Abstract
Introduction:
Evidence on endovascular therapy (EVT) for symptomatic intracranial stenosis (sICAS) from randomized-controlled clinical trials (RCTs) is conflicting. While prior RCTs on percutaneous transluminal angioplasty and stenting (PTAS) demonstrated harm or no benefit over best medical treatment (BMT), recent data suggest that submaximal balloon angioplasty with BMT may be superior to BMT alone.
Patients and methods:
A systematic review and meta-analysis of RCTs was conducted to evaluate the safety and efficacy of elective EVT plus BMT compared to BMT alone for sICAS.
Results:
Six RCTs (5 on PTAS and 1 on balloon-angioplasty) comprising 1606 patients were included. EVT increased the risk of any stroke or death (RR = 2.68; 95% CI: 1.72–4.19; I2 = 0%), ischemic stroke within the territory of the qualifying artery (RR = 2.51; 95% CI: 1.36–4.61; I2 = 0%), any ischemic stroke (RR = 1.99; 95% CI: 1.17–3.38; I2 = 0%), intracranial hemorrhage (RR = 6.23; 95% CI: 1.92–20.2; I2 = 0%), and mortality (RR = 3.52; 95% CI: 1.04–11.88; I2 = 0%) within 30 days. No significant benefit from EVT was detected regarding the risk of any stroke or death (RR = 0.29, 95% CI: 0.06–1.38; I2 = 68%), ischemic stroke in the territory of the qualifying artery (RR = 0.44, 95% CI: 0.14–1.33; I2 = 59%) and mortality (RR = 0.49, 95% CI: 0.16–1.55; I2 = 0%) beyond 30 days through 1 year.
Discussion and conclusion:
EVT is associated with adverse early outcomes, without reducing the risk of long-term stroke recurrence or mortality compared to BMT. Further research is warranted to identify high-risk subgroups who may benefit from EVT for sICAS and refine interventions to minimize periprocedural risks.

Keywords
Introduction
Intracranial atherosclerotic stenosis (ICAS) represents a major cause of acute ischemic stroke globally, with a nearly twofold higher prevalence among Asian compared to non-Asian populations and a high risk of stroke recurrence, affecting up to one-third of high-risk patients within 1 year.1,2 Despite risk factor modification and best medical treatment (BMT), the risk of breakthrough or recurrent stroke remains exceptionally high, leading to increased mortality and disability rates, as well as incremental healthcare costs, particularly in high-prevalence countries.3,4 Deployment of percutaneous transluminal angioplasty and stenting (PTAS) for symptomatic ICAS (sICAS) has fueled widespread interest in the past years, though enthusiasm has gradually receded, as randomized-controlled clinical trials (RCTs) have largely disproven the superiority of PTAS over BMT. 5
With the accrual of RCT data, factors associated with increased risk of stroke recurrence, including hemodynamic compromise, poor collateral status, and border zone or arterio-arterial embolic infarct patterns, have been increasingly recognized and utilized for selection of high-risk patients that are most likely to benefit from endovascular therapy (EVT).4,6,7 In addition, leveraging insights gained from early RCTs (e.g. the SAMMPRIS: Stenting and Aggressive Medical Therapy for Preventing Recurrent Stroke in Intracranial Stenosis trial), 6 standardized BMT approaches – now consolidated in clinical practice guidelines – have been implemented to ensure balanced and stringent control of risk factors in both interventional and control arms.7,8 With increasing awareness that the potential benefits of EVT in earlier trials may have been offset by periprocedural risks linked to interventionalists’ skills or specific devices (e.g. Wingspan stent), 9 robust selection criteria (e.g. high-volume sites and experienced interventionalists), newer-generation stents, and novel interventional approaches have been investigated to optimize outcomes.
In East Asia, sICAS accounts for 30%–50% of ischemic strokes, compared to 5%–10% in Western populations, largely attributable to genetic predisposition, environmental factors (e.g. high dietary sodium intake), and vascular risk factors (e.g. hypertension).1,10 Despite ongoing efforts to optimize risk factor management and adherence to BMT, real-world data outside RCTs indicate suboptimal control of vascular risk factors, particularly hypertension, alongside a rising incidence of sICAS and recurrent stroke.11,12 Given the substantial socioeconomic burden imposed by these epidemiological trends, there is an increasing real-world shift toward EVT in East Asian countries, driving growing expertise in angioplasty and stenting among neurointerventionalists. 13
Recently, the publication of the Balloon Angioplasty for Symptomatic Intracranial Artery Stenosis (BASIS) trial has reignited controversy regarding the potential efficacy of EVT in sICAS. 14 Submaximal balloon angioplasty without stenting has long been debated as a less invasive alternative to PTAS, minimizing arterial wall trauma and periprocedural risk, while achieving sufficient lumen widening to restore blood flow and attenuating stroke recurrence. 15 BASIS, a randomized, open-label, blinded-endpoint trial conducted at 31 centers across China, investigated submaximal balloon angioplasty plus BMT versus BMT alone in 512 patients with stroke or transient ischemic attack (TIA) due to severe sICAS (70%–99% stenosis). In this trial, balloon angioplasty significantly reduced the risk of the composite outcome of any stroke or death within 30 days, or ischemic stroke or revascularization of the qualifying artery after 30 days through 12 months. 14
A previous meta-analysis by our group, 16 which incorporated data from RCTs comparing PTAS plus BMT with BMT alone, identified significantly elevated risks of ischemic stroke, intracranial hemorrhage, and mortality associated with PTAS. In light of the recently published BASIS results, the current systematic review and meta-analysis aims to synthesize updated evidence from all available RCTs, providing a comprehensive assessment of the safety and efficacy of elective EVT in comparison with BMT alone in sICAS.
