Abstract
Cyclophosphamide has been the cornerstone of treatment of systemic sclerosis for a long time and is the first-choice therapy for treating systemic sclerosis–associated interstitial lung disease according to the European League Against Rheumatism recommendations on treatment of systemic sclerosis. However, new therapeutic options are emerging, and treatment with cyclophosphamide is hampered by its toxicity and restricted possible treatment duration. This review has a focus on the evidence of efficacy of cyclophosphamide in different aspects of systemic sclerosis and its organ involvements, reviews its toxicity, and will answer the question whether there is still a role for cyclophosphamide in the treatment of systemic sclerosis, taking the evidence and current therapeutic options into account.
Introduction
Treating patients with systemic sclerosis (SSc) is challenging due to the heterogeneity and clinical complexity of this disease. SSc is a generalized immune modulated disease characterized by inflammation, micro-vasculopathy, and fibrosis, affecting skin and internal organs and it causes significant morbidity and mortality. Treatment options are limited; immune-modulating agents are the cornerstone of treatment in SSc; however, there is a high unmet need for disease-modifying therapies resulting in high efficacy and manageable toxicity. In the last years, several trials investigating different immune-modulating therapies for early diffuse cutaneous SSc (dcSSc) and interstitial lung disease associated to SSc (SSc-ILD) have been published with variable results. Some of those trials have increased the therapeutic possibilities for early disease and/or organ involvement.
Therapeutic trials in SSc are hampered by its rarity and the lack of disease activity measures and validated outcome measures, beside the modified Rodnan skin score (mRSS). Since the 1990s, the mRSS was the primary outcome measure in dcSSc. 1 In the studies focused on lung involvement, the primary endpoint is the forced vital capacity as percentage predicted (FVC% pred.) in line with the primary endpoint of other ILDs. However, the complexity and heterogeneity of SSc necessitate an outcome measure that captures multiple organ involvement as well as patient-reported outcomes. Therefore, more recent trials chose the composite response index in dcSSc (CRISS) as the primary endpoint for clinical trials. 2
Cyclophosphamide is an alkylating agent which can be administered both orally and intravenously. It acts as a cytotoxic immunosuppressive agent through modulation of lymphocytic function that leads to depression of the inflammatory response and less fibrosis. 3 The last version of the European League Against Rheumatism (EULAR) treatment recommendations for SSc, published in 2017, describes treatment with cyclophosphamide as key for skin involvement and SSc-ILD. 4 At the time the last EULAR treatment recommendations were developed, the available evidence for the use of cyclophosphamide was strong. But with new therapeutic options emerging, one could debate whether cyclophosphamide is still the cornerstone of treatment in SSc. This review will discuss the use and evidence of cyclophosphamide in SSc, in different organ involvements as well as its safety issues.
Cyclophosphamide and ILD
ILD is the leading cause of death in patients with SSc; this holds true for both limited cutaneous systemic sclerosis (lcSSc) and dcSSc patients.5,6 The incidence of ILD in SSc varies from 25% to 90% depending on the method used to identify ILD. 7 Current expert opinion on the detection of SSc-ILD consist of a combination of pulmonary function tests, often showing a decrease in FVC% pred., and the presence of fibrosis on high-resolution computed tomography (HRCT) were the extend of fibrosis predicts progression of SSc-ILD. 8 Decline in lung function is a predictor of mortality in patients with SSc-ILD. 9 A clinical relevant decline in lung function in connective tissue diseases was found to be ⩾10% or ⩾5 to <10% relative decline in FVC% pred. and ⩾15% relative decline in diffusion lung capacity of carbon monoxide (DLCO)% pred. 10 Of note is that the clinical course of SSc-ILD can be variable and difficult to predict and longer follow-up with pulmonary function tests after 1 year are often needed to establish clinically relevant disease progression. 11 Furthermore, the role of repeated HRCT scans including the indication, time interval, and value of progression on the images has to be determined yet. 11
Currently, there is no established treatment algorithm for SSc-ILD, but patients with advanced or progressive disease receive immunosuppression. 12 Among these immunosuppressive drugs is cyclophosphamide, which was the first to show effect on SSc-ILD.
