Abstract
Objective:
Decreased maximal mouth opening is a common and disabling manifestation in systemic sclerosis patients. We aimed to study the course of maximal mouth opening, determinants of smaller maximal mouth opening over time and the burden of smaller maximal mouth opening on mouth handicap.
Methods:
Consecutive systemic sclerosis patients participating in the prospective Leiden Combined Care in systemic sclerosis cohort were included. Annual clinical assessment included maximal mouth opening measurement and mouth handicap evaluation (Mouth Handicap in Systemic Sclerosis scale). Presence of microstomia (maximal mouth opening < 30 mm) was studied. Maximal mouth opening over time was assessed on group level and for all patients individually. Baseline characteristics were analysed for their association with smaller maximal mouth opening over time (linear mixed-effects models). Furthermore, cross-sectional association between maximal mouth opening with Mouth Handicap in Systemic Sclerosis scale was assessed (linear regression analysis).
Results:
A total of 382 systemic sclerosis patients were studied with median follow-up time of 2.0 years (interquartile range = 0.0–3.0). At baseline, mean maximal mouth opening was 42.2 ± 8.0 mm and 7% suffered from microstomia. Annual decrease of > 5.0 mm in maximal mouth opening during follow-up occurred in 63 patients and was accompanied by increase in disease severity. Disease characteristics at baseline independently predictive for smaller maximal mouth opening over time were: more extended skin subtype; peripheral vasculopathy; pulmonary, renal and gastrointestinal involvement. Smaller maximal mouth opening was significantly associated with more reported mouth handicap.
Conclusion:
The course of maximal mouth opening is stable in a majority of systemic sclerosis patients. Still, maximal mouth opening over time was smaller in patients with more severe organ involvement. Although microstomia was infrequent, a smaller maximal mouth opening was significantly associated with more mouth handicap, indicating the importance to address maximal mouth opening in routine care of systemic sclerosis patients.
Introduction
Systemic sclerosis (SSc) is an inflammatory disease characterized by fibrosis and vasculopathy involving the skin and vital organs. 1 A large humanistic burden to SSc patients is imposed by the disease and its manifestations, including mouth handicap. 2 Mouth handicap in SSc patients occurs frequently and comprises several aspects including xerostomia, reduced maximal mouth opening (MMO), widening of the periodontal ligament space, resorption of the mandibula and synovitis of the temporomandibular joint (TMJ).2,3
The mouth handicap of interest in this study is the MMO, which is measured by the maximal interincisal distance. There is no normal value for MMO defined in literature since it depends on age, sex and ethnic origin, but an MMO < 30.0 mm is defined as microstomia.4–6 MMO in SSc patients is declined compared to healthy people as a consequence of fibrosis and loss of tissue elasticity. 3 A smaller MMO has been correlated with disease severity in cross-sectional research and with lower health-related quality of life over time.7–9 Apart from the mouth handicap, the fibrotic process in the face and mouth causes considerable concerns about disfigurement and lower self-esteem.10–12
Exercise programmes seem to have some positive effect on mouth opening, although content and dosage of exercise programmes vary between studies. 13 The natural history of MMO in SSc has not been described, which contributes to a difficult interpretability of intervention studies on MMO. Furthermore, it is unknown which patients are prone to develop a decrease in MMO. One could hypothesize that higher disease activity in SSc involves reduced MMO, but since SSc is a heterogeneous disease with varied outcomes and different patterns of organ-based complications, it is of interest to assess more specifically which patients are at risk. These considerations prompted this study in which we aimed to describe the course of MMO in SSc patients and to analyse disease characteristics that are associated with a smaller MMO over time. Second, we aimed to assess the burden of MMO on self-reported mouth handicap.
Methods
Patients and study design
For this study, we used data of the Combined Care in Systemic Sclerosis (CCISS) prospective cohort of the Leiden University Medical Center. 14 Patient with a clinical SSc diagnosis with at least one MMO measurement available and who were enrolled in the CCISS prospective cohort between April 2009 and December 2015 were evaluated in this study. Patients were classified as SSc using the American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2013 classification criteria and were classified as limited or diffuse cutaneous SSc (lcSSc, dcSSc) according to the classification system described by Van den Hoogen et al. and LeRoy and Medsger15,16 Patients without skin fibrosis were classified as non-cutaneous SSc.
