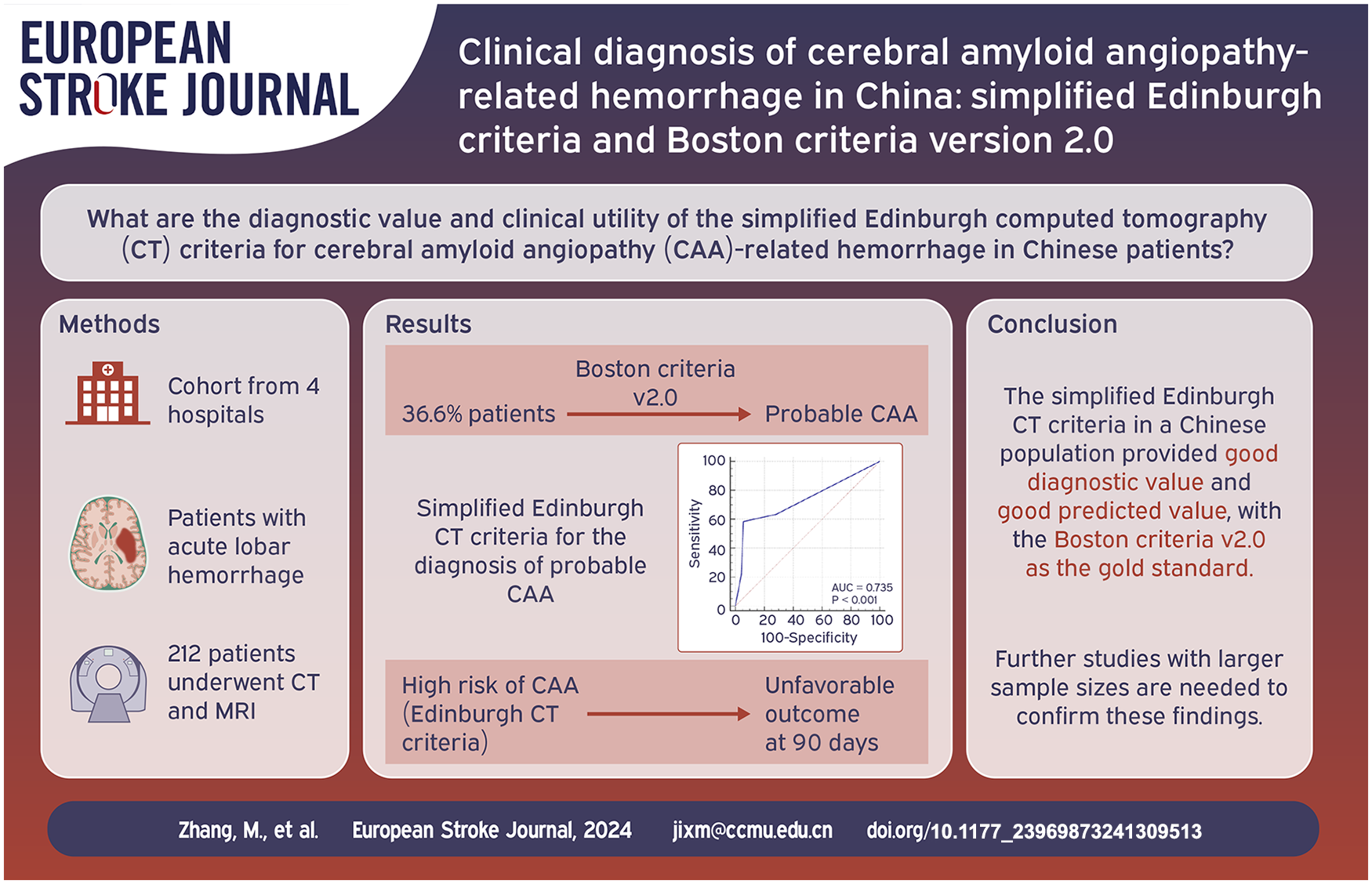
Abstract
Background:
Accurate diagnosis of cerebral amyloid angiopathy (CAA) in surviving patients is indispensable for making treatment decisions and conducting clinical trials. We aimed to evaluate the diagnostic value and clinical utility of the simplified Edinburgh computed tomography (CT) criteria for CAA-related hemorrhage in Chinese patients.
Methods:
We analyzed 212 patients with lobar hemorrhage who underwent brain CT and magnetic resonance imaging (MRI) from a multicentre cohort. Using the Boston criteria version 2.0 (v2.0) as the gold standard, we assessed the application value of the simplified Edinburgh CT criteria, and investigated whether the Edinburgh CT criteria predict patient outcomes.
Results:
Patients with probable CAA accounted for 36.6% according to the Boston criteria v2.0. The Edinburgh CT criteria indicated an area under the receiver operating characteristic curves (AUC) of 0.735 for the diagnosis of probable CAA, and it performed better when there was a high-risk threshold of CAA in the decision curve analysis. Patients with a high risk of CAA based on the Edinburgh CT criteria had poorer outcomes at 90-day after adjusting for confounding factors (p = 0.034). Finger-like projections in the Edinburgh CT criteria were associated with lobar microbleeds, cortical superficial siderosis, and multispot white matter hyperintensity according to the Boston criteria.
Conclusions:
Taking the Boston criteria v2.0 as the gold standard, the Edinburgh CT criteria demonstrated good diagnostic value and predicted outcomes well at 90-day in Chinese patients with lobar hemorrhage. Further studies with larger sample sizes are required to confirm these findings.
Introduction
Cerebral amyloid angiopathy (CAA) is an age-related small vessel disease that causes recurrent intracerebral hemorrhage (ICH) and progressive cognitive impairment. 1 As the main cause of non-hypertensive ICH, CAA accounts for 5%–20% of all spontaneous ICH in the elderly. 2 The recurrence rate of hemorrhage in CAA is 6.9% each year, leading to higher disability and mortality. 3 Early diagnosis of CAA in the living is crucial to avoid harmful treatments, which may improve the outcomes of patients.
