Abstract
Background:
There is paucity of data on the long-term outcomes after acute ischemic posterior circulation stroke (PCS). Additionally, the long-term prognostic value of the New England Medical Center-Posterior Circulation Registry (NEMC-PCR) classification of PCS has not been studied.
Patients and methods:
All consecutive patients with PCS registered in the Athens Stroke Registry between 01/1993 and 12/2012 were prospectively followed for up to 10 years and included in the analysis. The NEMC-PCR criteria were applied to classify them in relation to topography. The main studied outcomes were all cause mortality, stroke recurrence and major adverse cardiovascular events (MACEs).
Results:
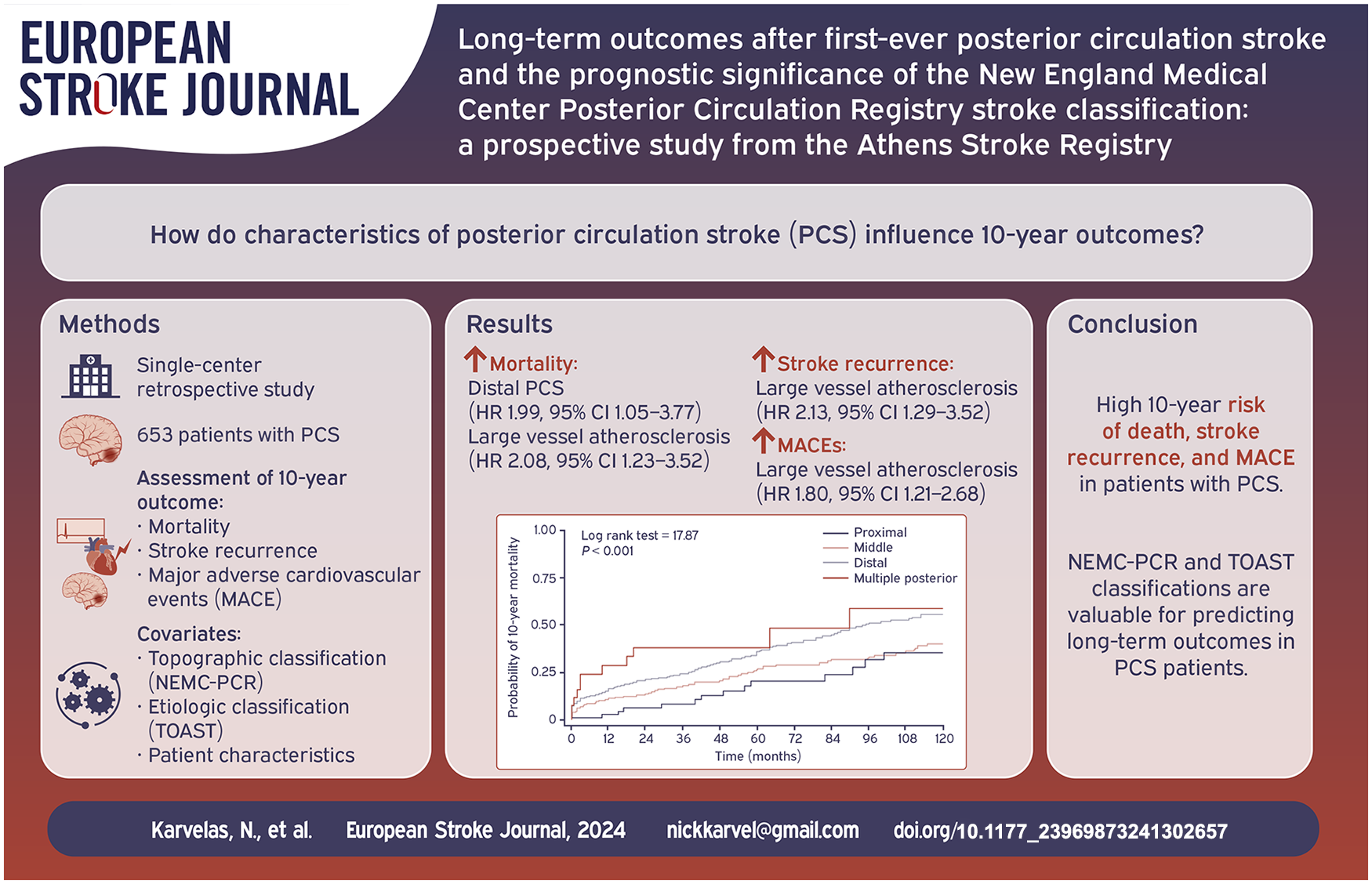
A total of 653 patients with PCS (455 men, mean age 68.06 years) were followed up for 52.8 ± 44.0 months. Seventy-four (11.3%), 219 (33.5%), 335 (51.3%), and 25 (3.8%) patients had proximal, middle, distal, and multiple territories PCS, respectively. During the 10-year follow-up period, 217 patients died (7.6 per 100 patient years), 127 developed recurrent stroke (4.2 per 100 patient years), and 209 had a MACE (7.3 per 100 patient years). The cumulative 10-year mortality was higher in distal and multiple territories PCS compared to middle and proximal PCS (55.6%, 58.8%, 40.0%, 35.5%, respectively, p < 0.001 by log-rank test). Patients with distal location PCS had almost twofold increased 10-year risk of mortality compared to proximal location patients after adjusting for all confounding variables (HR 1.99, 95% CI 1.05–3.77). Per TOAST classification, large artery atherosclerosis was associated with almost two-fold increase in risk of mortality, stroke recurrence and MACEs.
Discussion and conclusion:
A large proportion of PCS patients experienced 10-year death, stroke and MACE occurrence after PCS. NEMC-PCR topographic classification was found to have significant prognostic value, with distal and middle PCS having worse long-term outcomes than proximal PCS.
