Abstract
Introduction:
Stroke is a leading cause of death and disability. The European Stroke Organization (ESO) developed Stroke Unit certification criteria to standardize and improve stroke care quality. This study evaluates the impact of implementing these criteria on outcomes for acute ischemic stroke patients treated with intravenous thrombolysis (IV alteplase) in an Emergency Hospital in Bucharest, Romania.
Methods:
Our retrospective observational study compared outcomes before (2017–2020) and after (2021–2024) implementation of ESO Stroke Unit certification protocols. Data from 568 patients receiving IV alteplase were analyzed. Our analysis included in-hospital mortality, modified Rankin Scale (mRS) and NIH Stroke Scale (NIHSS) at discharge, NIHSS shift, and key time metrics (door-to-CT, neuro-to-needle, door-to-needle).
Results:
Post-intervention, the annual IV alteplase rate increased significantly (15.47% vs 4.51%,
Discussion:
Implementing ESO Stroke Unit certification criteria was associated with significant improvements in thrombolysis rates, key time metrics, and patient outcomes.
Conclusions:
ESO Stroke Unit certification may lead to improved stroke care and patient outcomes, particularly in emerging healthcare systems.

Introduction
Significant changes have taken place in the last two decades regarding the management of stroke patients, with substantial advancements in both diagnosis tools and available treatments.1–5 Revascularization therapies, such as intravenous thrombolysis and mechanical thrombectomy, along with the implementation of standardized stroke protocols in acute care, have drastically changed the prognosis for these patients.5–7
The complex medical care needed for stroke patients has led to the development of modern stroke units, encompassing a multidisciplinary approach, dedicated staff trained in managing stroke, closer monitoring and rapid access to treatment and investigations.6,8 Recent studies have shown that admitting acute stroke patients in stroke units significantly increases their chance of survival and leads to a reduction in long-term disability.9,10
Certification criteria for stroke units differ between countries.11,12 In an effort to define an international standard for all stroke units to adhere to, the European Stroke Organization (ESO) established a Stroke Unit Certification Committee, providing a model and a clear pathway for all centers aiming for excellence in stroke care.6,13,14
Despite the growing number of ESO certified centers, only a few hospitals have published data regarding the impact and benefits of obtaining this certification, mostly from Germany and Switzerland, demonstrating a positive effect on stroke-associated disabilities and broader access to intravenous thrombolysis.9,15,16
Following the ESO Certification of our Stroke Unit, located in Bucharest, Romania, we wanted to analyze its impact on outcomes for acute ischemic stroke patients that underwent intravenous thrombolysis (IV alteplase).
Methods
Study design
We conducted a retrospective observational cohort study to assess the impact of the ESO certification process on the outcomes of stroke patients receiving IV alteplase in our center between 2017 and 2024, split into two distinct groups, defined as Pre- and Post-intervention, as detailed below.
Intervention
The ESO Stroke Unit Certification Criteria can be found on the dedicated website (https://eso-certification.org/). Currently, centers can apply as either a Stroke Unit (a primary stroke unit, usually restricted to IV alteplase) or a Stroke Center (a more comprehensive stroke center, managing all aspects of stroke care including mechanical thrombectomy, research etc.).17,18
While our stroke unit has held national certification since 2014, we decided to begin the ESO certification process in 2020. An internal analysis regarding current stroke care settings was started, including a comparison with current ESO certification criteria and preparation of dedicated protocols and administrative changes.
The intervention – starting with January 1st 2021, all criteria of ESO certification were implemented as recommended. These included dedicated protocols regarding the Lead Stroke Physician and Personnel, General Infrastructure (dedicated stroke pathways), Investigations (priority access for stroke patients), Interventions and Monitoring, Teaching, Meetings, and Research, as well as monitoring of Quality Indicators (the seven categories of criteria for ESO certification).
