Abstract
Gastrointestinal symptoms affect the great majority of patients with systemic sclerosis. Management of these complications is often challenging as any region of the gastrointestinal tract may be involved, and significant heterogeneity exists in clinical presentation, kinetics, and outcomes. Here, we highlight new findings relevant to the management of systemic sclerosis-related gastrointestinal disease (lights) and consider areas that we have yet to elucidate (shadows).
Keywords
Introduction
Systemic sclerosis (SSc) is an autoimmune connective tissue disease characterized by progressive fibrosis, vasculopathy, and internal organ dysfunction. 1 The gastrointestinal (GI) tract is frequently affected, with over 90% of SSc patients experiencing GI complications. 2 Every region of the GI tract can be involved, resulting in diverse and variable disease manifestations. While some patients only experience mild gastroesophageal reflux disease (GERD) throughout their disease course, others may experience more severe complications, such as recurrent pseudo-obstruction and malnutrition.2–4 These complications are associated with substantial morbidity, including hospitalizations and the need for total parenteral nutrition (TPN). Currently, there are no standardized guidelines to assess and manage these complications. Here, we sought to shed light on the key articles related to SSc-related GI disease published in the last 4 years and to position these discoveries in the context of the existing literature. We will also discuss areas of focus for future studies and touch on the shadows that remain to be illuminated in SSc-related GI disease.
Methods
We conducted a literature search on PubMed with the terms “gastrointestinal” and “scleroderma” using the following filters: Classical Article, Clinical Study, Clinical Trial, Clinical Trial Protocol, Clinical Trial, Phase I, Clinical Trial, Phase II, Clinical Trail, Phase III, Clinical Trial, Phase IV, Comparative Study, Consensus Development Conference, Consensus Development conference, NIH, Controlled Clinical Trial, Meta-Analysis, Multicenter Study, Observational Study, Randomized-Controlled Trial, Review, Systematic Reviews, Validation Study, and in the last 4 years. A search on clinicaltrials.gov using the terms and phrases “scleroderma” and “gastrointestinal” was performed to identify ongoing trials in these areas. A total of 82 publications and clinical trials were identified. Single case reports were excluded, however, interesting case series are mentioned. We then prioritized the strongest studies and/or those with the most novel findings for discussion in this article.
Oropharynx
The oropharynx is commonly affected in SSc. 5 Patients with SSc often suffer from xerostomia, dental caries, loosening teeth, and microstomia. Due to the negative effects of oropharyngeal complications on quality of life, experts recommend early dental care referrals to determine the need for interventions (e.g. fluoride treatments, tooth stabilization procedures), and for the completion of oral exams to rule out tongue and pharyngeal carcinoma.5–7
In particular, microstomia presents challenges in maintaining good oral hygiene, which may culminate in tooth decay and impaired nutrition. 6 Prior studies determined that stretching exercises can improve inter-incisor distance and the size of the oral aperture in SSc patients with severe microstomia (oral opening < 30 mm). 8 Recently, in a single-blind randomized-controlled study, Uras et al. 9 compared changes in oral aperture size using educational materials alone (brochure and DVD) versus educational materials plus face-to-face teaching at baseline and at each subsequent follow-up (3, 6, 9, and 12 months). Experienced nurses held teaching sessions where they explained the exercises, and observed and corrected exercise performance. Patients were expected to complete 25 min of these exercises daily. Using an intention-to-treat analysis (n = 63), the experimental group had a non-significant improvement in oral aperture compared to the control group (p = 0.10). In a post hoc per-protocol analysis, including only those who completed the protocol for their allocated group (n = 39; 18 patients in the control group and 21 patients in the experimental group), there was a significant difference in the oral aperture size between groups (p = 0.02) with the experimental group experiencing a greater degree of improvement than the comparator group. The investigators ultimately determined that face-to-face nursing rehabilitation training improved microstomia more than providing patients with educational materials alone.
Key points
Face-to-face training combined with educational materials detailing stretching exercises may improve oral aperture size in patients with SSc.
Esophagus
Esophageal involvement is common in SSc with up to 90% of patients experiencing symptoms, such as dysphagia and GERD.2,10 Dysphagia, particularly to both solids and liquids, is often a sign of esophageal dysmotility; while symptoms of GERD are less specific and may be attributable to esophageal or gastric dysmotility and/or lower esophageal sphincter (LES) dysfunction.11,12 In patients with SSc, GERD may be associated with mild to severe symptoms. 11 If left untreated, GERD may lead to the development of strictures, esophagitis, Barrett’s esophagus, or esophageal carcinoma. Moreover, chronic uncontrolled GERD has been implicated in causing severe interstitial lung disease due to recurrent microaspiration.13–15
The treatment of GERD often begins with dietary and lifestyle modification, but these approaches are typically inadequate in controlling symptoms. 16 Proton-pump inhibitors (PPIs) are standard for care for patients with GERD and are recommended in the 2016 European League against Rheumatism (EULAR) recommendations. 17 Among the PPIs, there are no data to suggest that one is superior to another and the standard dosages for different PPIs are described in other reviews.18,19
Nevertheless, a subset of patients continues to experience reflux symptoms despite high-dose PPI therapy. To identify predictors of PPI-partial response (PPI-PR) in SSc patients with GERD, Foocharoen et al. 20 studied 243 patients with SSc treated with omeprazole twice daily. The severity of symptom-grading by visual analogue scale (VAS) and frequency scale for symptoms of GERD (FSSG) were measured at baseline and at 4 weeks. They defined PPI-PR as < 50% improvement in symptom severity and in acid reflux scores, and determined that 54% of their cohort had PPI-PR GERD. After adjusting for potential confounders, they found that esophageal dysphagia was the only predictor of PPI-PR GERD. Although these results need further validation, they suggest that screening patients for esophageal dysphagia may identify SSc patients who may require a more aggressive GERD regimen and may benefit from combination therapy.