Given the extreme difficulty in acquiring brain tissue samples, the in vivo diagnostic standard for CAA relies on neuroimaging markers obtained through magnetic resonance imaging (MRI). The Boston criteria v2.0 have been updated with the addition of emerging CAA MRI markers to improve the sensitivity without reducing the specificity obviously. 4 However, most patients undergo a computed tomography (CT) scan first upon admission, and MRI is not always suitable for patients with ICH during the hyperacute and acute phases because of contraindications, the inability to cooperate with the examination due to severe illness, and economic conditions.
Most recently, the simplified Edinburgh CT criteria have been shown to have the potential for diagnostic accuracy and timely decision-making in the clinical management in the Western populations.5,6 It is necessary to conduct research in Eastern populations because of the discrepancy in the incidence rate of CAA between Eastern and Western general populations. 7 We performed this study to explore the diagnostic and clinical value of the simplified Edinburgh CT criteria for CAA in patients with lobar hemorrhage compared with the Boston criteria v2.0.
Materials and methods
Participants
We retrospectively assessed 212 consecutive patients with lobar ICH from a multicentre cohort study (Clinicaltrials.gov ID: NCT05760950). The study was conducted in accordance with the principles of the Declaration of Helsinki. Patients who meet the following criteria were enrolled: (1) lobar hemorrhage confirmed by brain CT; (2) underwent MRI within 6 months of onset. 5 The exclusion criteria were as follows: (1) age < 50 year-old; (2) the image quality of CT and MRI was too poor to be evaluated; (3) clinical data were incomplete. This study was approved by the Ethics Committee of Xuanwu Hospital, Capital Medical University, and informed consent was provided from patients or their legal representatives.
Data Collection
Clinical data of patients, including age, sex, comorbid conditions, toxic habits, use of medications, systolic and diastolic blood pressure, Glasgow Coma Scale (GCS) score, National Institutes of Health Stroke Scale (NIHSS) score on admission, and modified Rankin Scale (mRS) scores at discharge and 90-day were obtained. Follow-up was performed via face-to-face interviews or standardized telephone conversations with the patients or their relatives. A score of 4–6 represented an unfavorable outcome.
The simplified Edinburgh CT criteria were as follows: the patients with both subarachnoid hemorrhage (SAH) and finger-like projections (FLPs) were considered as high risk of CAA, and those with either SAH or FLPs were considered as medium risk of CAA, and those with neither SAH nor FLPs were considered as low risk of CAA. 5 The investigators assessed FLPs and SAH by applying the three-dimensional reconstruction of head CT images after training. Hematoma shape was classified as round/ellipsoid, irregular, or multilobular form. Hematoma volumes were calculated by the areas of each layer and slice thickness. The white matter lesion (WML) grades on CT and on MRI were rated using the van Swieten et al. 8 Scale and the Fazekas scores respectively. Cerebral microbleeds (CMBs) and superficial cortical siderosis (cSS) were identified using susceptibility-weighted imaging (SWI). The four patterns of WML comprised multiple subcortical, peri-basal ganglia, large posterior subcortical patches, and large anterior subcortical patches on FLAIR MRI as previously reported. 9 Enlarged perivascular spaces (EPVS) were recorded on T2-weighted MRI by using a recommended 4-point visual rating scale (0 = no, 1 ⩽ 10, 2 = 11–20, 3 = 21–40, and 4 ⩾ 40). 10 All assessments of imaging markers for small vessel disease were conducted in accordance with the Standards for Reporting Vascular Changes on Neuroimaging 2 (STRIVE-2). 11
Patients with lobar hemorrhage were divided into no CAA, possible CAA, or probable CAA groups based on the modified Boston criteria version2.0 as previously described. 12 Imaging examinations were performed using 3T MR scanners. The retrospective multicenter study did not allow completely consistent standardized scanning parameters. In the routine MRI brain imaging protocol, the most frequently sequences of T1w, T2w, FLAIR and SWI were acquired using the following parameters: T1w: repetition time (TR)/echo time (TE) 120/2.1 ms, slice thickness 5 mm; T2w: TR/TE 5304/121.9 ms, slice thickness 5 mm; FLAIR: TR/TE 8500/92.8 ms, slice thickness 5 mm. SWI: TR/TE 37.8/23 ms, slice thickness 5 mm.
An experienced radiologist (Xu J, with 20 years of experience) and a trained neurologist (Zhao W, with 9 years of experience) blinded to the MRI independently evaluated the CT images, and they were supposed to achieve a consensus if there were disagreements. MRI analyses were performed by other two radiologists (Ma S and Liu X, with 10 and 5 years of experience, respectively) according to the same process as above. All the evaluators were blinded to the clinical data and outcomes.
Outcomes
The outcomes were as follows: (1) evaluation of the diagnostic value of the simplified Edinburgh CT criteria by comparing it with the modified Boston criteria v2.0 in CAA-related hemorrhage; (2) the applicability of the Edinburgh CT criteria in predicting the mRS at discharge and 90-day; (3) the relationship between neuroimaging markers on the Edinburgh CT criteria with markers on the Boston criteria.
Statistical analysis
Statistical significance was set at p < 0.05 and all tests were two-tailed. According to the distribution and results of the Kolmogorov–Smirnov test, continuous variables were presented as mean ± standard deviation or median and interquartile range, and categorical variables were expressed as percentages. The t-test or Mann-Whitney U test was used to compare continuous variables due to whether variables were normally distributed. Pearson’s or continuous correction chi-square tests were performed, as appropriate, to compare categorical variables. Inter-observer variability of evaluating the neuroimaging markers on CT and MRI was analyzed using Cohen κ statistic. The model for predicting the probability of CAA was proposed by Rodrigues et al. 5 We evaluated the diagnostic accuracy of simplified Edinburgh CT criteria by receiver operating curve (ROC) using MedCalc (version 20.0). The decision curve analysis (DCA) was performed to assess the net clinical benefit of the Edinburgh criteria using the R statistical package (version 4.2). Logistic regression analysis was performed to assess the value of the Edinburgh CT criteria for predicting functional outcome. The multivariable logistic regression included the confounding variables with a p value less than 0.10 at baseline and other factors that may impact on the outcome according to other studies.13,14 All the above statistical analyses were conducted using SPSS (version 25.0).
