Abstract
Introduction:
Endovascular thrombectomy (EVT) is an effective treatment for basilar artery occlusion (BAO) stroke in select patients. While there is a growing body of literature suggesting that advanced imaging modalities such as computed tomography perfusion (CTP) and magnetic resonance (MR) may not be necessary for selecting anterior circulation large vessel occlusion stroke patients for EVT, whether advanced imaging may be superior to conventional imaging (non-contrast CT and CT angiography) in identifying good treatment candidates among BAO patients is less clear.
Patients and methods:
This was a multicenter retrospective cohort study of BAO EVT patients treated from 2013 to 2022 in the Stroke Thrombectomy and Aneurysm Registry. Patients selected for EVT by advanced imaging (CTP or MR) were matched with those selected by conventional imaging using propensity score matching (PSM) accounting for possible confounders. Primary outcome was functional independence at 90 days. Other outcomes include bedridden state or death at 90-days and symptomatic intracranial hemorrhage (sICH).
Results:
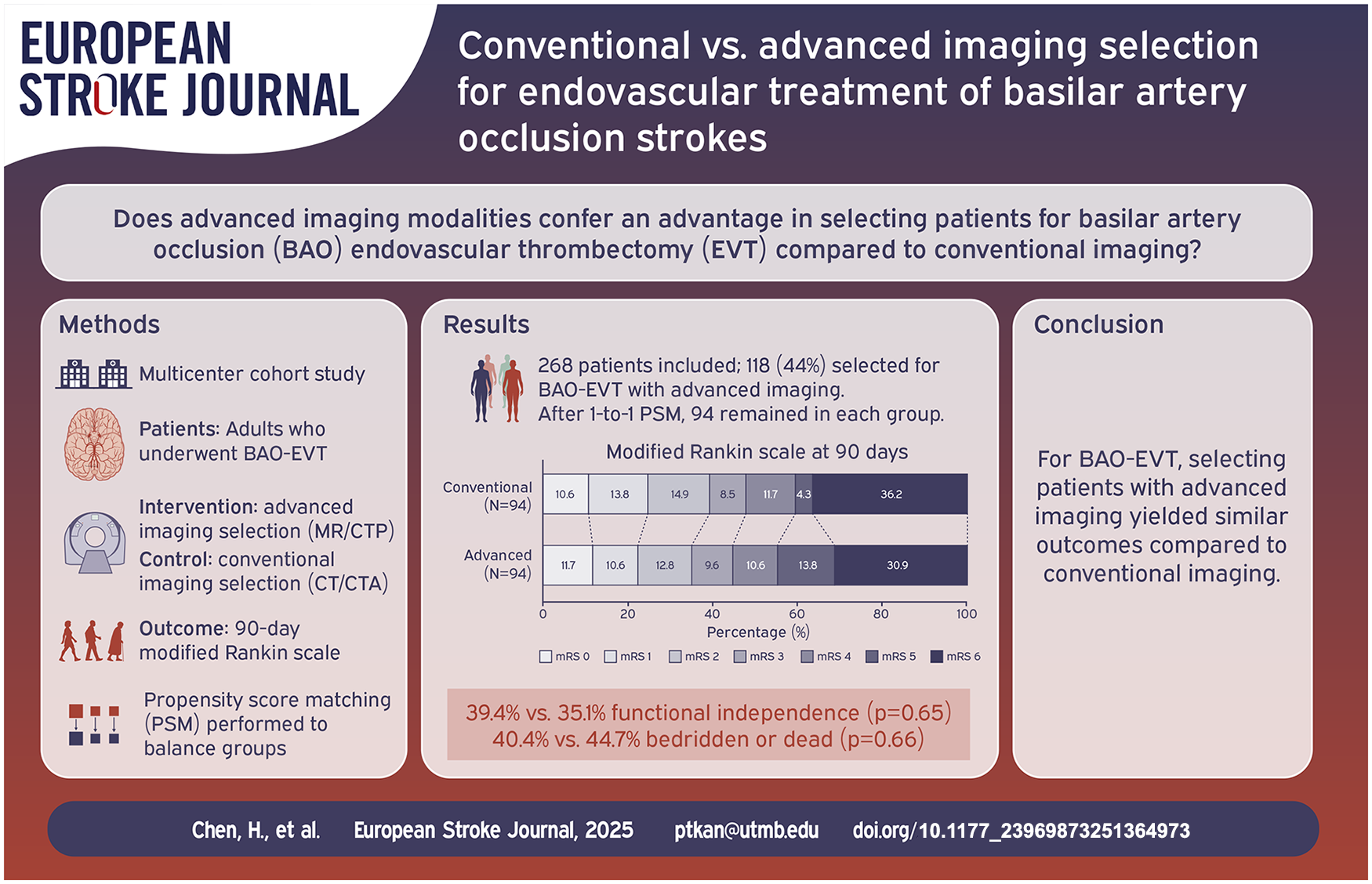
268 patients were included. 150 patients were selected for BAO EVT by conventional imaging, 86 by CTP, and 32 by MR. Patients selected by advanced imaging were significantly older than those selected by conventional imaging (median age 71 vs 64 years, p = 0.001); patient characteristics were otherwise similar between cohorts. After PSM, 90-day outcomes were similar between the two cohorts (p = 0.56), with similar rates of functional independence (39.4% vs 35.1%, p = 0.65), bedridden state or death (40.4% vs 44.7%, p = 0.66), and sICH (3.3% vs 5.7%, p = 0.49) for conventional and advanced imaging groups, respectively. Results were similar across treatment time windows (all p > 0.05).