The effectiveness of different combination therapies was recently explored in a randomized placebo-controlled trial. This trial sought to evaluate the effectiveness of add-on domperidone 10 mg three times daily versus alginic acid one chewing tablet three times daily in SSc patients with PPI-PR GERD on omeprazole 20 mg two times daily. 20 Alginic acid forms a suspension that floats above gastric contents in the stomach and works as a barrier to protect against reflux. SSc patients with GERD who were PPI-PR and not receiving domperidone or alginic acid for 2 weeks prior to baseline evaluation were included. VAS, FSSG, and the EuroQoL five-dimension questionnaires were obtained to assess symptom response. Thirty-eight patients were randomized to the domperidone and omeprazole treatment group and 37 patients received alginic acid and omeprazole. All three symptom scores improved significantly in both groups after 4 weeks of treatment. There was no significant difference in symptom-grading between the two groups at 4 weeks, and five patients (13.2%) in the domperidone group and eight patients (21.6%) in the alginic acid group did not respond (non-response was defined as symptom improvement < 50% after 4 weeks of combination treatment). This suggests that both domperidone and alginic acid are equally effective as add-on therapies to omeprazole.
Buspirone, a 5-HT1A receptor agonist that was initially approved for treatment of depression and anxiety, has been shown to increase LES pressure (Table 1).21,22 The effect of buspirone on esophageal symptoms in patients with SSc was recently evaluated in a 4-week open-label trial. 23 In this study, patients underwent high-resolution esophageal manometry and chest computed tomography (CT) to assess esophageal motor function and esophageal diameter, respectively. Patients’ self-assessment of symptoms (dysphagia, heartburn, regurgitation, and chest pain) and severity, as measured by VAS score, were also documented. All patients were on a single dose of PPI before and during the study, and received 20 mg of buspirone daily as the intervention. Of the 30 patients enrolled, 22 completed the trial. After 4 weeks, LES resting pressure increased significantly from 7.7 ± 3.9 to 12.2 ± 4.6 mmHg (p = 0.00002). Notably, there was a significant negative correlation between individual increases in resting LES pressure and supra-aortic esophageal diameter. This suggested that patients with less severe esophageal dilatation responded more to buspirone. In addition, both heartburn and regurgitation scores by VAS improved significantly from baseline after 4 weeks of buspirone. Thus, buspirone may be an effective add-on therapy to PPI for SSc patients with refractory GERD symptoms, particularly in the context of a hypotensive LES.
Prokinetic agents used in the management of SSc-related GI dysmotility.
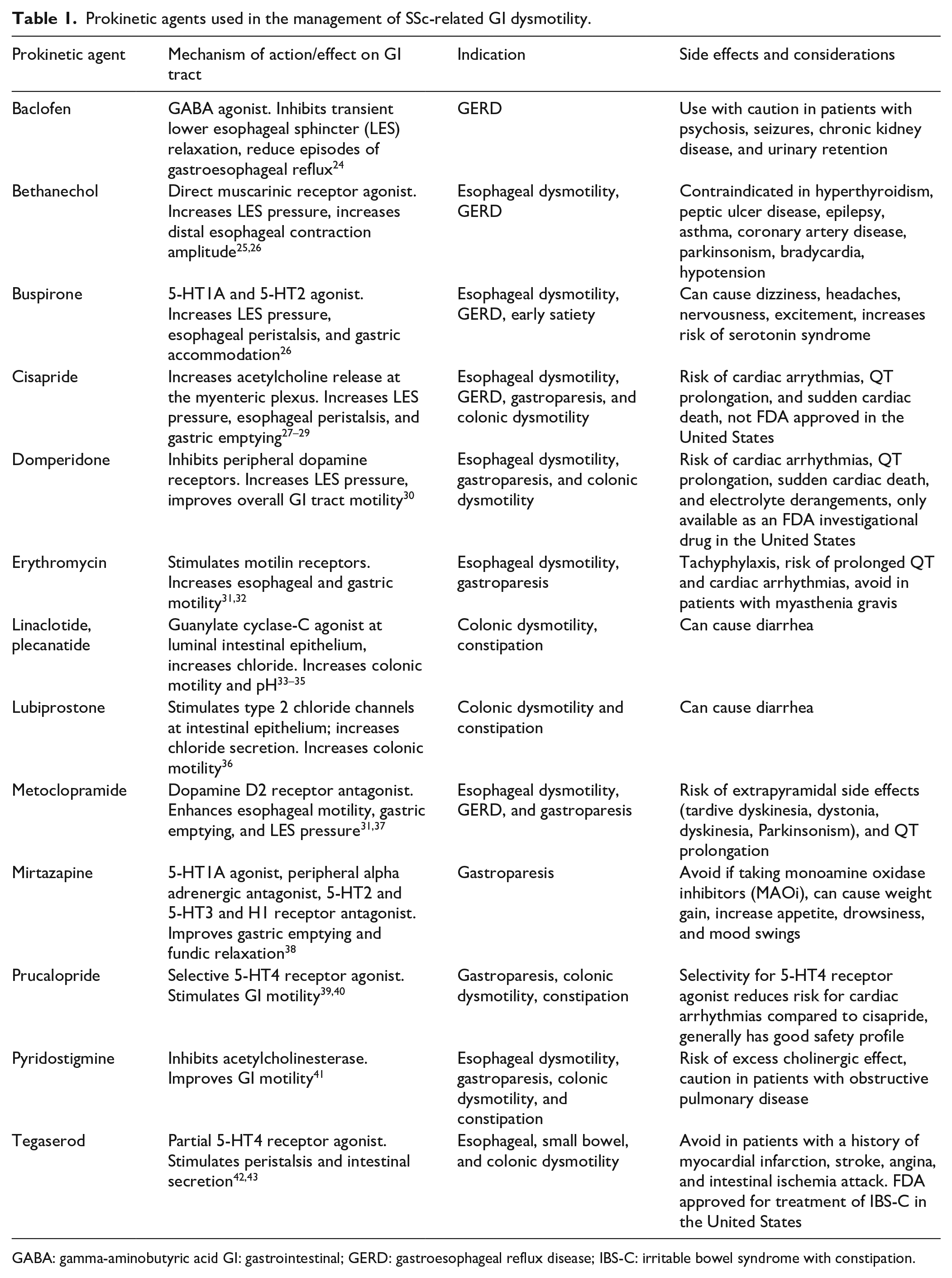
GABA: gamma-aminobutyric acid GI: gastrointestinal; GERD: gastroesophageal reflux disease; IBS-C: irritable bowel syndrome with constipation.