Results
This cohort study finally included 212 patients with lobar ICH from 2017 to 2022. The study flow chart was presented in Figure S1. Of all the patients who underwent MRI, 112 patients underwent SWI, and 204 patients underwent FLAIR and T2. The baseline characteristics of those 112 patients were displayed in Table 1. The baseline characteristics of 212 patients who underwent CT were represented in Table 2.
Characteristics of lobar ICH associated with modified Boston criteria v2.0.
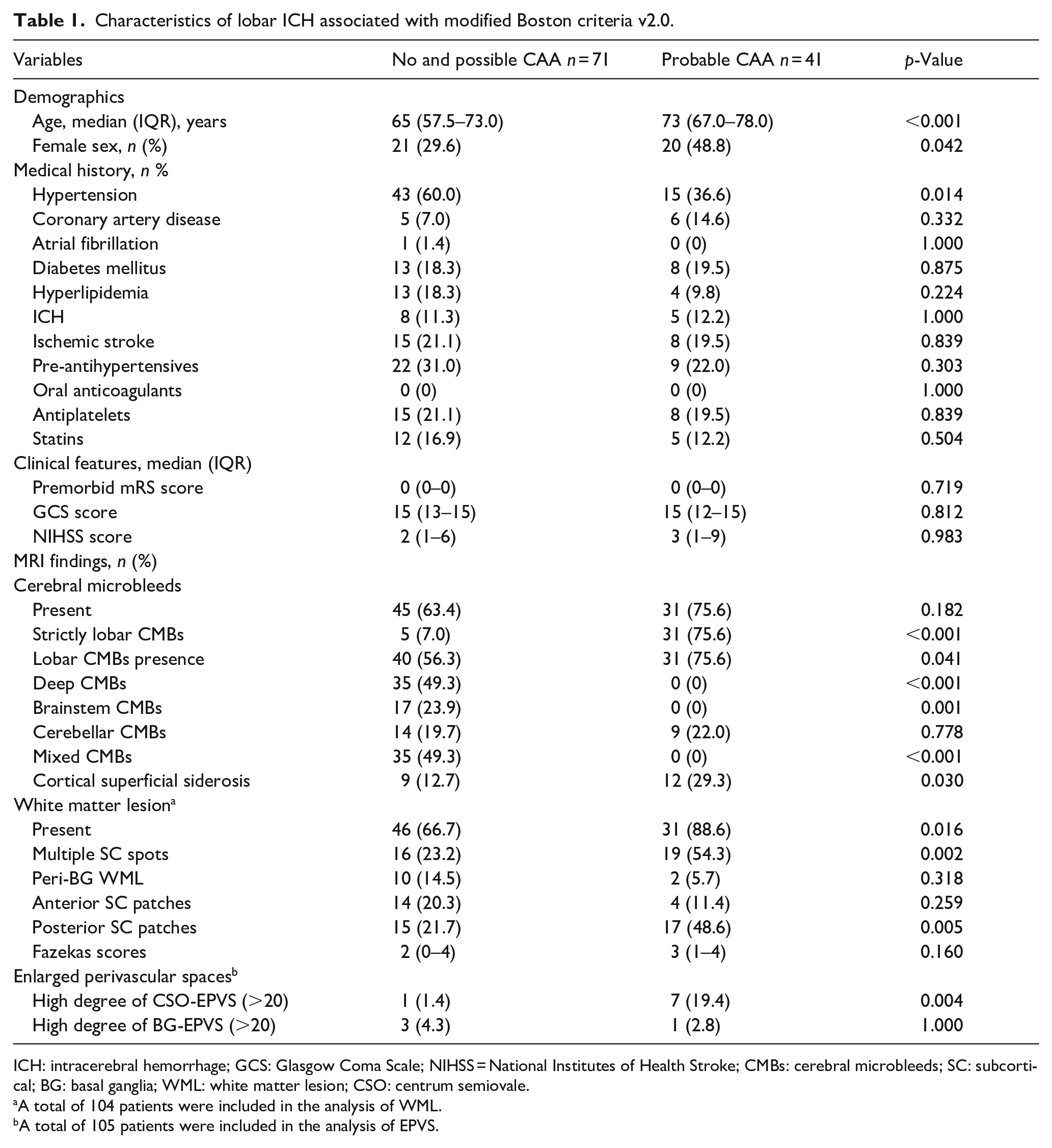
ICH: intracerebral hemorrhage; GCS: Glasgow Coma Scale; NIHSS = National Institutes of Health Stroke; CMBs: cerebral microbleeds; SC: subcortical; BG: basal ganglia; WML: white matter lesion; CSO: centrum semiovale.
A total of 104 patients were included in the analysis of WML.
A total of 105 patients were included in the analysis of EPVS.
Characteristics of lobar ICH associated with simplified Edinburgh CT Criteria.