Conclusions:
Selecting patients for basilar EVT using conventional versus advanced imaging did not result in different clinical outcomes, regardless of treatment time windows. Conventional imaging appears sufficient as a first-line tool for selecting basilar EVT patients in routine clinical practice.
Keywords
Introduction
Four landmark randomized controlled trials have investigated the efficacy and safety of endovascular thrombectomy (EVT) for acute ischemic stroke due to basilar artery occlusion (BAO).1–4 While pooled analyses demonstrated overall treatment benefit of BAO-EVT, only two of the four trials (ATTENTION and BAOCHE) met their primary efficacy endpoint. 5 The heterogeneity of basilar EVT’s treatment benefit may be due in part to differences in radiographic features of trial participants. 6 In the ATTENTION and BAOCHE trials, a minority of patients were selected for trial inclusion based on magnetic resonance (MR)2,3; however, whether these patients had different outcomes compared patients selected by conventional computed tomography (CT) and CT angiography (CTA) was not reported. To date, whether advanced imaging modalities such as MR or computed tomographic perfusion (CTP) confer an advantage over conventional CT/CTA when selecting BAO-EVT patients is overall unclear.
In this retrospective analysis of an international, multi-center database of endovascular stroke treatments, we investigate clinical outcomes of BAO-EVT patients selected by conventional imaging versus advanced imaging modalities. Given that advanced imaging modalities may not confer a significant clinical advantage in selecting anterior circulation EVT patients,7–12 we hypothesize that patient selection using conventional neuroimaging may yield similar clinical outcomes compared to advanced modalities for BAO-EVT patients.
Methods
Database characteristics
This was a retrospective cohort study of the Stroke Thrombectomy and Aneurysm Registry (STAR). 13 The registry includes centers from the U.S, Europe, South America, and Asia. A database of stroke patients who underwent EVT at 32 stroke centers participating in STAR from January 2013 to December 2022 was used for the current study. The study was approved by the institutional review board at each participating institution; patient consent was waived. The data at each institution were obtained retrospectively and collected according to a standardized protocol. Verification, de-identification, and attestation of data accuracy were performed by investigators at each contributing institution. Individual patient data from each contributing institution were pooled by investigators at STAR.
Patients and clinical variables
Adult patients who underwent EVT for BAO with available information on imaging modality used for patient selection were included. Exclusion criteria include: (1) concomitant anterior circulation vascular occlusion, (2) lack of information regarding imaging modality used for treatment selection, (3) treatment selection based on imaging modality other than CT/CTA, CTP, or MR, and (4) lack of 90-day clinical follow-up. Patients were divided into two study cohorts: Those selected for treatment by conventional imaging (CT and CTA) and those selected by advanced imaging (CTP or MR). The choice of imaging modality and processing software for patient selection was per local institutional protocols. The reasons underlying the choice of front-line imaging modality and criteria for pursuing additional second-line imaging are not recorded in the STAR database.
Patient demographic data included age, sex, medical comorbidities, and pre-stroke disability measured by modified Rankin scale 14 (mRS). Clinical characteristics included admission National Institutes of Health stroke scale (NIHSS), administration of intravenous thrombolysis, additional sites of vascular occlusion (posterior cerebral artery or vertebral artery), and symptom onset or last-known-well time to arteriotomy were also captured. Procedural data included additional endovascular procedures (angioplasty, intracranial stenting, or intra-arterial thrombolysis) and successful revascularization (modified treatment in cerebral ischemia score 15 of 2b or greater).
Study outcomes
Primary study outcome was the rate of good 90-day (±20 days) outcomes (mRS 0-2, implying functional independence). Secondary outcomes included rates of acceptable 90-day outcomes (mRS 0-3, implying ambulatory independence), poor 90-day outcomes (mRS 5 or 6, implying bedridden state or death), any intracranial hemorrhage (ICH), and symptomatic ICH (sICH, defined as presence of ICH and neurological worsening of 4 points or greater on the NIHSS). 16
Statistical analysis
Descriptive statistics were presented as median (Q1-Q3) for continuous variables or percentage for categorical variables. Missing data for pre-stroke mRS were imputed with zero (assuming no pre-existing disability), and missing data for prior intravenous thrombolysis were imputed as no (assuming no prior treatment).
Propensity score matching (PSM) was performed to balance the conventional and advanced imaging cohorts. Propensity scores were calculated using a binary logistic regression model including all captured clinical variables including sex, age, medical comorbidities, pre-stroke disability, admissions NIHSS, additional sites of vascular occlusion, intravenous thrombolysis treatment, time from stroke onset to arteriotomy (categorized into: 0–6 h, 6–12 h, 12–18 h, 18–24 h, more than 24 h, or unknown), additional endovascular procedures, and successful revascularization. Then, patients from the advanced imaging cohort were matched with patients from the conventional imaging cohort based on propensity scores with one-to-one nearest neighbor matching and a maximum allowable distance of 0.1. PSM performance was evaluated by standardized mean differences (SMD) of matched variables, where SMD less than 0.1 was deemed adequately balanced.
