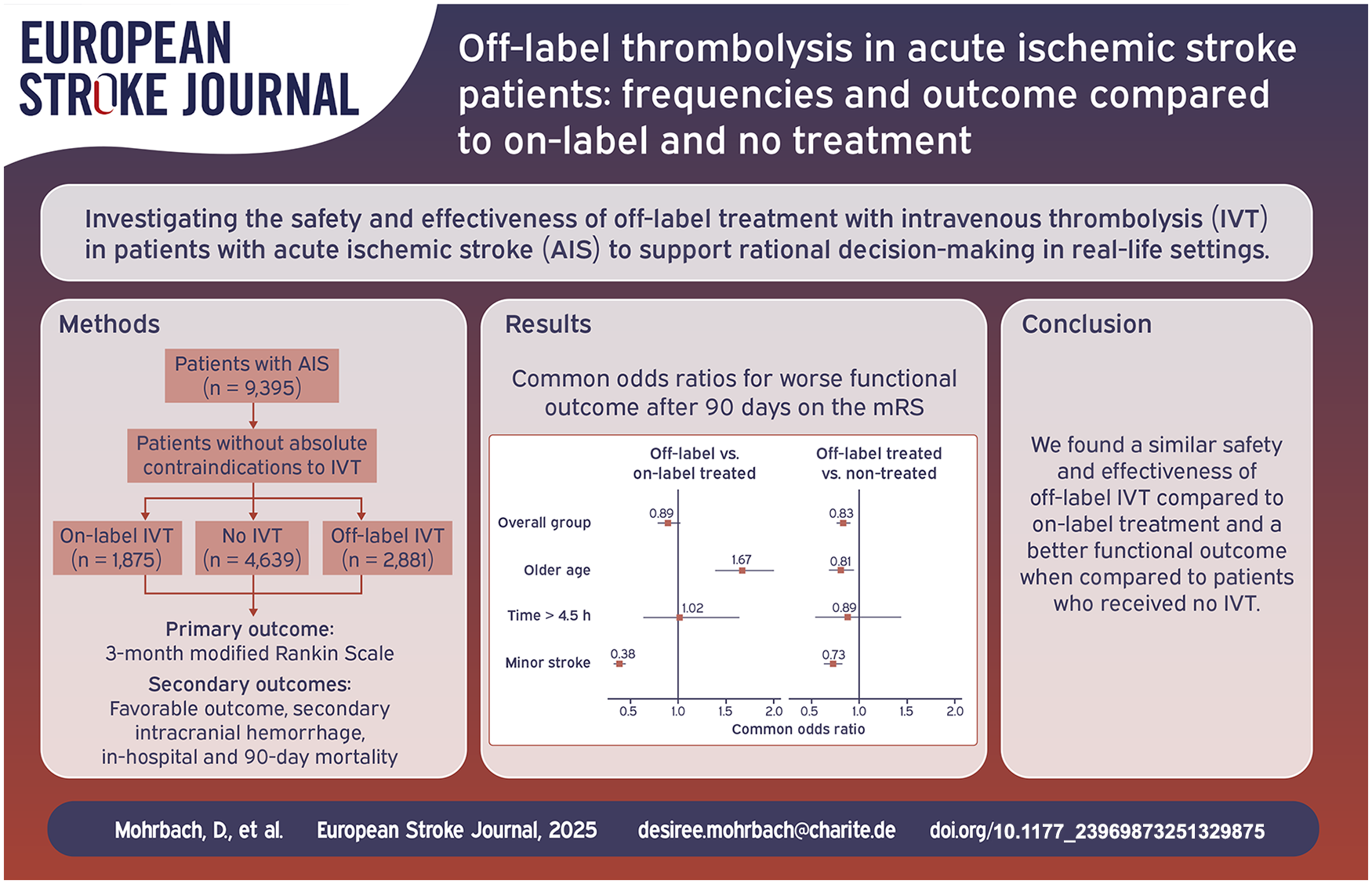
Abstract
Background:
Intravenous thrombolysis (IVT) is not approved for acute ischemic stroke in certain patient groups due to in- and exclusion criteria from mostly early clinical trials. In a real-life setting, we analyzed frequencies, outcomes and complications of off-label-IVT compared to on-label-IVT and no IVT to contribute to rational decision-making regarding patient selection for IVT.
Methods:
Patients in the Berlin Specific Acute Therapy in Ischemic or hAemorrhagic stroke with Long-term follow-up registry were divided into three groups (on-label, off-label, non-IVT-treated). Off-label criteria were age > 80 years, time from onset >4.5 h, mild (National Institutes of Health Stroke Scale [NIHSS] < 5) or severe (NIHSS > 25) stroke, and a history of prior stroke and diabetes mellitus, as those criteria applied during the study period. Outcomes of interest were the 90-day modified Rankin Scale (mRS), symptomatic intracranial hemorrhage (sICH), in-hospital- and 90-day-mortality. After multiple imputation, we performed multivariable logistic regression to calculate adjusted common odds ratios (cOR) with 95% confidence intervals (CI) for mRS and risk ratios for secondary outcome measures.
Results:
We analyzed 1875 on-label, 2881 off-label, and 4639 non-treated patients. The most common off-label-criteria were mild symptoms (44.6%) and older age (33.6%). Off-label-treated patients had no worse functional outcome when compared to on-label-treated patients (cOR 0.89, CI 0.78–1.02), but better outcome compared to non-treated patients (cOR 0.83, CI 0.76–0.91). No associations were found for sICH or mortality.