Recently, the effects of vonoprazan, a competitive H +/K + ATPase inhibitor, were studied in Japan for the treatment of SSc-related GERD. 44 Unlike PPIs, vonoprazan accumulates in parietal cells and can provide constant acid suppression. In this study, FSSG scores were collected in 15 SSc patients before and after treatment. Fourteen of the 15 patients were previously on a PPI or H-2 blocker. Patients were followed for 4–48 weeks after vonoprazan initiation. Of the 15 patients, seven received 20 mg/day of vonoprazan and eight received 10 mg/day. However, four out of eight of those receiving 10 mg/day eventually increased their dose to 20 mg/day due to insufficient effect on the lower dose. Total FSSG scores before treatment were high at 25.2 ± 10.7 (SD), and after treatment, mean scores decreased significantly to 9.6 ± 7.0 (SD; p = 0.0004). Significant decreases in mean acid reflux (p < 0.0001) and dysmotility (p = 0.0022) scores were also reported. Although vonoprazan is currently not available outside of Asia, we anticipate that it will play a significant role in the treatment of GERD in the coming years as more studies emerge.
In summary, the treatment of SSc-related esophageal symptoms is complex, and it can be helpful to determine whether esophageal symptoms are primarily due to esophageal dysmotility, LES dysfunction, or both. Those with decreased LES pressure may benefit from the addition of buspirone to PPI therapy. 23 Patients with dysmotility may benefit from prokinetics, and a trial of alginic acid may be effective in those who cannot tolerate prokinetics. 20
Key points
Distal esophageal dysphagia is a predictor of PPI-PR GERD.
Domperidone and alginic acid are both equally effective as add-on agents to omeprazole for improving symptoms of PPI-PR GERD.
Buspirone improves LES resting pressure and improves heartburn and regurgitation when added to PPI for treatment of GERD.
Vonoprazan at 20 mg/day improves mean total FSSG, acid reflux, and dysmotility scores.
Stomach
Gastric abnormalities are common in SSc and are reported in 30%–50% of patients. 45 Gastric antral vascular ectasia (i.e. GAVE) is reported in up to 35% of patients and may result in recurrent upper GI bleeding. 46 Gastroparesis and the loss of gastric accommodation may culminate in symptoms of early satiety, nausea, vomiting, and regurgitation, and refractory GERD. Initial treatment involves lifestyle modification, such as consuming smaller meals and avoiding foods that aggravate symptoms. Some patients may require pro-motility agents, such as metoclopramide, domperidone, and erythromycin to control symptoms. 47 However, the use of metoclopramide is limited due to the risk of extrapyramidal symptoms and the use of domperidone and erythromycin are limited due to cardiac risks and tachyphylaxis, respectively.