Primary and secondary outcomes were compared between the PSM cohorts with Fisher’s exact tests. Ordinal regression analysis was used to compare 90-day mRS outcomes between groups, and tests of parallel lines were used to confirm that the proportional odds assumption was not violated. For doubly robust analyses, additional adjustments of confounders were made for variables that remain unbalanced after PSM (SMD 0.1 or greater) as well as known predictors for post-EVT outcomes (age, admission NIHSS, pre-stroke mRS, and time from stroke onset to arteriotomy) 17 using logistic regression models assessing differences in clinical outcomes between the PSM cohorts. Subgroup analyses include patients treated in the early window (time from stroke onset to arteriotomy within 6 h), patients treated in the late window (time from stroke onset to arteriotomy beyond 6 h or unknown), patients selected by CTP (and their corresponding PSM patients who underwent conventional imaging), and patients selected by MR (and their corresponding PSM controls).
p-Value less than 0.05 was deemed statistically significant for the primary outcome. Adjustments for multiple comparisons were not performed for secondary outcomes, subgroup analyses, or sensitivity analyses due to the explorative nature of these additional comparisons. Statistical analyses were conducted using SPSS v29.0.
Results
A total of 373 adult patients were identified for inclusion. Nine patients with concomitant anterior circulation vascular occlusion, 48 without information on post-EVT revascularization, 41 without information on 90-day outcomes, and 7 selected for treatment with an imaging modality other than CT/CTA, CTP or MR were excluded. A total of 268 were included in the study, of whom 150 were selected for basilar EVT using conventional imaging, and 118 using advanced imaging (86 by CTP, and 32 by MR). The study flowchart is presented in Supplemental Figure S1.
Patient characteristics
Patient demographic, clinical and procedural characteristics are shown in Table 1. Patients selected for treatment by conventional imaging were significantly younger than those selected by advanced imaging (median age 64 vs 71 years, p < 0.001), and less likely to have been treated with angioplasty (2.7% v. 9.3%, p = 0.029). Patient characteristics were otherwise not significantly different between the two groups.
Patient characteristics.
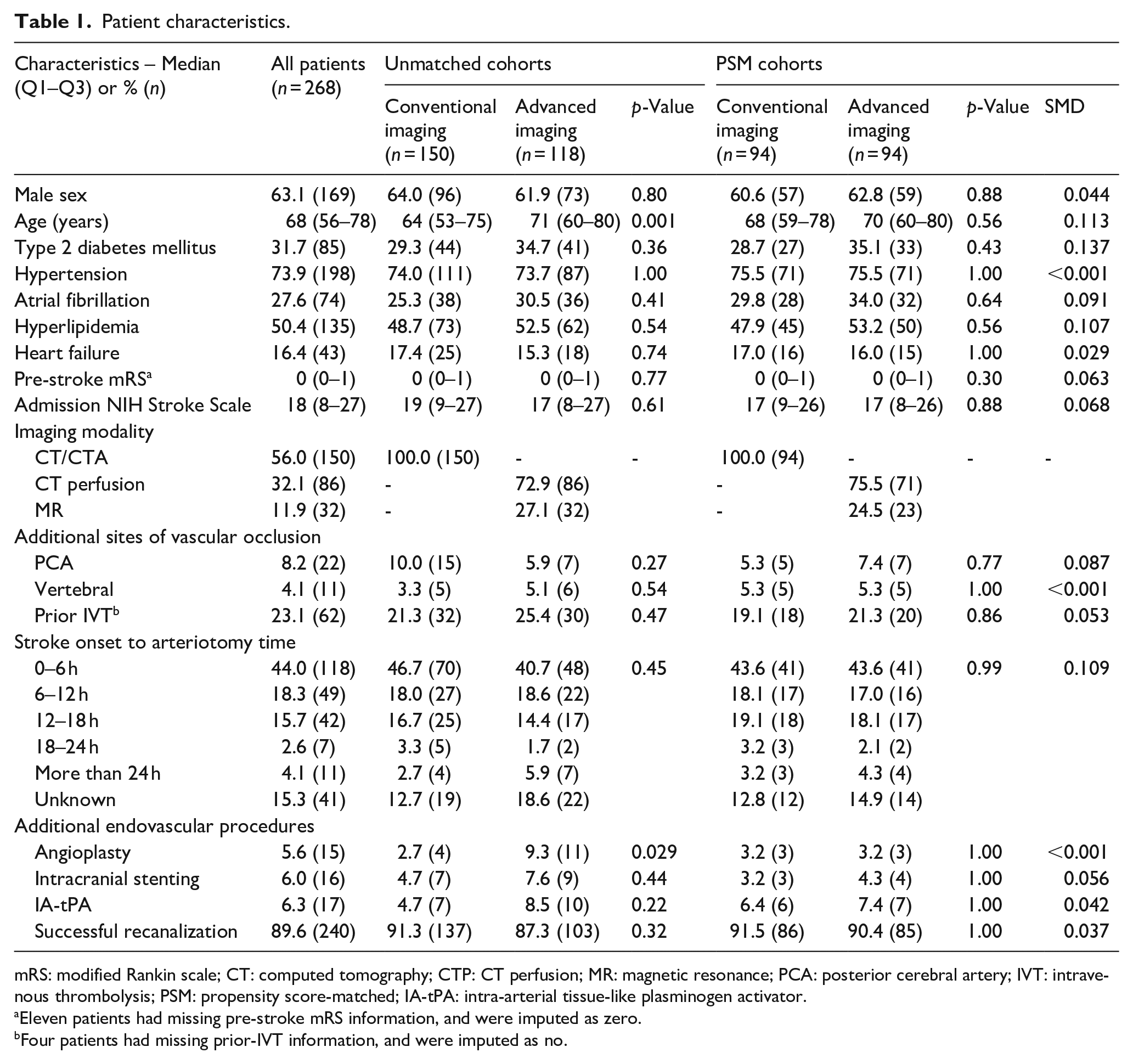
mRS: modified Rankin scale; CT: computed tomography; CTP: CT perfusion; MR: magnetic resonance; PCA: posterior cerebral artery; IVT: intravenous thrombolysis; PSM: propensity score-matched; IA-tPA: intra-arterial tissue-like plasminogen activator.
