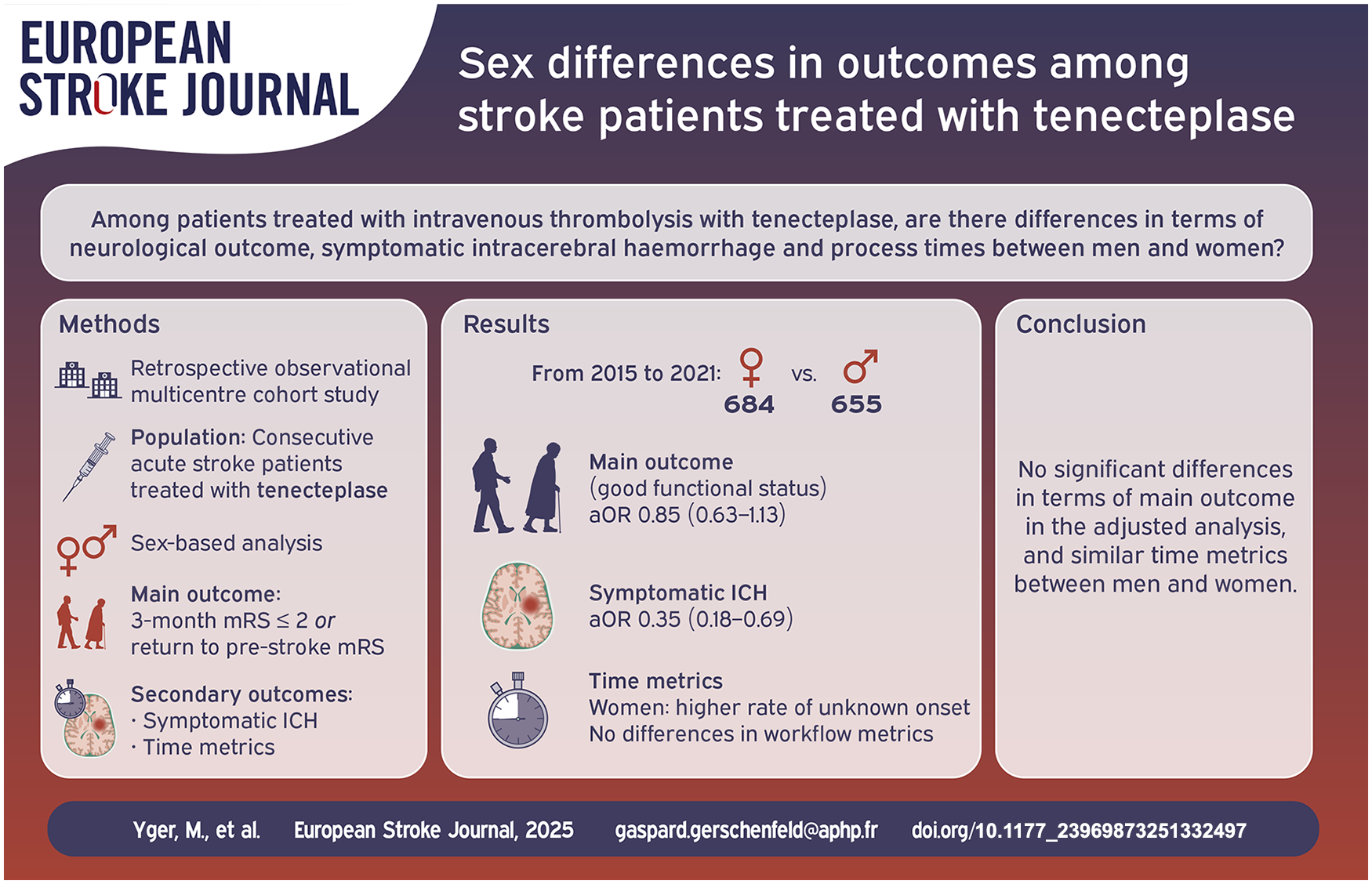
Abstract
Introduction:
A better understanding of sex-related differences remains key to improve stroke care for women. We aimed to assess the differences of prognosis, symptomatic intracranial hemorrhage (sICH) rates and process times between men and women suffering from ischemic stroke.