After 3 years of consistency in quality of care, our center decided to apply for ESO Stroke Unit certification in 2023. The final dossier was submitted in December 2023, and certification was received in June 2024.
Study population
We retrospectively collected data from 593 patients who underwent IV thrombolysis with alteplase for acute ischemic stroke at our center during the selected timeframe. After excluding patients with incomplete data (18 patients) and outliers (3 patients from each group based on admission NIHSS, as assessed by visual analysis of box-plot graphs), the final study lot included 568 patients. We defined two groups for our study – stroke patients receiving IV alteplase in the 4 years prior to implementing certification protocols (January 1st 2017–December 31st 2020) - called “Pre-intervention Group,” and patients treated after implementing ESO certification criteria (January 1st 2021–December 1st 2024) - called “Post-intervention Group.”
Data source and variables
The data were obtained from the National Thrombolysis Registry, The Registry of Stroke Care Quality (RES-Q) and the local stroke registry. We analyzed demographic data (age, gender) and clinical data, including modified Rankin Score (mRS) prior to admission, NIH Stroke Scale (NIHSS) at admission and TOAST classification of Stroke. Primary outcomes included in-hospital mortality, NIHSS and modified Rankin Scale at discharge, NIHSS shift (defined as the difference between NIHSS on admission and at discharge), and key time metrics such as Onset-to-Door, Door-to-CT time, Neuro-to-needle time, and Door-to-needle times (Neuro-to-needle time was defined as the duration from the neurologist’s first contact with the patient to the administration of intravenous alteplase). Favorable outcome was defined as mRS of 0–1. 19 Secondary outcomes that were analyzed include thrombolysis-associated complications and recurrent stroke. Evaluation of hemorrhagic transformation was conducted using computed tomography (CT) at 24 h or sooner in case of clinical deterioration. Hemorrhagic transformation was classified according to ECASS III criteria (symptomatic hemorrhagic transformation was considered any intracranial hemorrhage associated with clinical deterioration, as defined by an increase of 4 or more points on NIHSS score, or that led to death and that was identified as the predominant cause of the neurological deterioration). 20 Symptomatic extracerebral hemorrhage is defined as any bleeding that required intervention, caused a change in the patient’s condition, or exceeded a volume of 100 ml. Recurrent stroke was defined as any stroke occurring during hospitalization. A detailed list of variables used can be found in the Supplemental Material.
Statistical analysis
We used the Shapiro–Wilk test to check for normality in small-sized continuous variables and
Results
The Pre-intervention group comprised 184 patients, with 80 (43.5%) women and 104 (56.5%) men, and the post-intervention group comprised 384 patients, 171 (44.5%) women and 213 (55.5%) men. The median age for the pre-intervention group was 69 years (25;92) and 70 years (32;93) for the post-intervention group. Table 1 shows the main demographic and clinical characteristics of the group of patients.
Cohort demographics and clinical characteristics at admission.

mRS: modified Rankin Scale; NIHSS: The National Institutes of Health Stroke Scale; SD: standard deviation; TOAST: Trial of ORG 10172 in Acute Stroke Treatment.
Mann-Whitney
Pearson-Chi square test.
There was no significant difference between the two groups regarding age, gender and mRS prior admission.
A total of 3914 ischemic strokes were documented in hospital records during the pre-certification period, while 2811 strokes were recorded during the post-certification period. The annual IV alteplase rate increased for the post-intervention group, from 4.51% to 15.47%, with patients being over three times more likely to receive IV alteplase (OR = 3.54,
Onset-to-door for the pre-intervention group was lower than for the post-intervention group (96 min vs 114 min,
Key time metrics regarding the administering of IV alteplase can be seen in Table 2. Definitions for each time metric can be found in the Supplemental File.
Comparison of IV thrombolysis times between the two groups.