Eleven patients had missing pre-stroke mRS information, and were imputed as zero.
Four patients had missing prior-IVT information, and were imputed as no.
Patients in the two study arms were matched using propensity scores calculated with all captured clinical variables. After PSM, n = 94 patients remained in each arm, and there were no significant differences in patient characteristics. SMD measurements were acceptably low for all clinical variables except for age (SMD 0.113), type 2 diabetes mellitus (T2DM; SMD 0.137), hyperlipidemia (SMD 0.107), and time from stroke onset to arteriotomy (SMD 0.109).
Patient outcomes
Patient outcomes are shown in Table 2. Between the PSM cohorts, there were no statistically significant differences in terms of 90-day functional independence (39.4% vs 35.1%, p = 0.65), 90-day independent ambulation (47.9% vs 44.7%, p = 0.77), 90-day bedridden state or death (40.4% vs 44.7%, p = 0.66), ICH (9.7% vs 13.3%, p = 0.49), or sICH (3.3% vs 5.7%, p = 0.49; Table 2). Ninety-day outcomes measured by mRS were also not statistically different in ordinal regression analysis (common odds ratio [cOR] 0.98, 95% confidence interval [CI] 0.59 to 1.63, p = 0.95; Figure 1). After additional adjustments for age, NIHSS, pre-stroke mRS, time from stroke onset to arteriotomy, T2DM, and hyperlipidemia, advanced imaging selection was not associated with significantly different odds of functional independence (adjusted odds ratio (aOR) 0.80, [95% CI 0.40 to 1.60], p = 0.53), independent ambulation (aOR 0.84 [95% CI 0.43 to 1.63], p = 0.60), or bedridden state or death (aOR 1.22 [95% CI 0.64 to 2.32], p = 0.55).
Patient outcomes of PSM cohorts without and with additional multivariable adjustments.
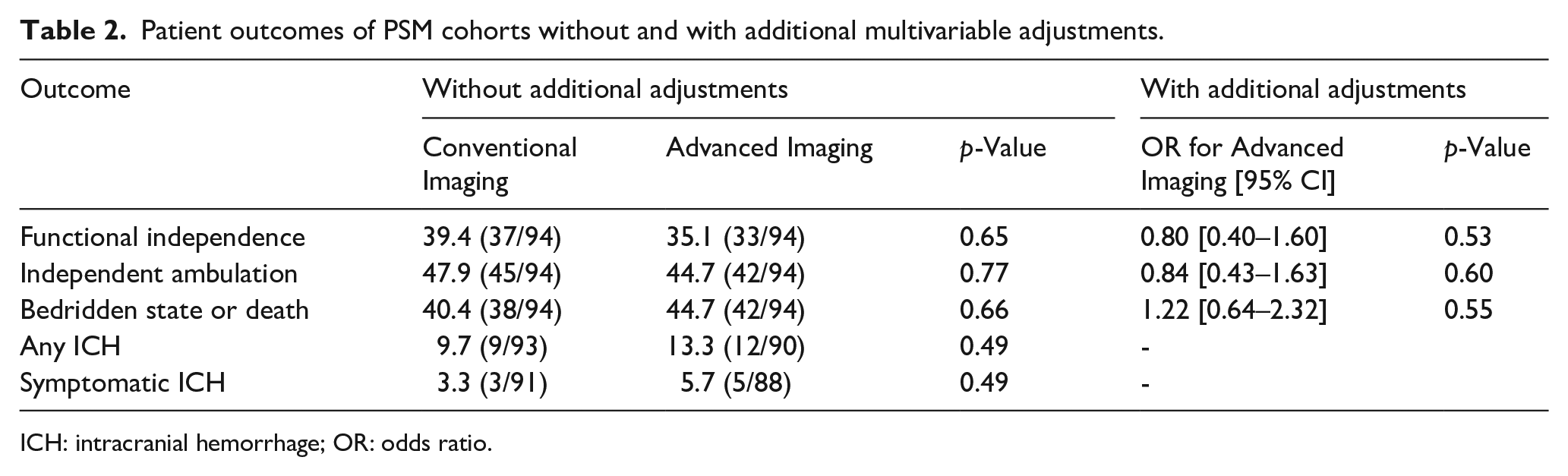
ICH: intracranial hemorrhage; OR: odds ratio.