Introduction
Each year, approximately 795,000 people experience an ischemic stroke, 1 with approximately 20%–25% of these cases being posterior circulation strokes (PCS) caused by an infarction in the territories supplied by the vertebrobasilar arterial system.2,3 Unlike anterior circulation strokes (ACS), PCS are more likely to manifest with non-specific symptoms and non-focal neurologic findings leading to more frequent misdiagnosis or delayed diagnosis and treatment.4–6 In addition, the National Institutes of Health Stroke Scale (NIHSS), the most widespread clinical tool used in the acute stroke setting to quantitatively measure neurologic deficits and predict stroke severity, is skewed toward anterior circulation and might overestimate the outcome for patients with PCS.7–9 However, there is a paucity of data on the long-term outcomes in PCS, especially regarding recurrence and major cardiovascular events, as well as independent prognostic factors.
The Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification of subtypes of acute ischemic stroke has been the predominant system of classification of strokes since 1993. 10 TOAST is an etiologic classification of strokes that is convenient, with good inter-observer agreement and significant prognostic value.11–13 However, TOAST classifies strokes solely based on the presumed etiologic mechanism without considering the anatomic components. The New England Medical Center-Posterior Circulation Registry (NEMC-PCR) classifies ischemic infarcts as involving proximal, middle, and distal intracranial posterior circulation territories.14,15 However, the long-term prognostic value of this vascular-related topographic PCS classification has not been studied.
The purpose of this study is to investigate the long-term outcomes of PCS and to examine their prognostic factors, particularly focusing on the prognostic value of the NEMC-PCR vascular topographic ischemic stroke classification.
Materials and methods
Study design and data sources
All consecutive patients who experienced a first-ever acute stroke with symptoms onset within 24 h and admitted to “Alexandra” University Hospital between January 1993 and December 2011 were included in the study. 16 Patients were admitted from the emergency department and received treatment either in the acute stroke unit or in general medical wards when stroke unit beds were unavailable. Patients with transient ischemic attack (TIA), subarachnoid hemorrhage, and recurrent stroke were not included in the registry. Detailed data were prospectively recorded for each patient, including demographics, medical history, associated cardiovascular risk factors, clinical findings, and neurological severity on admission (assessed by NIHSS score). Additionally, laboratory investigations, and administered treatments were documented. Upon admission, a brain computed tomography scan and a 12-lead electrocardiogram were performed. A second CT or magnetic resonance imaging (MRI) of the brain was performed within the first week, and further investigations were carried out based on the protocol. The underlying ischemic stroke pathogenic mechanisms were determined using the TOAST classification. 10 The study’s neuroradiologist re-reviewed all available brain imaging studies, recording the ischemic stroke lesions in detail. To classify the territorial distribution, the criteria of NEMC-PCR were applied, categorizing strokes as proximal, middle, and distal (Figure 1).14,15 Branch diseases identified on imaging studies were included for statistical purposes in the small vessel disease (SVD) group. The data were processed and analyzed without any personal identifiers to ensure patient confidentiality. The study received approval from the institutional review board and the ethics committee of the “Alexandra” Hospital, adhering to the ethical principles outlined in the Declaration of Helsinki for medical research involving human subjects.
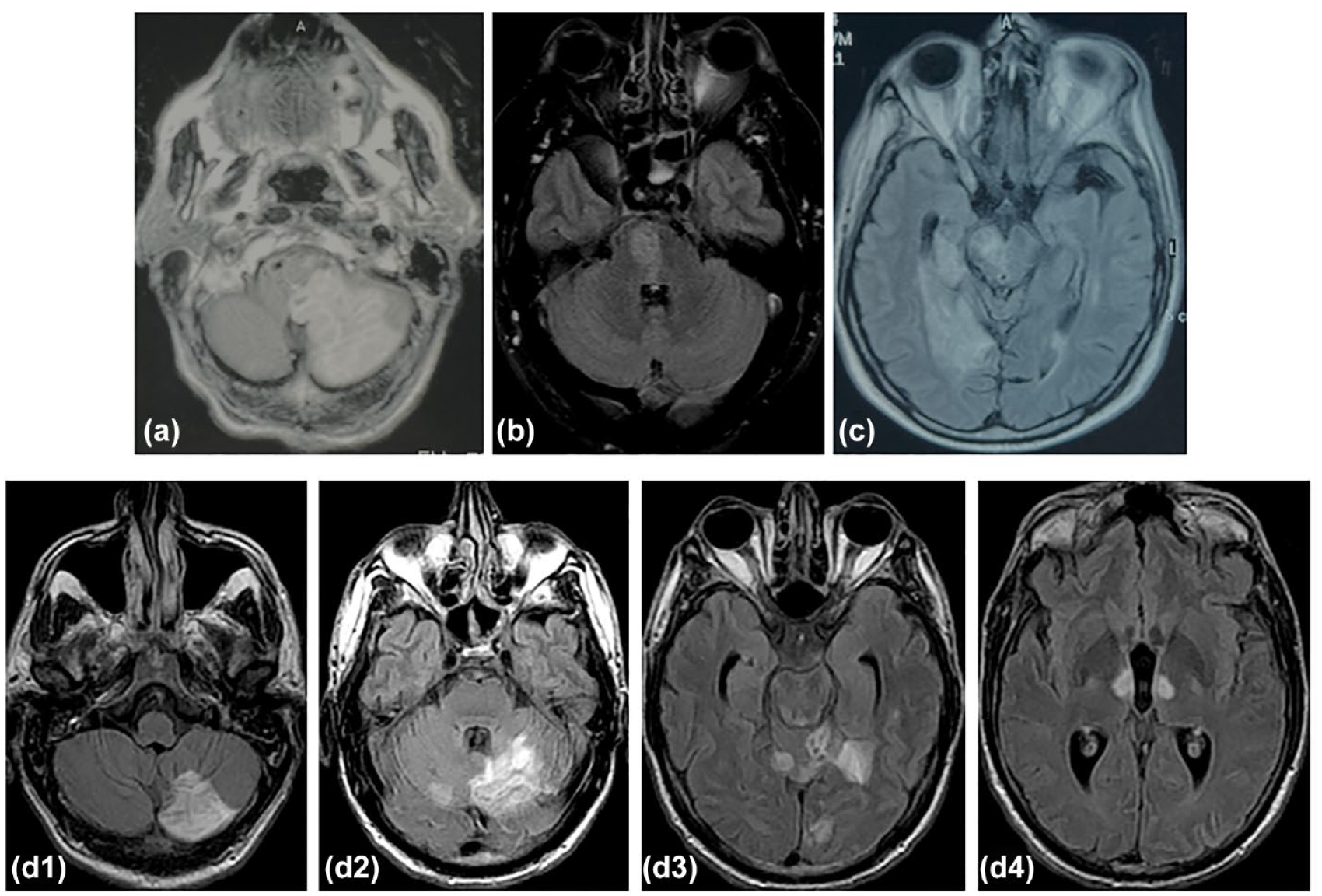
Examples of magnetic resonance (MRI) fluid-attenuated inversion recovery (FLAIR) imaging findings from PCS in the Athens Stroke Registry based on the New England Medical Centre Posterior Circulation Registry (NEMC-PCR) classification: Proximal (a), Middle (b), Distal (c) and multiple posterior territories in a patient with vertebral and basilar occlusions (d1–d4).
