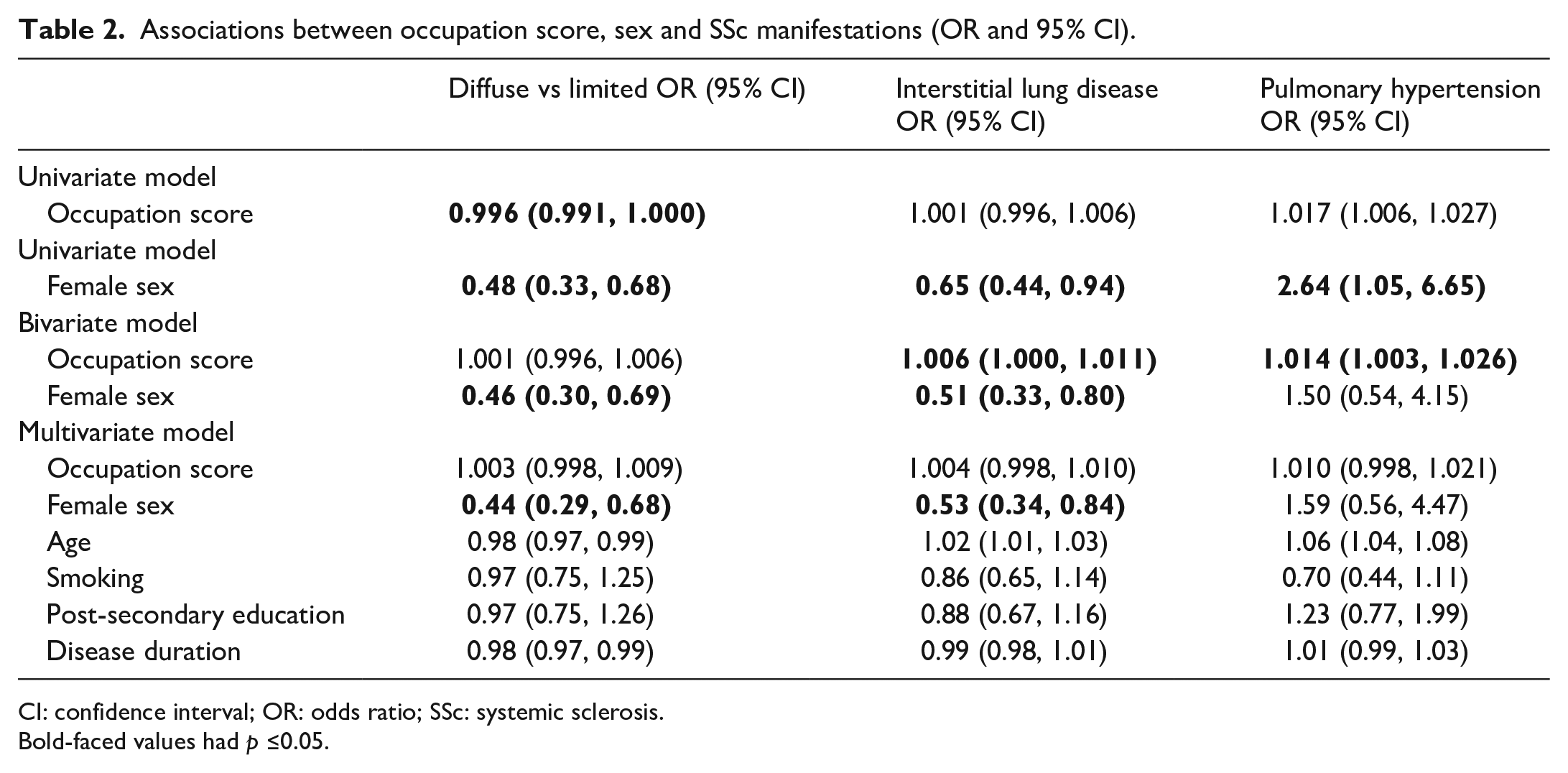
CI: confidence interval; OR: odds ratio; SSc: systemic sclerosis.
Bold-faced values had p ≤0.05.
Interestingly, occupation scores were associated with ILD (OR 1.006; 95% CI 1.000, 1.011) and PH (OR 1.014; 95% CI 1.003, 1.026) in bivariate analyses adjusting only for sex. However, these effects were lost after adjusting for smoking and education, in addition to age and disease duration in multivariate models.