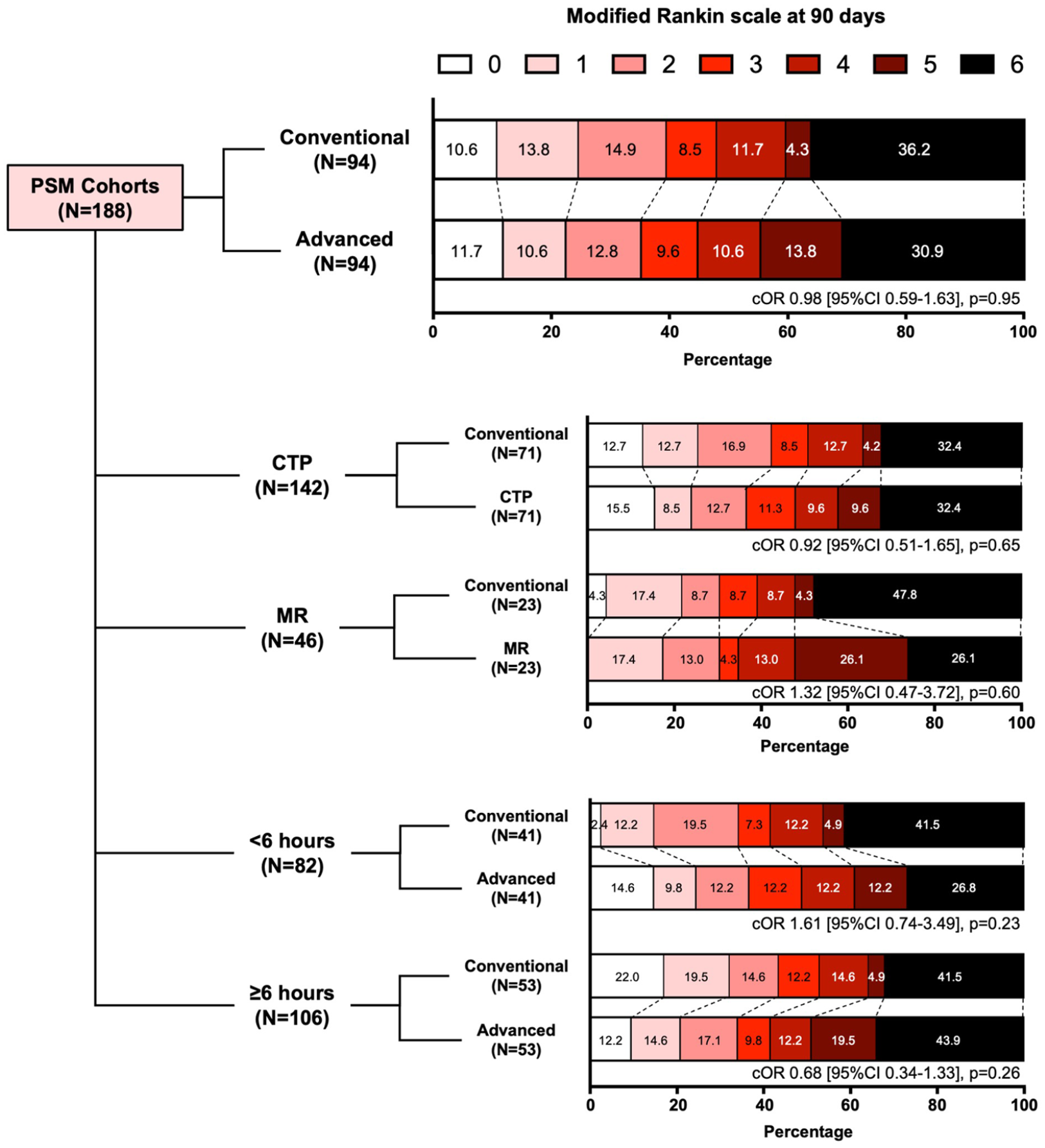
Ninety-day functional outcomes of basilar thrombectomy patients selected by conventional and advanced imaging in the propensity score-matched (PSM) cohorts and subgroups stratified by advanced imaging modality and time from stroke onset to arteriotomy. Patients who received computed tomography perfusion (CTP) or magnetic resonance (MR) imaging were compared with their PSM counterparts in the conventional imaging arm. Patients with unknown time of stroke onset were included in the ⩾6 h subgroup. Ordinal regression analyses were conducted for comparisons between groups; tests of parallel lines confirmed that proportional odds assumption was not violated. p-Values < 0.05 were deemed statistically significant.
Subgroup analyses
Subgroup analyses of CTP or MR selection are shown in Table 3. Patients selected by CTP or MR did not have significantly different 90-day clinical outcomes compared to their PSM counterparts. The imaging modality used for patient selection also did not lead to difference in patient outcomes for those treated in the early (within 6 h of stroke onset) or late (beyond 6 h or unknown time of stroke onset) time windows (Table 4). Distributions of 90-day mRS scores for subgroup analyses are presented in Figure 1, and ordinal regression analyses did not reveal any statistically significant differences in 90-day mRS across all comparisons (all p > 0.05).
Subgroup analyses of CTP and MRI selected patients.
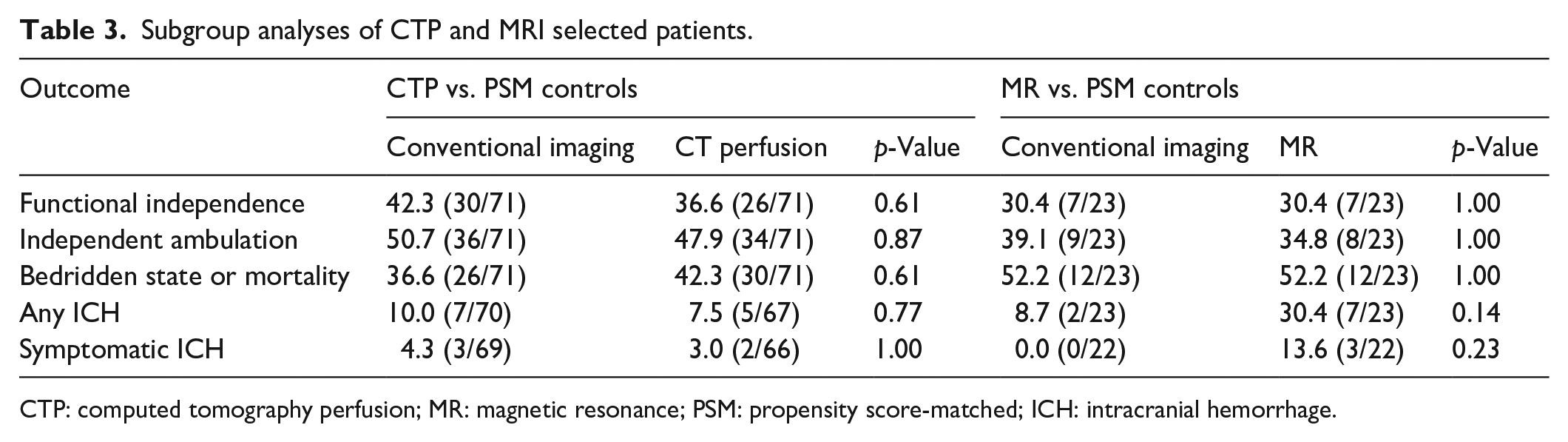
CTP: computed tomography perfusion; MR: magnetic resonance; PSM: propensity score-matched; ICH: intracranial hemorrhage.