Methods
Standard protocol approvals and registrations
This systematic review and meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 17 As per study design (systematic review and meta-analysis), approval from an Ethics Committee was not required. The study protocol, incorporating a predefined PICOS framework (Population, Intervention, Comparison, Outcome, and Study design), was developed prospectively and registered with the PROSPERO database (CRD42024628023). The authors affirm that all data supporting the results are provided within the manuscript and its Supplemental Materials.
Data sources and searches
In this systematic review and meta-analysis, four independent reviewers (MIS, EP, LP, AT) conducted a literature search for published RCTs evaluating the efficacy and safety of elective EVT plus BMT compared to BMT alone in patients with sICAS. Eligible RCTs were identified through a systematic search of the MEDLINE (via PubMed) and Scopus databases, conducted on November 29th, 2024. The search terms used across all databases included: “angioplasty,” “stent,” “medical,” “intracranial,” “vertebral,” “middle cerebral,” “basilar,” “stenosis,” “atherosclerosis,” “atherosclerotic,” “stroke,” “cerebral ischemia,” and “randomized controlled trial.” No language or other restrictions were applied. The complete search algorithm for all databases is provided in the Supplement.
Following the identification of relevant studies, a systematic screening process was conducted based on the titles and abstracts of the articles. The following inclusion criteria were applied: (i) studies involving patients with intracranial stenosis of 50%–99%,7,18 (ii) studies focusing on sICAS, defined as ischemic stroke or TIA in the territory of the stenotic artery, (iii) a minimum follow-up period of 3 months, (iv) RCTs comparing elective EVT plus BMT vs BMT alone, according to current clinical practice guidelines.7,8 Elective EVT was defined as angioplasty, with or without stenting, performed beyond 24 h from symptom onset in patients with sICAS presenting with ischemic stroke or TIA. Studies were excluded if they met any of the following criteria: (i) studies reporting only intracranial arteries with total occlusion, (ii) studies that did not report any of the predefined outcomes of interest, (iii) trials that did not include a comparator arm, and (iv) narrative reviews, observational studies, systematic reviews, case reports, case series, commentaries, non-peer-reviewed studies, pre-prints, or conference abstract.
To confirm eligibility, the full-text articles of selected studies were thoroughly reviewed, and those that did not meet our pre-defined inclusion criteria were excluded. In cases of overlapping datasets, the study with the most comprehensive dataset was retained. All studies were independently assessed by four authors (MIS, EP, LP, AT), with any disagreements resolved through consensus after consultation with a fifth tie-breaking author (GT). Only publicly available published studies were used for meta-analysis.
Quality control, bias assessment, and data extraction
The risk of bias for relevant domains in each included study was evaluated using the Cochrane Collaboration’s Risk of Bias Tool (RoB 2). 19 Quality control and bias assessment were conducted independently by four reviewers (MIS, EP, LP, AT), with any disagreements settled by consensus after discussion with the corresponding author (GT). For further analyses, data including the year of publication, study design, type of intervention, population characteristics, location of the study, follow-up period, sample size, and outcomes were extracted from individual studies in structured reports.
Outcomes
A pairwise meta-analyses of aggregate data was conducted, including all identified RCTs comparing outcomes in patients undergoing elective EVT plus BMT versus BMT alone. The primary efficacy outcomes of interest comprised: (i) any stroke or death within 30 days of enrollment or EVT; (ii) acute ischemic stroke in the territory of qualifying artery within 30 days; (iii) any ischemic stroke within 30 days. The primary safety outcomes included: (i) intracranial hemorrhage within 30 days; and (ii) death within 30 days. Secondary outcomes comprised: (i) any stroke or death beyond 30 days through 1 year and within 1 year; (ii) ischemic stroke in the territory of the qualifying artery beyond 30 days through 1 year; (iii) death beyond 30 days through 1 year; (iv) disabling or fatal stroke within 1 year; (v) intracranial hemorrhage within 1 year. The previous outcomes of interest were also assessed in subgroup analyses after stratification according to trial setting and population (i.e. Chinese vs non-Chinese). A sensitivity analysis was conducted excluding the VAST trial, as this RCT investigated the safety and feasibility of stenting for both extra- and intracranial symptomatic vertebral artery stenosis, with only the subgroup referring to the V4 (intracranial) segment of the vertebral artery included in the primary analyses.20,21 In addition, subgroup analyses for the primary outcomes of interest were performed according to (i) stent type, including only RCTs on PTAS, stratified by self-expandable versus balloon-expandable stent; and (ii) timing of intervention, including all identified RCTs, stratified by early versus delayed intervention. Early and delayed intervention were defined as a median time interval between symptom onset and randomization of ⩽10 days and >10 days, respectively.