Several uncontrolled studies performed in the 1990s and the 2000s revealed a positive effect on pulmonary function in patients with SSc-ILD receiving cyclophosphamide.13–16 The first randomized controlled trial with cyclophosphamide for SSc-ILD, the Scleroderma Lung Study I (SLS I), using oral cyclophosphamide for 1 year showed a small positive effect on pulmonary function and quality of life after 12 months compared to placebo but the effects waned after monitoring patients for another year off therapy.17,18 In the SLS I, 60% of the included patients had diffuse subtype. The second randomized controlled trial, the UK lung study, showed a trend toward improvement of pulmonary function with six intravenous pulses of cyclophosphamide combined with low-dose prednisolone followed by azathioprine compared to placebo after 12 months. 19 The third study was the Scleroderma Lung Study II (SLS II), comparing oral cyclophosphamide for 12 months followed by placebo with mycophenolate mofetil (MMF) for 24 months. Forty-two percent of the included patients in the SLS II study had dcSSc. Both cyclophosphamide and MMF proven to be equally effective in SSc-ILD after 24 months, but MMF was better tolerated. 20 The above-mentioned trials used different routes of administration of cyclophosphamide (oral and intravenous), different dosages, and different treatment durations. The results of the randomized controlled trials are summarized in Table 1. To further understand the effects of cyclophosphamide on SSc-ILD, several additional studies were performed. The efficacy of treatment with 1 year of cyclophosphamide of both SLS I and SLS II was found to be equal, but this effect was not sustained after discontinuation, underscoring the necessity of prolonged treatment of SSc-ILD. 21 This prolonged treatment was applied in an observational study of patients treated with 12 monthly intravenous pulses with cyclophosphamide followed by maintenance therapy consisting of azathioprine or MMF. In this trial, a stabilization of pulmonary function in patients with SSc-ILD over a 3-year period was found. 22
Summary of the randomized controlled trials with cyclophosphamide for systemic sclerosis–associated interstitial lung disease.
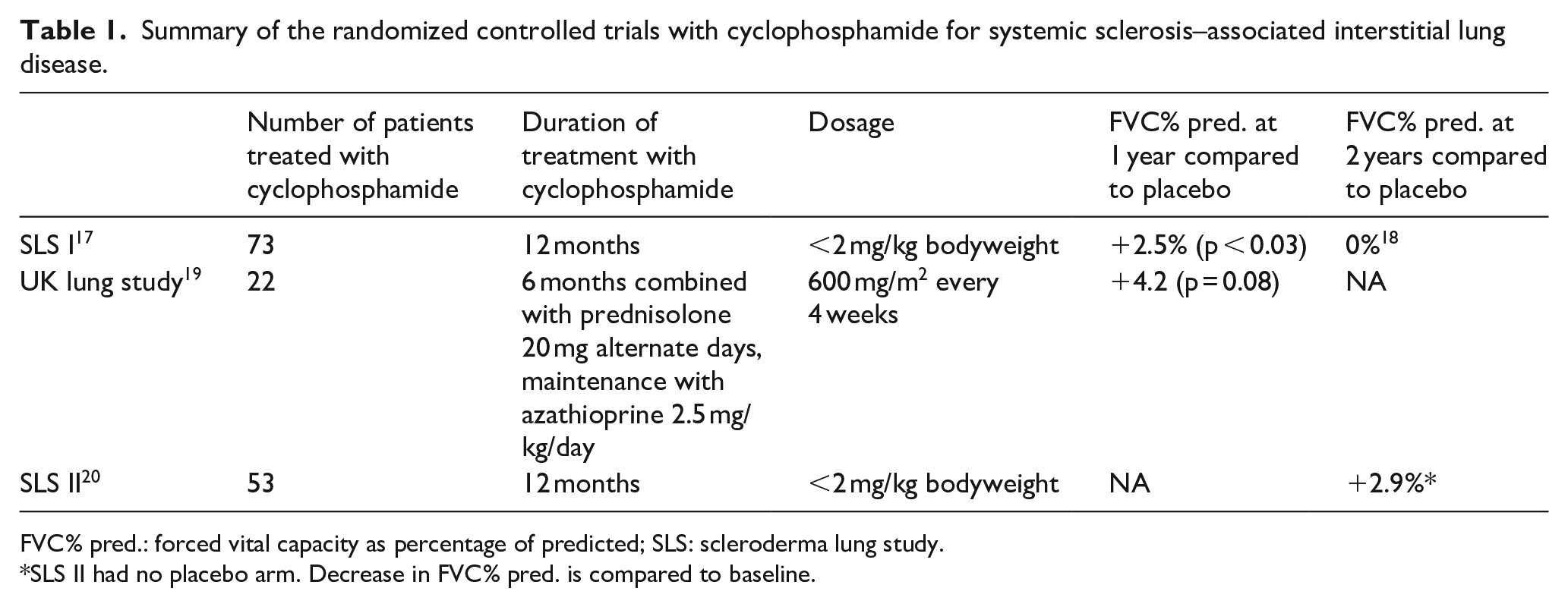
FVC% pred.: forced vital capacity as percentage of predicted; SLS: scleroderma lung study.