The CCISS cohort has previously been described in detail. 14 In short, all patients in the cohort undergo annual extensive screening during a one- or two-day healthcare programme, including detailed physical examination, a modified Rodnan skin score (mRSS) assessment, laboratory testing, echocardiography, electrocardiogram (ECG), Holter evaluation, pulmonary function test and high-resolution computed tomography (CT) for evaluating interstitial lung disease. The MMO was measured annually by a trained physical therapist using a digital calliper. 17 In addition, mouth handicap was assessed annually by the Mouth Handicap in Systemic Sclerosis scale (MHISS) questionnaire. The MHISS has been validated previously and is acknowledged in the EULAR Outcome Measures Library. The MHISS was used to evaluate the degree of mouth handicap in SSc according to the patient in three domains: mouth opening restriction, mouth dryness and aesthetic concerns.12,18 The study protocol for the CCISS cohort (REU 043/ SH/sh) was approved by the Ethics Committee and all patients provided written informed consent.
Organ involvement
For this study, clinical characteristics of patients were evaluated and included seven organ domains: 1: skin subtype (non-cutaneous SSc, limited cutaneous SSc and diffuse cutaneous SSc), 2: peripheral vasculopathy [yes/no] (presence of pitting scars and/or digital ulcers), 3: heart [yes/no] (reduced left ventricular ejection fraction (LVEF < 54%) and/or conduction abnormalities detected during ECG or Holter registration), 4: lung [yes/no] (presence of interstitial lung disease, defined as alveolitis and/or fibrosis detected on CT-imaging in combination with decreased lung function (forced vital capacity (FVC) < 80% and/or diffusing capacity of the lung for carbon monoxide (DLCO) < 70%)), 5: renal [yes/no] (presence of proteinuria of ⩾ 500 mg per 24 h and/or renal crisis in the past), 6: gastrointestinal [yes/no] (presence of at least one of the following characteristics: dysphagia, vomiting, gastric antral vascular ectasia, intestinal distension and faecal incontinence) and 7: musculoskeletal [yes/no] (presence of at least one of the following characteristics: synovitis, friction rubs and proximal muscle weakness).
Observer variability in MMO measurements
To be able to evaluate changes in MMO over time, intra- and interobserver variability of MMO measurements were assessed. Twenty-six healthy volunteers (73% female, mean age 30 ± 10 years) had their MMO measured at two time points by two physical therapists involved in the CCISS healthcare programme. The volunteers presented in random order and the physical therapists were unaware of each other’s and their own previous measurements. The mean MMO of 104 measurements in the healthy volunteers was 53.3 ± 9.4 mm. The intraclass correlation coefficients (ICCs) for interobserver variability were 0.903 (t = 1) and 0.947 (t = 2) for absolute agreement on single measures in a two-way random model. The ICC for intra-observer variability were 0.947 (physical therapist 1) and 0.945 (physical therapist 2). The mean differences between the physical therapists’ MMO measurements per volunteer (physical therapist 1 – physical therapist 2) were 3.1 ± 2.8 mm (p < .001) for t = 1 and 1.8 ± 2.8 mm (p = .004) for t = 2, according to the Bland and Altman 19 method. We, therefore, chose a difference of > 5.0 mm to represent a real change in MMO between two measurements.
Statistical analyses
We evaluated the course of MMO on group and on individual level. On group level, the course of MMO was analysed separately for early, intermediate and late SSc. Disease duration was classified based on the time since first non-Raynaud symptom at baseline: early SSc: < 2 years, intermediate SSc: 2–5 years and long-standing SSc: > 5 years. On individual level, change in MMO within a patient during annual follow-up visits was determined.
Because decreasing MMO is worrisome, we assessed characteristics of patients with decreasing MMO in two ways. First, we evaluated characteristics of patients that showed a decrease of MMO of > 5.0 mm over 1 year at some point during follow-up. In these patients, we analysed changes in disease severity (mRSS, FVC and DLCO) during the year in which the MMO decrease occurred, using Wilcoxon signed ranks tests and paired t-tests where appropriate. In addition, the number of prescribed immunosuppressive therapies and vasoactive agents in the year before and after the MMO decrease was analysed. Mortality in patients with decreasing MMO was compared to mortality in patients without decreasing MMO using Pearson’s chi-squared test. Second, linear mixed-effects models were used to identify SSc characteristics associating with a smaller MMO during follow-up, while incorporating repeated measurement within a patient. This analysis was performed univariably including longitudinal MMO measurement of all SSc patients as dependent factor, and organ involvement at baseline as independent factor. Because SSc can manifest in different patterns of organ-based complications, and because antibody status and sex have previously been correlated with MMO, we also performed a multivariable linear mixed-effects model including all organ involvements, antibody status and sex. 8 Three patients expressed two SSc-specific auto-antibodies and were excluded from only this analysis, because this is unusual in SSc and would possibly blur the evaluation of the association between auto-antibodies and the course of MMO. 20
Finally, we evaluated the burden of smaller MMO on reported mouth handicap as measured by the MHISS. This analysis was performed cross-sectionally at baseline and was assessed in separate multivariable linear regression models with total MHISS and its three subdomains as outcome, corrected for sex.