Statistical analysis
For the aggregate meta-analysis, we calculated for each dichotomous outcome of interest the corresponding risk ratios (RRs) and 95% confidence intervals (95% CI). The random-effects model (DerSimonian and Laird) was employed to estimate pooled effect sizes. 22 In cases that no events were observed for assessed outcomes in included studies, a continuity correction was performed in accordance with the Cochrane Handbook. Subgroup differences were evaluated using the Q test. Heterogeneity was assessed with the I2 and Cochran’s Q statistics. 23 For qualitative interpretation, I2 values exceeding 50% and 75% were considered indicative of substantial and considerable heterogeneity, respectively. A significance threshold of 0.1 was applied for the Q statistic, while pooled estimates were considered statistically significant at a two-tailed p-value of <0.05, as determined by the equivalent z-test. Publication bias across individual studies was graphically assessed using funnel plot inspection and the equivalent z test for each pooled estimate with a two-tailed p-value <0.05 was considered statistically significant. All meta-analyses were conducted using R software (version 3.5.0) with the meta and metafor packages.
Results
Literature search and included studies
The systematic review of PubMed and Scopus identified 327 articles, of which 64 duplicates were removed. Of the remaining 263 articles, 224 were excluded based on title or abstract screening, and 39 underwent full-text assessment against the predefined inclusion and exclusion criteria. Subsequently, 33 were excluded (Supplemental Table S1), leaving six RCTs that were in line with our inclusion/exclusion criteria (seven records in total, as the results of the VAST trial were reported in two records)20,21 comprising 1606 patients with sICAS (800 and 806 patients, who underwent elective EVT plus BMT vs BMT alone, respectively).6,14,21,24–26 Key study characteristics are summarized in Supplemental Table S2, and the PRISMA flowchart is presented in Figure 1.
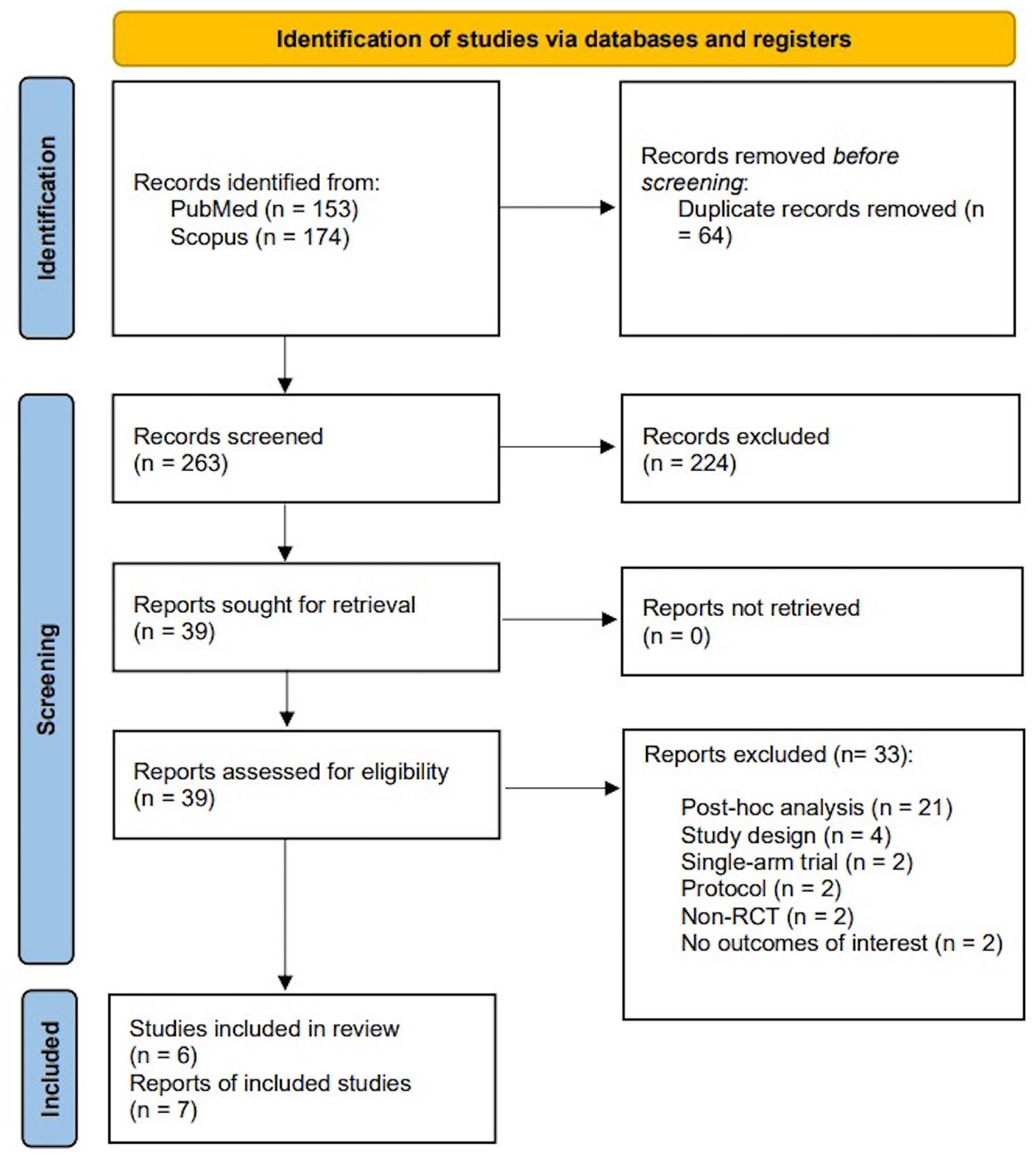
PRISMA flowchart diagram presenting the selection of eligible studies.
