Abstract
Introduction:
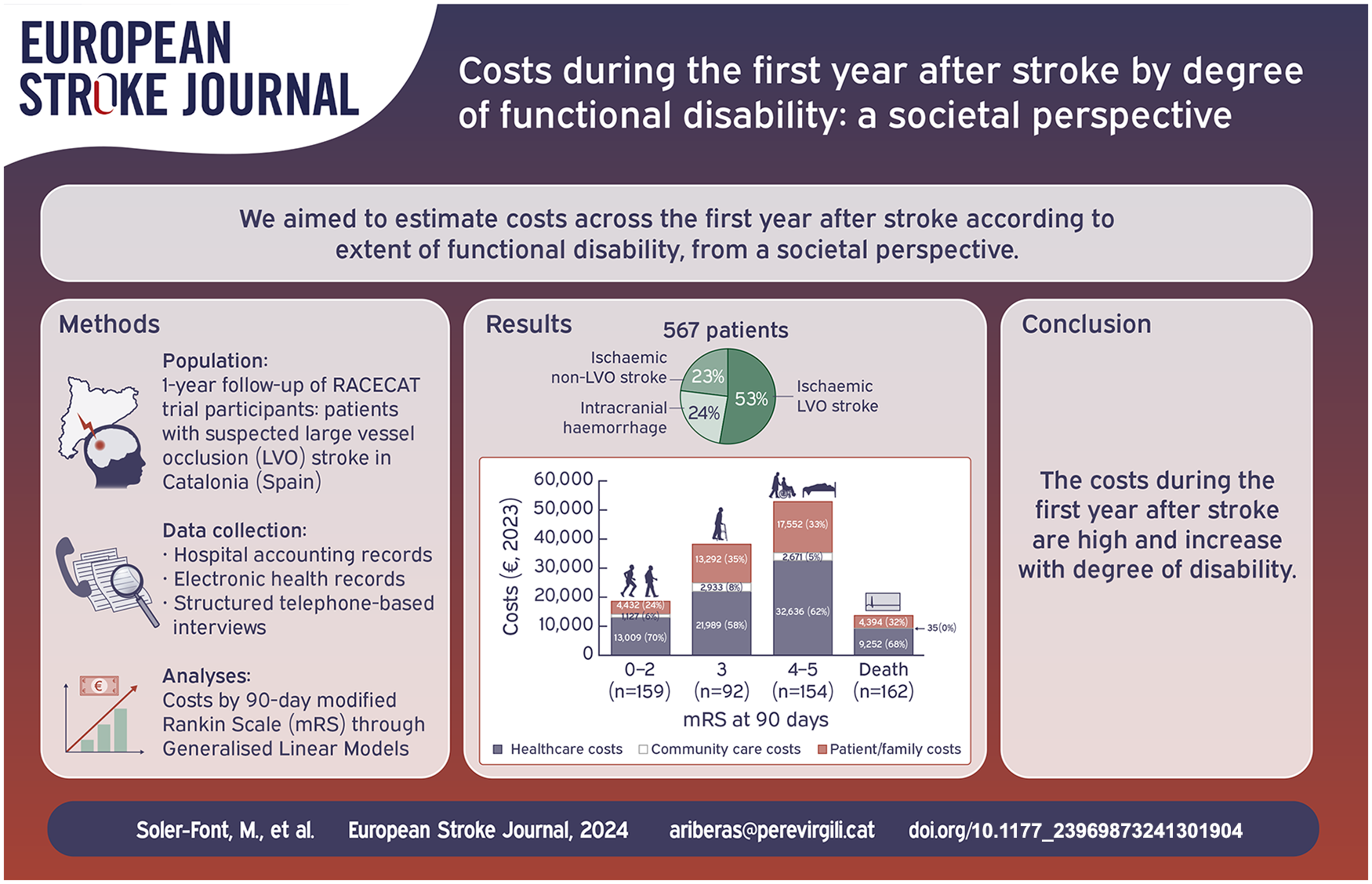
The aim of this study was to estimate societal costs during the first year after stroke by degree of functional disability.