Outcomes and follow-up
Patients were prospectively followed-up at 1, 3, and 6 months after discharge and yearly thereafter for up to 10 years after stroke or until death by stroke physicians participating in the study. Follow-up was routinely performed in the outpatient clinic. Severely handicapped patients were monitored at their residence or via telephone interview. Death, cause of death, stroke recurrence, and major adverse cardiovascular events (MACEs) were ascertained by using death certificates, patient records, information from general practitioners, and family physicians’ records. Overall mortality during follow-up was the primary end point. The secondary end points were stroke recurrence and MACEs, defined as the composite cardiovascular event of recurrent stroke, new myocardial infarction or unstable angina, new peripheral arterial diseases or embolism, aortic aneurysm rupture and sudden death with or without resuscitation. Recurrent stroke was defined as a cerebrovascular event of sudden onset, lasting more than 24 h, occurring subsequent to the initial stroke, resulting in a new neurological deficit or deterioration of the previous neurological deficit, as demonstrated on brain imaging. For patients lost during follow-up, survival data were censored at the last time known they were alive. Patients who experienced more than one MACEs during the follow-up were censored at the time of the first event.
Statistical analysis
Patients were classified into four groups based on territory of the index PCS: proximal, middle, distal, and multiple posterior strokes. Continuous data are presented as mean with standard deviation (SD) or as median with interquartile range (IQR). Categorical data are presented as absolute and relative frequencies. The ANOVA test was used to compare the continuous variables and the chi-square test was applied to compare discrete variables. Cumulative survival was determined using Kaplan–Meier estimates and outcomes between groups was compared with the log-rank test. Patients who were lost during follow-up were censored in survival analyses at the date of their last visit. In case of more than one stroke or MACE recurrence during the follow-up period, patients were censored at the time of the first event. Univariate and multivariate analyses were conducted for each one of the studied outcomes. Univariate Cox regression models were used to identify baseline variables associated with 10-year mortality, stroke recurrence and incidence of MACEs. Factors that were significantly (p ≤ 0.05) associated with these outcomes were retained for the multivariate models. Two sets of multivariate models were computed: the first one on the entire study population (Model 1) and the second one only on patients that survived at least until discharge (Model 2). In Model 2 post-discharge treatment regimens were additionally included as covariates. Results from Model 1 were compared against results from Model 2 to ensure that the latter did not reflect survival bias. Proximal stroke was used as the reference category in order to perform dichotomous comparisons with patients in the other PCS groups. The results of regression models are given as hazards ratio (HR) with the 95% confidence interval (CI). As sensitivity analyses, the Fine and Gray competing risk model was used to estimate subhazard ratios assuming death as a competing risk, using the R package cmprsk. 17 The threshold of statistical significance was p ⩽ 0.05. All analyses were performed using STATA software (version 14·1; STATA Corporation, College Station, TX, USA) and R, version 4.2.1 (R Foundation for Statistical Computing, 2022).
Results
Baseline characteristics
From the cohort of 2815 patients with acute first-ever ischemic stroke, 653 (23.2%) exclusively involved the posterior territory, 2076 (73.7%) the anterior territory and 85 (3.0%) both territories. The mean elapsed time from symptoms onset to emergency room in patients with PCS was 4.2 ± 2.3 h. There were 455 (69.7%) men and 198 (30.3%) women with a mean age of 66.9 ± 11.2 (SD) and 70.7 ± 12.4 (SD), respectively (Table 1). There were 74 (11.3%) cases with proximal posterior vascular territory infarction, 219 (33.5%) with middle, 335 (51.3%) with distal, and 25 (3.8%) with infarcts in multiple territories. Single stroke location was found in the majority of cases (82.4%) and more than one ischemic lesion in the same vascular territory in 13.8% of cases (Supplemental Table 1). Large artery atherosclerosis documented (LAA) by angiography studies (>50% stenosis) were found in 126 (19.3%) cases (98 intracranial). The predominant etiopathogenic mechanism of proximal PCS was intracranial or extracranial atherosclerosis (52.7%) whereas SVD or branch disease was the most common mechanism in middle territory PCS (65.8%), and cardioembolism in the distal territory (33.1%).
Baseline characteristics of 653 patients with acute posterior circulation ischemic stroke (PCS).