Patients and methods:
We performed a sex-based retrospective analysis of the French Tenecteplase Treatment in Stroke multicentric registry from 2015 to 2021. We assessed sex differences in terms of 3-month good neurological outcome (defined as modified Rankin score ⩽ 2 or back to its pre-stroke value), occurrence of sICH and process times with multivariable logistic regression analyses.
Results:
1339 consecutive patients (684 women, 655 men) received tenecteplase between 2015 and 2021, among whom 873 underwent thrombectomy. Both groups differed in terms of demographic characteristics, etiology, and unclear onset. In multivariable logistic regression analyses, women and men did not differ in terms of 3-month good neurological outcome (aOR = 0.85 [0.63–1.13]). Women were less likely to have a sICH after tenecteplase (aOR = 0.35 [0.18–0.69]) and more likely to have a stroke of unknown onset (aOR 1.44 [1.07–1.95]), but workflow metrics did not differ once stroke code was triggered.
Discussion and conclusion:
In our study, we did not find a significant difference between women and men in terms of 3-month good outcome. However, our study size may hinder its detection. Interestingly, we observed that women had a lower rate of sICH after tenecteplase. Women’s workflow metrics were similar to men’s, except for a higher rate of stroke with an unknown onset.
Introduction
Stroke in women is a preoccupying and rising pathology, as they carry a higher burden than men.1,2 While this may be due in part to their longer life expectancy, several studies have pointed out differences in baseline characteristics, severity, and medical management between women and men treated for acute ischemic stroke (AIS).3–6 Women suffering from AIS are older,4,7–10 smoke less frequently,4,8,9 and more often have atrial fibrillation and high blood pressure than men.4,9,10 Before the advent of reperfusion therapies, women had been reported to have a worse prognosis than men. 11 However, these outcome discrepancies have narrowed over time with intravenous thrombolysis (IVT) and mechanical thrombectomy (MT).4,12 Intrinsic biological differences and sex-related differences in medical management depending on local societal considerations have been hypothesized as factors of these discrepancies.11–17
Available data from historical clinical trials and large registries are conflicting on the possibility of a differential treatment effect between women and men.4,6,7,9,15,18,19 Except for two studies which used pro-urokinase and tenecteplase,3,8 the thrombolytic used was alteplase. Tenecteplase is a genetic variant of alteplase with higher fibrin specificity and is now considered as a reasonable alternative to alteplase in AIS patients. 20 Sex-focused data on patients treated with tenecteplase are limited. A secondary analysis of the NOR-TEST trial, which compared tenecteplase (at a high dose of 0.4 mg/kg) to alteplase found no significant difference in terms of clinical outcomes between sexes. 3 More recently, a sex-disaggregated secondary analysis of the AcT trial, a large non-inferiority trial which compared 0.25 mg/kg tenecteplase and alteplase in non-selected AIS patients, found no differences in terms of workflow metrics between sexes. 21
In this study, our aim was to assess the influence of sex on clinical outcome, rate of 24h-symptomatic intracranial hemorrhage and process times in AIS patients treated in a real-world setting, with tenecteplase with or without additional MT.
Patients and methods
Patients
We conducted a retrospective analysis of the large real-life observational multicenter Tenecteplase Treatment in Ischemic Stroke (TETRIS) registry between May 2015 to June 2021. Details on TETRIS have been previously published. 22 Briefly, TETRIS is a multicenter registry based on prospective local databases in six French stroke centers, which includes consecutive patients who received IVT with tenecteplase (0.25 mg/kg, maximum 25 mg) for AIS since May 2015, with or without additional MT. IVT could be administered: (i) within 4.5 h of symptoms onset; within 4.5 and 9 h of symptoms onset if they had salvageable brain regions on perfusion imaging according to the EXTEND criteria 23 ; or in the presence of a magnetic resonance imaging mismatch between diffusion-weighted imaging and fluid-attenuated inversion recovery in case of unknown onset. 24
Clinical and imaging data
We extracted from the registry demographic data (age, biological sex, vascular risk factors, baseline medication and mRS), stroke characteristics (National Institutes of Health Stroke Scale (NIHSS) score, intracranial occlusion site, Alberta Stroke Program Early CT Score (ASPECTS)), recanalization status (defined as a modified Thrombolysis in Cerebral Infarction score of 2b or3) for patients who underwent MT, process times and final determined cause of stroke at the 3-month consultation according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification. 25 Stroke with unwitnessed onset and wake-up stroke were classified as stroke of unknown onset. We collected the following outcome data: symptomatic intracranial hemorrhage (sICH, defined below) and 3-month mRS score. Cerebral imaging data were assessed retrospectively by experienced stroke neurologists and neuroradiologists.