Recent studies have suggested that prucalopride, a selective 5-hydroxytryptamine-4 (5-HT4) receptor agonist approved for the treatment of chronic idiopathic constipation, may benefit patients with gastroparesis (Figure 1).47,48 A double-blind, randomized, placebo-controlled crossover study by Carbone et al. investigated the effect of prucalopride 2 mg daily on gastric emptying rate and symptoms in non-SSc patients with gastroparesis (n = 34). Their primary endpoint was a change in symptom severity as assessed by the gastroparesis cardinal symptom index (GCSI), and their secondary end points were the symptom measures captured from the patient assessment of upper gastrointestinal disorders—symptom severity index, the patient assessment of upper gastrointestinal disorders—quality of life, and daily diaries. Gastric emptying rate was assessed with the C-octanoic breath test. Importantly, patients treated with prucalopride reported significantly improved total GCSI and the subscales of fullness/satiety, nausea/vomiting, and bloating/distension. Patient’s overall patient assessment of upper gastrointestinal disorders—quality of life score and gastric half-emptying time also significantly improved. Another study examined the effects of prucalopride in a small population of patients with SSc. In this open-label cross-over study (PROGASS), the investigators identified improvement in both reflux and bloating symptoms after treatment with prucalopride though, this study was primarily focused on the colon and is discussed in more detail below. 49
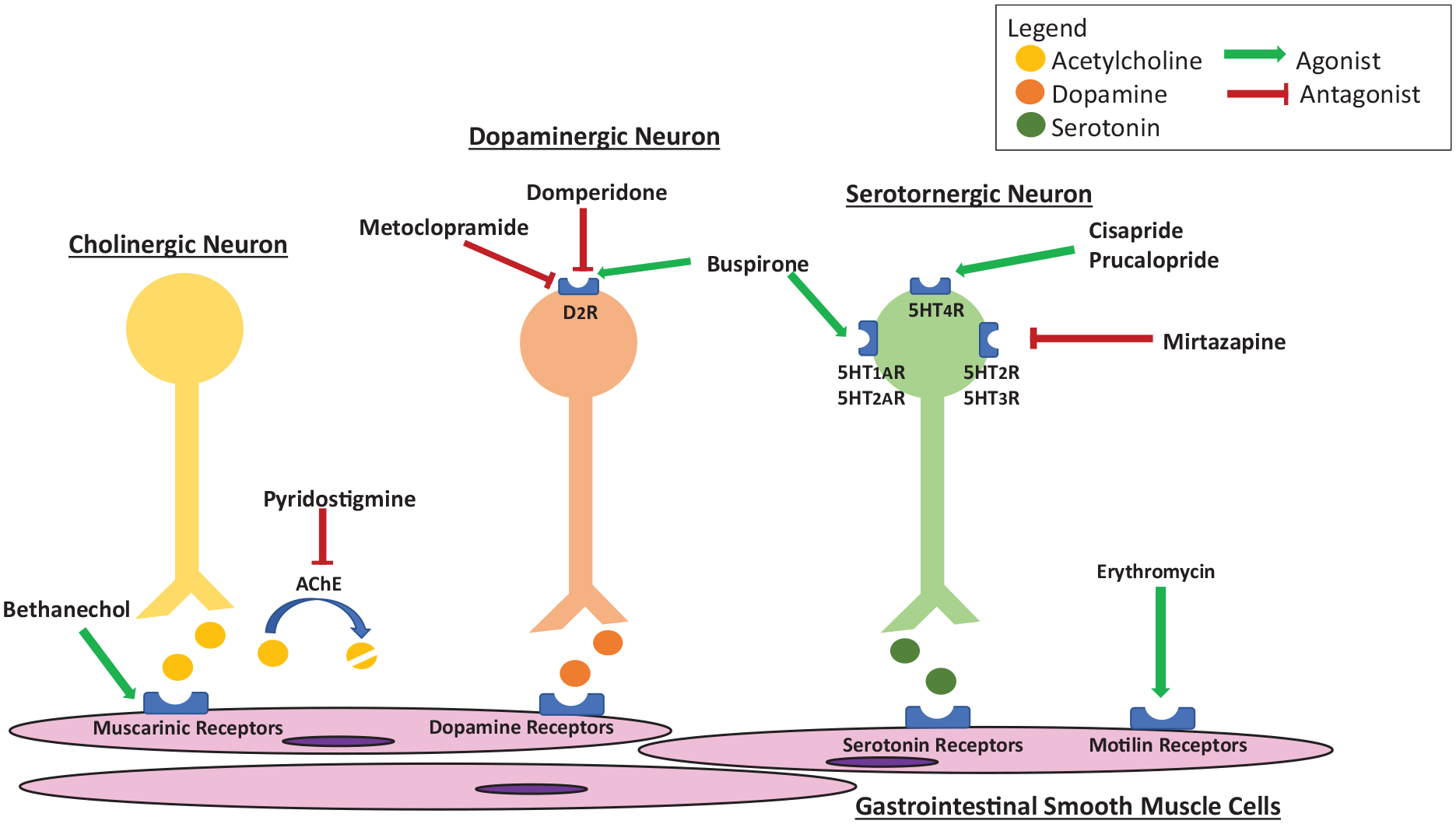
Action of pro-motility agents at the GI tract.
Recently, an open-label prospective study sought to assess the efficacy and safety of mirtazapine in non-SSc patients with gastroparesis. 38 In this study, 30 patients took mirtazapine 15 mg at bedtime daily for 4 weeks. Data on symptom severity were collected using the GCSI and clinical patient grading assessment scale (CPGAS) at pretreatment, 2, and 4 weeks. The primary and secondary end points were changes in nausea and vomiting in response to mirtazapine as measured by the GCSI and CPGAS, respectively. Of the 30 patients, 24 completed the study. Fourteen patients reported adverse effects, and 6/14 discontinued therapy. A statistically significant improvement in nausea, vomiting, retching, and perceived loss of appetite was observed at 2 and 4 weeks relative to baseline. Statistically significant improvements in CPGAS scores at weeks 2 and 4 were also noted. This supports the potential benefit of mirtazapine in patients with gastroparesis and symptoms of nausea and vomiting. Mirtazapine may be particularly helpful for patients with poor intake and weight loss as it is also an appetite stimulant. Future studies evaluating the effects of mirtazapine on patients with SSc are warranted.