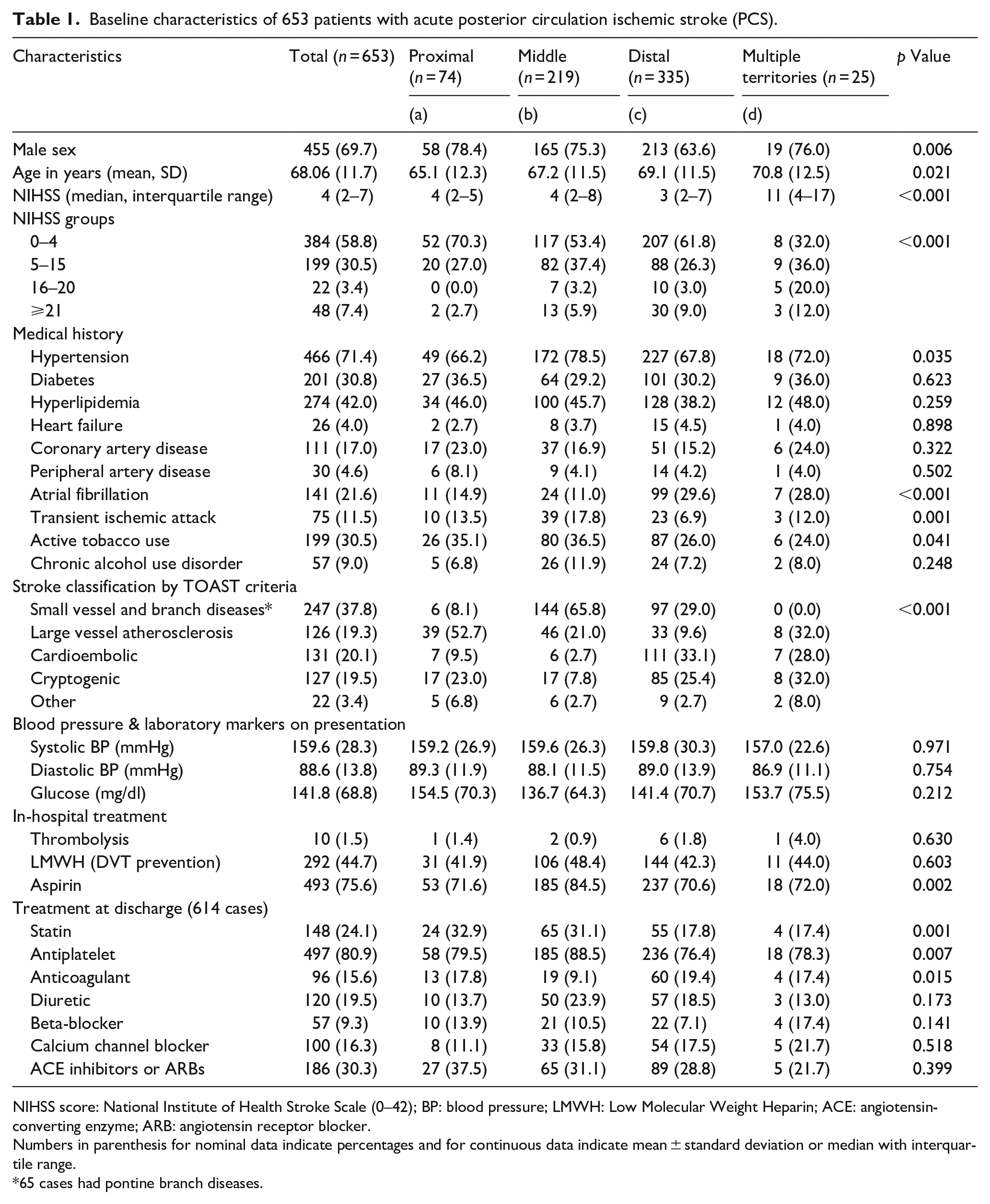
NIHSS score: National Institute of Health Stroke Scale (0–42); BP: blood pressure; LMWH: Low Molecular Weight Heparin; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker.
Numbers in parenthesis for nominal data indicate percentages and for continuous data indicate mean ± standard deviation or median with interquartile range.
65 cases had pontine branch diseases.
Follow-up and 10-year outcomes
Study patients were followed for a mean period of 52.8 ± 44.0 (SD) months, corresponding to a total of 2873 patient-years (median 3.46 years, IQR 1–8 years). Sixty-four (9.8%) patients were lost to the scheduled follow-up (from 1 to 117 months). During the follow-up period, 217 (33.2%) patients died (7.6 per 100 patient years), 127 (19.4%) developed recurrent stroke (4.2 per 100 patient years) and 209 (32%) had a MACE (7.3 per 100 patient years); recurrent stroke 127, myocardial infarction or unstable angina 55, new peripheral arterial diseases or embolism 9, aortic aneurysm rupture 4 and sudden death 14. Twenty-one patients experienced more than one MACE.
The total 10-year cumulative probability of mortality was 48.2% (95% CI 43.1–53.3) and was higher in distal PCS and multiple territories PCS compared to middle and proximal PCS (55.6%, 58.8%, 40%, 35.5%, respectively, p < 0.001 by log-rank test) (Table 2 and Figure 2). The overall 10-year stroke recurrence and MACE cumulative probabilities were 31.4% (95% CI 26.5–36.3) and 46.8% (95% CI 41.0–52.7), respectively. Similarly, 90-days mortality was higher in distal and multiple locations compared to proximal and middle. No statistically significant differences were observed on stroke recurrence and MACE between different stroke locations based on the NEMC-PCR criteria (Table 2, Figure 2).
Cumulative 90-days and 10-year probabilities (%) of stroke outcomes in patients with acute posterior circulation ischemic stroke (PCS) according to the New England Medical Center Posterior Circulation Registry (NEMC-PCR) criteria.