Conclusions:
Our results suggest a beneficial effect of off-label treatment in clinical practice, which warrants further investigation into treatment in relevant subgroups and an extension of the product license.
Introduction
Over the past decades, the use of alteplase for IVT (intravenous thrombolysis) of AIS (acute ischemic stroke) has become part of clinical routines and increased in frequency with broader patient selection. 1 This trend is mirrored in the guidelines for AIS management, with an extension of patient eligibility criteria for IVT. 2 Eligibility criteria for the European product license for Actilyse© (alteplase) being effective at the start time of our study were principally based on in- and exclusion criteria of the early randomized controlled trials. 3 Even though some changes have been made to the product license, it has not been updated according to findings of more recent trials and guideline recommendations.2,4 This may in part be related to concerns as to how far the results for the safety and efficacy of IVT are applicable to daily clinical practice.1,5
The heterogeneity of daily practice in IVT of AIS has been addressed in many studies investigating its benefits and potential harms in different patient groups.3,4,6 Outcomes of off-label treatment have only been investigated in a small number of studies and part of these studies only evaluated short-term or safety outcomes.7–12 Further, analyses were conducted on off-label versus on-label treatment, disregarding patients not receiving IVT. In order to monitor and improve healthcare of AIS patients, it is critical that both on-label and off-label use as well as non-treated patients are considered.
In the current study, we analyzed the frequency, complications and 3-month functional outcome of off-label treatment with IVT in general as well as for each off-label reason separately in patients admitted to Berlin-based hospitals. Our aim is to support rational decision-making in a real-life setting regarding patient selection for IVT.
Methods
Study design and setting
B-SPATIAL (Berlin Specific Acute Therapy in Ischemic or hAemorrhagic stroke with Long-term follow-up, Clinicaltrials.gov identifier: NCT03027453) is a prospective, multicenter registry of patients with AIS or hemorrhagic stroke admitted to 15 Berlin-based hospital with a stroke-unit meeting the following inclusion criteria: age ⩾ 18 years, symptom onset within 6 h prior to hospital arrival, and persistent neurological symptoms at ambulance or hospital arrival. Data was collected by a team of dedicated study assistants. Patients were not required to sign an informed consent prior to inclusion and received a follow-up call or written questionnaire to assess the modified Rankin Scale (mRS) after 3 months, when they could also decide to opt out. Objection to the use of their complete data was possible at multiple points.
We included data from patients admitted between January 2016 and December 2021 with the discharge diagnosis according to ICD-10 (International Statistical Classification of Diseases and Related Health Problems) of acute ischemic stroke (I63). We excluded all patients with an interruption of IVT or missing data for IVT. As we considered the time period between 2016 and 2021, we used the European product license for Actilyse© from 2018 as reference. 13 Accordingly, off-label criteria were defined as time to treatment between 4.5 and 6 h (from symptom onset or last-seen-well), age > 80 years, mild symptoms (using the ECASS II definition with a NIHSS score < 5, as the product license does not mention a specific cut-off), 14 severe stroke with a NIHSS (National Institutes of Health Stroke Scale) score > 25, or a combination of diabetes and prior stroke. Patients who met at least one of the off-label criteria were assigned to the off-label group if they had received IVT, or to the no-treatment group if they had not received thrombolytic treatment. On-label use was defined as treatment in patients who met none of the previously defined off-label criteria. In case data were not complete on one of those criteria, patients were considered to be on-label for the missing criterion.
Measurements
We analyzed the outcome of off-label treatment compared to on-label and no-treatment, in respective analyses. Our primary outcome measure was the mRS 90 days after the event. Secondary outcome measures were favorable clinical outcome (mRS 0–2), symptomatic intracranial hemorrhage (sICH), in-hospital mortality and 3-month mortality, by using a score of 6 on the mRS in the follow-up. SICH was reported and defined as any new hemorrhage on brain imaging in the context of any decline in neurological status. In Germany, a (possible) causal relationship is usually considered during the first 36 h after initiation of thrombolysis.
Statistical analysis
We used STATA© version 17.0 for the statistical analyses and graphs.
For our analyses, we first conducted analyses on the overall treatment groups (on-label, off-label, and no-treatment) and subsequently subdivided the off-label and no-treatment group into five categories (i.e. representing the respective non-label criterion, as well as one category for patients with multiple reasons of off-label use). We used different comparison groups as the reference group, depending on the analysis. For the on-label versus off-label analyses, the overall on-label group was used as a reference category, as subdivision was not possible. For our analyses regarding patients off-label and no-treatment, analyses were conducted on the overall group, as well as each subgroup.
In all analyses focusing on the mRS, we calculated unadjusted and adjusted common odds ratios (cOR) for higher mRS as well as their corresponding 95% confidence intervals (CI) through ordinal logistic regression. Potential sources of confounding were addressed by propensity score (PS) adjustment. For each comparison, we calculated a different PS, as the score is dependent on the observations included in the analysis. The PS indicates a patient’s probability of being exposed (i.e. receiving treatment) by combining the information measured from all confounders in one score. The set of variables to create the PS was the same, and included age (continuous), sex (binary), previous care dependency (binary), previous walking ability (binary), performed additional endovascular treatment (ET, binary), NIHSS score at admission (continuous), and time from onset to treatment (in hours; only for the on-label vs off-label analyses). We performed multiple imputation for mRS at 3 months and confounding variables. Sensitivity analyses were performed by excluding patients who received ET, as well as excluding older age from off-label criteria, according to the updated product license from June 2018.