All statistical analyses were performed using SPSS version 25. The p-values < 0.05 were considered statistically significant. Standard deviation, interquartile range (IQR) and 95% confidence intervals (CIs) were computed where appropriate.
Results
Baseline characteristics
Eighteen patients were excluded because of no available MMO measurement. In total, 382 consecutive SSc patients were included in this study of whom baseline characteristics are presented in Table 1. The mean age was 54 years and the vast majority was female (83%). Ninety percent (n = 342) of patients met the ACR/EULAR 2013 Systemic Sclerosis Classification Criteria and 85% (n = 323) met the Van den Hoogen et al., and LeRoy and Medsger criteria.15,16 LcSSc was present in 58% (n = 223) and dcSSc in 25% (n = 95). Median follow-up time was 2.0 years (IQR = 0.0–3.0) and the total range of follow-up time was 0.0–6.0 years. The median number of MMO measurements per patient was 2.0 (IQR 1.0-3.0; total range 1.0–7.0). The percentages of patients positive for anti-topoisomerase I antibodies (ATA), anti-centromere antibodies (ACA) and RNA polymerase III were 24% (n = 91), 37% (n = 140) and 5% (n = 18), respectively.
Baseline characteristics of all included SSc patients.

Data are n (%), mean ± SD or median (interquartile range (IQR)).
ACA: anti-centromere antibodies; ATA: anti-topoisomerase I antibodies; MHISS: mouth handicap in systemic sclerosis; SSc: systemic sclerosis.
SSc subgroups were defined according to duration of non-Raynaud symptoms: early SSc: < 2 years, intermediate SSc: 2–5 years and long-standing SSc: > 5 years.
Course of MMO
At baseline, mean MMO was 42.2 ± 8.0 mm and 7% of patients (n = 27) suffered from microstomia. In line with the general healthy population, men had larger MMO than women: mean baseline MMO in male patients was 45.0 ± 7.8 mm versus 41.6 ± 7.9 mm in female patients (p = .002). During follow-up, overall mean percentage of patients with microstomia was 9%, range 6%–12%. In Figure 1, the mean MMO during follow-up is shown for early, intermediate and long-standing SSc separately. At group-level, mean MMO was relatively stable over time independent of disease duration. Annual change in MMO on individual level during the first year of follow-up, according to the disease duration (time since first non-Raynaud symptom), is shown in Figure 2. Although a decrease in MMO > 5.0 mm between two consecutive visits occurred in patients with different disease durations, a change in MMO occurred more often in patients with a short disease duration.
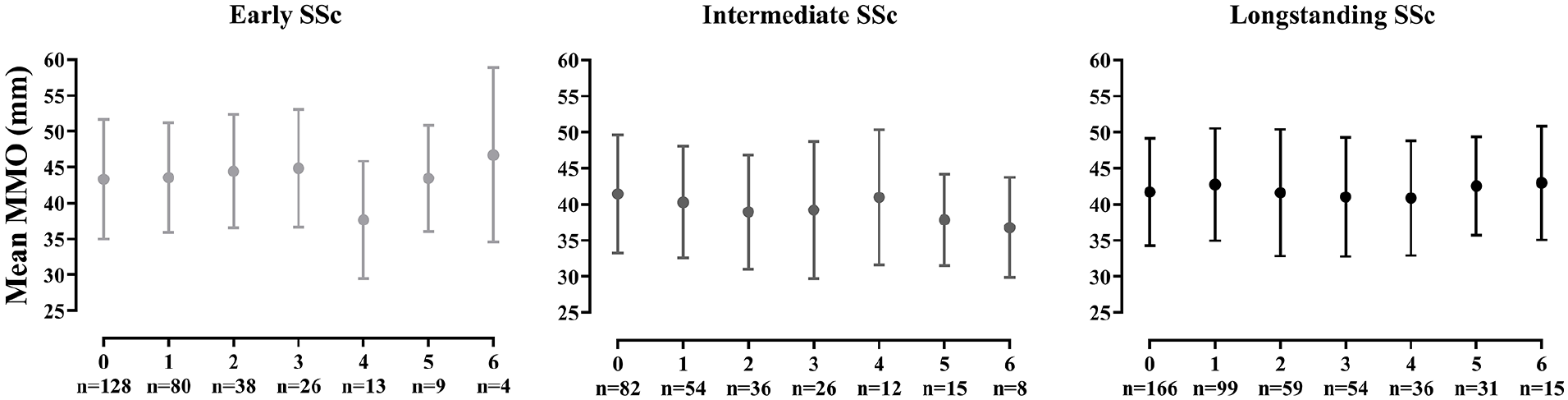
Mean MMO over time in SSc patients, stratified according to disease duration.