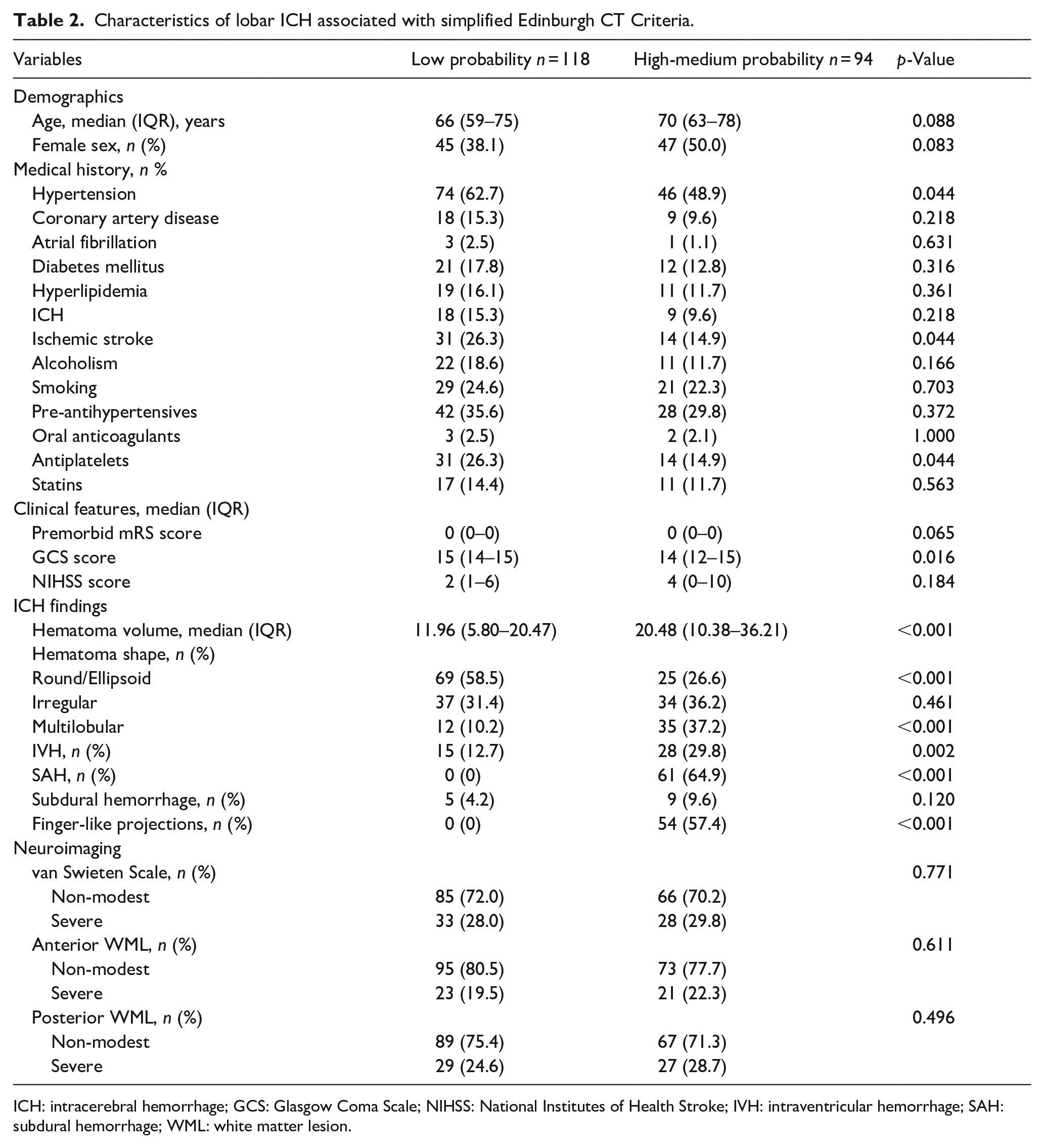
ICH: intracerebral hemorrhage; GCS: Glasgow Coma Scale; NIHSS: National Institutes of Health Stroke; IVH: intraventricular hemorrhage; SAH: subdural hemorrhage; WML: white matter lesion.
Based on the Boston criteria v2.0, 41 (36.6%) patients met probable CAA. The patients with probable CAA were older (73 vs 65, p < 0.001), female (48.8% vs 29.6%, p = 0.042), had strictly lobar CMBs (75.6% vs 7.0%, p < 0.001), posterior SC patches (48.6% vs 21.7%, p = 0.005), and multiple subcortical spots patterns of WML (54.3% vs 23.2%, p = 0.002) than non-probable CAA group. The patients with probable CAA were less likely to have a history of hypertension (36.6% vs 60.0%, p = 0.014) (Table 1).
According to the simplified Edinburgh CT criteria, 94 (44.34%) patients were classified as having a high-medium probability of CAA. Compared to the patients with low CAA probability, patients with high and medium risk of CAA were less likely to have a history of hypertension (48.9% vs 62.7%, p = 0.044), ischemic stroke (14.9% vs 26.3%, p = 0.044), medication history of antiplatelets (14.9% vs 26.3%, p = 0.044), and round-shaped hematoma (26.6% vs 58.5%, p < 0.001). Moreover, they had lower GCS scores (14 vs 15, p = 0.016), larger hematoma volume (20.48 ml vs 11.96 ml, p < 0.001), multilobular hemorrhage (37.2% vs 10.2%, p < 0.001), and intraventricular hemorrhage (29.8% vs 12.7%, p = 0.002) (Table 2).
External validation of the Edinburgh mode
One hundred twelve patients with SWI were selected for ROC and DCA curve analysis for external verification more accurately. Taking the Boston criteria v2.0 as the reference, the specificity of high-risk grading based on the Edinburgh CT criteria to rule in probable CAA-relate ICH was 94.37% (95% confidence interval (CI) 86.2–98.4), and the sensitivity of diagnosis of medium and low risk CAA was 58.54% (95% CI 42.1–73.7). The area under ROC (AUC) was calculated at 0.735 (95% CI 0.643–0.814), with a positive likelihood ratio of 10.39 (95% CI 3.88–27.86) and a negative likelihood ratio of 0.44 (95% CI 0.30–0.63). The high and medium probability of CAA demonstrated a specificity of 98.15% (95% CI 90.1–100.0), and a sensitivity of 46.55% (95% CI 33.3–60.1). The AUC of Edinburgh CT criteria (high or intermediate risk vs low risk) for CAA according to the Boston criteria (possible or probable vs non-CAA) was 0.739 (95% CI 0.648-0.818). The positive likelihood ratio was 25.14 (95% CI 3.54–178.69) and the negative likelihood ratio was 0.54 (95% CI 0.43–0.69) (Figure 1).