For the analyses focusing on binary outcome measures (i.e. favorable clinical outcome, sICH, in-hospital and 3-month mortality), we used generalized linear modelling with log-link to obtain Risk Ratios (RR) and their corresponding 95% CI. We adjusted for age, sex, ET, NIHSS score at admission, pre-stroke walking ability and care dependency, as well as time from onset to treatment (for the off-label vs on-label analyses).
Results
Between January 2016 and December 2021, a total of 14,425 patients were registered in B-SPATIAL. Of those, 10,560 patients had suffered AIS. A total of 9395 patients met our selection criteria and were subject to our analyses (1875 in the on-label, 2881 in the off-label, and 4639 in the non-treated group). The selection of patients according to diagnosis and IVT is shown in Figure 1. Baseline data are presented in Table 1. In short, the overall cohort had a mean age of 73 years. Three-month mRS follow-up was available in 78.2% of patients.
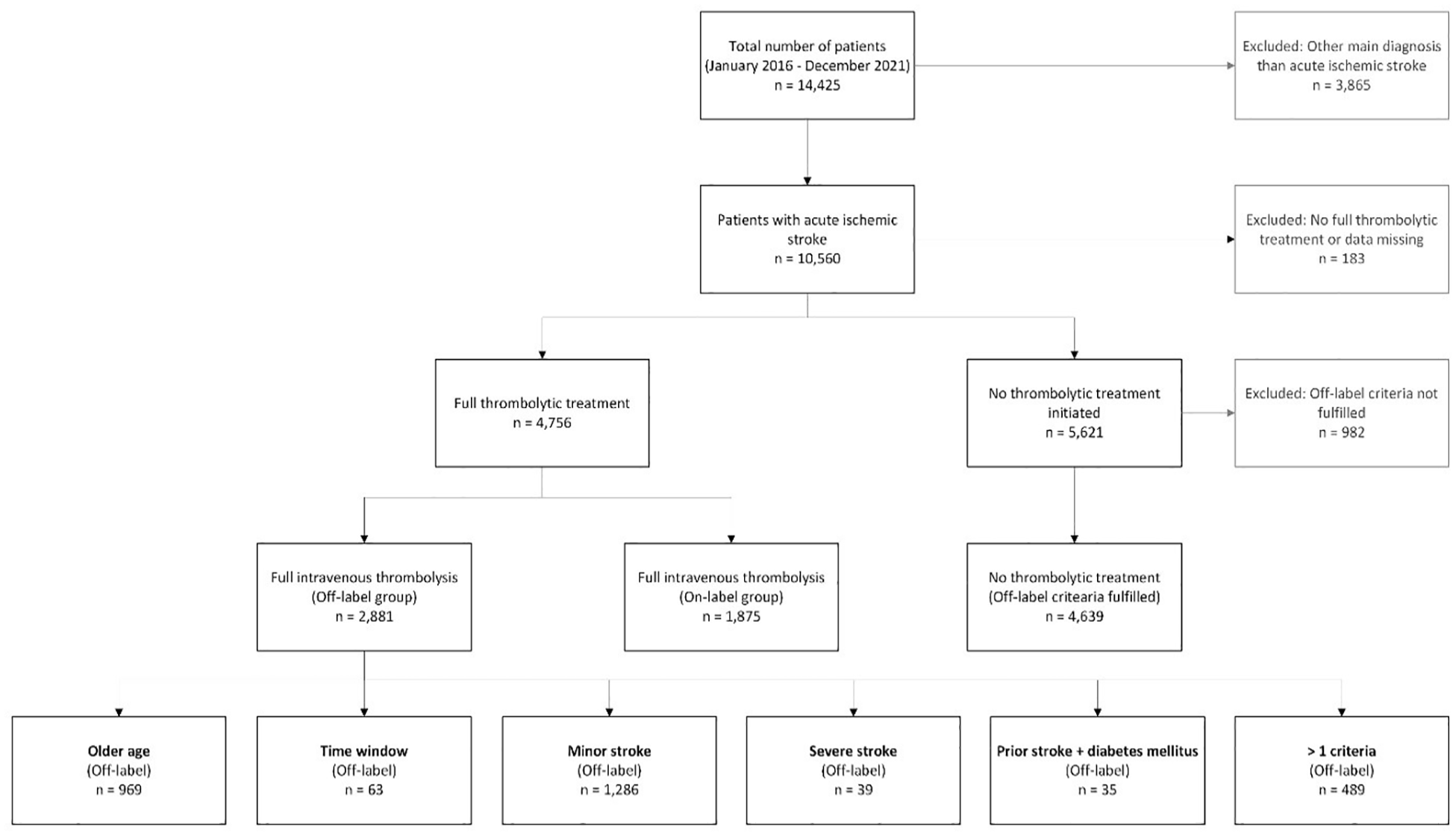
Selection process of patients.
Baseline clinical and treatment characteristics for all patients as well as the overall treatment groups.