Transcutaneous electroacupuncture has also been studied in SSc patients with refractory upper GI symptoms and/or gastroparesis. A recent study utilized patient-reported outcomes (scleroderma health assessment questionnaire (SHAQ), the University of California Los Angeles Scleroderma Clinical Trial Consortium Gastrointestinal Tract Instrument 2.0 (UCLA GIT 2.0), and short-form survey 36 (SF-36)) assessed at baseline and after 14 days to measure the effects of applying transcutaneous electrical nerve stimulation (TENS) using a commercially available TENS device at GI acupoints—ST36 (Zusanli) and PC6 (Neiguan). 50 Specific parameters of the electrical stimulus are described in the original paper. The investigators determined that prolonged TENS application decreased GI symptom scores (p = 0.02). Although more studies are needed for validation, TENS may be a novel option for treatment of upper GI symptoms. 50
Key points
Prucalopride at 2 mg daily can improve symptoms associated with gastroparesis and improve gastric half-emptying times in patients with SSc.
Mirtazapine at 15 mg nightly, can improve symptoms of nausea and vomiting associated with gastroparesis, while also increasing appetite.
TENS may be a novel adjunct to the treatment of SSc-related GI disease, improving symptoms and physical functioning scores.
Small bowel
Approximately, 8%–50% of patients with SSc experience small bowel dysfunction. 51 Small intestinal bacterial overgrowth (SIBO) and abnormal bowel transit are the main complications experienced that result in symptoms, such as bloating, diarrhea, weight loss, malnutrition, and decreased quality of life.52–54 The prevalence of SIBO is estimated to be 30%–62% and identifying clinical features associated with SIBO remains an important priority for risk stratification. 55 Some studies have found that a global symptomatic score of digestive symptoms ⩾ 5, disease duration > 5 years, significant weight loss in past 6 months (defined as ⩾ 5% of total body weight), and a higher UCLA GIT score are associated with SIBO in SSc patients.54,56,57 Furthermore, our group previously demonstrated that male sex, myopathy, and sicca symptoms were associated with severe GI dysmotility (defined as the need for TPN). 58 Subsequent studies went on to show that among SSc patients who required TPN during their disease course, all had objective delayed small bowel transit by whole gut transit (WGT) scintigraphy, highlighting the fact that small bowel dysfunction is enriched among patients with SSc who require TPN. 59
Currently, there is no standardized approach to managing SIBO in SSc, and patients are often treated empirically with cyclic antibiotics.2,10 Rifaximin has been shown to be effective in the management of diarrhea, other abdominal symptoms, and in normalizing lactulose hydrogen breath tests in SSc patients with SIBO. 60 The American Gastroenterological Association recommends a total daily rifaximin dose of 800–1200 mg a day with treatment lasting 1–4 weeks. 61 Unfortunately, recurrence of SIBO is common and patients may need multiple courses of antibiotics to control symptoms. This is not without risks, however, as antibiotics promote GI dysbiosis which may exacerbate symptoms. 62 Once patients are treated for SIBO, management of small and/or large bowel dysmotility, when present, is important to optimize function and possibly reduce SIBO recurrence. Small bowel dysmotility is usually treated with octreotide, though some reports also support the use of prucalopride and pyridostigmine.19,41,63–66 Of note, the pathogenesis of small bowel dysmotility is thought to involve atrophy of the muscularis propria and neuropathy.67,68 Thus, it is important to identify and treat dysmotility early, while the smooth muscle of the GI tract is still functional and responsive to prokinetic agents. 2
Gut dysbiosis has also been shown to play a role in the GI symptoms, and prior work demonstrates that SSc patients have alterations in their gut microbiome.69,70 Building upon this idea, novel treatments, such as fecal microbiota transplantation (FMT) are being studied in SSc. In a single-center, randomized, double-blind, placebo-controlled 16-week study, Fretheim et al. 71 explored the safety and efficacy of FMT using commercially available anaerobic cultivated human intestinal microbiota (ACHIM) in patients with SSc. Ten female patients were randomized to FMT by gastroduodenoscopy (n = 5) or placebo (n= 5). Primary endpoints were safety and clinical efficacy on GI symptoms, measured using the minimally clinically important difference (MCID) UCLA GIT 2.0, at weeks 4 and 16. 72 Symptoms that were reported as “somewhat better” were considered clinically meaningful. Two of the control patients experienced procedure-related serious adverse effects. Symptoms of diarrhea, distension/bloating, and/or fecal incontinence improved in four out of five patients in the FMT group compared to two out of four in the placebo group. Based on the findings of this pilot study, the investigators launched the ReSScue trial, a phase II, multicenter, placebo-controlled, randomized 20-week trial to evaluate the safety and efficacy of FMT on lower GI symptoms. 73 While this study is still ongoing, it will be interesting to see if their results will further support FMT as a potential treatment for SSc patients with GI symptoms (Table 2).
Ongoing clinical trials related to management of GI disease in SSc.