Outcomes and measures
The primary outcome was 3-month good neurological outcome, defined either by a modified Rankin Scale score (mRS) ⩽2 or a return to the pre-stroke mRS score. Secondary outcomes were sICH, defined as an increase of at least 4 points in the NIHSS score within 36 h of IVT associated with a local or remote type 2 PH, 26 and 3-month mortality.
We used the following end points for process times: time of symptoms onset (when known); time when IVT was started; time of arterial puncture in neuroangiography suite; and time of first successful reperfusion (when a modified thrombolysis in cerebral infarction score of at least 2b was first achieved for patients who underwent MT).
Statistical analysis
Quantitative variables were described as medians (interquartile ranges (IQR)), and qualitative parameters as counts and percentages. Categorical variables were compared using the Chi-square or Fisher’s exact test, and continuous variables were compared using the Student’s t-test or Wilcoxon rank-sum test, as appropriate. To handle missing values, multiple imputations were performed under the missing-at-random assumption using the MICE package in R. Results from the analysis of each imputed dataset (n = 10) were combined using Rubin’s rules.
We evaluated the influence of sex on the primary outcome (3-month good outcome), and secondary outcomes (3-month mortality and sICH). In order to have an adjusted model that would be adapted for all outcomes, including sICH which were expected to have a few events, we first made a multivariable logistic regression analysis adjusted for age, as it is a main influencer on stroke outcome, and inclusion center (Model 1). Due to the small number of centers (6), to avoid convergence issues with a mixed model the inclusion center was added as a fixed effect in the statistics models. To account of other confounders, we performed a second multivariable logistic regression analysis (Model 2) adjusted for age, inclusion center, NIHSS at admission, pre-stroke hypertension, pre-stroke diabetes mellitus, current smoking, history of stroke, history of myocardial infarction, pre-stroke atrial fibrillation, pre-stroke mRS (⩽ 2 or >2), delay from onset to intravenous thrombolysis (<120, 120–180, >180 min, unclear), occlusion site, treatment with MT, and stroke etiology. Log-linearity was assessed for continuous variables. If not met, continuous variables were categorized into tertiles. Potential collinearity was checked using the variance inflation factor. Subgroup analyses were performed according to age (using tertiles as age cut-offs), delay from symptoms to IVT, occurrence of MT, and stroke etiology by adding an interaction term between these variables and sex in the multivariable model. P-values for interaction terms were estimated using the log-likelihood ratio test (D3 function in the MICE package for R).
To further reduce the unbalance in demographic and clinical characteristics between men and women, we performed an additional sensitivity analysis using propensity-weighted analyses (Model 3). The propensity score was estimated with multivariable logistic regression (package WeightIt in R), with sex as the dependent variable, and including as independent variables: age, pre-stroke hypertension, pre-stroke diabetes mellitus, current smoking, history of stroke, history of myocardial infarction, pre-stroke atrial fibrillation, pre-stroke mRS, NIHSS at admission, occlusion site, delay from onset to intravenous thrombolysis, treatment with MT, stroke etiology, and inclusion center. A propensity score-weighted analysis was performed using inverse propensity score weights and the entire dataset to estimate the average impact of sex in the overall population. Weights were stabilized by multiplying each unit’s weight by the proportion of units of their group (proportion of males and females). Covariate balance between the two groups was assessed before and after weighting, and an absolute standardized difference (ASD) of less than 0.1 was considered evidence of balance. The standard errors of the estimated odds ratios (ORs) were calculated using a robust standard error estimator.
We assessed the influence of sex on process times using a linear regression model, adjusting for age (categorized into tertiles), pre-stroke mRS (⩽2 or >2), baseline NIHSS score, type of arterial occlusion, and inclusion center. Missing values were not imputed in this analysis. We checked for linearity, collinearity, and normality of the residuals. To ensure normality of the residuals, the dependent variable was log-transformed. Finally, we also assessed the influence of sex on the eventuality of an unclear onset of stroke symptoms using a dedicated multivariable logistic regression analysis adjusted for the same variables.