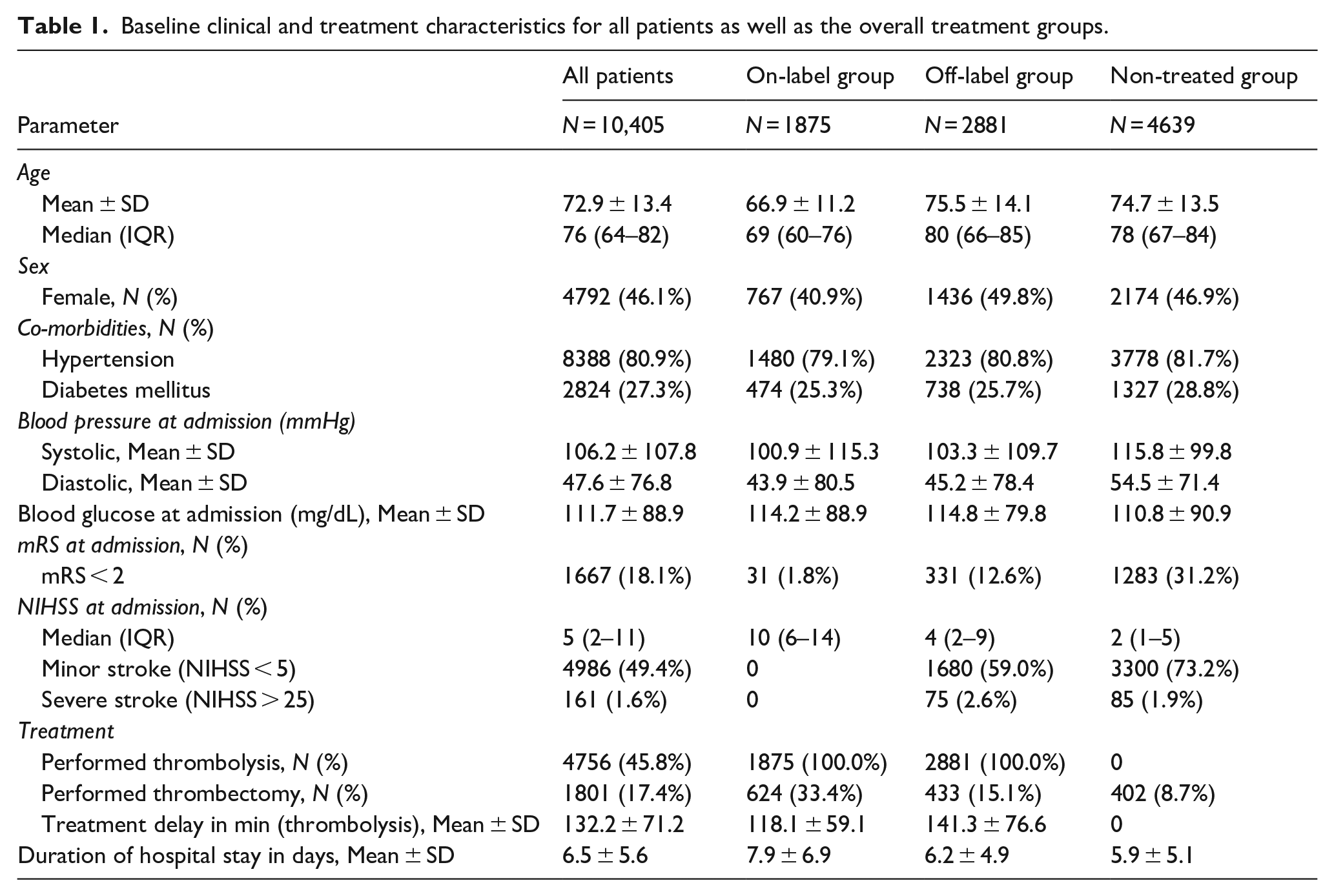
Regarding stroke severity, almost half of patients (49.4%,
Patient demographics slightly differed when comparing on- and off-label as well as no-treatment groups. The off-label group had a higher proportion of females compared to the on-label group (49.8%,
Treatment rates
Of all patients, 45.8% (
Of those patients who received IVT, 60.6% (
Off-label treatment and functional outcome
Off- versus on-label patient groups
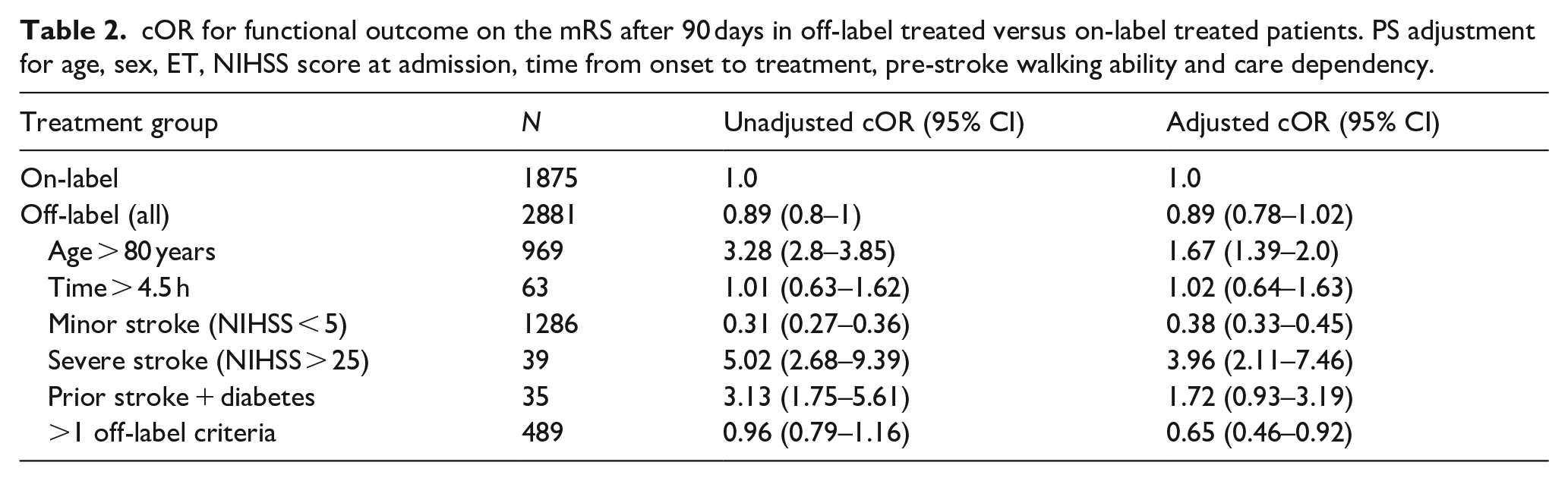
The off-label group had no statistically significant different clinical outcome compared to the on-label group (adjusted cOR 0.89, CI 0.78–1.02). However, outcomes varied between subgroups. Unadjusted and adjusted cOR as well as their respective CI for the overall as well as each subgroup are presented in Table 2. Results were mostly unchanged after sensitivity analyses (see Table A and C in Supplemental Appendix).