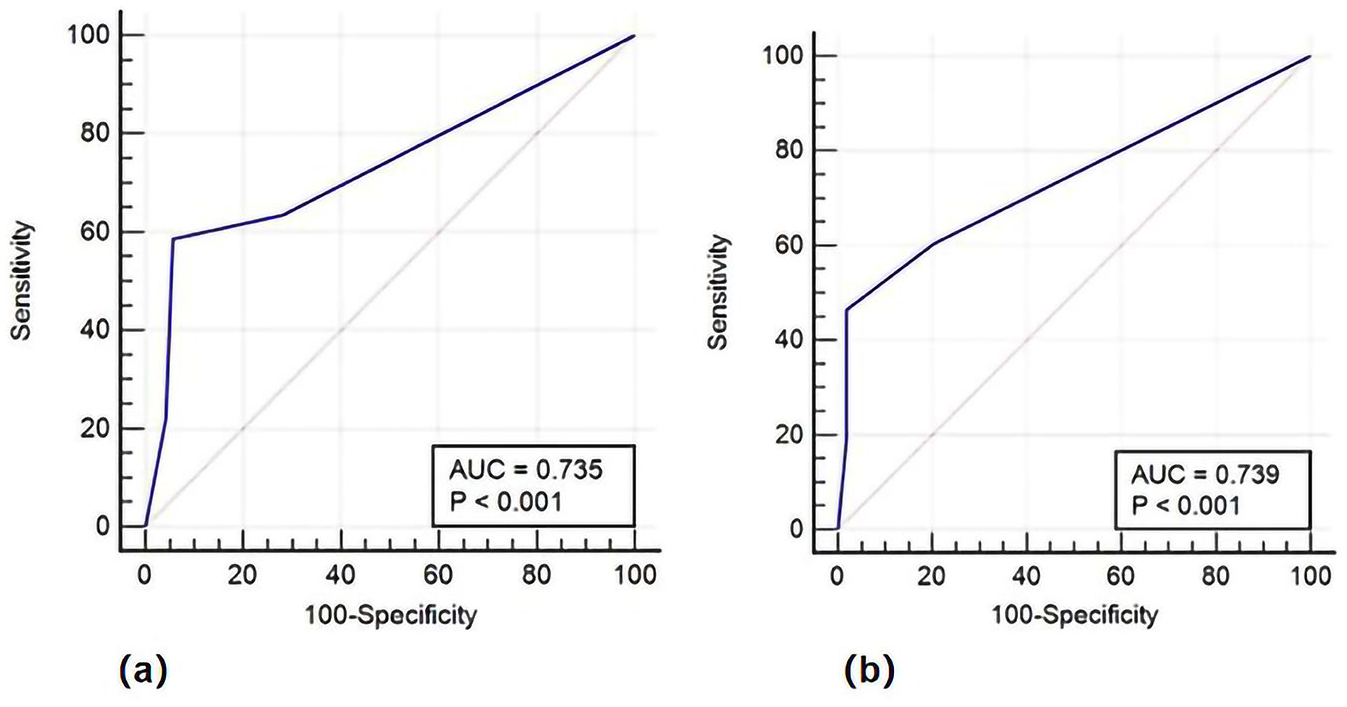
Discrimination of the simplified Edinburgh CT criteria: (a) discrimination, sensitivity, and specificity of high risk of CAA (vs Intermediate/low risk) for probable CAA (vs non-CAA and possible CAA) and (b) discrimination, sensitivity, and specificity of high/intermediate risk of CAA (vs low risk) comparing with probable and possible CAA (vs non-CAA).
DCA was performed to evaluate the clinical utility of the Edinburgh CT criteria to predict CAA. The criteria demonstrated its clinical net benefit to avoid both false negative and positive, but failed to show good value when compared with the strategy of supposing that all patients have CAA at lower risk thresholds. The Edinburgh mode indicated clinical net benefit at high threshold probabilities for the diagnosis of probable or possible CAA (Figure 2).
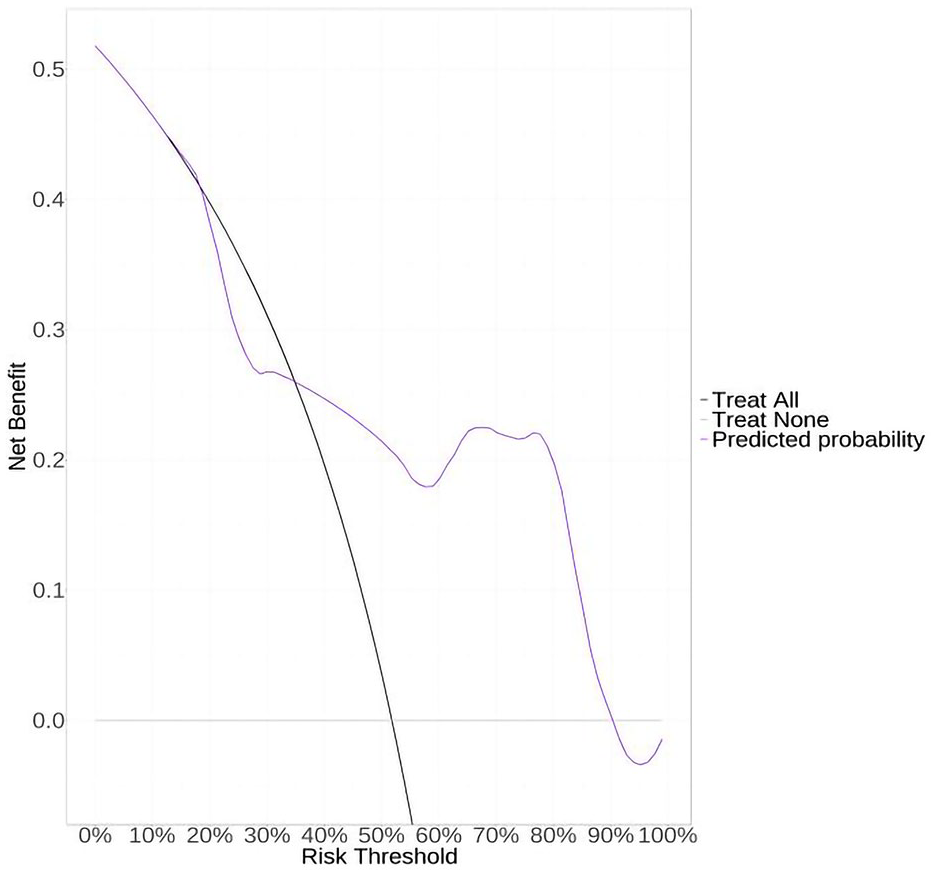
Decision curve analysis of the simplified Edinburgh CT criteria. Decision curve analysis assessing the ability of the simplified Edinburgh CT criteria to rule in or rule out probable and possible CAA.