Subgroup analyses of patients in the early (<6 h) and late (⩾6 h) time window.
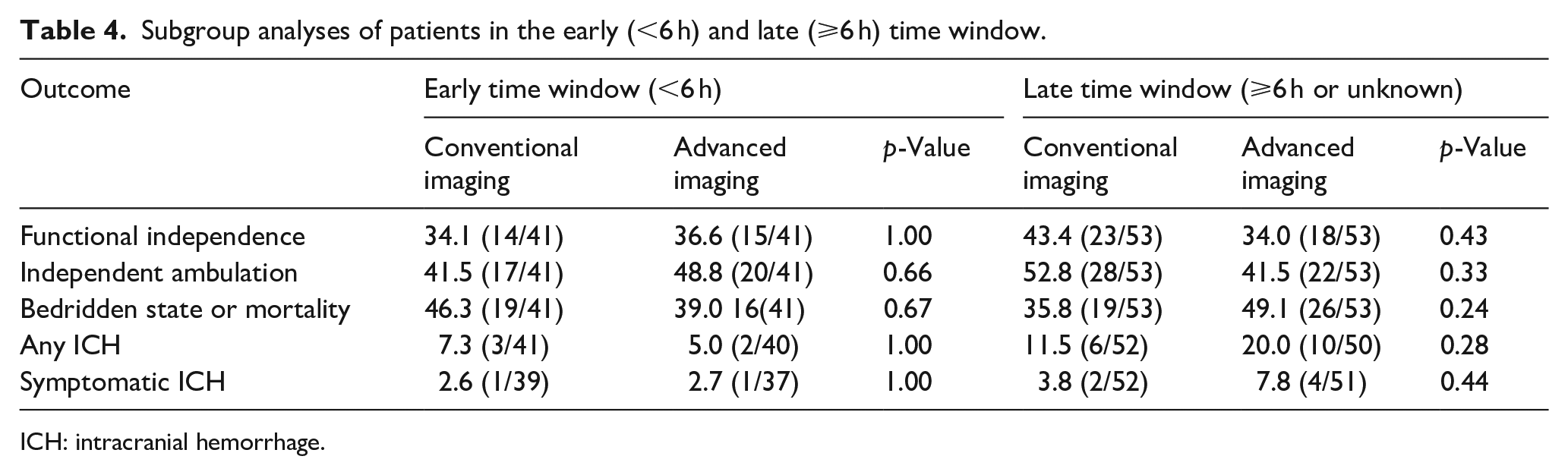
ICH: intracranial hemorrhage.
Discussion
In this international multi-center retrospective study of BAO stroke patients, we found that conventional or advanced imaging selection of EVT patients did not result in significant differences in 90-day clinical outcomes or rates of ICH. These findings persisted in subgroup analyses across treatment time windows and advanced imaging modality used (CTP or MR). Overall, results of the current study suggest that conventional imaging (CT and CTA) may be sufficient for selecting BAO-EVT patients in routine clinical practice, and that the use of advanced imaging modalities such as CTP or MR may not be necessary.