No adjustment for multiple comparisons was made. All statistical analyses were performed using R software, version 4.1.1 (R Project for Statistical Computing).
Results
Baseline characteristics
Between May 2015 and June 2021, 1339 AIS patients were treated with tenecteplase (684 [51%] women and 655 [49%] men) in the participating centers (patients characteristics shown in Table 1). Among them, 873 patients (462 [53%] women and 411 [47%] men) also underwent MT. Successful recanalization rates did not differ between women and men (87% vs 85%, p = 0.254). Among the 1084 patients (533 [49%] women and 551 [51%] men) with known onset, only 43 patients (13 [2.4%] women and 30 [5.4%] men) received tenecteplase within 4.5–9 h according to the EXTEND criteria.
Patient characteristics according to sex subgroups.

ASPECTS: Alberta stroke program early computed tomography Score; IQR: interquartile range; LAA: large-artery atherosclerosis; MCA: middle cerebral artery; mRS: modified Rankin scale; MT: mechanical thrombectomy; NIHSS: National Institutes of Health stroke score; ODE: other determined etiology; MT: mechanical thrombectomy; mTICI: modified Thrombolysis in Cerebral Infarction score; TOAST: Trial of Org 10172 in Acute Stroke Treatment; UDE: undetermined etiology.
Data is presented as No. (%) unless indicated otherwise.
Primary outcome
In the unadjusted analysis, women had a lower rate of 3-month good outcome than men (48% vs 60%, OR 0.61 [95% CI 0.49-0.76], p < 0.001). This association was no longer statistically significant in multivariable logistic regression analyses (Figure 1, Table 2) adjusted on age and inclusion center (model 1; adjusted OR [aOR] 0.90 [95% CI 0.70-1.15], p = 0.385) or with additional variables (model 2, see Methods; aOR 0.85 [95% CI 0.63-1.13], p = 0.267). The absence of statistically significant association of sex with 3-month good outcome persisted across different subgroup analyses according to age, onset-to-IVT times, whether patients underwent MT or not, and across stroke etiologies according to the TOAST classification (Supplemental eFigure 2).
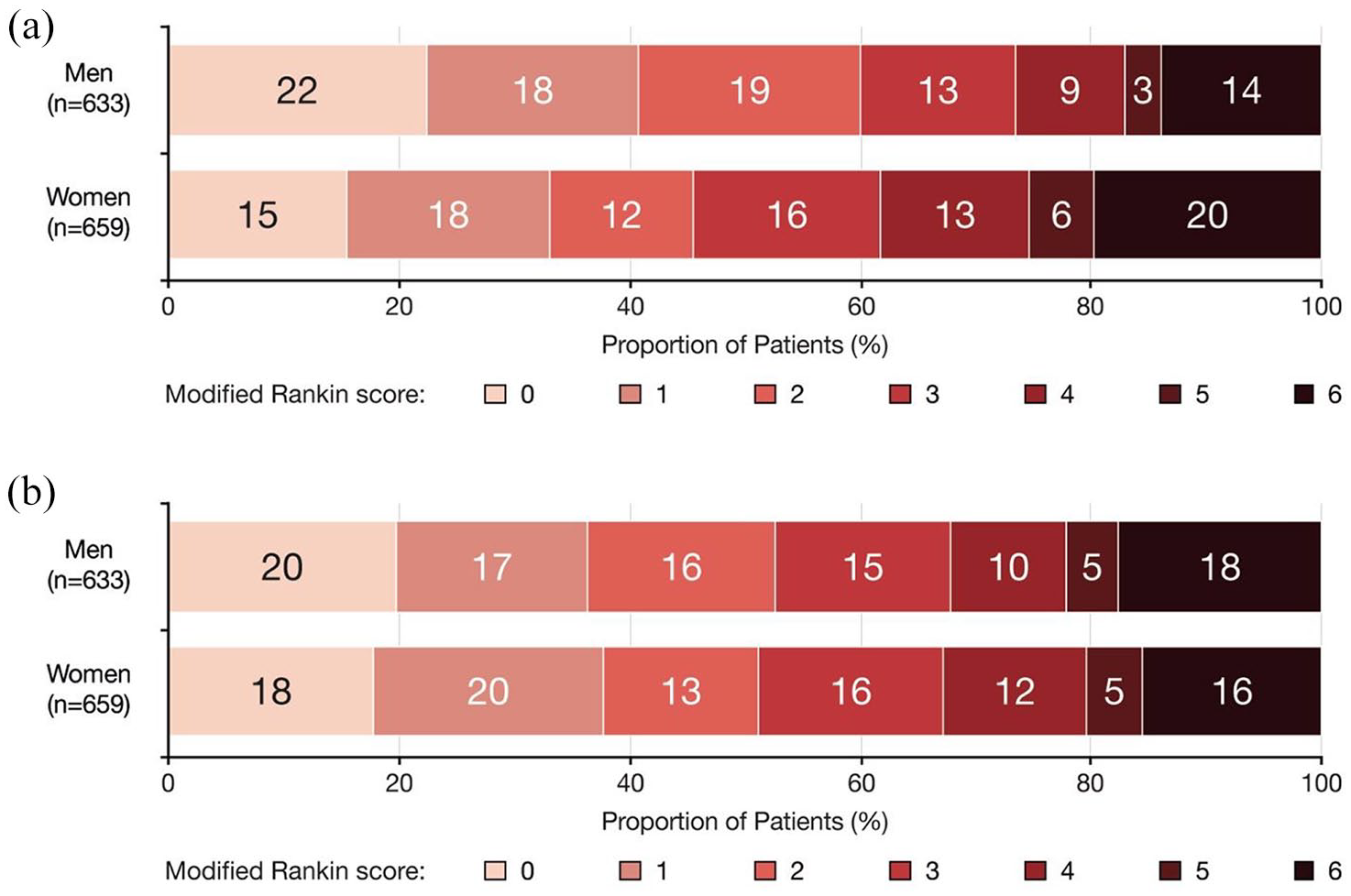
Three-month functional outcome according to sex subgroups: (a) 3-month mRS (without propensity score weighting) and (b) 3-month mRS (with propensity score weighting).