Mann-Whitney
The time metrics for IV alteplase, including door-to-CT (
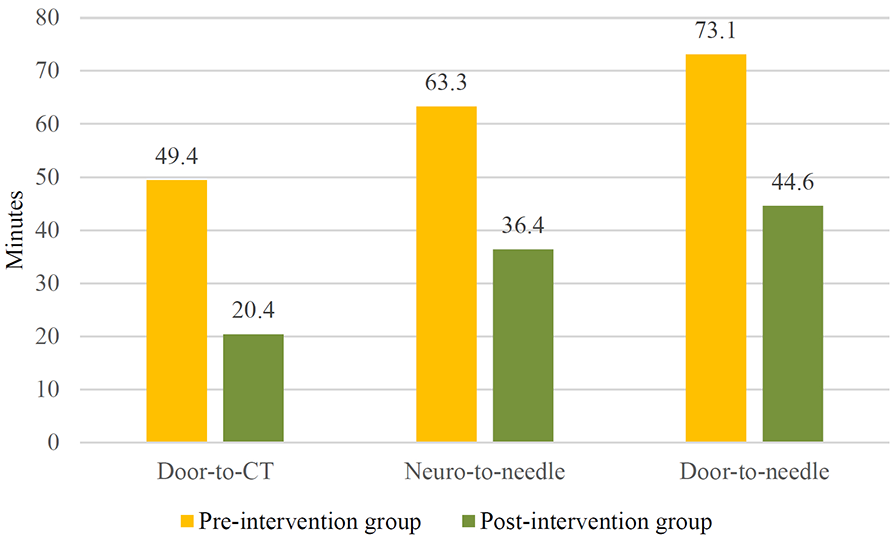
Comparison of time metrics for the two groups.
The comparison regarding main outcomes, as assessed by mRS, NIHSS score at discharge, NIHSS shift, thrombolysis rates, and complications, are shown in Table 3.
Comparison of thrombolysis outcomes across two groups.

aOR: adjusted OR; mRS: modified Rankin Scale; NIHSS: The National Institutes of Health Stroke Scale; OR: Odds Ratio; SD: standard deviation; TOAST: Trial of ORG 10172 in Acute Stroke Treatment.
Independent
Mann-Whitney
Pearson-Chi square test; aOR obtained by multiple regression adjusting for confounders (Onset-to-door, NIHSS at admission, stroke etiology).
NIHSS at discharge was lower for the post-intervention group versus pre-intervention group (with a median of 3 vs 4,
A higher proportion of patients in the post-intervention group had a favorable outcome (44.8%) compared to the pre-intervention group (33.2%),
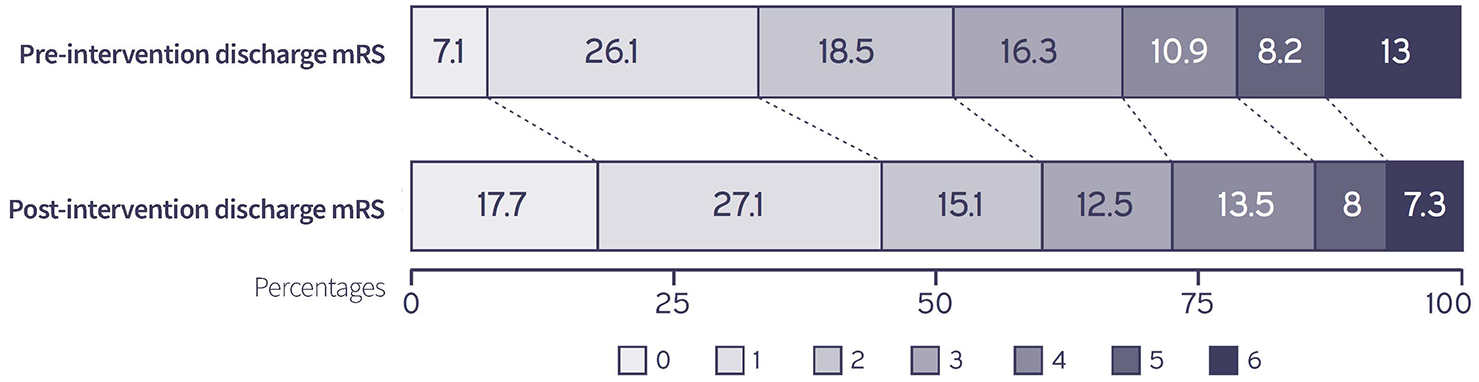
mRS at discharge for the two groups.