Edinburgh criteria and Boston criteria for predicting functional outcome
According to the simplified Edinburgh CT criteria, patients with high probability of CAA achieved more unfavorable outcomes than those with low (47.26% vs 14.58%, p = 0.002) and medium (47.62% vs 20.33%, p = 0.016) probability at 90-day (Figure 3). Similarly, patients with high risk on CT had higher rates of unfavorable outcomes at discharge than patients with low risk (66.66% vs 34.75%, p = 0.006) (Figure S2). The multivariate logistic regression analysis showed that higher risk of CAA (odds ratio [OR] 3.578, 95% CI 1.099–11.656, p = 0.034) was an independent predictor associated with unfavorable outcomes at 90-day (Table 3) rather than at discharge (Table S1).
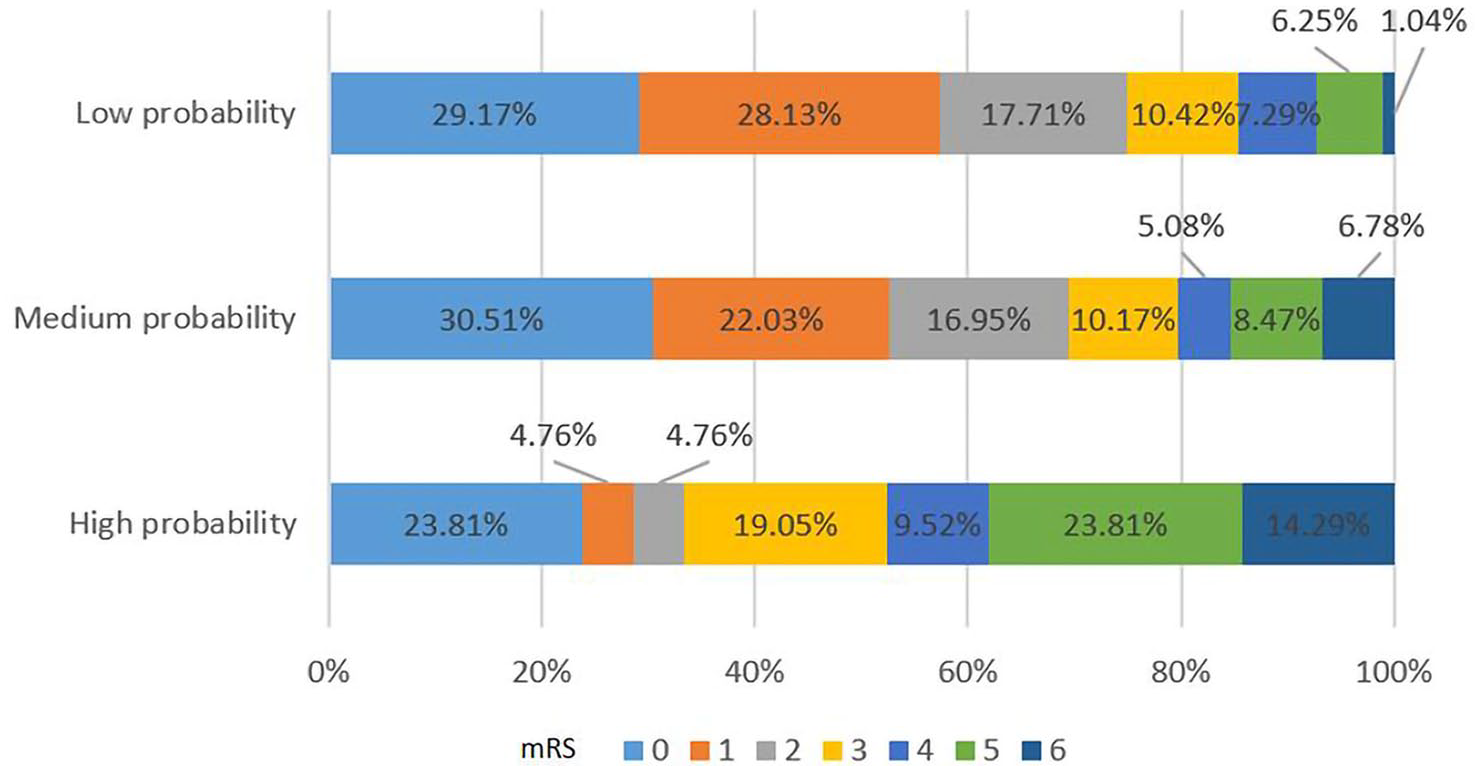
mRS at 90-day based on the Edinburgh CT criteria.
Multivariate logistic regression analyses for functional outcome at 90-day.