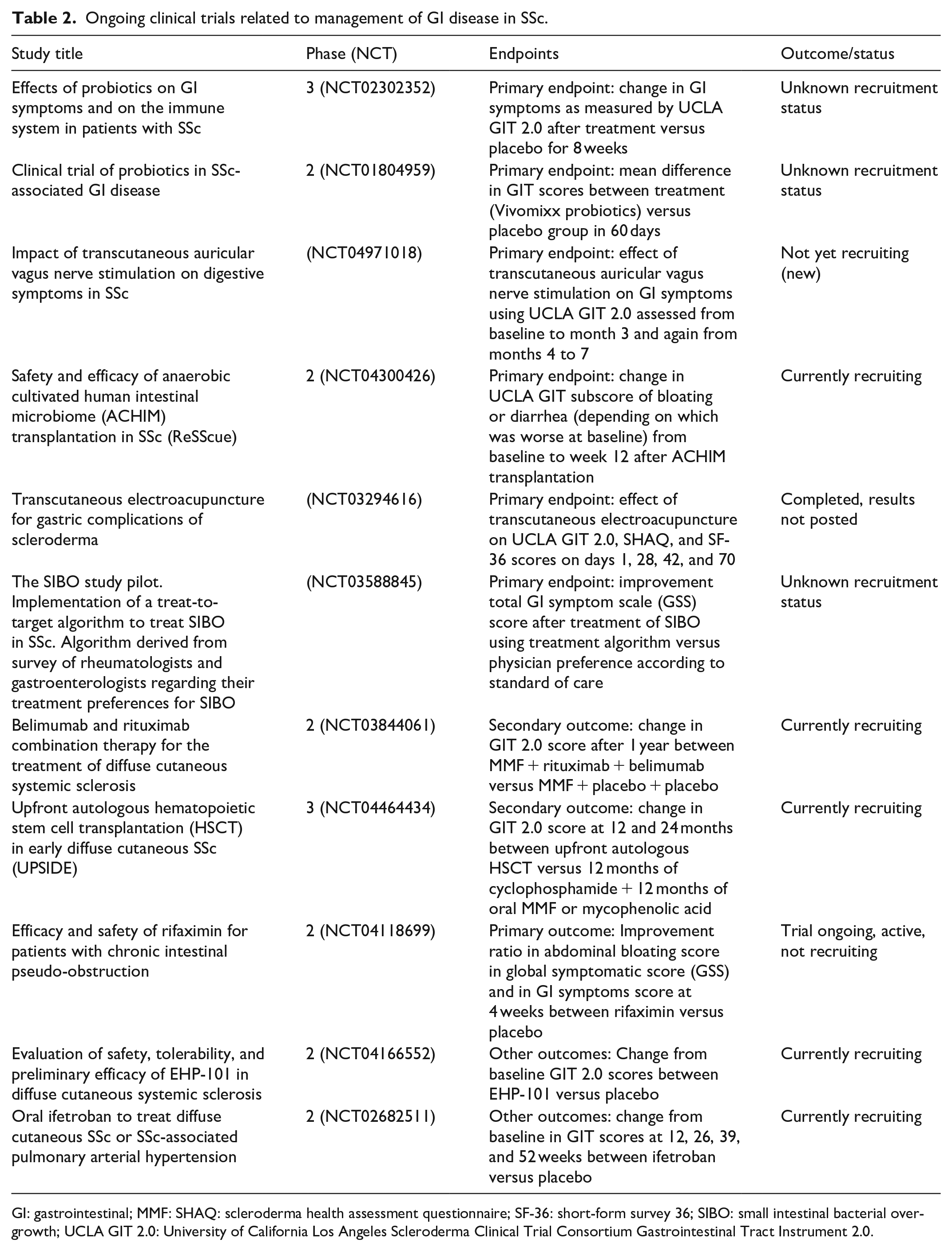
GI: gastrointestinal; MMF: SHAQ: scleroderma health assessment questionnaire; SF-36: short-form survey 36; SIBO: small intestinal bacterial overgrowth; UCLA GIT 2.0: University of California Los Angeles Scleroderma Clinical Trial Consortium Gastrointestinal Tract Instrument 2.0.
Probiotics are also being explored in the management of SSc-related GI symptoms. A recent open-label pilot clinical trial sought to evaluate the efficacy and safety of Saccharomyces boulardii for the treatment of GI symptoms and SIBO in SSc. 74 S. boulardii was selected because it is resistant to antibiotics and it can be given with antibiotics used to treat SIBO. In this study, 40 patients with SSc were randomized to metronidazole treatment only (M), S. boulardii only (SB), or M and SB. Patients were followed for 2 months. The primary outcome was the effect of the treatment on GI symptoms evaluated by the National Institute of Health Patient-Reported Outcomes Measurement Information Systems (NIH PROMIS) and hydrogen breath test results. After 2 months of treatment, 55% of patients receiving M + SB therapy were eradicated of SIBO compared to 33% of patients receiving SB only, and 25% of patients receiving M only. Patients in the M + SB and SB groups also had decreased diarrhea, abdominal pain, bloating, gas, and flatulence. These symptoms were unchanged in those receiving M only. These data suggest that combination therapy or monotherapy with SB can improve SSc GI symptoms. Although validation studies are needed, we suggest that, in patients with refractory GI symptoms, a short-term, empiric trial of S. boulardii can be considered.
Key points
FMT with commercially available anaerobic cultivated human intestinal microbiota improved symptoms of diarrhea, distension/bloating, and/or fecal incontinence in a small study of SSc patients, and larger studies are underway.
Combination of S. boulardii (SB) and metronidazole treatment was 22% more effective in eradicating SIBO that SB treatment alone and 30% more effective that metronidazole treatment alone.