The differences between the mean annual in-hospital mortality rates were statistically significant between the two groups (12.5% for the pre-intervention group vs 7.3% for the post-intervention group,
The post-intervention group showed a significant reduction in cerebral hemorrhagic transformation (8.6% vs 16.3%,
Discussion
This study is the first to analyze the effects of the ESO Stroke Unit Certification process on outcomes for acute ischemic stroke patients receiving IV thrombolysis in an Emergency Hospital in Bucharest, Romania.
No major differences were observed between the two groups regarding demographic data, except for Onset-to-door, NIHSS at admission and TOAST Stroke etiology, which were included as potential confounders when comparing outcomes.
Onset-to-door shows a marked increase for the post-intervention group (113.85 min vs 95.60 min,
Most patients that receive IV alteplase have either a cardioembolic or an atherothrombotic etiology of stroke, as previously shown by multiple studies.21,22 While our study confirms these findings, a significant shift in etiology can be seen between the two groups, with an increase in strokes due to large vessel disease and a decrease in cardioembolic strokes for the post-intervention group. This may be explained by the fact that cardioembolic stroke patients are more prone to having contraindications due to ongoing anticoagulant therapy, while the implementation of acute stroke protocols led to an increase in CT angiography imaging being performed, with improved likelihood of detecting large vessel disease.23,24
The process of ESO Certification is complex, spanning seven different categories of evaluation and including protocols for prioritizing acute stroke in the ER, interdisciplinary cooperation, availability of paraclinical investigations, implementation of acute stroke pathways, key time metrics, quality of care, dedicated protocols for all phases of stroke, early rehabilitation, dedicated registries, and many more.17,18
The effects of implementing these guidelines can be seen in the evolution of the key time metrics, with a focus on door-to-CT (DTC), neuro-to-needle (NTN), and door-to-needle (DTN), which have been proven to have a positive influence on patients’ outcome.25,26All time metrics evaluated in our study significantly improved for the post-intervention group, with DTN decreasing by 38.9% from a mean time of 73.07 ± 27.85 min in the pre-certification group to 44.64 ± 20.73 min in the post-certification group (
We decided to include in our analysis the less common time metric of Neuro-to-needle. Defined as the duration from the first contact of the neurologist with the patient to administering IV alteplase, this metric evaluates the performance of the neurologist leading the case independent of other factors related to the Emergency Department (ED) such as late symptoms recognition, logistics etc. As most of the patients in this study were transported directly to CT after initial ED evaluation, the Neuro-to-Needle time evaluates the rapidity of the stroke neurologist in evaluating the patient, analyzing imaging and coordinating the team to administer IV alteplase. Its marked improvement suggests that the protocols associated with our intervention may lead to faster decision times.
Importantly, the increase in IV alteplase rate and decrease in time metrics did not come at the cost of patient safety. While no differences were seen between the two groups regarding significant extracerebral hemorrhage or recurrence of strokes, patients in the post-intervention group had a 49% lower probability of hemorrhagic transformations (8.6% vs 16.3%,
Significant improvements were seen on the NIHSS score, both in NIHSS at discharge and the NIHSS shift for the post-intervention group. Since our study included only patients that had received IV alteplase, the improvement should be attributed to both the decrease in key time metrics in the ER, resulting in a faster DTN, 29 but also the increase in quality of care during hospitalization.