Quality control and publication bias of included studies
The risk of bias of the included studies was assessed using the RoB 2 and is presented in Figure S1 and Figure S2 of the Supplemental Material. While all studies employed randomization processes that generally reduced selection bias, there were concerns in some trials, such as unclear allocation concealment, which contributed to ambiguity in their randomization procedures.14,21,24,25 Performance bias was uniformly high across all trials due to their open-label designs, as both participants and treating physicians were aware of treatment assignments. Although detection bias was well-controlled through blinded outcome evaluation in all studies, the lack of blinding during treatment administration raises concerns about consistency in care. Generalizability was limited in two trials,24,25 where geographic concentration and reliance on highly skilled operators limited applicability to broader populations. Missing data was minimal across all trials, reducing attrition bias. However, funding sources and potential conflicts of interest in one study may have influenced study design and reporting. 26 Additionally, early termination of two RCTs reduced statistical power and limited the comprehensiveness of long-term outcomes.6,26,27
All funnel plots assessing publication bias among trials reporting the outcomes, both overall and in subgroups, are compiled in the Supplemental Material. Overall, included trials were unlikely to suffer from significant publication bias, as evidenced by the symmetrical funnel plots across all assessed outcomes.
Primary outcomes
The overall results of our meta-analysis are presented in Table 1. Within the first 30 days, EVT was associated with a significantly elevated risk of any stroke or death (RR = 2.68, 95% CI: 1.72–4.19; I2 = 0%; p for Cochran Q = 0.95; Figure 2), along with increased risk of ischemic stroke within the territory of the qualifying artery (RR = 2.51, 95% CI: 1.36–4.61; I2 = 0%; p for Cochran Q = 0.93; Figure 3(a)), any ischemic stroke (RR = 1.99; 95% CI: 1.17–3.38; I2 = 0%; p for Cochran Q = 0.81; Figure 3(b)), intracranial hemorrhage (RR = 6.23, 95% CI: 1.92–20.20; I2 = 0%; p for Cochran Q = 0.60; Figure 4), and mortality (RR = 3.52, 95% CI: 1.04–11.88; I2 = 0%; p for Cochran Q = 0.87; Figure 5).
Overview of primary and secondary outcomes in patients with symptomatic intracranial stenosis treated with EVT plus BMT versus BMT alone.

RR: risk ratio; CI: confidence interval; EVT: endovascular therapy; BMT: best medical therapy.
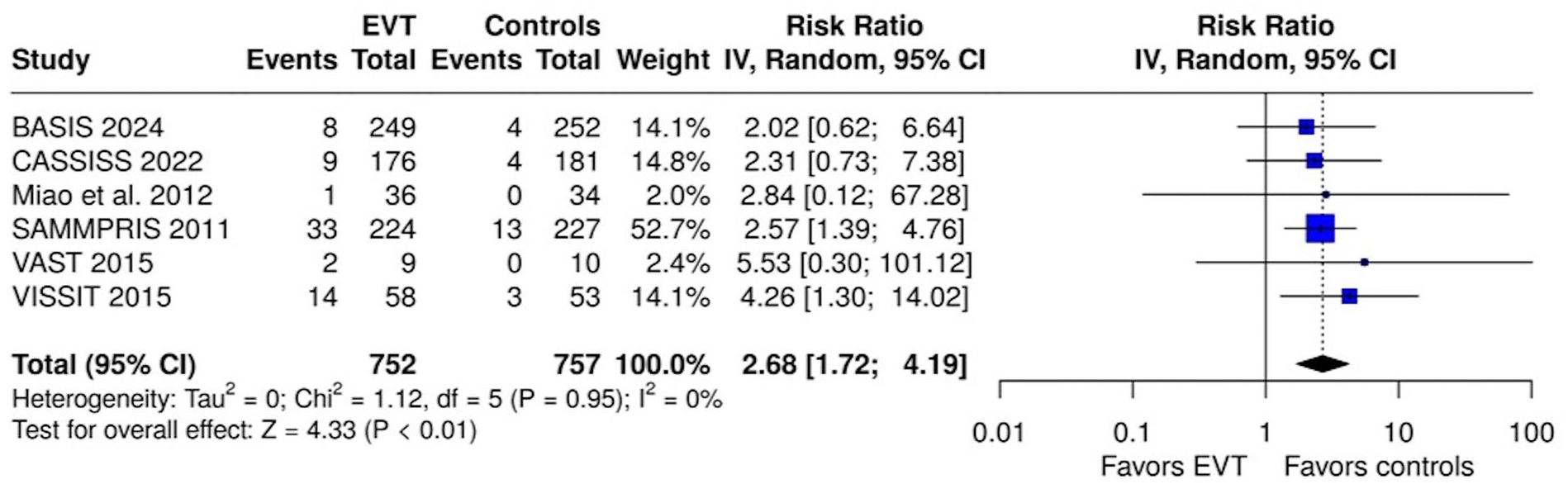
Forest plot comparing the risk of any stroke or death within 30 days in EVT and control group.