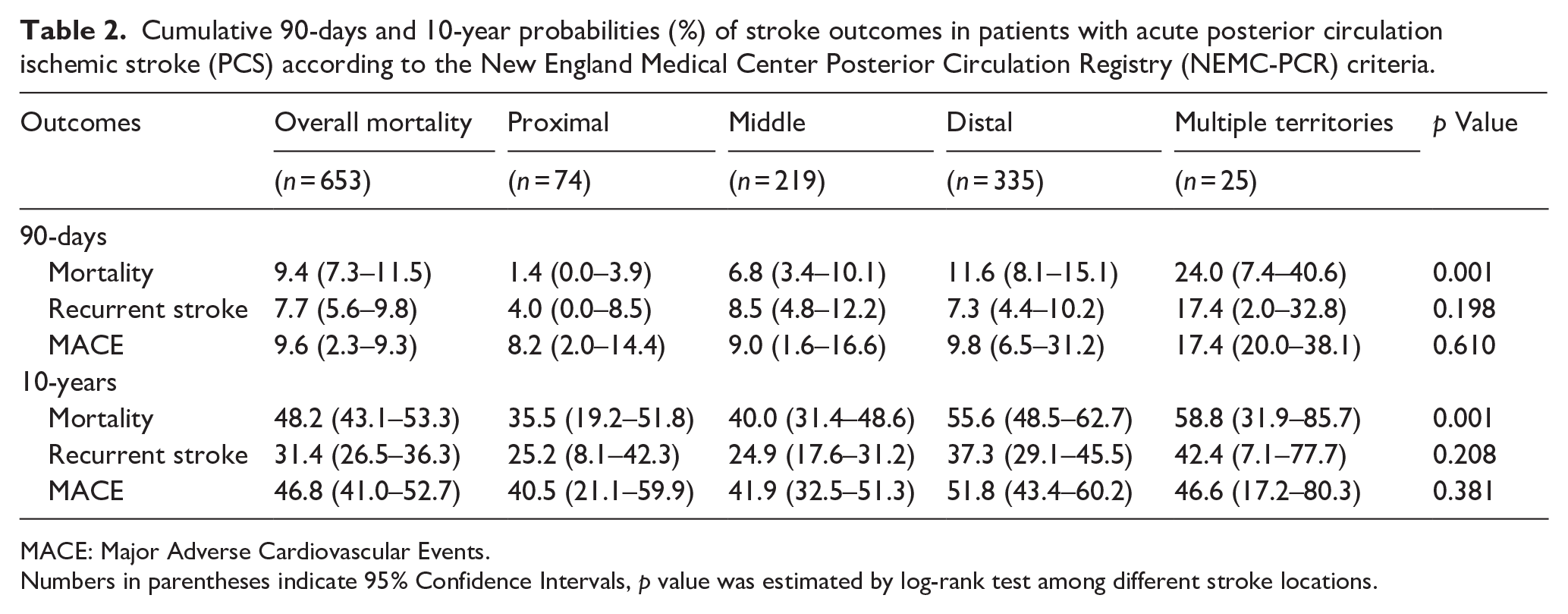
MACE: Major Adverse Cardiovascular Events.
Numbers in parentheses indicate 95% Confidence Intervals, p value was estimated by log-rank test among different stroke locations.
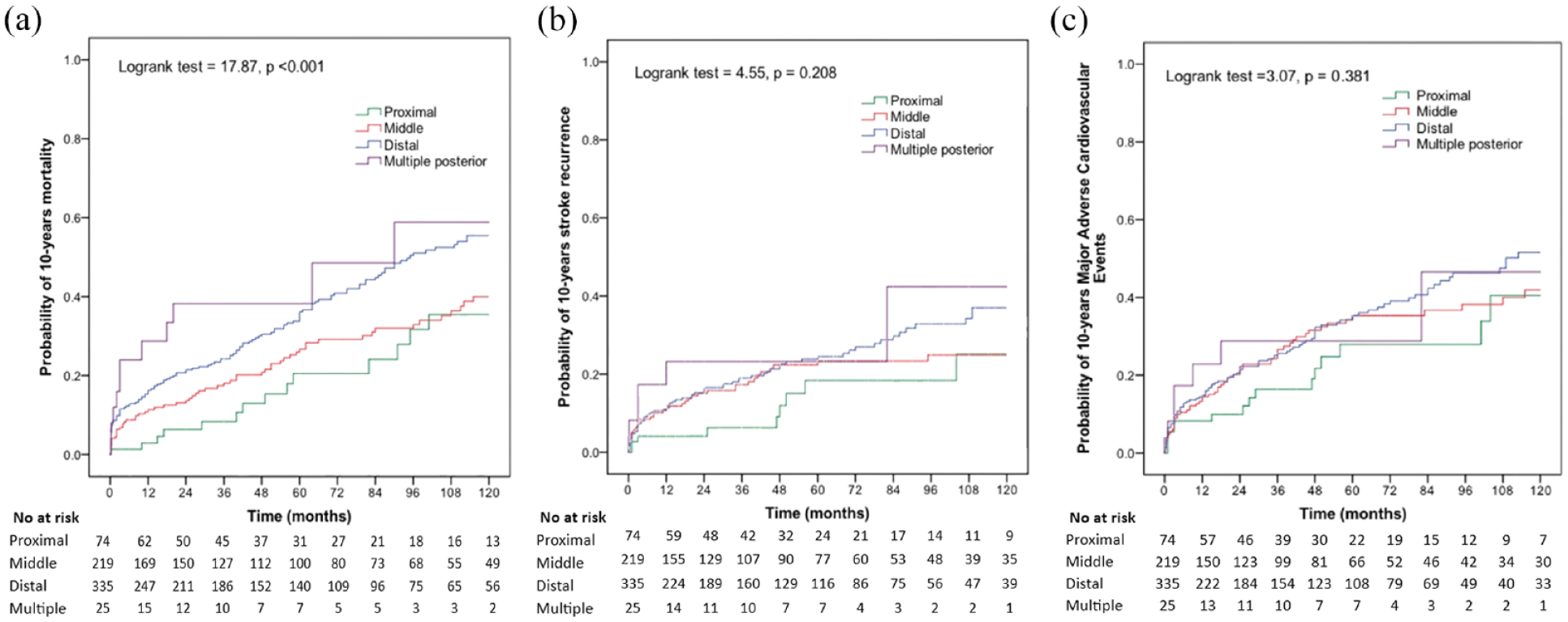
Kaplan–Meier estimates of probability of death (a), stroke recurrence (b) and major adverse cardiovascular events (c) over a period of 10 years after acute posterior circulation ischemic stroke (PCS) according to New England Medical Centre Posterior Circulation Registry (NEMC-PCR) criteria.