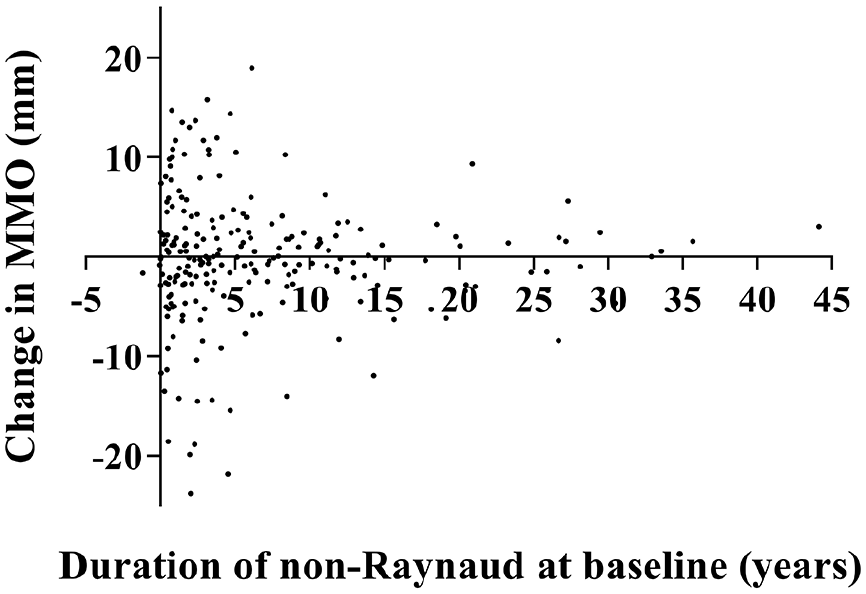
Change in MMO during the first year of follow-up, according to duration of non-Raynaud symptoms.
Annual decrease in MMO and characteristics in patients with decreasing MMO
During follow-up, the percentage of patients showing an MMO decrease of > 5.0 mm ranged from 5% to 16%, mean 10%. This accounts for in total 63 patients (median follow-up 3.0 years (IQR = 2.0–5.0)) that showed a decrease in MMO of > 5.0 mm at some point during follow-up. This MMO decrease occurred between baseline and the first year of follow-up in 56% (n = 35) of these patients. Of the 63 patients, 22% had early SSc (n = 14; 11% of the total early SSc group), 31% had intermediate SSc (n = 20; 24% of the total intermediate SSc group) and 44% had long-standing SSc (n = 29; 17% of the total long-standing SSc group). Figure 3 shows the course of MMO of these 63 patients starting at the year before the MMO decrease occurred. Stabilization of MMO was observed in a majority of patients after the decrease had occurred. Only two patients showed a second decrease > 5.0 mm over 1 year during follow-up. Of the 63 patients showing a decrease in MMO, 16% (n = 10) developed microstomia. The MMO decrease was accompanied by clinical increase of disease severity: delta mRSS = +2.1, p = .052, delta DLCO = −2.4%, p = .007. In addition, immunosuppressive therapies and vasoactive agents were started more often in the year after the MMO decrease had occurred (Supplemental Material 1). Mortality was not higher in the group of 63 SSc patients with a decrease in MMO of > 5.0 mm: of 382 included patients, 32 patients died after a mean follow-up of 2.8 ± 1.7 years, of which four patients had shown a decrease in MMO at some point (p = .453).