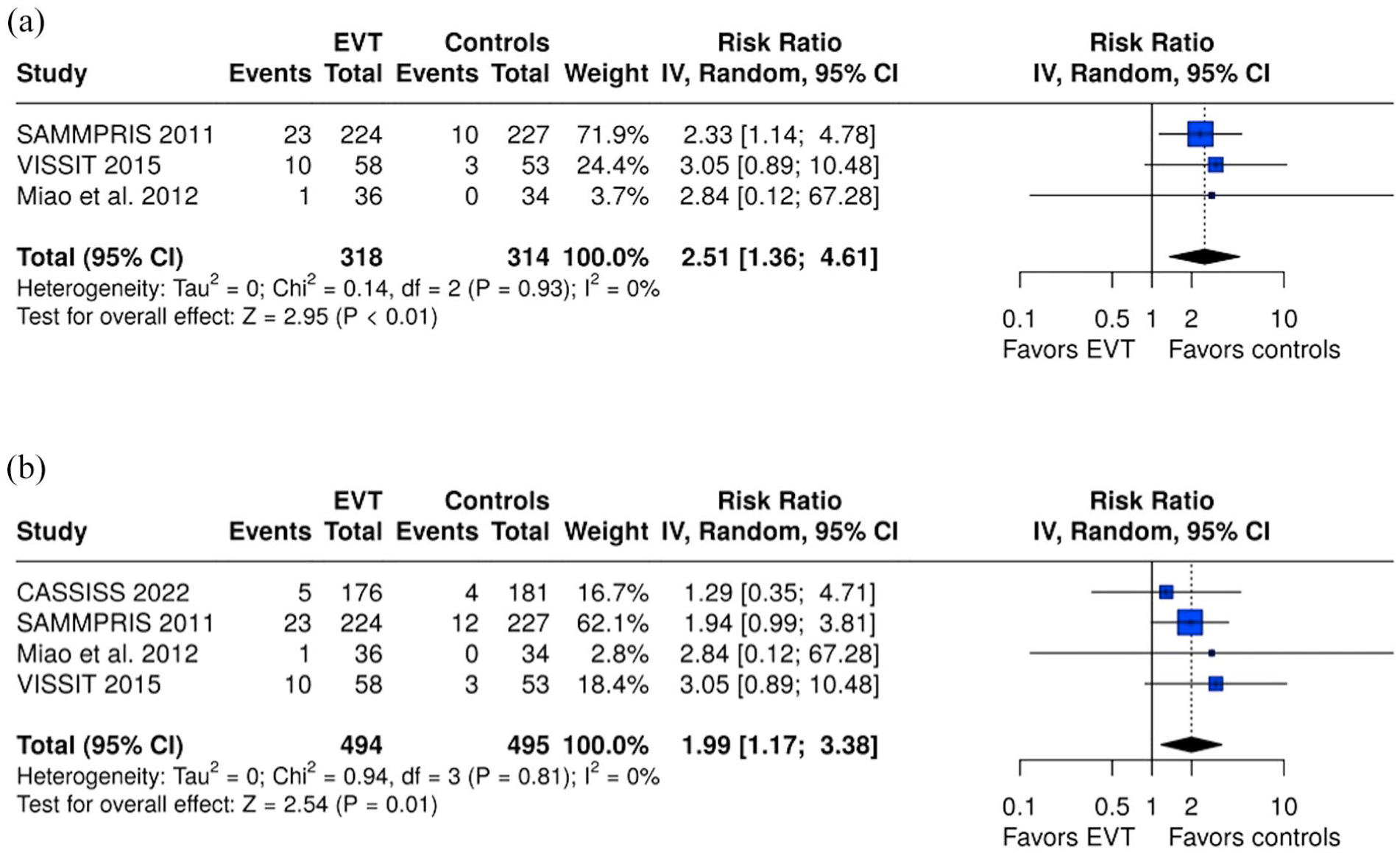
(a) Forest plot comparing the risk of ischemic stroke in the territory of the qualifying artery within 30 days in EVT and control group and (b) forest plot comparing the risk of any ischemic stroke within 30 days in EVT and control group.