cOR for functional outcome on the mRS after 90 days in off-label treated versus on-label treated patients. PS adjustment for age, sex, ET, NIHSS score at admission, time from onset to treatment, pre-stroke walking ability and care dependency.
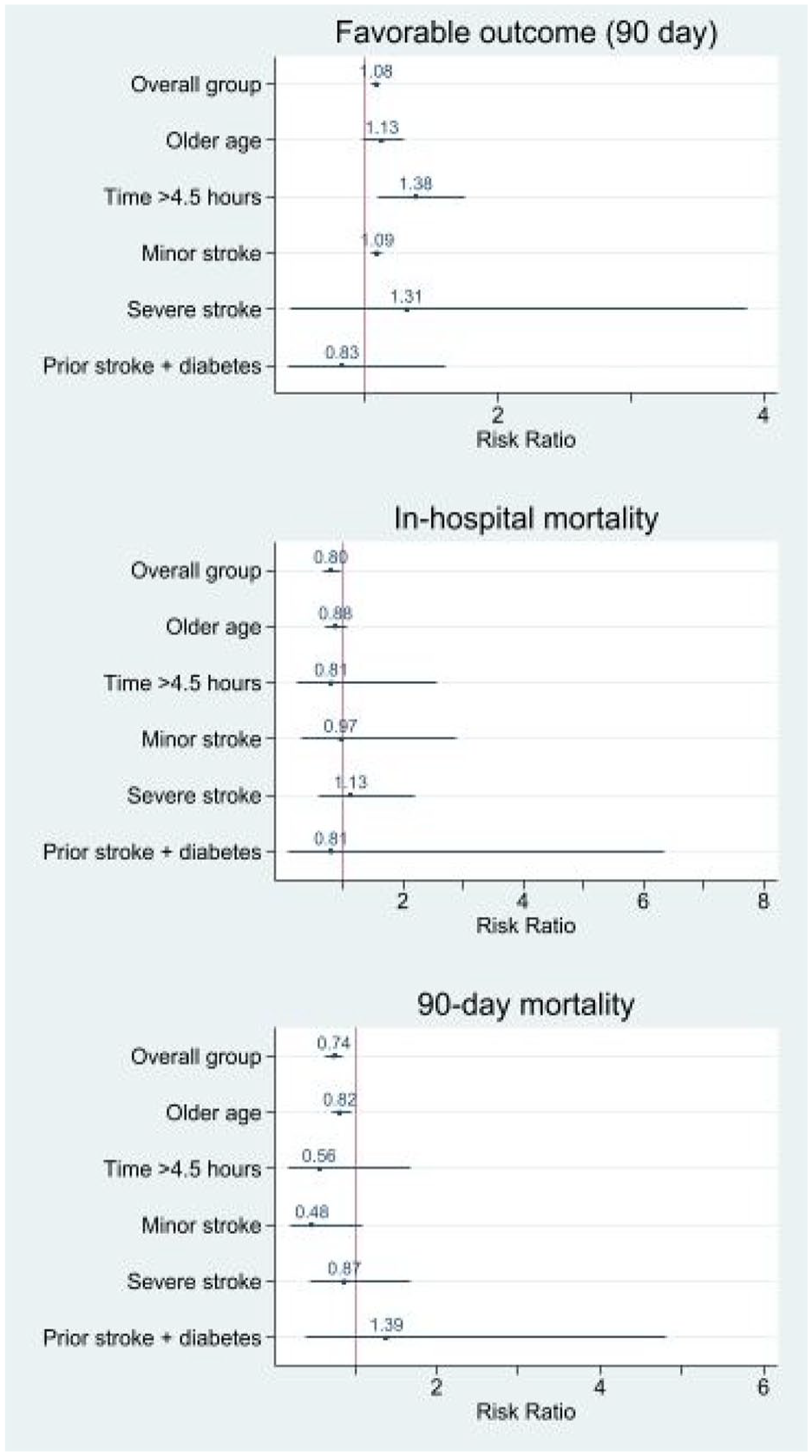
Off-label versus non-treated patients
Patients treated off-label had in general a better 3-month outcome when compared to patients who met one of the non-label criteria and did not receive IVT, as there were lower odds for a high score on the mRS at 90 days (adjusted cOR 0.83, CI 0.76–0.91). The results for the overall as well as each subgroup are presented in Table 3. Sensitivity analyses did not lead to meaningful differences (see Table B and D in Supplemental Appendix).
cOR for functional outcome on the mRS after 90 days in off-label treated versus non-treated patients. PS adjustment for age, sex, ET, NIHSS score at admission, pre-stroke walking ability and care dependency.