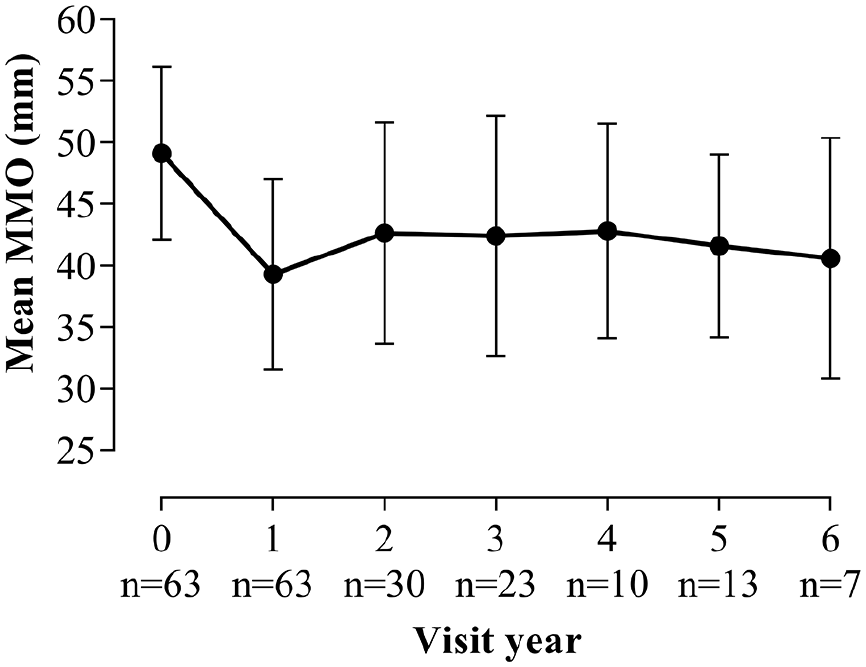
The course of MMO in the group of 63 SSc patients with decreasing MMO during follow-up.
Baseline determinants of a smaller MMO at baseline and over time
All seven organ involvements at baseline, except for cardiac involvement, were associated with a smaller baseline MMO (Supplemental Material 2). When we studied baseline organ involvement with MMO over time in univariable linear mixed-effects models, sex and all seven organ involvements were significantly associated with smaller MMO over time (Table 2). Patients seropositive for RNA polymerase III antibodies and ATA showed smaller MMO than seronegative patients (β = −5.9, p = .001 and β = −2.1, p = .037, respectively). Univariable association between MMO over time with antibody status, using ATA as reference category instead of seronegative patients, showed a significantly larger MMO over time in ACA-positive patients compared to ATA-positive patients (β = 3.6, 95% CI = 1.7–5.6, p < .001). Multivariable analysis including all seven organ-systems, sex and antibody status, demonstrated that sex, a more extended skin subtype, peripheral vasculopathy, pulmonary, renal and gastrointestinal involvement at baseline were associated with smaller MMO over time. The mean MMO over time in patients with lcSSc was 2.9 mm smaller compared to those with non-cutaneous SSc (β = −2.9, p = .004). Significant associations between MMO over time and baseline characteristics were also seen for peripheral vasculopathy (β = −2.4, p = .001), pulmonary involvement (β = −1.8, p = .027), renal involvement (β = −3.4, p = .031) and gastrointestinal involvement (β = −3.0, p < .001; Table 2). There was no significant association between antibody status with MMO over time in multivariable analysis.
Univariable and multivariable linear mixed-effects models of baseline disease characteristics associating with MMO over time in SSc patients.