Patients and methods:
Descriptive study of the cumulative costs incurred during 1-year follow-up of a cohort of patients with stroke in Catalonia (Spain) participating in a multicentre, population-based, cluster-randomised trial (RACECAT). Patients were recruited between September 2017 and January 2019. Costs were collected for each patient from stroke onset to 1-year follow-up through hospital accounting records, electronic healthcare records and structured telephone-based interviews at 6 and 12-months follow-up. Disability was assessed using the 90-day modified Rankin Scale (mRS). Healthcare, community care, and patient/family costs were included. We used complete data from 567 eligible participants. Cost data were analysed using generalised linear models (GLMs) with gamma distributions and log link functions. For variables with >10% zero values, two-part models were applied. We performed sensitivity analyses modifying unit costs for patient/family costs.
Results:
Of the 567 patients included, 53% had ischaemic large vessel oclusion (LVO) stroke, 24% intracranial haemorrhage and 23% ischaemic non-LVO stroke. Mean cost per patient during the first year after stroke was €29,673 ± 28,632, and increased with degree of disability (mRS 0–2: €18,568 ± 12,244; mRS 3: €38,214 ± 28,172; mRS 4–5: €52,859 ± 36,383). Healthcare costs represented the highest proportion of total costs (63%; €18,724/patient) across all disability levels, with index hospitalisation being the highest (€12,319 ± 17,675); however, community care and patient/family costs represented over 40% of total cost in patients with higher disability levels.
Discussion and conclusion:
Our results are in line with other studies; the costs during the first year after stroke are high and increase with disability. These results are valuable for calculating the cost of severe stroke cases.
Introduction
Stroke has a significant impact on a person’s physical, cognitive and functional capacity. Disability due to stroke can affect work productivity and result in a need for care and support. 1 Consequently, there is a substantial economic burden on individuals, families, and society as a whole. 2
Most studies focus on healthcare costs, but a few take a societal perspective emphasising the importance of the burden stroke poses on patient/family costs, especially for those patients with high disability, and among younger adults due to productivity loss.3,4 In 2017, the cost of stroke was estimated at a total of €60bn, of which, €27bn were healthcare costs, €5bn were community costs, and €28bn were patient/family costs (of these, €16bn were caregiving and €12bn were patient productivity loss) in 32 European countries. 5 In 2016, a Spanish study concluded that the mean cost per patient was €27,711 during the first year, of which more than two-thirds corresponded to patient/family costs (mainly caregiving). 6
The modified Rankin Scale (mRS) at 90 days is considered the main tool to assess functional disability post-stroke. 7 Cost estimates relating to 90-day mRS categories may be valuable for healthcare economic evaluations of stroke.8,9
The aim of this study was to estimate costs during the first year after stroke by the degree of functional disability measured by 90-day mRS, from a societal perspective in a cohort of patients in Catalonia (Spain).
Methods
Study context and population
We assessed a sub-cohort of patients recruited in the RACECAT trial (ClinicalTrials.gov NCT02795962). RACECAT was a multicentre, population-based, cluster-randomised trial involving patients in Catalonia (Spain), with suspected large vessel occlusion (LVO) stroke attended by emergency medical services (EMS) in areas where the closest local stroke centre was not capable of performing thrombectomy. Details of the RACECAT trial are reported elsewhere.10,11
We included all consecutive patients between September 2017 and January 2019 (n = 629). We included 567 patients after excluding stroke mimics (n = 42), transient ischaemic attacks (TIAs; n = 14) and six patients lost to follow-up (i.e. patients who did not answer 6- and 12-month follow-up questionnaires as they could not be located; Figure 1). Loss to follow-up patients did not show significant differences in sex, age, mRS at 90 days and diagnosis, compared to included patients.
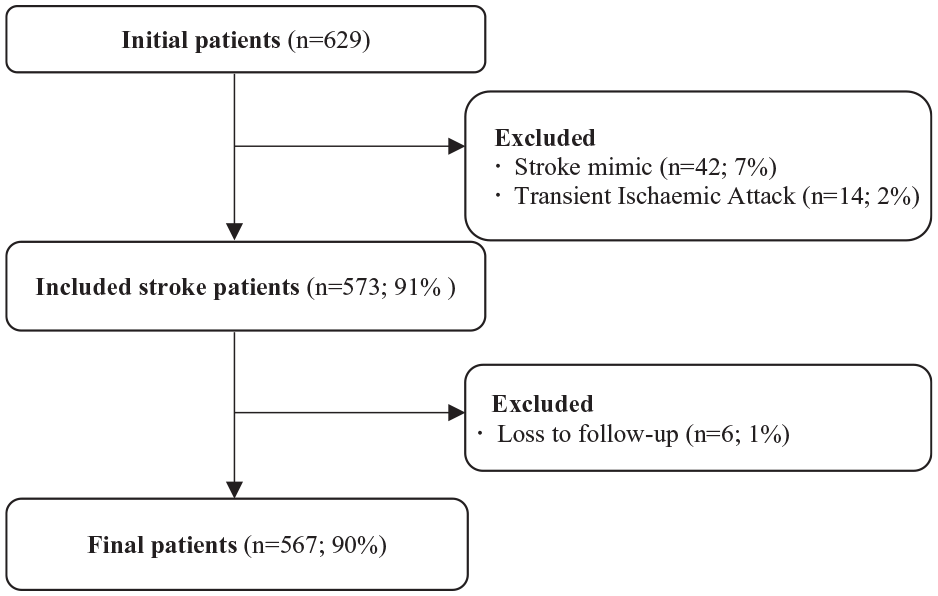
Study flowchart.