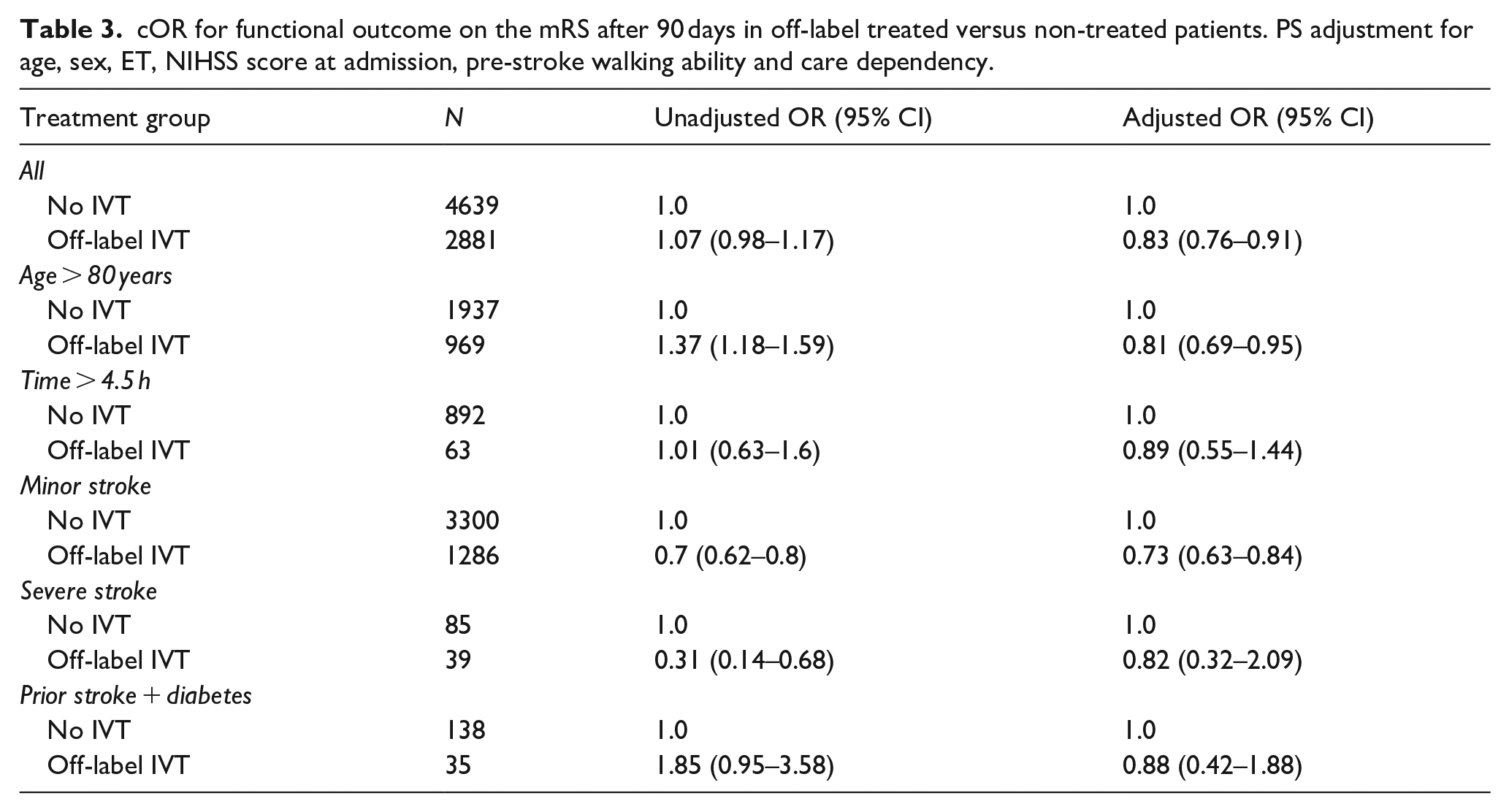
Secondary outcome measures
We obtained similar results for secondary outcome measures (favorable clinical outcome, sICH, in-hospital mortality, 3-month mortality), which are shown in Figure 2. Overall, the off-label group had no significant difference in the probability of favorable functional outcome at 90 days, sICH, in-hospital mortality and 3-month mortality when compared to the on-label group.
Calculated RR for secondary outcome measures in patients treated off-label versus on-label. Adjusted for age, sex, time from onset to treatment, NIHSS score at admission, ET, pre-stroke walking ability and care dependency.