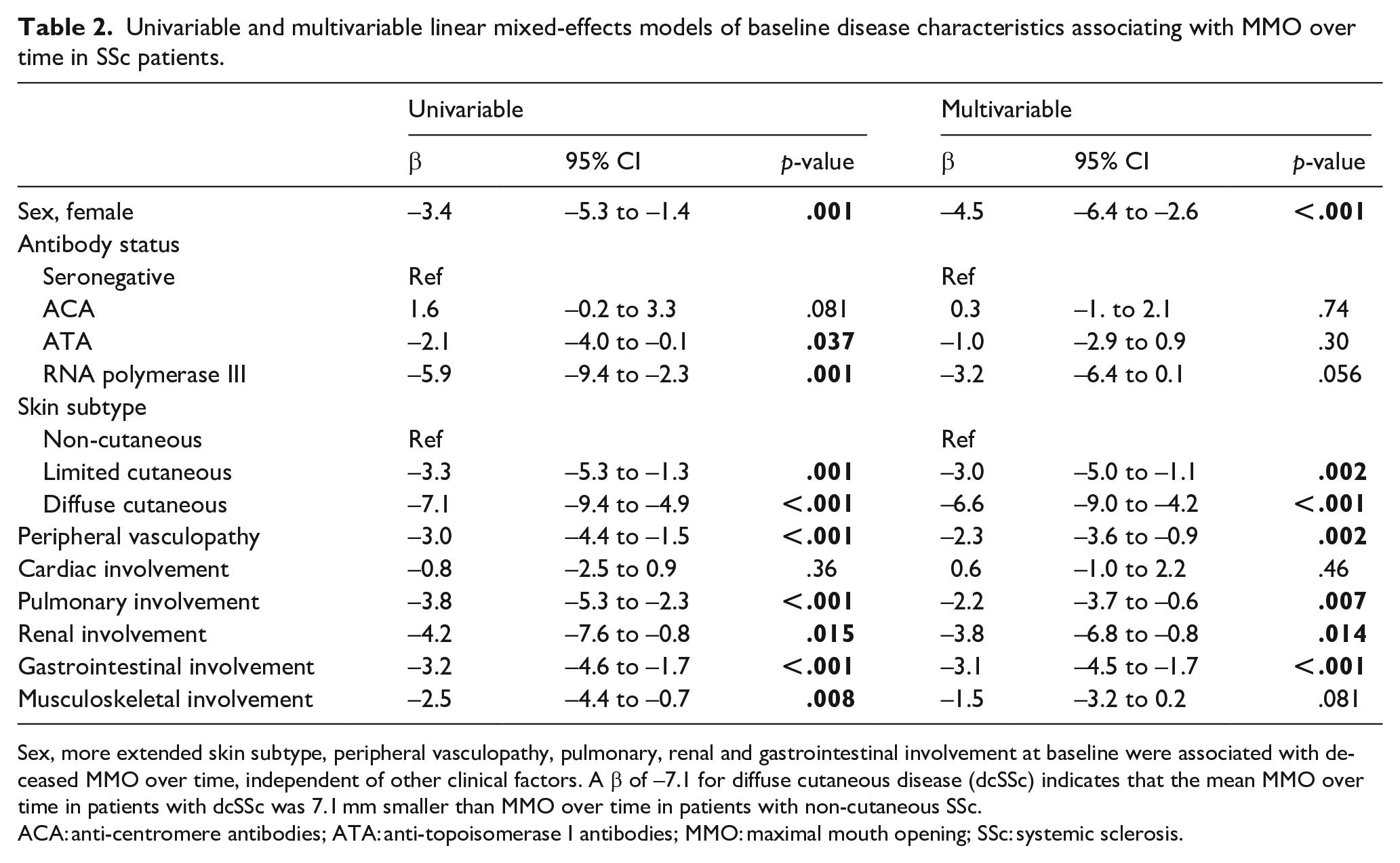
Sex, more extended skin subtype, peripheral vasculopathy, pulmonary, renal and gastrointestinal involvement at baseline were associated with deceased MMO over time, independent of other clinical factors. A β of −7.1 for diffuse cutaneous disease (dcSSc) indicates that the mean MMO over time in patients with dcSSc was 7.1 mm smaller than MMO over time in patients with non-cutaneous SSc.
ACA: anti-centromere antibodies; ATA: anti-topoisomerase I antibodies; MMO: maximal mouth opening; SSc: systemic sclerosis.
The burden of a small MMO on mouth handicap
Cross-sectional analysis of baseline MMO and baseline MHISS revealed that MHISS was significantly lower, thus indicating less mouth handicap in patients with larger MMO (β = −0.5, 95% CI = −0.6 to −0.3, p < .001; Table 3). This indicates that the mouth handicap score increases with 0.5 point per mm decrease in MMO. The three subdomains of the MHISS concerning mouth opening restriction, mouth dryness and aesthetic concerns separately, were also significantly associated with MMO (Table 3).
Linear regression analyses between baseline MMO and mouth handicap in SSc patients, corrected for sex.