Colon
Colonic dysmotility affects up to 50% of patients with SSc. 75 Patients often present with constipation, pain, distension, and some patients experience severe complications, such as recurrent pseudo-obstruction. 4 Factors associated with increased risk of SSc-related pseudo-obstruction include older age, male sex, diffuse cutaneous SSc, myopathy, and opioid use. 4 Interestingly, anti-RNA polymerase-3 antibodies are negatively associated with risk of pseudo-obstruction in SSc. For patients with evidence of slow colonic transit, pro-motility agents, such as prucalopride, neostigmine, and pyridostigmine may improve symptoms of constipation.19,76–78 Congruent with the mechanism of pro-motility agents, patients with less smooth muscle atrophy and less severe GI symptoms may respond better.11,67,68,77 Therefore, prompt diagnosis and intervention with pro-motility agents is important in managing symptoms. 76
The PROGASS study is an open-label, cross-over study that evaluated the efficacy of prucalopride in SSc patients with mild to moderately severe GI symptoms. 49 In this study, 40 SSc patients who met Rome III criteria for functional constipation with self-reported mild to moderately severe constipation (score of 1–3 based on a 5-point Likert-type scale) were randomized 1:1 to prucalopride 2 mg daily or no treatment for 1 month and vice versa with a 2-week washout period. Patient-reported symptoms using the UCLA GIT 2.0 were collected before and after each sequence. Lactulose breath tests were also performed in a subgroup of patients to determine oro-cecal transit time (OCTT). Among the 29 patients who completed the study, prucalopride was significantly associated with more intestinal evacuations (p < 0.001), improvement of the UCLA GIT 2.0 constipation (p < 0.001), reflux (p < 0.005), and bloating scores (p = 0.01). Seventy-two percent of patients ranked prucalopride as “moderately” to “more than moderately” effective. OCTT was also significantly reduced when taking prucalopride (p = 0.035). This suggests that prucalopride may improve constipation, reflux, and bloating in SSc patients with mild to severe symptoms.
Linaclotide is a pro-secretory and pro-motility agent that has been studied as a treatment for constipation in several diseases, however, it has not been well-studied in SSc.33,79,80 A recent, small, retrospective case series included 31 patients and sought to report clinical experience with linaclotide in treating refractory symptoms of constipation in SSc. 34 In this series, 28 (90.3%) patients reported a clinical response to linaclotide. Eighteen patients (18/31) were taking a low dose of linaclotide (⩽ 145 mcg/day) and 17/18 reported a response; high-dose linaclotide (> 145 mcg/day) was effective in 11 out of the 13 patients on high-dose therapy. This suggests that linaclotide may also be effective in treating refractory constipation in SSc, though randomized, placebo-controlled studies are needed in this population. In another retrospective case series, 31 SSc patients with refractory GI symptoms were on pyridostigmine for at least 4 weeks, and 51.6% of patients reported symptomatic improvement. 41 Fifteen out of 31 patients experienced adverse effects, with diarrhea being the most common adverse effect, suggesting it may be more useful for patients with constipation. In a series of observational studies, the use of intravenous immunoglobulin (IVIG) as an adjunct in SSc treatment showed significant improvement in GIT 2.0 scores from baseline (p = 0.002), suggesting that IVIG may also provide therapeutic relief for GI dysmotility in SSc. 81 The rationale behind this therapy pertains to the presence of anti-muscarinic3 receptor antibodies which are reported in a subset of patients with SSc with rapidly progressive SSc GI disease and pseudo-obstruction. 82 These antibodies negatively impact GI motility and their effects may be reversible with human immunoglobulin. Nevertheless, evidence from these smaller studies is limited and future studies evaluating these therapies in patients with SSc and defining the subpopulation of patients that would benefit most are warranted.
Key points
Prucalopride is effective in improving UCLA GIT 2.0 parameters of constipation, reflux, and bloating. Prucalopride treatment also significantly reduced OCTT, as measured by lactulose breath test.
Small studies suggest that IVIG, linaclotide, and pyridostigmine may improve GI symptoms, especially constipation, in patients with SSc.
Anorectum
The anorectum is the second most commonly affected region of the GI tract in SSc. 75 Fecal incontinence in particular affects up to 50% of patients and is associated with a decreased quality of life.83,84 SSc-related anorectal dysfunction is attributed to neuropathy resulting in decreased recto-anal inhibitory reflex (RAIR) as well as thinning and atrophy of the internal anal sphincter. 85 Furthermore, chronic constipation and straining can lead to rectal prolapse, further worsening fecal incontinence. 75 When pelvic floor dysfunction is present, treatment for SSc-related anorectal dysfunction usually involves pelvic physical therapy and biofeedback. 86 Sacral nerve stimulation (SNS) has been recommended by the American College of Gastroenterology (ACG) for treatment of fecal incontinence in those who do not respond to conservative therapy. 87 Prior case studies exploring the efficacy of SNS in patients with SSc are controversial, with some studies demonstrating a benefit, while others do not.88,89 More recently, in a study comparing percutaneous tibial nerve stimulation (PTNS) and SNS in non-SSc patients, the investigators found no significant difference between fecal incontinence and quality of life scores, suggesting that both approaches are equally efficacious. 90
Key points
A recent study suggests that both PTNS and SNS are equally effective in treating fecal incontinence, potentially offering new alternatives for treating patients with refractory symptoms.
In the shadows of SSc GI disease management: identifying key points that need illumination
Our understanding of SSc GI disease has grown in the past two decades with new discoveries related to pathogenic autoantibodies, studies focused on GI dysbiosis, and advancements in technologies to better diagnose and manage GI dysmotility. Therefore, the optimal approach to managing GI symptoms and complications in SSc is continuously evolving. Here, we will discuss several key questions related to SSc GI disease and highlight important areas of focus for discovery.
Should we screen for and treat asymptomatic GERD in patients with SSc?