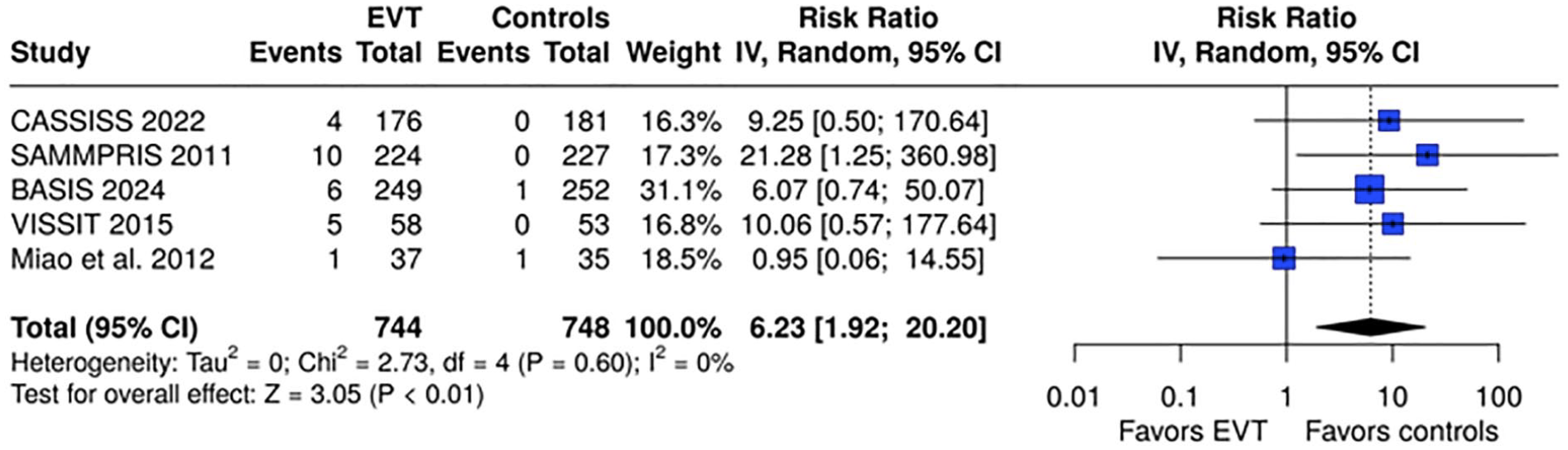
Forest plot comparing the risk of intracranial hemorrhage within 30 days in EVT and control group.
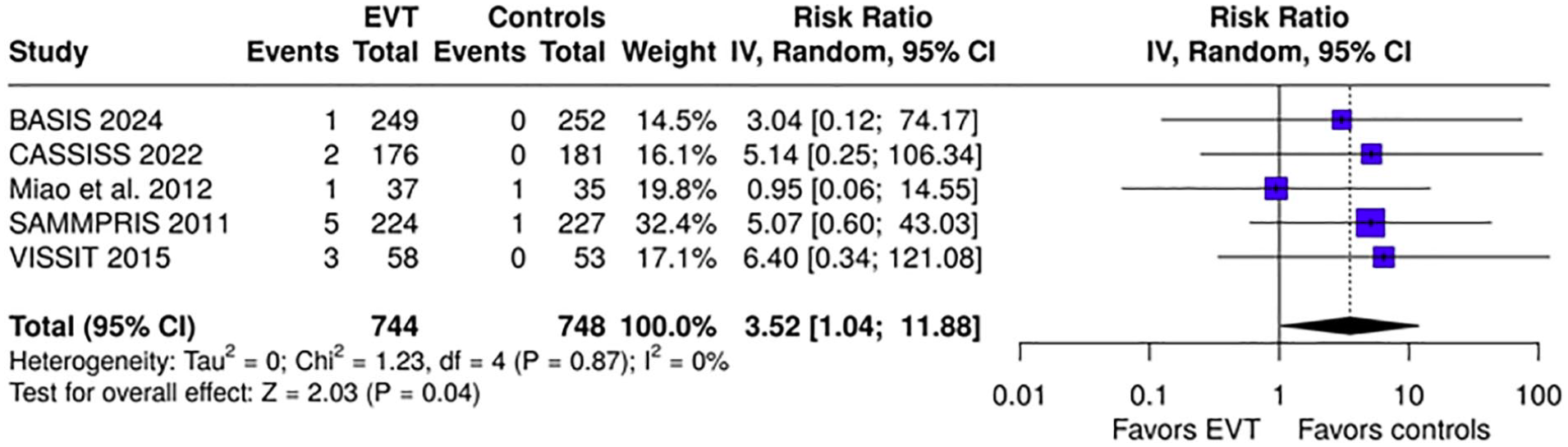
Forest plot comparing the risk of death within 30 days in EVT and control group.