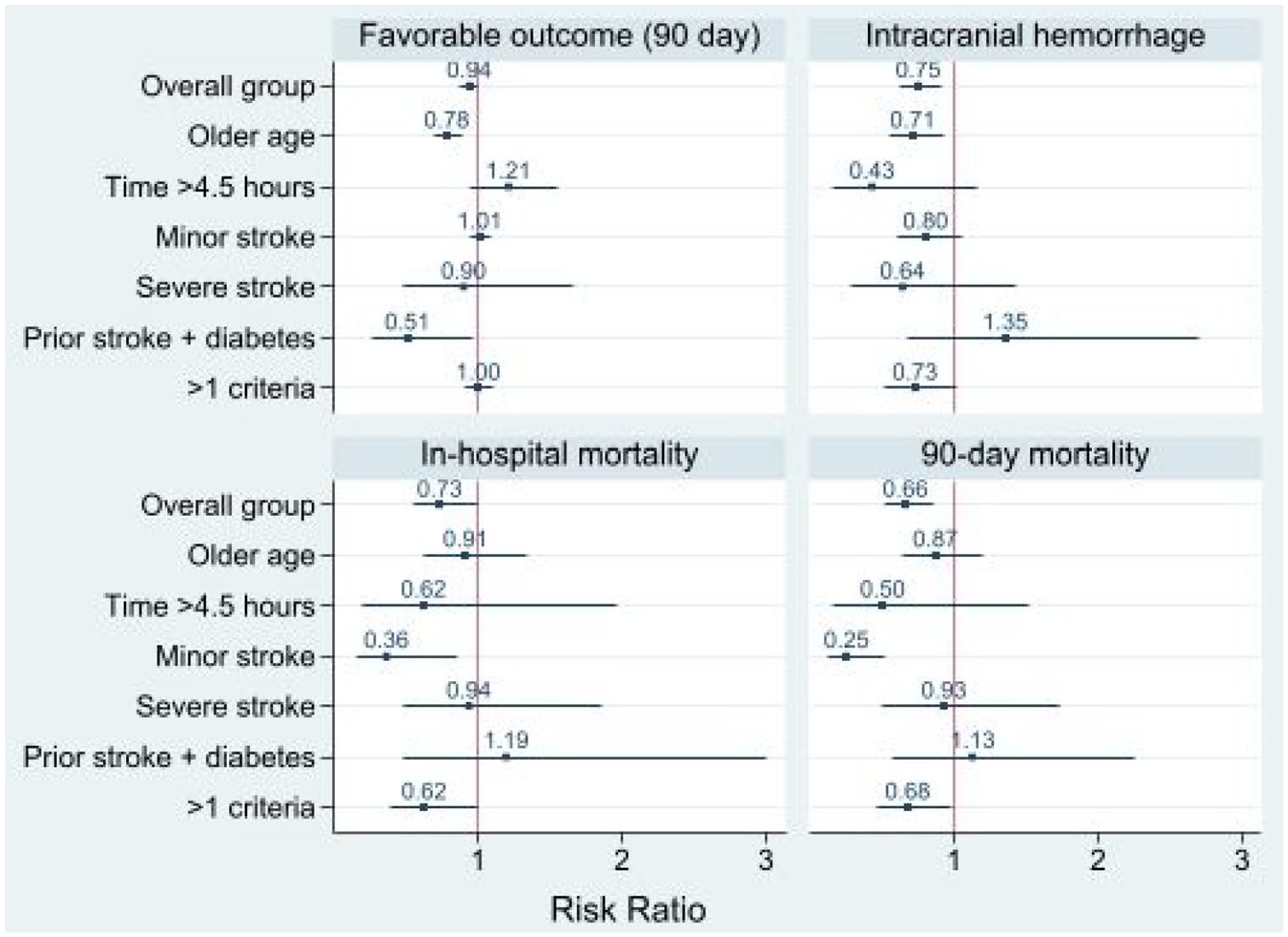
Compared to patients not treated with IVT, off-label treated patients had a higher probability of a good outcome and a lower risk regarding 90-day mortality. Adjusted RR for the subgroups are shown in Figure 3.
RR for secondary outcomes in off-label treated versus non-treated patients. Adjusted for age, sex, NIHSS score at admission, ET, pre-stroke walking ability and care dependency.
Discussion
In this Berlin-based registry, almost half of AIS patients presenting with neurological symptoms within less than 6 h from onset received IVT. Interestingly, most of these patients had at least one of the pre-defined criteria that did not allow IVT according to European licensing criteria. The most common reasons for off-label treatment were mild stroke and older age. Overall, our data suggest that the off-label group did not have worse outcomes compared to the on-label group, but better outcomes than the non-treated group.
Studies comparing off-label treatment with non-IVT-treated patients are scarce. Our data are in line with previous studies reporting an increased off-label use of IVT, particularly in patients >80 years or minor stroke.1,8,15 With regard to beneficial and harmful effects, our findings partially confirm results from previous studies, suggesting that off-label treatment has similar outcomes as on-label treatment after adjustment for confounders.6,9–11,16,17 A clear benefit of IVT was seen in the randomized IST-3 trial. 4 We found a beneficial effect of IVT in patients with minor stroke, which at first sight might be seen as contradicting the results of the PRISMS trial that did not find better outcomes in patients with minor, non-disabling strokes. 18 Of note, NIHSS < 5 did not mean ‘non-disabling’ in our registry. Further, we found higher odds for a worse 90-day functional outcome in older age compared to treatment according to the label, which was contrary to findings from ISTR, VISTA and Bluhmki et al., 9,19,20 but found lower odds for a worse outcome when compared to no treatment. Our data was further consistent with SITS-ISTR regarding patients who received IVT within 4.5 to 6 h, as we did not find worse functional outcome at 3-months. 11 Contrary to data from ISTR and to the individual participant data meta-analysis of randomized controlled trials by Emberson et al., 6 patients with severe stroke had worse functional outcome when compared to the on-label group, but not when compared to the group not treated. The association of a combination of prior stroke and diabetes or a violation of multiple on-label criteria has been investigated rarely so far, but indicated that off-label treated patients with such characteristics have no higher probability for worse functional outcome or IVT-specific safety outcomes.12,21 Our present results show no evidence for a beneficial or harmful effect of IVT in patients with prior stroke and diabetes compared to patients treated on-label or patients not treated.