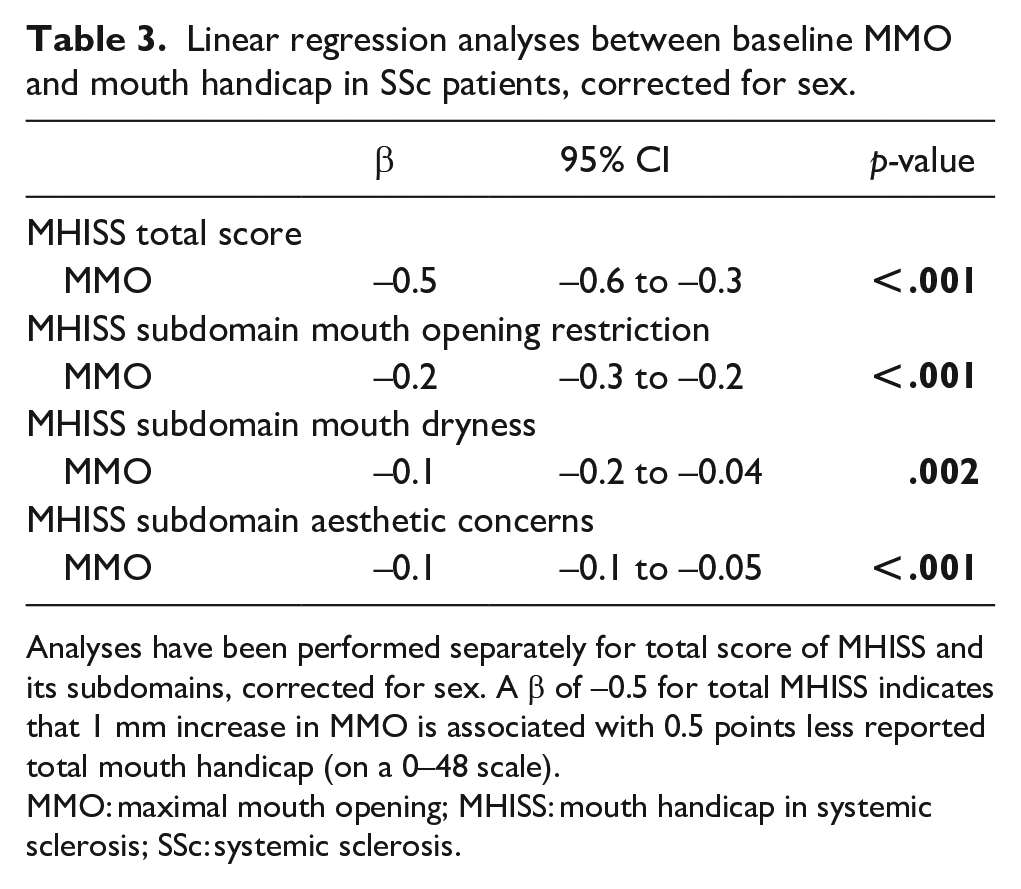
Analyses have been performed separately for total score of MHISS and its subdomains, corrected for sex. A β of −0.5 for total MHISS indicates that 1 mm increase in MMO is associated with 0.5 points less reported total mouth handicap (on a 0–48 scale).
MMO: maximal mouth opening; MHISS: mouth handicap in systemic sclerosis; SSc: systemic sclerosis.
Sensitivity analysis with patients meeting classification criteria
Analysis of MMO over time according to baseline organ involvement was repeated in patients fulfilling the ACR/EULAR 2013 Systemic Sclerosis Classification Criteria at baseline (n = 342). Results were comparable to the total SSc group (Supplemental Material 3).
Discussion
Reduced mouth opening occurs frequently in SSc patients and is disabling. To the best of our knowledge, this is the first study that described the course of MMO in a large group of SSc patients over time. Using data from a prospective cohort of SSc patients, we showed that mean MMO over time was relatively stable in a majority of patients and microstomia occurred in 6–12% of patients. In patients with a significant MMO decrease, an increase in disease severity was observed (worsening of mRSS and DLCO and increase in prescribed immunosuppressive and vasoactive agents). Furthermore, independent of other variables, more extended skin subtype, presence of peripheral vasculopathy and involvement of the pulmonary, gastrointestinal and renal system at baseline were associated with smaller MMO over time. Finally, a smaller MMO was significantly associated with more severe mouth handicap including restricted mouth opening, dry mouth and aesthetic concerns as experienced by patients.