The optimal use of advanced neuroimaging modalities such as CTP and MR during acute stroke triage to select patients for reperfusion therapy has been a topic of debate. In anterior circulation stroke, studies have demonstrated that EVT selection based on conventional CT alone may lead to similar outcomes, even in the extended time window.7,12,18–21 Recent positive results from low-ASPECTS thrombectomy trials for anterior circulation LVOs, negative trial results from distal and medium vessel occlusion strokes, and a growing body of literature advocating for direct-to-angiosuite systems may further obviate the need for advanced imaging during acute stroke triage in general.18,21–25 Despite these trends, whether advanced imaging selection for EVT for BAO strokes may be advantageous is less clear. CT-based imaging markers are important for selecting basilar EVT patients, and scoring systems are available for quantifying early ischemia in posterior circulation strokes using non-contrast CT (e.g. pc-ASPECTS 26 and MPI 27 ). However, unlike ASPECTS for anterior circulation strokes, 28 pc-ASPECTS and MPI are lesser-known, and more prone to poor inter-rater reliability and signal artifacts in the posterior fossa. 29 The heterogeneity of pc-ASPECTS areas in terms of clinical implications are also substantial (e.g. discrepancies in clinical deficit of pontine vs occipital lobe infarcts), further limiting its clinical use. Furthermore, while collaterals can be assessed for anterior circulation LVOs to assist patient selection, there is currently no validated tool to assess collateral status in the basilar territory. Thus, it is possible that advanced imaging modalities such as CTP and MR may provide more reliable tissue-level information, and their use may confer an advantage when optimizing patient selection.30–33
While some studies have suggested that CTP can identify hypo-perfused tissue for BAO strokes,34–36 the use of CTP in the posterior circulation is overall unvalidated and has not demonstrated clinical reliability in large clinical trials. In our study, CTP use did not appear to improve patient selection. Data on the reliability of CTP in the posterior circulation is limited,32,37 and the lack of significant difference in our study may be due to multiple factors. The reliability of CTP is known to be poor in subcortical areas (e.g. brain stem, thalami) compared to the cortex 38 ; thus, CTP may not be as reliable for BAO strokes compared to anterior circulation occlusions. Beam hardening artifacts from the petrous bone may also limit the reliability of CTP in the brainstem. 29 Furthermore, well-established thresholds for CTP metrics such as cerebral blood flow and time to peak were derived based on hemodynamic characteristics of the anterior cerebral circulation, and they may not be applicable to BAO strokes.39,40
Reasons underlying the lack of difference between conventional imaging and MR may be more nuanced. On one hand, DWI quantification of ischemic burden may not be reflective of permanent tissue damage, as these lesions can be reversible 41 and may be more benign compared to ischemic changes on CT. 42 Thus, MR-selected patients may be more likely to have favorable clinical outcomes. On the other hand, MR may have been selectively used as a second-line imaging modality at some institutions. Thus, some MR-selected patients may be associated with worse outcomes as they may have had limited or unfavorable CT scans. These possible and competing phenomena may have influenced the overall similar outcomes between patients selected for BAO-EVT by CT/CTA and MR in our study, and results must be interpreted with caution.
Despite the above uncertainties, our results suggest that BAO-EVT patients selected by CT/CTA in routine clinical practice have equivalent clinical outcomes compared to patients selected by CTP or MR. Given that it is less resource and time-intensive compared to advanced imaging modalities, CT/CTA may be appropriate and sufficient as a front-line selection for BAO-EVT. However, inherent limitations of CT/CTA for BAO strokes may lead to under-identification of patients who may derive significant benefit from EVT. Thus, for patients who appear to be poor candidates for basilar EVT due to limited or unfavorable CT/CTA findings, additional advanced imaging modalities may still be valuable as a second-line selection tool to potentially “rule in” their eligibility.
Our study has several limitations. Quantification of early ischemic signs (e.g. pc-ASPECTS or MPI) was not available in the STAR database, and it is unclear if patients had similar ischemic burdens between groups. Choice of imaging modality was based on local institutional policies, and the reasons for opting conventional versus advanced imaging selection was not recorded in the STAR database. It is possible that some patients who were selected for BAO-EVT may have undergone first-line conventional imaging that revealed concerning or uncertain findings. Thus, while our findings suggest that first-line conventional imaging may perform similarly well compared to advanced imaging modalities, we cannot rule out a clinical benefit of second-line advanced imaging for select BAO-EVT candidates. A large portion of BAO EVT patients in the STAR database did not have available information regarding the imaging modalities used to select each patient for treatment; thus, our study is vulnerable to ascertainment bias. Furthermore, while our study is the first to report a large series of patient outcomes of BAO EVT selected by conventional versus advanced imaging modalities, sample size is modest, and we may lack statistical power to detect more subtle differences. Finally, as a retrospective observational study with self-reported clinical and imaging outcomes without core lab adjudication, there may be uncaptured and hidden confounders that could not be accounted for.
Conclusions
In patients with BAO, outcomes after EVT were similar in patients selected using conventional versus advanced imaging modalities. Thus, conventional imaging appears sufficient as a first-line tool for selecting basilar EVT patients in routine clinical practice. Future prospective studies are needed to investigate the role of advanced imaging modalities for BAO during acute triage, especially as a second-line modality for patients with equivocal or unfavorable conventional imaging findings.