Prognostic factors
Regarding stroke localization based on NEMC classification, the distal PCS territory was associated with a two-fold increased mortality compared to proximal territory in model 1 (HR 1.91, 95% CI 1.05–3.46, p < 0.05) and model 2, (HR 1.99, 95% CI 1.05–3.77, p < 0.05). No associations emerged in univariate models between NEMC classification and stroke recurrence or MACE (Supplemental Tables 3 and 4). PCS secondary to LAA was found to be independently associated with mortality (HR 2.08, 95% CI 1.23–3.52, p < 0.01), higher stroke recurrence (HR: 2.13 95% CI: 1.29–3.52, p < 0.01) and MACEs (HR 1.80, 95% CI 1.21–2.68, p < 0.01) compared to SVD stroke in multivariate models respectively in Model 2 (Table 3). Cryptogenic stroke was associated with increased mortality (HR 2.61, 95% CI 1.58–4.31, p < 0.001) (Table 3). In addition, the presence of atrial fibrillation was found an independent predictor of stroke recurrence.
Final multivariate Cox regression analysis of 10-years outcomes in patients that survived hospitalization (n = 615) with acute ischemic stroke in posterior circulation.
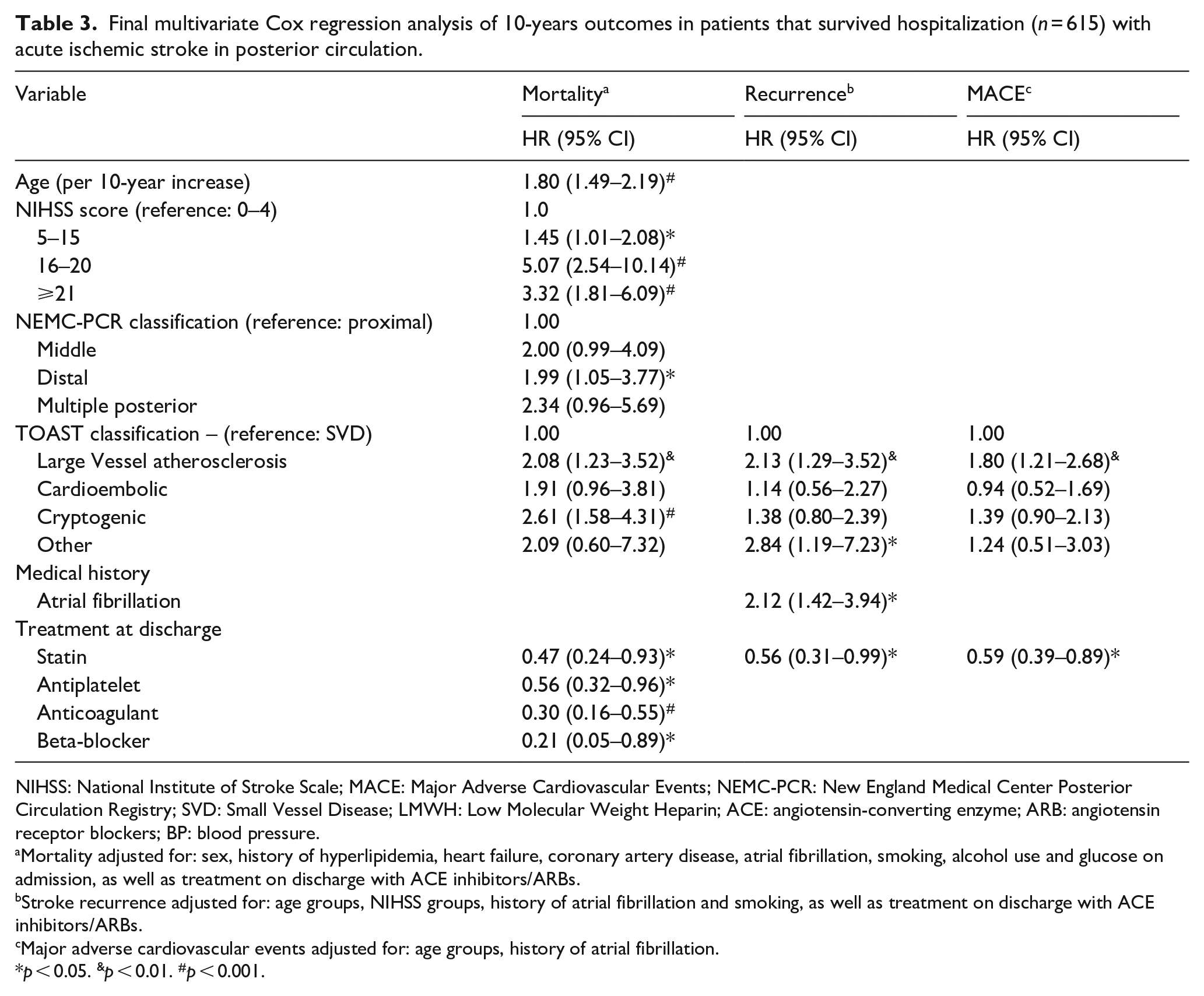
NIHSS: National Institute of Stroke Scale; MACE: Major Adverse Cardiovascular Events; NEMC-PCR: New England Medical Center Posterior Circulation Registry; SVD: Small Vessel Disease; LMWH: Low Molecular Weight Heparin; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blockers; BP: blood pressure.