SLS II had no placebo arm. Decrease in FVC% pred. is compared to baseline.
Cyclophosphamide and skin involvement
The severity and distribution of skin thickening in SSc can be quantified using the mRSS. 1 It has been shown to differentiate potentially disease-modifying drugs from placebo in randomized controlled trials. However, there is no international consensus on mRSS response criteria. Most used in literature are an improvement of ⩾5 units and ⩾25% from baseline.23,24
In the above-mentioned early, observational trials using cyclophosphamide for SSc-ILD, an improvement of skin involvement was found in patients with dcSSc. 16 The presumed efficacy on both lung and skin involvement resulted in cyclophosphamide as control treatment in the trials using hematopoietic stem cell transplantation in patients with severe progressive dcSSc.25,26 In these trials, patients randomized to the control treatment with 12 monthly pulses of intravenous cyclophosphamide were later found to have experienced a moderate decrease in mRSS as well. The two SLSs, using oral cyclophosphamide, revealed a statistically significant decrease in mRSS from 21.1 (±9.7) to −5.4 (±0.8) at 12 and −7.2 (±0.8) at 24 months. 27 With the efficacy of cyclophosphamide on skin involvement established in these trials, a big European observational trial was fostered by the EULAR.
The European Scleroderma Observational Study (ESOS) aimed to compare the effectiveness of four treatment options in the early management of patients with dcSSc. 28 Patients with dcSSc, a disease duration from onset of skin thickening of <3 years and no or <4 months of immunosuppressive therapy, were included in 50 centers. The treatment protocol consisted of no immunosuppression, methotrexate, MMF, or cyclophosphamide, and the choice of treatment was made by treating physician and patient. In this study, 326 patients were included, and 87 patients were treated with cyclophosphamide, either as intravenous monthly pulses with a minimum dose of 500 mg/m2 for 6–12 months or oral with a dosage of 1–2 mg/kg/day with a recommended duration of 12 months. Patients treated with cyclophosphamide received maintenance treatment with methotrexate, MMF, or azathioprine after the 6–12 months of cyclophosphamide. Based on a weighted regression model to correct for confounding, there were statistically significant reductions in mRSS in all four treatment groups, with no differences between groups. In the cyclophosphamide group, the mRSS reduced with 3.3 point after 12 months compared to −2.2 points in the group not treated with immunosuppression. 28 The results of treatment with cyclophosphamide on skin involvement are summarized in Table 2.
Summary of the controlled trials with cyclophosphamide for systemic sclerosis on skin involvement (no reported mean mRSS results of the SCOT trial). 26 .

mRSS: modified Rodnan skin score; SD: standard deviation; SLS: scleroderma lung study; ESOS: European scleroderma observational study; CI: confidence interval; SCOT: short course oncology therapy; ASTIS: autologous stem cell transplantation international scleroderma.