Our data underline the necessity of awareness for MMO as a relevant health problem in SSc patients and emphasize the need for interventions improving MMO in SSc, especially in patients who are at risk of having a smaller MMO. In addition, by describing course of MMO over time, our results can be of help when interpreting results of studies focussing on improving MMO in SSc. As such, in intervention studies on MMO, it should be taken into account that a majority of SSc patients remain stable in MMO and that observer variability is present in MMO measurements.
It is not clear why some patients develop a declined MMO and others do not. Our results show that increased disease severity including a more extended skin subtype was predictive for a smaller MMO over time. Intuitively, this seems logical as skin fibrosis can cause declining MMO. This is also in agreement with previous cross-sectional research showing that skin involvement, as measured by mRSS, was negatively associated with MMO. 8 Overall disease severity may represent the amount of fibrosis and microangiopathy, not only in the areas affecting mouth opening such as facial skin and the structures surrounding the TMJ, but also the internal oral organs such as the tongue. 21 Unfortunately, data on TMJ involvement were not available for our cohort, but TMJ involvement has been described frequently in SSc and was presumably present in our study as well. 22 Strikingly, although microstomia was present only in a minority of patients, still a significant association between MMO and aesthetic concerns was observed indicating the impact of reduced MMO.
A previous study showed that presence of ACA was associated with a larger MMO based on a multivariable regression analysis using ATA as reference group. 8 Although our univariable analysis revealed that ACA-positive patients had larger MMO over time than ATA-positive patients, we did not confirm an association between antibody and MMO in our multivariable analysis. A likely explanation for this finding is that adding skin subtype in the multivariable analysis overruled the association between auto-antibody and MMO. 23 Indeed, in the study by Baron et al., 8 neither skin subtype nor mRSS were included in the multivariable model. Remarkably, we found that patients seropositive for RNA polymerase III had a significantly smaller MMO at baseline and over time. This is in line with previous research associating RNA polymerase III antibodies with a very rapid onset of disease and skin thickening progression in SSc. 24
This study has several limitations that should be considered, such as its cohort design. Patients with more severe disease were probably more likely to drop-out the annual care programme due to inability to participate. On the contrary, all patients with a clinical diagnosis were included and as such our data reflect changes in MMO in the broad SSc population. A second limitation is that the choice for a minimum difference of 5.0 mm implying a real change in MMO is beforehand arbitrary. Although we confirmed the validity of our cut-off value by determining the ICC of the physical therapists performing the MMO measurements, the observer variability is studied in healthy volunteers and could be different in SSc patients who already show more impaired MMO. It also must be pointed out that it is unknown what change in MMO is clinically relevant for SSc patients. Further research on the minimal clinical important difference in MMO is required. A third limitation is the possible influence of pharmacological and exercise therapy on the course of MMO, which is not addressed in this study. However, all included patients participated in the same cohort and received the same information about mouth exercises during the annual visit to the care programme. As for the pharmacological therapies, little is known on its effect on MMO. 6 Still, this study’s strength lies in its longitudinal design and overall high quality of prospectively collected data.
In summary, we set out to determine the course of MMO over time in SSc patients and its determinants. We showed that MMO over time was relatively stable in a majority of SSc patients. Patients with a more severe disease at baseline were at risk for a smaller MMO over time. Because a smaller MMO was also associated with self-reported mouth handicap by patients, we advocate that MMO is addressed in routine care of SSc patients and emphasize the importance of research on the treatment of reduced mouth opening.
Supplemental Material
sj-pdf-1-jso-10.1177_23971983221138177 – Supplemental material for Mouth opening in systemic sclerosis: its course over time, determinants and impact on mouth handicap
Supplemental material, sj-pdf-1-jso-10.1177_23971983221138177 for Mouth opening in systemic sclerosis: its course over time, determinants and impact on mouth handicap by Sarah JH Khidir, Maaike Boonstra, Sytske Anne Bergstra, Gerry WM Boerrigter, Elles M Voogt-van der Harst, Maarten K Ninaber, Nina Ajmone Marsan, Tom WJ Huizinga, Annette HM van der Helm-van Mil and Jeska K de Vries-Bouwstra in Journal of Scleroderma and Related Disorders
Footnotes
Acknowledgements
The authors would like to thank C.H. Kromme for managing the data and E.C. de Moel for her explanation on linear mixed-effects models.
Data availability statement
The data underlying this article are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