Mortality adjusted for: sex, history of hyperlipidemia, heart failure, coronary artery disease, atrial fibrillation, smoking, alcohol use and glucose on admission, as well as treatment on discharge with ACE inhibitors/ARBs.
Stroke recurrence adjusted for: age groups, NIHSS groups, history of atrial fibrillation and smoking, as well as treatment on discharge with ACE inhibitors/ARBs.
Major adverse cardiovascular events adjusted for: age groups, history of atrial fibrillation.
p < 0.05. &p < 0.01. #p < 0.001.
Treatment with statins post-discharge decreased the risk of mortality (HR 0.47, 95% CI 0.24–0.94, p < 0.05), stroke recurrence (HR 0.56, 95% CI 0.31–0.99, p < 0.05) and MACE occurrence (HR 0.59, 95% CI 0.39–0.89, p < 0.05) in multivariate models (Table 3). Other treatments that attenuated the 10-year risk of death were antiplatelet therapy (HR 0.51, 95% CI 0.32–0.84, p < 0.01), anticoagulants (HR 0.21, 95% CI 0.12–0.39, p < 0.001) and beta-blockers (HR 0.21, 95% CI 0.05–0.89, p < 0.05) (Table 3). Competing risk analyses for recurrent stroke and MACEs, with death as competing risk, did not differ significantly from the Cox regression models (Supplemental Tables 5 and 6).
Discussion
Our study showed that in PCS, the probabilities of mortality, stroke recurrence, and MACE occurrence were 48.2%, 31.4%, and 46.8%, respectively, during the 10-year follow-up period. Distal PCS, as per NEMC-PCR criteria, was significantly associated with a higher 10-year mortality. Additionally, stroke secondary to LAA and cryptogenic stroke were independently associated with a higher 10-year risk of death.
To the best of our knowledge, our study is the first to report on 10-year mortality, recurrent stroke, and MACEs after first-ever ischemic PCS. A few other studies have investigated mortality after PCS, albeit for shorter timeframes. The 3-month mortality rates reported in these studies range widely between 1.3% and 11.0%.18–20 Data from the Acute STroke Registry and Analysis of Lausanne, a prospective cohort of consecutive acute ischemic stroke patients from 2003 to 2008, reported a 10.5% 90-day mortality rate, similar to our 9.4%. 21 In contrast, a multicenter European study conducted in 22 hospitals found a higher 3-month mortality rate among PCS patients (16.1%) compared to ours. 22 In the landmark study of the NEMC-PCR, the 30-day mortality rate was quite low (3.6%).15,23 However, the results from the NEMC-PCR study were affected by referral bias, as patients with severe strokes were less likely to be referred, and the sample population was skewed toward patients with TIA. The differences found are mainly related to the patients’ age, severity at admission based on the NIHSS scale, the selection criteria of the cases (stroke and/or TIAs), and stroke etiopathogenic mechanism.
Approximately one-third of our study patients had a probability of stroke recurrence rate during a 10-year follow-up period. No statistical differences were observed based on the topographical NEMC-PCR classification of patients. This could be partially explained by the differences in the distribution of the etiopathogenic stroke mechanism and the risk factor profile of the patients in our study, as well as treatment efficacy during secondary prevention. Results from a systematic review and meta-analysis showed a 39.2% cumulative stroke recurrence rate at 10 years, results similar to ours of PCS although including anterior and posterior circulation patients. 24 In the recently published CHANCE-2 study, which included patients with minor ischemic stroke and TIAs, 7.4% of patients with PCS experienced recurrence within 90 days compared to a similar range of 8.3% in the anterior circulation. 25 Almost half of our patients had the probability to develop MACE during the follow-up study period. To the best of our knowledge, this was the first study to estimate the long-term cumulative incidence of MACEs after PCS, while a few studies have reported on long-term MACE after ischemic stroke in general. The 5-year cardiovascular event occurrence rate was 13.5% for patients with mild-to-moderate ischemic stroke in a recently published cohort from China. 26 The five-year risk of myocardial infarction or vascular death after ischemic stroke was 17.4% in the Northern Manhattan Study. 27 It should be mentioned that our findings cannot be directly compared to other studies because the definitions of MACE and the follow-up period have differed between studies. However, our results indicate the need for intensive and sustained control of cardiovascular risk factors after PCS.
This is the first study to assess the possible role of topographic classification per NEMC-PCR criteria in the long-term prognosis after PCS, and our findings suggest that this classification may have significant long-term prognostic value. In the NEMC-PCR, Professor Caplan and his team categorized PCS brain lesions as involving proximal, middle, and distal posterior circulation territories and reported that embolism was the most common underlying etiologic mechanism leading to distal events, SVD for middle PCS, and LAA for proximal events. 15 These findings are largely confirmed by our study. While TOAST etiopathogenic classification remains of paramount clinical and prognostic importance, our results indicate that NEMC-PCR topographic classification should be used complementary to TOAST for PCS. Importantly, the increasingly widespread availability of CT angiography (CTA) enables precise occlusion localization, in which setting the NEMC-PCR could be utilized for acute management. Despite patients with distal PCS presenting with a lower median NIHSS score than middle and proximal territory PCS, probability of death was significantly higher for distal strokes, indicating that these patients might have later deteriorated to moderate or severe deficits (NIHSS > 10). The majority of PCS patients with mild deficits (NIHSS ⩽ 10) receiving either thrombolysis or endovascular therapy has been shown to have favorable outcomes,28,29 thus patients with distal PCS should be considered for early intervention. Additional imaging parameters that have been shown to predict outcomes and could complement the predictive ability of the NEMC-PCR classification are the extent of the lesion, as quantified by the posterior circulation Alberta stroke program early CT score (pc-ASPECTS), 30 and the presence of collateral circulation. 31 Furthermore, patients with distal territory involvement may need to undergo a more meticulous work-up for cardioembolic risk factors and more intensive management in terms of secondary prevention.