The efficacy of intravenous cyclophosphamide in daily clinical practice was described in a mono-center retrospective analysis in 99 dcSSc patients. The median disease duration was 3.8 months from first non-Raynaud symptom. This study showed a mean change in mRSS of −3.9 at 12 months, −5.2 at 24 months, and −6.6 at 36 months. Multivariate analyses showed that the only significant predictor of non-response at month 12 was non-response at 6 months, suggesting that a decision to continue with this therapy or change to an alternative therapy can be made at 6 months. 29
Cyclophosphamide and the heart
Cardiac involvement in SSc is a well-recognized complication with an estimated prevalence of 25%–35%. 30 Cardiac manifestations can affect all structures of the heart and may result in pericardial effusion, arrhythmias, conduction system defects, valvular impairment and ischemia. Myocardial fibrosis is the hallmark for SSc-associated cardiac disease. 31 The updated treatment recommendations for SSc do not provide recommendations for cardiac disease. 4 Several cardiac manifestations of SSc seem to be associated to micro-vascular pathology; however, patients with active myocarditis are recommended to be treated with aggressive immune-modulating therapy. 30 The evidence of this recommendation is poor, as only case series have been reported. The most commonly used treatment of myocarditis consists of a combination of high dose glucocorticoids and cyclophosphamide.32,33 On contrary, of note is that treatment with high dosage cyclophosphamide during the procedure of hematopoietic stem cell transplantation may rarely result in acute cardiac failure, an exceptional cause of treatment-related mortality. 34
Cyclophosphamide and other manifestations of SSc
Anecdotally reports are available on the effect of cyclophosphamide on other manifestations of SSc, mostly on its effects on complications of the intestinal tract. One of the early observational trials with cyclophosphamide in SSc showed no effect on oesophageal function, but two case reports describe a possible positive effect on gastric antral vascular ectasia associated to SSc.16,35,36
Cyclophosphamide and survival benefit
Treatment with cyclophosphamide has shown survival benefit in patients with SSc-ILD in a retrospective study; however, a survival benefit of 1-year treatment with oral cyclophosphamide was not found in the SLS I and SLS II.37,38 In the ESOS study, comparing different treatment protocols in real life, no statistically significant survival benefit was found for any one of the treatment arms. The weighted regression model, to correct for confounding, revealed a 12- and 24-month survival of 91.7% and 90.1% in the patients treated with cyclophosphamide compared to 88.6% and 84.0% in the no immunosuppression group. 28 Also the hematopoietic stem cell transplantation trials provide no evidence for a survival benefit as no placebo arm was used in these trials.25,26
Side effects of cyclophosphamide
Treatment with cyclophosphamide is associated with several site effects. Well known is the risk of pancytopenia resulting in septic and hemorrhagic complications. Further potential toxicities include nausea, vomiting, diarrhea, alopecia, malaise, and hemorrhagic cystitis (Figure 1). High cumulative doses of oral cyclophosphamide have been associated with a higher long-term risk of bladder- and skin cancer and both male and female infertility, which may be irreversible. 4 Especially, the risk of infertility should be regarded with care in fertile patients and the opportunity to preserve fertility should be discussed.
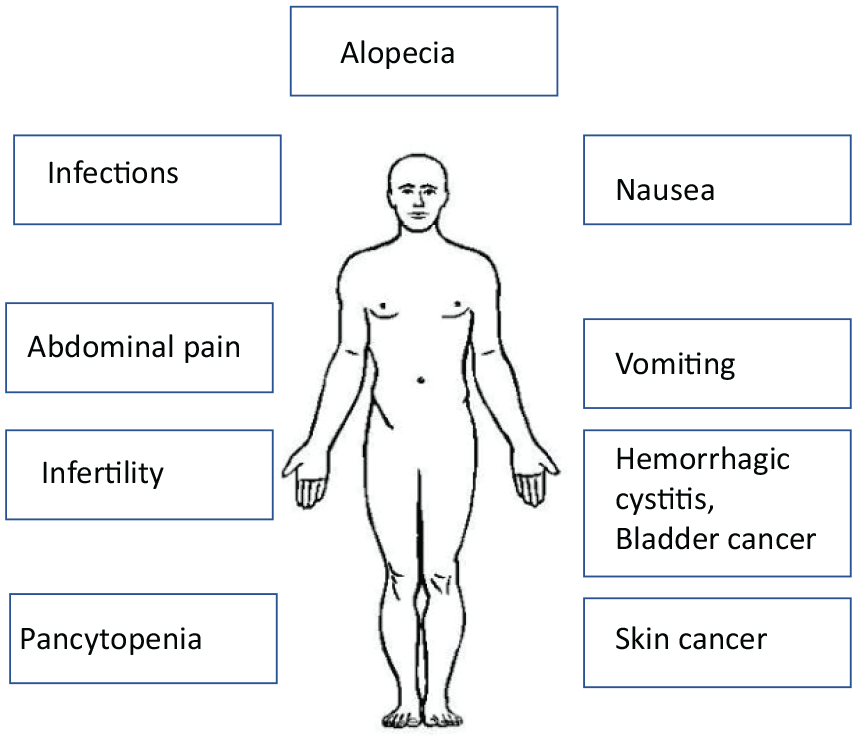
Schematic overview of possible side effects of cyclophosphamide treatment.