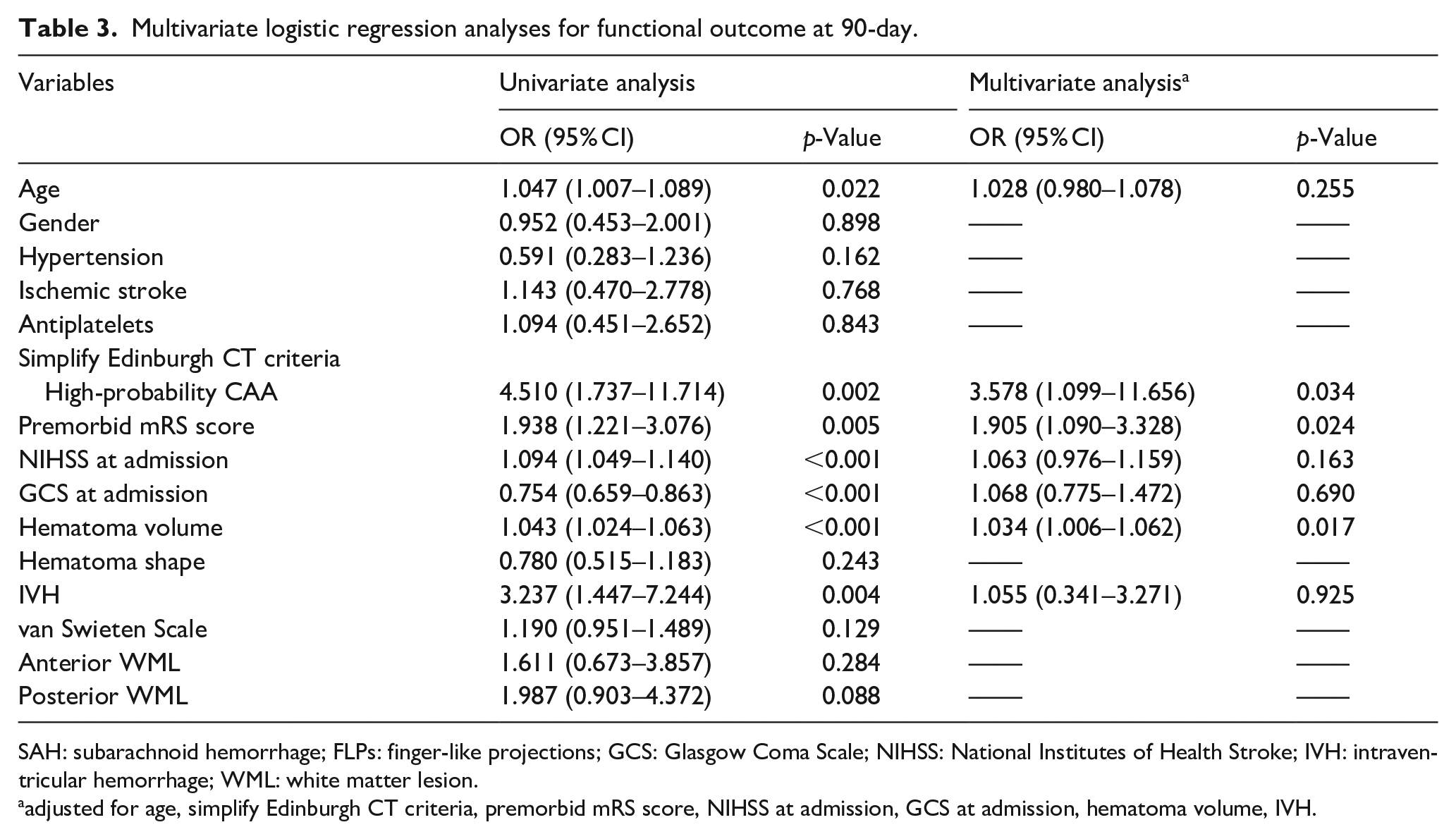
SAH: subarachnoid hemorrhage; FLPs: finger-like projections; GCS: Glasgow Coma Scale; NIHSS: National Institutes of Health Stroke; IVH: intraventricular hemorrhage; WML: white matter lesion.
adjusted for age, simplify Edinburgh CT criteria, premorbid mRS score, NIHSS at admission, GCS at admission, hematoma volume, IVH.
According to the Boston v2.0 criteria, patients with probable CAA had no significant difference in outcomes at 90-day compared with those with possible or no CAA respectively (Figure S3). After adjusting for potential confounders, patients with probable CAA showed no correlation with the outcome at 90-day (Table S2).
Association between markers on MRI and CT
The inter-observer variability of the main imaging markers was listed in Table S4. The presence of FLPs was related to strictly lobar CMBs (67.9% vs 20.2%, p < 0.001), cSS (32.1% vs 14.3, p = 0.036) and multiple subcortical spots WML patterns (52.0% vs 32.5%, p = 0.013). SAH showed no significant association with any of the MRI indicators (Table S3).
Discussion
This study was the largest cohort of patients diagnosed with CAA-related hemorrhage in lobar ICH in the Eastern population in the real world since the Boston criteria were updated. Based on the Boston criteria v2.0, the simplified Edinburgh CT criteria showed a nearly good discriminative capacity to distinguish CAA-related ICH from other causes. The practical clinical value was ideal when false positives were avoided instead of avoiding false negatives according to the DCA. Patients with a high-risk of CAA based on the Edinburgh CT criteria may achieve unfavorable outcomes at 90-day.
A series of autopsy studies have shown that the prevalence rate of CAA-related ICH in Eastern cohorts was lower than that in Western cohorts due to the geographical and ethno-racial discrepancy. 7 In our study, probable CAA constituted 36.6% in lobar ICH over 50 year-old, which was lower than the proportion in a recent study. 6 Other studies showed a similar percentage of patients with probable CAA (34%–38.6%).15–17 This discrepancy is due to differences in ethnicities, age restrictions, and versions of the diagnostic criteria applied. Therefore, the effectiveness of the same diagnostic criteria may differ among populations.
It is a critical step to identify the probability of a given patient with CAA-related ICH in acute phase. It will impact the decision-making process for surgical treatment and the use of antithrombotic drugs and statins in patients with deep venous thrombosis, a history of stent implantation, atrial fibrillation, and other comorbidities. These interventions may increase the risk of bleeding and could potentially lead to a poor prognosis in cases of CAA-related ICH.18–21 On account of the APOE genotyping is not available worldwide, especially in the acute-phase of ICH, the simplified Edinburgh CT criteria proposed by Rodrigues et al. 5 included two CT markers: SAH and FLPs. The specificity of the simplified Edinburgh CT model to diagnose probable CAA and distinguish CAA-related ICH from other causes based on Boston criteria v2.0 was higher than that of the other two studies based on Boston criteria v1.5.6,17 A previous study demonstrated that the simplified Edinburgh CT criteria had a specificity of 87.1% and a sensitivity of 58.5% to diagnose the probable CAA-related ICH. 6 The other study showed the Edinburgh criteria had a specificity of 98% and a sensitivity of 33% to differentiate CAA from non-CAA. 17 This discrepancy may be due to different participants and diagnostic criteria. In the previous study of Rodrigues et al., there may be bias in selecting older patients and more serious diseases in autopsy. Compared with their participants, the patients in our study were younger and had smaller hematoma volumes, which further validate the application of the Edinburgh CT criteria.