Secondary outcomes
Regarding secondary outcomes, EVT was associated with a non-significant trend toward reduced risk of any stroke or death beyond 30 days through 1 year (RR = 0.29, 95% CI: 0.06–1.38; I2 = 68%; p for Cochran Q = 0.04; Figure S7) and within 1 year (RR = 0.80, 95% CI: 0.25–2.64; I2 = 82%; p for Cochran Q < 0.01; Figure S8); yet, as reflected by the evidence of significant heterogeneity, this trend was mainly driven by the findings of the BASIS trial favoring EVT. Similarly, although there was a trend toward reduced risk of ischemic stroke in the territory of the qualifying artery beyond 30 days through 1 year this did not reach statistical significance (RR = 0.44, 95% CI: 0.14–1.33; I2 = 59%; p for Cochran Q = 0.06; Figure S9). Regarding mortality beyond 30 days through 1 year, no association with EVT was uncovered (RR = 0.49, 95% CI: 0.16–1.55; I2 = 0%; p for Cochran Q = 0.86; Figure S11). The risk of intracranial hemorrhage within 1 year remained significantly higher among EVT-treated patients (RR = 4.91, 95% CI: 1.35–17.91; I2 = 0%; p for Cochran Q = 0.38; Figure S13), though only one additional event beyond the 30-day period was reported in the control group of the SAMMPRIS trial. No association was uncovered between EVT and disabling or fatal stroke within 1 year (RR = 0.91, 95% CI: 0.43–1.93; I2 = 55%; p for Cochran Q = 0.08; Figure S14).
In subgroup analyses (Figures S16–S25), no significant subgroup effect was uncovered for any of the primary or secondary outcomes of interest, with the exception of the risk of any stroke or death beyond 30 days through 1 year (p for subgroup difference = 0.02; Figure S20) and within 1 year (p < 0.01; Figure S21), which appeared significantly lower in Chinese compared to non-Chinese studies, heavily influenced by the findings of the BASIS trial. In sensitivity analysis excluding the VAST trial, EVT was associated with a significantly elevated risk of any stroke or death within the first 30 days (RR = 2.63, 95% CI: 1.68–4.14; I2 = 0%; p for Cochran Q = 0.93; Figure S26). No significant subgroup effects emerged for any of the primary outcomes of interest (Figures S27–S36) after stratification based on (i) stent type (self-expandable vs balloon-expandable) and (ii) timing of intervention (early vs delayed).
Discussion
The findings of the present systematic review and meta-analysis demonstrate worse clinical outcomes with EVT in patients with sICAS, as reflected by the significantly elevated risk of early ischemic and hemorrhagic events, as well as the significantly higher 30-day mortality among patients undergoing elective EVT plus BMT compared to BMT alone. Extending these findings beyond the 30-day period, despite evidence from the BASIS trial favoring EVT, 14 the pooled efficacy estimates remained non-significant during a long-term follow-up period of 1 year, consistent with prior meta-analyses that failed to demonstrate the efficacy of EVT in reducing the risk of long-term stroke recurrence or death associated with sICAS.16,28
The findings of significant intergroup differences in the subgroup analyses stratified by RCT setting and population (Chinese vs non-Chinese) merit further consideration. Although the risk of stroke or death within the first 30 days was comparable between subgroups, significant differences emerged in the timeframe beyond 30 days to 1 year, indicating more favorable EVT outcomes in Chinese compared to non-Chinese trials. While significant inter-racial differences in the prevalence and prognosis of sICAS have been previously acknowledged in the literature,1,29 neutral results were reported in a prior RCT on PTAS in a low-risk Chinese population 24 and the findings of the present subgroup meta-analyses appear heavily reliant on the BASIS trial. 14 This association likely suggests that the design of BASIS, 30 rather than inherent population characteristics, is primarily accountable for the observed effects. This observation also creates uncertainty regarding the generalizability of the findings of this trial.
Notably, despite the proof-of-concept evidence on the technical feasibility of submaximal balloon angioplasty in sICAS, extensive criticism has surrounded the design of BASIS,15,30 comprising: (i) the inclusion of patients presenting with TIA in an unblinded trial, a factor that arguably introduced major biases favoring revascularization, given that both treating physicians and patients were aware of allocation to the control arm; (ii) the controversial inclusion of revascularization as a component of the composite primary outcome; (iii) the prolonged interval from the qualifying event to revascularization (on average exceeding 30 days from the index event), which augments the likelihood of overestimating the efficacy or safety of EVT by excluding patients at the highest risk of early recurrent stroke (i.e. those with ICAS more vulnerable to rupture); and (iv) the use of less aggressive BMT compared to previous trials (100 mg of aspirin throughout the follow-up period compared to the 325 mg dose of SAMMPRIS), 6 coupled with a lack of reported adherence to BMT, particularly in hypertension management that represents an important predisposing factor for sICAS progression and stroke recurrence in Asian populations.31–33