Cyclophosphamide can be applied either orally or intravenously. The oral route delivers a higher cumulative dose, and therefore could result in a higher long-term risk as mentioned above. The side effects of intravenous cyclophosphamide pulse therapy in auto-immune diseases have been reported in several case series. In a retrospective study in 65 patients treated with intravenous pulses for various autoimmune diseases, 60% experienced side effects, with no association between the frequency of side effects on treatment duration, age, diagnosis, or dose reduction. 39 Of note is that in the SLS II, 44% of the patients randomized in the cyclophosphamide arm discontinued prematurely due to side effects. 20 In a retrospective trial in 75 patients treated with intravenous cyclophosphamide for SSc-ILD, adverse events occurred in 23% of the cases, consisting of pneumonia, anemia, leucopenia, hematuria, renal insufficiency, and allergic exanthema. 22 The much lower rate of adverse events when applying intravenous cyclophosphamide suggests that, among other evidence, intravenous cyclophosphamide might be safer and better tolerated.
Reviewing the evidence and drawbacks of cyclophosphamide
Cyclophosphamide has been the first-line therapy in SSc for a long time. Cyclophosphamide is one of the few immunomodulating agents to have proven effect in a series of randomized controlled trials and can prevent disease progression and improve organ-related outcomes.21,28 However, treatment with cyclophosphamide is hampered by several problems.
First, despite this efficacy, a survival benefit of cyclophosphamide for SSc-ILD has not been established to date. 38 Second, because of the high toxicity of cyclophosphamide over MMF and the comparable efficacy on skin and lung involvement, MMF is often the first choice of treatment based on expert opinion. 40 In view of evidence suggesting that intravenous cyclophosphamide might be safer and better tolerated, this route of administration could be preferred, but a randomized controlled trial comparing intravenous cyclophosphamide to MMF is lacking. Third, given its toxicity, the cyclophosphamide treatment period is restricted and maintenance therapy with another immune-modulating treatment is often followed. Future studies are needed to determine the optimal duration of immunomodulation. Fourth, it could be that treatment of SSc should be initiated much earlier than is currently the case, for example, in patients with very early SSc and signs of adverse prognosis. Evidence for treatment of these patients is expected soon from the very early diagnosis of systemic sclerosis (VEDOSS) cohort and the hit hard and early trial.41,42
With the emergence of promising new therapies for SSc-ILD consisting of antifibrotics and the combination of antifibrotics and immune-modulating agents, the role of cyclophosphamide could decrease even more in the near future. To date, numerous trials are ongoing for SSc and SSc-ILD with immune-modulating agents, antifibrotics, and the combination of both, addressing the need of an effective treatment for SSc. Some of these trials have the CRISS as primary outcome, some pulmonary function, mRSS, inhibition of progression, or adverse events. 43 With the diversity of primary outcome of those trials, we are gaining new knowledge of this complex disease. Patients with SSc and doctors treating these patients are curiously waiting for the results.
Conclusion
As the paradigm in treating SSc shifts, treatment recommendations for SSc are quickly changing and combine both evidence and expert opinion. However, taking the big unmet need for disease-modifying therapies resulting in high efficacy and manageable toxicity for SSc into account, eligible patients should be included in randomized trials as much as possible. Cyclophosphamide has already lost its cornerstone position for treatment of SSc to MMF, because of the favorable toxicity and equal efficacy. It could be possible that, however, treatment with cyclophosphamide may be preserved as second- or third-line treatment for SSc. In my opinion, based on the evidence reviewed above, there is still a role for cyclophosphamide in the treatment of SSc.
Footnotes
Acknowledgements
The Editor/Editorial Board Member of JSRD is an author of this paper, therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Board member had no involvement in the decision-making process.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