Primary and secondary outcomes.

NA: not applicable.
Data are presented as No. (%) [Number of patients with non-available information] unless indicated otherwise. Three-month good outcome was defined as a modified Rankin score (mRS) ⩽2 or stability of the pre-stroke mRS. Symptomatic intra cranial hemorrhage (sICH) was defined as an increase of at least 4 points in the NIHSS score within 36 h of IVT associated with a local or remote type 2 PH. Odd ratios were estimated with imputation of missing data (including outcomes). The weighted analysis was propensity score weighted (average treatment effect estimated).
Variables included in each model:
Model 1: age and inclusion center.
Model 2 and 3: model 1 plus baseline NIHSS, pre-stroke hypertension, pre-stroke diabetes mellitus, current smoking, history of stroke, history of myocardial infarction, pre-stroke atrial fibrillation, pre-stroke mRS, delay from onset to intravenous thrombolysis, occlusion site, treatment with MT, and stroke etiology.
For mortality and sICH analysis, if the number of events was too low to support logistic regression models with many variables, some multivariable models were not performed.
Missing data: 3-month mRS, 14 (2.1%) men and 25 (3.7%) women; sICH 6 (0.9%) men and 11 (1.6%) women.
Secondary outcomes
We performed a safety analysis of sICH rates and 3-month mortality. In the unadjusted analysis, sICH rates were nominally lower among women (2.1%, n = 22/684) compared to men (4.0%, n = 29/655; OR 0.54 [0.28–1.05], p = 0.069). This association persisted after adjusting on age and inclusion center (model 1; aOR 0.35 [95% CI 0.18-0.69], p = 0.002). Regarding 3-month mortality, while in the unadjusted analysis women had a higher mortality rate (20%, n = 130/684, OR 1.55 [1.15–2.09], p = 0.004), this association was not confirmed in the adjusted analysis (model 1; aOR 0.93 [0.67–1.28], p = 0.640).
Regarding workflow metrics, in the unadjusted analysis while both groups did not differ in terms of symptoms onset-to-IVT times (160 vs 156 min, p = 0.260), women had longer IVT-to-puncture times than men (80 vs 75 min, p = 0.043). After adjusting on age, pre-stroke mRS, baseline NIHSS score, type of intracranial occlusion and inclusion center, there were no significant differences in terms of process times between sexes (Table 3).