There were no independent associations between occupation score and mortality, pain and response to treatment, after adjusting for sex, age, smoking, education and disease duration (Supplementary Tables 1–3).
Discussion
In this study, we examined the effect of gender on SSc. In the absence of a validated measure of gender in the CSRG data collection protocol, occupation was used as a surrogate measure. Each patient was assigned an occupation score based on their self-reported occupation. While our data confirmed existing evidence of sex differences in SSc (e.g. male patients had higher rates of diffuse disease2,6,23 and ILD,24–26 and higher risk of mortality), 27 occupation, a gender-related variable, was not associated with disease subset, ILD, PH, pain, response to treatment or mortality in multivariate analyses.
There is a paucity of data on the effect of gender SSc clinical outcomes. This is certainly in part due to the lack of a standardized measure of gender in most existing datasets, including the CSRG. Given the increasing interest in gender, we constructed a gender variable based on self-reported occupation. Occupation has been included in published composite measures of gender.15–17 However, using this surrogate variable, we found no independent association with outcomes of interest.
It is interesting to note, though, that gender did have an effect on ILD and PH in the bivariate model. It is possible that adjustment for smoking and education, which are also gender-related variables, may have resulted in over-fitting of the model. This would suggest that a composite measure including multiple surrogate markers might be a more sensitive measure of gender. On the other hand, it could also be that occupation is a poor surrogate measure for gender. Indeed, gender has been shown to play a more significant role in chronic pain than sex.28,29 However, in our study, occupation was not associated with pain.
This study is not without limitations. Our measure of gender using occupation as a surrogate marker was ad hoc. A validated measure of gender could have yielded different results. In addition, occupation scores were not available for 594 patients who reported being retired (without reporting their previous occupation), students, self-employed, unemployed, or volunteers, or for whom occupation was not reported. These individuals were excluded from the analysis. Nonetheless, our cohort included 1104 patients and we believe that this sample was sufficient to generate robust exploratory data. Finally, CRISS scores are only measured in patients with dcSSc, of which the majority are males.
In conclusion, our data failed to find an independent association between occupation, a gender-related variable and various outcomes in SSc. These results, however, should be interpreted with caution as occupation may be a poor measure of gender. Future research using a validated measure of gender will be needed to generate robust data on the effect of gender in SSc.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983221143599 – Supplemental material for Occupation as a gendered-role and outcome in systemic sclerosis
Supplemental material, sj-pdf-1-jso-10.1177_23971983221143599 for Occupation as a gendered-role and outcome in systemic sclerosis by Fatema Alkhamees, Oriana Hoi Yun Yu, Mianbo Wang and Marie Hudson in Journal of Scleroderma and Related Disorders
Footnotes
Acknowledgements
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the members of the Canadian Scleroderma Research Group (CSRG): Janet E Pope (London, Ontario), Murray Baron (Montreal, Quebec), Marie Hudson (Montreal, Quebec), Geneviève Gyger (Montreal, Quebec), Maggie J Larché (Hamilton, Ontario), Nader A Khalidi (Hamilton, Ontario), Ariel Masetto (Sherbrooke, Quebec), Evelyn Sutton (Halifax, Nova Scotia), David Robinson (Winnipeg, Manitoba), Tatiana S Rodriguez-Reyna (Mexico City, Mexico), Nancy Maltez (Ottawa, Ontario), Doug Smith (Ottawa, Canada), Carter Thorne (Newmarket, Canada), Alena Ikic (Québec City, Québec), Paul R Fortin (Québec City, Québec) and Marvin J Fritzler (Calgary, Canada).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: The CSRG has been supported by the Canadian Institutes of Health Research, the Scleroderma Society of Canada and its Chapters of Ontario and Saskatchewan, Sclérodermie Québec, Cure Scleroderma Foundation, INOVA Diagnostics Inc. (San Diego, CA), Dr. FookeLaboratorien GmbH (Neuss, Germany), Euroimmun (Lubeck, Germany), Mikrogen GmbH (Neuried, Germany), Fonds de la recherche en santé du Québec, the Canadian Arthritis Network and the Lady Davis Institute of the Jewish General Hospital, Montreal, QC. The funders had no role in the design and conduct of the study, nor the decision to prepare and submit the manuscript for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