Overall, our data partly endorse findings from previous studies and suggest that product license restrictions should be reconsidered, as there is no evidence that IVT is associated with increased risks or attenuated effects regarding those off-label criteria.
As the product license does not mention a clear cut-off as to which NIHSS score or symptoms are to be classified as ‘minor’ stroke, we used the ECASS II definition with a NIHSS score < 5, as this definition was also used in other trials. 22 However, this cut-off does not exactly mirror real-life deficit severity. In fact, it is common practice that AIS patients should be treated if stroke related symptoms are disabling, regardless of the NIHSS score.
During the completion of this study, there have been adjustments to the product license. While in the first half of 2018, alteplase was not recommended for patients older than 80 years, it is now recommended for patients irrespective of their age. 13 We used the previous product license for our off-label subgroups as this was the available label during the course of the present study.
Strengths and limitations
A major strength of our registry is the uniform data collection in all hospitals with a stroke unit in Berlin. The results of the present study reflect the current state of IVT and its application in a real-life setting. Proportions of patients declining any use of their data or opting against follow-up assessment were low, and functional outcome follow-up rates were high compared to other registries.
As we made use of a registry-based data collection, the quality of our results highly depends on the accuracy of collection and documentation of routine care data. Given the observational nature of registries, there is some variation in IVT protocols, technical equipment, and staff between hospitals, which could possibly affect outcomes. We cannot make firm conclusions regarding true treatment effects due to the strong likelihood of confounding even after adjustment. This applies particularly to unobserved or undocumented factors that are present during the process of therapeutic decision-making and are summarized as ‘bias by indication’, such as refusal against treatment or mismatch- or penumbra-imaging, which has been a valuable decision tool regarding an extension of the time window for IVT application. 23 We note that we do not have information on whether patients with symptom onset or last-seen-well >4.5 h were treated based on advanced (mismatch) imaging. Still, we believe that the present methods represent a legitimate approach to investigate differences in possible risks and benefits of off-label IVT in various groups. Another limitation is that other contraindications were not included in the definition of the off-label group since these criteria are usually accepted as contraindications in guidelines and routine care. Furthermore, while we assigned patients treated after 4.5 h from onset to the off-label group, the inclusion criteria of B-SPATIAL (arrival to hospital within 6 h from onset) imposed a limit to the time window regarded. We therefore have no information on patients who were treated after 6 h from stroke onset. A final limitation is that our data were derived from Berlin-based hospitals, hence it does not necessarily reflect nationwide or international trends and routine use of IVT in AIS patients. However, similar trends have been reported from other research groups as well.1,8,11 Finally, the groups of patients with >4.5 h since onset or last seen well, NIHSS > 25 or the combination of prior stroke and diabetes mellitus were too small to draw firm conclusions.
Conclusion
More than half of AIS patients receiving IVT were treated ‘off-label’ according to the 2018 EU product license. Overall, our results suggest a similar safety and effectiveness compared to on-label treatment, and a better functional outcome when compared to patients who received no IVT. Our data provide valuable guidance for real-life treatment practices, where we suggest a broader patient selection for IVT. Our findings further add arguments that the product license should be updated in order to offer this effective treatment to more patients.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251329875 – Supplemental material for Off-label thrombolysis in acute ischemic stroke patients: Frequencies and outcome compared to on-label and no treatment
Supplemental material, sj-docx-1-eso-10.1177_23969873251329875 for Off-label thrombolysis in acute ischemic stroke patients: Frequencies and outcome compared to on-label and no treatment by Désirée Mohrbach, Lora Wagner, Christian H Nolte, Martin Ebinger, Irina Lorenz-Meyer, Ira Rohrpasser-Napierkowski, Peter Harmel, Georg Hagemann, Bruno-Marcel Mackert, Hans-Christian Koennecke, Bob Siegerink and Heinrich J Audebert in European Stroke Journal
Footnotes
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HJA received research grants from German Ministry of Research and Education and the German Research Foundation for the B-SPATIAL registry and B_PROUD study, and personal fees from Boehringer Ingelheim. CHN received research grants from German Ministry of Research and Education, German Center for Neurodegenerative Diseases, German Center for cardiovascular Research. All other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B-SPATIAL was funded by the Federal Ministry of Education and Research, German Ministry of Research and Education and the German Research Foundation granted to the Center for Stroke Research Berlin. The funder had no role in study design, analysis of the data, interpretation of the results, or drafting of the text.
Ethical approval
The B-SPATIAL study was approved by the local ethics committee of the Charité–University Medicine Berlin (EA1/208/21).
Informed consent
Since B-SPATIAL is a registry focused on the quality of care, patients were not required to sign an informed consent prior to inclusion. However, for follow-up procedures, patients or their legal representatives could decide to either opt out or object completely to the use of their data at several time points, in accordance with local data protection rules and regulations. Consent was not collected for specific research projects.
Trial registration
Not applicable.
Guarantor
Désirée Mohrbach.
Contributorship
DM, HJA and BS conceptualized the study. CHN, ME, ILM, IRN, PH, GH, BMM, HCK and HJA were involved in data acquisition. The statistical analyses were planned and conducted by DM and BS, also in consultation with LW. DM, HJA and BS interpreted the results and drafted the manuscript. DM created all figures and tables. HJA provided funding for the project and as well as project supervision together with BS. All authors critically revised the final version of the manuscript.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