Process times according to sex subgroups.

IQR: interquartile range; IVT: intravenous thrombolysis; MT: mechanical thrombectomy.
Data are presented as median [IQR] (number of patients with available data) unless indicated otherwise. Analysis was adjusted on age (tertile), pre-stroke mRS (⩽2 or >2), NIHSS score at admission, type of arterial occlusion, and inclusion center.
Only for patients with a known symptom onset time.
Finally, a higher proportion of women had a stroke of unclear onset (22% vs 16%, p = 0.004) compared to men. Characteristics of patients with unknown onset are shown in Supplemental eTable 1. Both sexes did not differ in terms of median last seen well-to-IVT time (470 vs 470 min, p = 0.945). After adjusting on age, pre-stroke mRS, baseline NIHSS score, type of intracranial occlusion and inclusion center, this association with an unclear onset of symptoms remained statistically significant (aOR 1.44, 95% CI 1.07–1.95; p = 0.017).
Sensitivity analysis
For the primary and safety outcomes, we performed a propensity score-weighted sensitivity analysis (model 3) based on the same variables as model 2 (see Methods). After weighting on propensity score, all variables were well balanced (ASD < 0.1; Supplemental eFigure 1). This analysis corroborated our findings from the multivariable logistic regression analyses with no statistically significant association of sex with 3-month good outcome and 3-month mortality, and a significantly lower rate of sICH among women (Figure 1, Table 2).
Discussion
Compared to men, women suffering from AIS were older, had a higher rate of cardioembolic strokes and appeared to have poorer neurological outcome. The association of female sex with poor 3-month outcome did not persist in the adjusted analyses. Notably, we observed that women had a lower sICH rate. Once stroke code was triggered, women and men did not differ in terms of process times. However, we found that women had a higher rate of stroke of unknown onset.
In our cohort, women were older than men (median age 80 vs 69 years), with higher rates of hypertension and atrial fibrillation history and lower rates of diabetes mellitus, in line with previous studies.1,11,27 In the analyses which took into account these demographic differences (models 2 and 3), there was no statistically significant association between female sex and 3-month good outcome. Interestingly, the main driver among these factors appears to be age as adjustment on this sole factor (model 1) yields similar results. Overall, our study does not support the hypothesis of clinically relevant differences of prognosis after IVT between sexes that was mentioned by previous reports.4,9,10,15 However, we cannot rule out that our cohort is too small and underpowered to detect a potentially clinically relevant difference in response to tenecteplase between sex.
We found that women were at lower risk of sICH than men after IVT with tenecteplase. This trend is in line with previous studies on IVT with alteplase. Indeed, an analysis of a large international registry (45,079 patients) found a lower risk of sICH among women (OR 0.8 [0.66–0.96]). 9 A similar trend was reported in two meta-analyses, one on differences of outcomes after IVT between sexes and another on risk factors for hemorrhagic transformation after AIS.18,28 However, our effect size is surprising and we believe it should be interpreted cautiously given the small number of events and the lack of data from tenecteplase trials supporting such a difference.3,29 One of the hypotheses generated to explain this trend was that men were more frequently treated by antiplatelets agents prior to AIS, which are known to increase the risk of sICH. 30 However, in our analysis which indirectly accounted for this variable (model 3), sICH rate remained lower in women. Another hypothesis could be that women, who tend to have lower body weights than men, receive lower doses of tenecteplase which in turn could lead to a lower sICH rate. Unfortunately, total body weight was not collected in our registry, which may represent a significant confounder. We can also speculate that there may be other sex-intrinsic differences that could explain this difference, such as that among postmenopausal women, circulating estrogens could influence neuro-inflammation despite their lower levels and thus sICH.31,32 Overall, the underlying reasons of a potentially lower sICH rate among women remain an open question.