To better explore the clinical application value of this model, we further plotted the DCA curve. We suggested that if the threshold probability of a patient is relatively high (>35% approximately), using this Edinburgh CT criteria to confirm the diagnosis adds more benefit than either treat as all negative or all positive. And same as previous study, our data indicated that the net benefit of Edinburgh CT criteria in rule out CAA was uncertain when there were no CT features. 6 That is to say, the criteria manifested no more clinical net benefit compared with taking all patients with lobar ICH as if they were CAA when making clinical decisions at low threshold probabilities. This result may be caused by several reasons. Firstly, the sample size of our study is smaller than that of western studies and only a subset of patients received SWI, which may have caused selection bias. Secondly, for the same disease, because of the discrepancies in ethnic and geographical factors, some pathological or imaging characteristics may be delicate different. Thirdly, for the new version of Boston criteria has added some markers, it remains to be supported by data form large sample study to verified and updated for CT diagnostic criteria.
As for the predictive value of the Edinburgh CT criteria in lobar ICH, multivariate regression showed that the Edinburgh grading was independently associated with clinical outcome. Correspondingly, baseline information suggested that patients at higher risk for the Edinburgh classification had more adverse factors, such as lower GCS, larger hematoma volume, worse hematoma shape, IVH, and severer atrophy. For the SAH and FLPs are easily identified on CT, it is also advantageous to physicians to make clinical decision in the super-acute or acute stage more conveniently, compared with the current ICH scores combined with multiple indicators. And the modified Boston criteria is likely to be more predictive of recurrent ICH in long-term rather than the short-term outcomes in lobar hemorrhage.22,23
Additionally, we found that a significant association between FLPs on CT and CMBs, cSS, and multispot WML on MRI. The cSS manifests as a linearly low signal along the sulci on the SWI. It usually caused by convexal SAH due to the rupture of vessels in the leptomeningeal or cortical surface affected by Aβ protein deposition. 24 This results from the expansion of lobar hemorrhage or blood migration after hemodiapedesis. 25 These two potential mechanisms correspondingly explain the possible reasons for the correlation between cSS and SAH and the association between cSS and FLPs. There was a trend, although not statistically significant between cSS and SAH, which is inconsistent with the results of two previous studies.6,16 This discrepancy was probably due to the proportion of patients with cSS (18.75%) was relatively small compared to other studies in Western population.6,16 CAA-related ICH tends to spread into the subarachnoid space with irregular boundaries, such as FLPs. 26 Unlike SAH, FLPs are likely to indicate parenchymal-predominant CAA driven by APOE e4. 16 Histopathological studies have proved that strictly lobar CMBs are linked to APOE e4, 27 indirectly suggesting that different image representations may be connected with the underlying APOE genotype. As the neuroimaging was updated in the Boston criteria, the multispot WML was confirmed to be associated with CAA-related ICH independently of age and the total WML burden. 9 The WML is associated with the changes in integrity of the vessel wall, disruption of the brain blood-barrier, and chronic ischemia or hypoperfusion.28,29 Hematoma growth is partly due to the disruption of blood-brain barrier after ICH. 30 This may explain why patients with multispot WML are more likely to have FLPs which are the elongated extensions arising from the hematoma.
Some limitations must be acknowledged. First, this was a retrospective study. We ruled out a large proportion of patients who didn’t undergo MRI, which perhaps cause some selection bias. Secondly, due to the lack of pathological data support, our findings may exist only in the terms of neuroimaging characteristics and do not definitely represent the true pathology. However, the diagnostic verification by imaging is also the best verification method for Chinese people, and because of the limitations of cultural traditions and medical habits, autopsy is usually not performed in China.
Conclusions
Our study suggests that the simplified Edinburgh CT criteria performed diagnostic accuracy of CAA in Chinese patients with lobar hemorrhage when compared with the Boston criteria v2.0, and showed good predictive value for functional outcome of CAA-related ICH at 90-day. Further prospective studies with larger scale are needed to verify our findings.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241309513 – Supplemental material for Clinical diagnosis of cerebral amyloid angiopathy related hemorrhage in China: Simplified Edinburgh criteria and Boston criteria version 2.0
Supplemental material, sj-docx-1-eso-10.1177_23969873241309513 for Clinical diagnosis of cerebral amyloid angiopathy related hemorrhage in China: Simplified Edinburgh criteria and Boston criteria version 2.0 by Mengke Zhang, Ruiwen Che, Xin Liu, Chengbei Hou, Zhongyue Wang, Sen Hu, Shengqi Fu, Yuan Kan, Hailiang Sun, Jianmin Xu, Shiliang Ma, Sijie Li, Changhong Ren, Wenbo Zhao, Milan Jia, Jingang Wang, Chuanjie Wu and Xunming Ji in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (Nos. 82071468 and 82271507), and National Key Research and Development Program (2022YFC3602401).
Ethical approval
The ethics the Ethics Committee of Xuanwu Hospital, Capital Medical University approved this study ([2020]142).
Informed consent
Verbal or written informed consent was provided at the time of admission from patients or their legal.
Trial registration
Clinicaltrials.gov ID: NCT05760950.
Guarantor
Mengke Zhang
Contributorship
Mengke Zhang, Chuanjie Wu, Xunming Ji designed the study. Ruiwen Che, Liu Xin, Zhongyue Wang, Sen Hu, Shengqi Fu, Yuan Kan, Hailiang Sun, Jianmin Xu, Shiliang Ma, Sijie Li, Changhong Ren, Wenbo Zhao, Milan Jia, Jingang Wang collected the data. Mengke Zhang, Chengbei Hou analyzed the data. Mengke Zhang drafted the manuscript. Chuanjie Wu, and Xunming Ji revised the manuscript.
Data availability
Data can be provided on reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