Clinical outcomes, as measured by mRS at discharge and mortality, showed improvement for the post-intervention group. Patients in this group had a significantly higher likelihood of a favorable outcome and a lower risk of mortality compared to the pre-intervention group.
Some, but not all the effects observed in our modeling, can be associated to a faster administration of treatment, as measured by Door-To-Needle time. The improved clinical outcome for the patients in the post-intervention group may be linked to the complex changes in stroke management, from the hyperacute stage to long-term follow-up, as regulated by the ESO Stroke Unit certification criteria. 30
Although our analysis demonstrates significant associations between ESO Stroke Unit certification and improved stroke outcomes, it does not establish direct causality. The observed improvements are likely multifactorial, influenced by factors such as increased physician experience over time, stroke awareness campaigns, and improvements in hospital facilities not quantified in this study, all of which may contribute to better patient outcomes. A prospective study design will be needed to better evaluate the direct impact of ESO certification.
The main limitation of this study is the retrospective collection of data. This may have caused selection bias by eliminating patients with incomplete data. Another limitation of the study is that it is based on a single center, and comparative data from non-ESO certified stroke units in the region were not available for inclusion. A key missing outcome is mRS at 3 months, as well as the causes of in-hospital mortality. While this data is available for the post-intervention group due to ESO guidelines implementation, it is missing for the pre-intervention group, which makes a direct comparison impossible. We consider this to be another argument in favor of ESO certification, leading to mandatory collection of such valuable metrics.
Conclusions
ESO Stroke Unit certification was associated with improvements in thrombolysis rates, thrombolysis-related times and patient functional outcomes at discharge, as measured by NIHSS at discharge, NIHSS shift, modified Rankin scale at discharge and in-hospital mortality. Almost all aspects of stroke care saw significant improvements after the implementation of ESO certification criteria on hospital level.
One of the most important aspects of our findings is that the benefits of certification can be seen from the moment of implementing the criteria, preceding in our case the actual ESO certification by almost 3 years. This underlines the importance of uniform application of guidelines and collaborative care models for patients with ischemic stroke. Our study highlights their importance and hopes to influence the decision of other centers to start the ESO accreditation process.
We believe that these data suggest that it is perhaps hospitals from emerging healthcare systems that stand to benefit most from ESO Stroke Unit Certification. While focusing on the Stroke Unit, the guidelines tackle collaboration with the Emergency Department, Radiology, Cardiology, Neurosurgery, Vascular Surgery, Internal Medicine, Neurointerventional, Rehabilitation, Speech and Swallow specialist, and Occupational Medicine, reaching far beyond the physical borders of the Stroke Units and fundamentally changing stroke care for the entire hospital and the region it serves.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251334285 – Supplemental material for Benefits of ESO stroke unit certification: A report from a Tertiary Stroke Center in Bucharest, Romania
Supplemental material, sj-docx-1-eso-10.1177_23969873251334285 for Benefits of ESO stroke unit certification: A report from a Tertiary Stroke Center in Bucharest, Romania by Vlad Tiu, Roxana Urdea, Cristian Ghiță, Bianca Chelaru, Simona Petrescu, Horia Nicolae, Diana Dobrin and Cristina Aura Panea in European Stroke Journal
Footnotes
Acknowledgements
The improvements in stroke care reported in this study are owed to the hundreds of people working in the Elias University Emergency Hospital, that have tirelessly challenged themselves to offer a better chance to all of our stroke patients. We are immensely grateful for their help.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Vlad Tiu is currently a member of the ESO Stroke Unit Certification Committee.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was waived by The Ethics Committee of the Elias University Hospital due to the retrospective, observational design of the study.
Informed consent
Not applicable (retrospective cohort registry analysis).
Guarantor
VT
Contributorship
VT, RU, CG, BC wrote the first draft and performed statistical analysis. BC, RU, HN and SP reviewed the literature. DD, CP and VT developed the protocol. VT, CP and DD conceived the study. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