Regarding etiologic classification, a previous study of PCS outcomes by etiology at 3 months found that LAA and cardioembolic strokes had fourfold higher mortality than other causes. 32 In long-term population-based studies that included ischemic strokes of anterior and posterior circulation, cardioembolic stroke has been associated with higher mortality, but no differences were found in stroke recurrence among stroke subtypes.12,33 In contrast, Amarenco et al., 34 in a large sample of patients with minor stroke or TIA, showed that LAA and cardioembolic stroke had a double probability compared to undetermined cause in experiencing stroke in the following 12 months. Recently published studies indicate that patients with intracranial atherosclerotic diseases in the posterior circulation are at higher risk for stroke recurrence. In a pooled analysis of 323 PCS patients with documented vertebrobasilar stenosis, 20.4% presented with recurrent stroke (11.4%) or TIA (10%) during a 90-day follow-up period. 35 The risk of recurrence was fivefold in cases with intracranial stenosis. In a Chinese study, following 218 patients with intracranial atherosclerosis, the reported recurrence rate over a 1-year period was higher in the posterior (25.5%) compared to anterior circulation (14.2%). 36 After adjusting for several confounders, we found that LAA stroke in PCS patients had a twofold increased risk of recurrence and MACE occurence compared to small vessel. The majority of these patients (82%) had angiographically documented intracranial atherosclerotic arteries. Critical stenosis of the large arteries in the posterior circulation and unstable atherosclerotic plaques persist after the onset of stroke and are causes of recurrent thromboembolic events. Studies with angioplasty and stenting in patients with symptomatic intracranial atherosclerosis have so far failed to improve patient outcomes. Aggressive management of risk factors and dual antiplatelet therapy is recommended for 90 days, which is then reduced to aspirin alone. 37 Moreover, our finding that atrial fibrillation was an independent predictor of recurrence in PCS patients suggests more efforts are needed to treat such patients with well-established anticoagulants.
A secondary finding of our study is that statin administration at hospital discharge after acute ischemic PCS is associated with at least an approximately 50% reduction not only in stroke recurrences and MACEs but also in mortality. Although our study did not assess patient adherence to treatment during long-term follow-up, the finding is important and suggests the necessity of aggressive cholesterol lowering in patients with acute PCS.38,39 Another interesting finding, the strong association of administration of β-blockers with a reduction in mortality, even though they are mild antihypertensive drugs, could perhaps be explained by their antiadrenergic effect. Many regions of the brain supplied by the posterior circulation are involved in the regulation of autonomic functions of the central nervous system. 40 Damage to these areas resulting in a massive release of catecholamines could lead to sudden neurogenic death.
The strengths of our study include its large consecutive patient population, its prospective design with a long follow-up period, detailed recording of etiopathogenic and topographic characteristics of events, as well as assessment of long-term hard outcomes, including major cardiovascular events that have scarcely been reported in the literature. On the other hand, our study has some limitations. First, this was a single-institution registry from a tertiary healthcare center; hence, our findings should be interpreted with caution before being applied to populations with different characteristics. A population-based setting with a specific catchment area would be more accurate for quantifying outcomes in the general population. Second, 9.8% of enrolled patients were lost to follow-up; although this is an acceptable rate, it may have introduced selection bias. Third, compliance and adherence to treatment after discharge was not universally documented and may have affected the outcomes of the study.
In conclusion, in this prospective cohort of patients after first-ever PCS, we found that half of PCS patients have the probability of dying or experiencing a major cardiovascular event and one third have the probability of experiencing a stroke during the 10-year follow-up period. NEMC-PCR topographic classification of PCS was found to have significant prognostic value after PCS. Distal and middle PCS were shown to have worse long-term outcomes than proximal PCS. Cardioembolic events were the most frequent etiopathogenic type in distal PCS. Our findings support the use of the NEMC-PCR topographic classification in clinical practice along with the TOAST etiopathogenic classification for patients with PCS and indicate the need for a more thorough work-up for embolism in patients with distal PCS and more intense secondary prevention strategies and follow-up in all patients with PCS, particularly in those with distal and proximal PCS.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241302657 – Supplemental material for Long-term outcomes after first-ever posterior circulation stroke and the prognostic significance of the New England Medical Center Posterior Circulation Registry stroke classification: A prospective study from the Athens Stroke Registry
Supplemental material, sj-docx-1-eso-10.1177_23969873241302657 for Long-term outcomes after first-ever posterior circulation stroke and the prognostic significance of the New England Medical Center Posterior Circulation Registry stroke classification: A prospective study from the Athens Stroke Registry by Nikolaos Karvelas, Leonidas Palaiodimos, Dimitrios Karamanis, Dimitrios Sagris, Anna-Maria Louka, Panagiotis Papanagiotou, Eleni Korompoki, George Ntaios and Konstantinos Vemmos in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the institutional review board and the ethics committee of the “Alexandra” Hospital, Athens, Greece.
Informed consent
Written informed consent was obtained from all patients before data collection.
Guarantor
KV
Contributorship
KV and GN conceived the study. KV was involved in protocol development, gaining ethical approval and patient recruitment. KV, NK, LS, and DK performed data analysis and interpretation of results. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
All de-identified data employed in this study are available upon reasonable request from any qualified investigator for replication of procedures and results.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