Notwithstanding the previous considerations, there is a well-grounded pathophysiological rationale behind the pursuit of submaximal balloon angioplasty in highly-selected patients with sICAS, with potential advantages over PTAS, including an assumingly reduced risk of the “snow-plowing effect” and local perforator occlusion, along with an expectedly lower risk of periprocedural intracranial hemorrhage due to vessel rupture, wire perforation, or reperfusion intraparenchymal injury from submaximal and gradual flow restoration relative to PTAS.34–36 Despite these intriguing hypotheses, our meta-analysis showed that periprocedural safety as determined by the risk of intracranial hemorrhage within 30 days did not differ in BASIS compared to previous PTAS trials (with the exception of SAMMRIS the limitations of which have since been addressed in subsequent trial designs).6,14 In the BASIS trial, rescue stent implantation was allowed in cases of (i) residual stenosis ⩾ 70% after balloon dilation or unstable antegrade flow (mTICI < 2b); (ii) arterial dissection impairing distal blood flow (mTICI < 2b); and (iii) thrombosis or embolization jeopardizing distal perfusion. The overall rate of rescue stenting in BASIS was 16.2% of the interventional arm (39 cases in total). The vast majority of the cases requiring rescue stenting were due to arterial dissection as an intra-procedural complication of balloon angioplasty (14.5% of the interventional arm), followed by thrombosis (1.7%) and vasospasm (1.2%). Moreover, the rate of restenosis of the qualifying artery within 1 year was reported at 15.7%, necessitating the acquisition of longer follow-up data before the potential benefits of submaximal balloon angioplasty can be weighed against its periprocedural risks, as well as its long-term efficacy and safety outcomes. 14
The present results are in accordance and expand the findings of previously published meta-analyses by our group and others,16,28 incorporating randomized data from a total population of 1606 patients with sICAS. These results are also aligned with current European Stroke Organization (ESO) and American Heart Association/American Stroke Association (AHA/ASA) guidelines that advocate against the use of EVT as first-line treatment in sICAS; though rescue EVT may be considered in highly-selected cases (e.g. in patients with recurrent stroke that are unresponsive to aggressive BMT, or in the presence of artery-to-artery embolism and hemodynamic failure with poor collaterals).7,8
Certain limitations of the present systematic review and meta-analysis should also be acknowledged. First, since this was an aggregate data meta-analysis, inferences regarding factors that could aid in selecting patients likely to benefit from EVT cannot be drawn from the available data; thus, individual patient data meta-analyses are required to provide insights into criteria for risk stratification and optimal patient selection for future RCTs. Second, the low event rate in the BMT arms of included RCTs presents significant challenges for translating these findings to real-world clinical practice. Previous meta-analyses have similarly emphasized the need for larger sample sizes and longer follow-up periods in sICAS trials, particularly to address the limited event rate with BMT alone. 37 Third, although baseline characteristics were balanced across groups as per trial design, there was substantial heterogeneity in inclusion criteria, patient populations, and the devices and procedures applied across trials. This heterogeneity renders data pooling susceptible to type I and II errors. 38 While random-effects models were employed and significant heterogeneity (linked to BASIS) was detected only in very few secondary outcomes, further RCTs investigating novel devices and interventional approaches (e.g. submaximal balloon angioplasty) are warranted to improve outcomes and ensure more homogeneous comparisons.39,40 Fourth, limitations and other sources of bias inherent to the included RCTs, as detailed in the risk of bias assessment, may have confounded the present results. 34 Given the promising signal of potential efficacy of submaximal balloon angioplasty from the BASIS trial, efforts to minimize periprocedural complications and effectively select high-risk patients should be prioritized in future trials.
In conclusion, EVT is associated with a significantly higher risk of adverse outcomes in the early post-stroke period and presents no benefit over BMT in reducing long-term stroke recurrence or mortality in patients with sICAS. These findings emphasize the need for further research to identify high-risk patient subgroups and implement strategies that mitigate periprocedural risks to improve sICAS management and prognosis.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251324863 – Supplemental material for Endovascular therapy versus best medical treatment for symptomatic intracranial atherosclerotic stenosis: A systematic review and meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873251324863 for Endovascular therapy versus best medical treatment for symptomatic intracranial atherosclerotic stenosis: A systematic review and meta-analysis by Maria-Ioanna Stefanou, Evangelos Panagiotopoulos, Lina Palaiodimou, Aikaterini Theodorou, George Magoufis, Stavros Spiliopoulos, Apostolos Safouris, Odysseas Kargiotis, Klearchos Psychogios, Tatiana Sidiropoulou, Frantzeska Frantzeskaki, Panayiotis D Mitsias, Katharina Feil, Annerose Mengel, Marios Themistocleous, Ulf Ziemann and Georgios Tsivgoulis in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent – Ethical approval
This study did not require an ethical board approval or written informed consent by the patients according to the study design (systematic review and meta-analysis).
Guarantor
GT
Contributorship
Dr. Stefanou participated in study concept and design, acquisition of data, analysis and interpretation, and prepared the first draft of the manuscript. Dr. Panagiotopoulos participated in acquisition of data, analysis and interpretation, and prepared the first draft of the manuscript. Drs. Palaiodimou and Theodorou contributed to acquisition of data, analysis, critical revision of the article for important intellectual content. Drs. Magoufis, Spiliopoulos, Safouris, Kargiotis, Psychogios and Themistocleous participated in critical revision of the article for important intellectual content. Drs. Sidiropoulou, Mitsias, Feil, Mengel and Ziemann contributed to critical revision of the article for important intellectual content and provided critical comments on data interpretation and synthesis. Dr. Tsivgoulis was responsible for study design, analysis and interpretation, critical revision of the article for important intellectual content.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