A key clinical question that SSc physicians often face is whether SSc patients should be screened and treated for asymptomatic GERD. Although this is not standard of care, there is evidence suggesting that it may be worth considering. Forty-eight to 80% of patient-reported symptoms are related to GERD symptoms, including classic (e.g. heartburn, regurgitation, or dysphagia) and atypical symptoms (e.g. hoarseness, chronic cough, or asthma-like symptoms). 91 Studies have shown that chronic uncontrolled GERD is associated with restrictive lung disease, and it is hypothesized that this is due to chronic microaspiration of gastric juices into the lung.13–15 However, PPI therapy is not without side effects, and chronic PPI use has been associated with infections (pneumonias and Clostridium difficile), osteoporosis, nutritional deficiencies, and SIBO.17,18 Determining whether screening and treating asymptomatic patients affects long term outcomes in the lung and in the gut will need to be a focus of future research.
What is the optimal standardized treatment approach for SIBO in SSc?
Treatment of SIBO in SSc has remained complex as patients are susceptible to recurrent episodes and the etiology is thought to be complex, including various types of bacterial overgrowth, GI dysmotility, and chronic PPI use. 92 Currently, rifaximin, an antibiotic that is not systemically absorbed, is often trialed initially. 93 However, there are no set guidelines on the use and timing of prokinetic agents, such as octreotide and pyridostigmine, or the application of different systemic antibiotics in the treatment of SIBO in SSc. Further studies exploring the use and escalation of these therapies individually or in combination can provide clinicians with a framework for treating recurrent SIBO in SSc.
What is the role of immunomodulation in the management and prevention of SSc GI dysfunction?
The role of immunomodulation in the management and/or prevention of progressive GI dysmotility is a controversial topic that is still being explored. There is increasing evidence suggesting that anti-muscarinic antibodies negatively impact GI motility in a subset of patients with SSc.94,95 It is hypothesized that these antibodies contribute to GI smooth muscle atrophy, which is a common finding throughout the SSc gut.2,96,97 However, screening tests for these antibodies are not clinically available, which makes it challenging to identify the subgroup of affected patients. Furthermore, there are no objective biomarkers that represent ongoing immune mediated disease activity in the gut, limiting our ability to distinguish disease activity from damage, and identify patients whose GI dysfunction may benefit from immunomodulation. This also poses a challenge in identifying patients for SSc-related GI clinical trials and in accurately assessing the benefits of immunosuppression in SSc-related GI dysfunction. Several small case series have suggested that immunomodulators, such as IVIG may improve SSc-related GI dysfunction, but treatment costs are high and the optimal patient population that would benefit from this intervention is not well defined. A phase 2 double-blind randomized-controlled trial (RCT) was previously planned to assess the effects of IVIG in early SSc (NCT04138485) had included the UCLA GIT 2.0 to assess the effect of IVIG on GI symptoms; however, the study has since been withdrawn.
Does the early application of pro-motility agents prevent progressive GI dysmotility in high-risk GI subgroups?
Another area of controversy is whether the early application of pro-motility agents or transcutaneous neuromodulation can improve GI outcomes in those that are high risk for severe and progressive GI dysmotility. The rationale for this proposed treatment strategy is built on the idea that smooth muscle atrophy, a common finding in the SSc, is associated with severe GI transit delays; therefore, interventions that stimulate GI muscle contractions may prevent progressive smooth muscle atrophy in those at risk for poor outcomes.85,98,99 Questions that need to be addressed are: (1) defining the patient subset that would benefit; (2) determining the overall risks and benefits of early intervention with pro-motility agents or neuromodulation; and (3) determining the optimal time of initiation and optimal duration of therapy.
What is the relationship between GI dysmotility and GI dysbiosis in SSc?
There is also growing evidence that SSc patients have GI dysbiosis.69,70 Currently, it is unknown whether there is a biologically important interaction between GI dysmotility and dysbiosis in SSc. Furthermore, the relative contribution(s) of dysbiosis to each patient’s clinical symptoms is also unclear. Further research delineating these relationships will be important in the development of novel, targeted therapeutic strategies.
What is the role of diet and nutrition in the management of SSc GI disease?
As discussed above, there is burgeoning interest in the role of GI dysbiosis in the pathogenesis of SSc GI disease. It is reasonable to consider that diet also plays a role in GI symptoms and in determining the composition of the GI microbiota in SSc. A systematic review recently explored the role of diet and nutrition in SSc GI disease, 16 and although the authors found improvement in patient-reported symptoms with a low FODMAP diet and probiotic use, the studies included were small and the evidence remains weak. Interesting questions to explore include what diets show a benefit in the management of SSc GI symptoms, and which subset of patients (either based on reported symptoms or clinical signs) would most likely benefit will be important in future studies.
Conclusion
As our understanding of SSc-related GI disease continues to grow and evolve, novel therapeutic strategies have been developed to manage GI complications. Despite these new advances, many gaps remain to be explored, such as the determination of biomarkers of GI disease activity and the need for targeted therapies. As we continue to uncover the answers in the field of SSc-related GI disease, we envision the use of targeted immunomodulatory therapies, prokinetic agents, electrostimulation, and agents that positively influence the GI microbiota (e.g. fecal transplant, probiotics) to be utilized individually or in combination in the strategic management of SSc-related GI complications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH/NIAMS K23 AR071473 to ZM; Rheumatology Research Foundation to ZM; K24 AR063120-06A1 to DK