Finally, in this study we observed that women were more likely to have a stroke of unknown onset, even after adjusting on age, pre-stroke mRS and baseline NIHSS. A possible explanation for this finding could be that as women get older, they are more isolated than men. 33 Unfortunately, this information was not collected in our database. Overall, this finding may reflect a current evolution in an aging European country, highlighting the need to adapt home care organizations and widen the use of easily triggered remote alarms in this population. Notably, once stroke code was triggered, we did not observe any additional treatment delay for women in the adjusted analysis. However, in the unadjusted analysis the IVT-to-puncture time was slightly longer for women, which may reflect that the management of these frailer patients is more complex and require more time to discuss the risks and benefits of MT. Overall, our findings are in line with the evolving practice reported in Europe over the last past years,12,34 and a recent workflow metrics analysis of the AcT trial. 21 However, this observation is likely to be highly dependent on local cultural and societal features and cannot be extrapolated to other regions in which delayed care for stroke in women remains an ongoing concern. 13
Strengths and limitations
Our study has several strengths. It is based on a large multicentric registry of AIS patients treated with tenecteplase with a low rate of missing data. Data are homogenous concerning care in our cohort due to a unique national recruitment. Our study also has limitations inherent to its retrospective design. It was based on retrospectively collected data, with unmeasured confounding factors that may influence our results. Importantly, its relatively small sample size may preclude the detection of small but significant difference in terms of treatment response between sexes. TETRIS is a national-based registry and data concerning treatment delay cannot thus be easily extrapolated to other areas. Finally, given that only patients treated with tenecteplase are included in TETRIS, our conclusions on the impact of sex on stroke outcome are not relevant for other stroke patients (patients not treated with IVT or IVT with other thrombolytic).
Conclusion
After adjustment on potential confounders, we did not observe a significant difference in terms of 3-month neurological outcome between women and men suffering from AIS who received tenecteplase. However, the existence of a clinically relevant difference that our sample size would be insufficient to detect cannot be excluded. In our analysis, we also observed that women have a lower sICH rate than men in this cohort. Women were at higher risk to have a stroke of unclear onset compared to men. However, once stroke code was triggered workflow metrics were similar between sexes.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251332497 – Supplemental material for Sex differences in outcomes and time to treatment in stroke patients treated with tenecteplase
Supplemental material, sj-docx-1-eso-10.1177_23969873251332497 for Sex differences in outcomes and time to treatment in stroke patients treated with tenecteplase by Marion Yger, Edouard Januel, Thomas Checkouri, Nicolas Chausson, Didier Smadja, Jildaz Caroff, Stéphane Olindo, Gaultier Marnat, Emmanuel Wiener, Fernando Pico, Guillaume Turc, Wagih Ben Hassen, Pierre Seners, Michel Piotin, Frédéric Clarençon, Sonia Alamowitch and Gaspard Gerschenfeld in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All reported disclosures were outside the submitted work. Dr Yger reports personal fees from Lundbeck. Dr. Marnat reports personal fees from Stryker, Medtronic and Microvention. Prof. Clarençon reports personal fees from Medtronic, Guerbet, Balt Extrusion, and Penumbra. Dr. Chausson reports a grant and personal fees from Boehringer Ingelheim and Bristol Myers Squibb and personal fees from Stryker. Prof. Sibon reports personal fees from Astra-Zeneca, Bayer, BMS-Pfizer, Boehringer Ingelheim, Elsevier, Novonordisk, Servier, and Medtronic. Prof. Alamowitch reports personal fees from the Astra-Zeneca, Bayer, BMS-Pfizer, and Elsevier. Dr Gerschenfeld reports personal fees from Truffle Capital. No other disclosures were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has received funding from the DMU Neurosciences of the APHP. Sorbonne Université and the “Investissements d’avenir” ANR-10-IAIHU-06.
Informed consent
As per current French law regarding retrospective studies of anonymized standard care data, patients were informed of their participation in this research and offered the possibility to withdraw, but no written consent was required.
Ethical approval
This research was approved by the Sorbonne University Research Ethics Committee (CER-2021-1053).
Guarantor
GG
Contributorship
MY, EJ, GG and SA researched literature and conceived the study. MY, GG, and SA were involved in protocol development and gaining ethical approval. MY, TC, NC, DS, JC, SO, GM, EW, FP, GT, WBH, PS, MP, FC, and GG were involved in patient recruitment and data collection. MY, EJ, GG, and SA were involved in data analysis. MY, EJ, and GG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
ORCID iDs
Trial registration
TETRIS Registry, NCT05534360
Data sharing statement
The data that support the findings of this study are available from the corresponding author upon agreeing to a data sharing agreement.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