Supplemental Material
sj-tiff-1-eso-10.1177_23969873251364973 – Supplemental material for Conventional versus advanced imaging selection for endovascular treatment of basilar artery occlusion strokes
Supplemental material, sj-tiff-1-eso-10.1177_23969873251364973 for Conventional versus advanced imaging selection for endovascular treatment of basilar artery occlusion strokes by Huanwen Chen, Marco Colasurdo, Hidetoshi Matsukawa, Conor Cunningham, Ilko Maier, Sami Al Kasab, Pascal Jabbour, Joon-Tae Kim, Stacey Quintero Wolfe, Ansaar Rai, Robert M Starke, Marios-Nikos Psychogios, Edgar A Samaniego, Nitin Goyal, Shinichi Yoshimura, Hugo Cuellar, Jonathan A Grossberg, Ali Alawieh, Ali Alaraj, Mohamad Ezzeldin, Daniele G Romano, Omar Tanweer, Justin Mascitelli, Isabel Fragata, Adam Polifka, Fazeel Siddiqui, Joshua Osbun, Roberto Crosa, Charles Matouk, Min S Park, Michael R Levitt, Waleed Brinjikji, Mark Moss, Travis Dumont, Ergun Daglioglu, Richard Williamson, Pedro Navia, Reade De Leacy, Shakeel Chowdhry, David J Altschul, Alejandro M Spiotta and Peter Kan in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Chen: None. Dr Colasurdo: None. Dr Matsukawa received a lecture fee from Daiichi-Sankyo and Stryker and consulting services fee from B. Braun. Dr Al Kasab: grant from Stryker for RESCUE-ICAS registry. Dr Cunningham: None. Dr Maier: Speakers honoraria from Pfizer and Bristol-Myers Squibb. Dr Jabbour: None. Dr Kim: None. Dr Wolfe: None. Dr Rai: None. Dr Starke: RMS research is supported by the NREF, Joe Niekro Foundation, Brain Aneurysm Foundation, Bee Foundation, Department of Health Biomedical Research Grant (21K02AWD-007000), and by National Institute of Health (R01NS111119-01A1) and (UL1TR002736, KL2TR002737) through the Miami Clinical and Translational Science Institute, from the National Center for Advancing Translational Sciences and the National Institute on Minority Health and Health Disparities. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. RMS has an unrestricted research grant from Medtronic and Balt and has consulting and teaching agreements with Penumbra, Abbott, Medtronic, Balt, InNeuroCo, Cerenovus, Naglreiter, Tonbridge, Von Medical, and Optimize Vascular. Dr Psychogios: Grants from the Swiss National Science Foundation (SNF) for the DISTAL trial (33IC30_198783) and TECNO trial (32003B_204977), Grant from Bangerter-Rhyner Stiftung for the DISTAL trial. Unrestricted Grants for the DISTAL trial from Stryker Neurovascular Inc., Phenox GmbH, Penumbra Inc. and Rapid Medical Inc., Sponsor-PI SPINNERS trial (Funded by a Siemens Healthineers AG Grant), Research agreement with Siemens Healthineers AG, Local PI for the ASSIST, EXCELLENT, TENSION, COATING, SURF, and ESCAPE-NEXT trials. Speaker fees: Stryker Neurovascular Inc., Medtronic Inc., Penumbra Inc., Acandis GmbH, Phenox GmbH, Siemens Healthineers AG. Dr Samaniego: None. Dr Arthur: Consultant for Arsenal, Balt, Johnson and Johnson, Medtronic, Microvention, Penumbra, Perfuze, Scientia, Siemens, Stryker. Research support from Balt, Medtronic, Microvention, Penumbra, and Siemens. Shareholder Azimuth, Bendit, Cerebrotech, Endostream, Magneto, Mentice, Neurogami, Neuros, Perfuze, Revbio, Scientia, Serenity, Synchron, Tulavi, Vastrax, VizAI. Dr Yoshimura received a lecture fee from Stryker, Medtronic, Johnson & Johnson, Kaneka Medics. Dr Cuellar: Dr Hugo Cuellar: Consultant for Medtronic, Penumbra and Microvention. Dr Grossberg: None. Dr Alawieh: None. Dr Tanweer: None. Dr Mascitelli: None. Dr Fragata: None. Dr Polifka: None. Dr Osbun: None. Dr Crosa: None. Dr Matouk: Consultant for Stryker, Medtronic, Microvention, Penumbra, and Silk Road Medical. Speaker for Penumbra and Silk Road Medical. Contact PI for NIH Grant R21NS128641. Dr Park: Consultant for Medtronic. Dr Levitt: Unrestricted educational grants from Medtronic and Stryker; consulting agreement with Medtronic, Aeaean Advisers and Metis Innovative; equity interest in Proprio, Stroke Diagnostics, Apertur, Stereotaxis, Fluid Biomed, and Hyperion Surgical; editorial board of Journal of NeuroInterventional Surgery, data safety monitoring board of Arsenal Medical. Dr Brinjikji: None. Dr Moss: None. Dr Dumont: None. Dr Williamson: Consultant for Medtronic, Stryker, and Synaptive Medical. Dr Navia: Consultant for Penumbra, Medtronic, Stryker, Cerenovus, and Balt. Dr Leacy: Research grants from Siemens Healthineers and Kaneka medical. Consultant for Cerenovus, Stryker Neurovascular and Scientia Vascular. Minor equity interest Vastrax, Borvo medical, Synchron, Endostream, Von Vascular. Dr Chowdhry: Consultant and proctor for Medtronic and Microvention. Dr Ezzeldin: Speaker for Viz.ai and has stocks in Galaxy Therapeutics. Dr Spiotta: Research support from Penumbra, Stryker, Medtronic, and RapidAI. Consultant for Penumbra, Stryker, Terumo, and RapidAI. Dr Kan: Grants from the NIH (1U18EB029353-01) and unrestricted educational grants from Medtronic and Siemens. Consultant for Imperative Care and Stryker Neurovascular. Stock ownership in Vena Medical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The STAR registry receives research support from Penumbra, Microvention, Medtronic, Stryker, RapidAI, Brain Aneurysm Foundation.
Informed consent
Patient consent was waived due to the retrospective nature of the study.
Ethical approval
The study was approved by the institutional review board at the Medical University of South Carolina (protocol number: Pro00090704) and at each participating institution. The data at each institution were obtained retrospectively and collected according to a standardized protocol.
Guarantor
PK is the guarantor of this study.
Contributorship
HChen, MC, AMS, and PK conceived the study idea. MC, HM, CC, IM, SAK, PJ, JTK, SQW, AR, RMS, MNP, EAS, NG, SY, HCuellar, JAG, AAlawieh, AAlaraj, ME, DGR, OT, JM, IF, AP, FS, JO, RC, CM, MSP, MRL, WB, MM, TD, ED, RW, PN, RDL, SC, and DJA collected the data. HChen, MC, and HM analyzed the data. HChen and MC wrote the manuscript. HM, MRL, AMS, and PK revised the manuscript. All authors approved the final submission.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