Data collection
We collected data during 12 months from stroke onset. Data were obtained from: (1) hospital accounting records; (2) electronic healthcare records; and (3) structured telephone-based interviews with the patient and/or caregiver or close relative. Interviews were conducted at 90 days to assess the mRS, and at 6 and 12 months after stroke to retrospectively collect data on use of community care and patient/family resources used since the stroke and up to 1-year. For patients who died between follow up periods, interviews were not conducted and thus community and patient/family resource consumptions during survival within these periods could not be assessed. With the exception of the six patients lost to follow-up (excluded), the database had no missing information.
Variables
Modified Rankin Scale (mRS) at 90 days
The mRS assesses the level of disability in people after stroke. 12 Outcome assessors assign a score from 0 (no symptoms) to 6 (death), with each score representing a different level of disability or functional impairment. We categorised the mRS at 90 days post-stroke into 4 levels according to clinical criteria13,14: 0–2 (no disability or slight disability), 3 (moderate disability), 4–5 (severe disability) and 6 (death).
Costs
No discount rate was applied because the cost description 15 had a 1-year follow-up period. All costs are expressed in euros (€) for the reference year 2023. Cost classifications followed Drummond et al. and ESO Health Economics recommendations.9,15
Healthcare costs
Acute phase included the Emergency Medical Service (EMS) and the index hospitalisation. EMS costs for transport for the stroke episode were obtained from accounting records, which take into account the availability of resources in each healthcare region, that is, the cost of each EMS service is lower [on average] in regions were the number of ambulances per capita is higher. Index hospitalisation costs were estimated from hospital accounting records through a micro-costing approach (detailed in Supplemental Material 1), including admission days to the emergency ward, general ward, stroke unit or intensive care unit, hospital medication and procedures. Costs were updated to 2023 using the Spanish Health-specific Consumer Price Index (CPI). 16
The follow-up phase captured the use of healthcare resources from hospital discharge to 12 months, including new hospitalisations (acute and intermediate care), outpatient rehabilitation, mental healthcare, minor outpatient surgery and other medical services. Data were obtained from the Catalan Health Surveillance System, an electronic health record system that tracks public health service utilisation and costs, weighted by Adjusted Morbidity Groups (AMG). 17 Since these data were provided in aggregated form, it was not possible to directly separate stroke-related and unrelated healthcare costs. To address this and minimise any potential inflation, a percentage was attributed to each cost category based on a previous study, 18 and these percentages were validated through consensus with clinical experts (Supplemental Table 1). Costs were calculated for the reference year 2023.
Nursing home use was assessed via telephone interviews, with costs derived from the published 2023 tariffs of the Catalan Government. 19
Community care costs
Use and costs of General Practitioner clinics, community medication and outpatient consultations were obtained from the Catalan Health Surveillance System 17 for 2023. These costs included both stroke-related and unrelated costs, to which we applied the same percentage allocation approach to avoid cost inflation, as done for all costs derived from the Catalan Health Surveillance System.
Use of telecare, home-care (public, private and home resident caregiver) and home modifications or adaptations were obtained from interviews. Telecare and public home healthcare costs were obtained from the published Catalan tariffs for the year 2023. 20 The unit cost for private home healthcare and home resident caregivers was the minimum wage in Spain for caregivers for the year 2023. 21 Home modifications and adaptation costs were obtained from standard quotes for 2018 of the Centre for Personal Autonomy (Centre de Vida Independent), a return-home pilot programme in Catalonia that included the costs for the adaptations and home modifications. 22 Home modifications and adaptations are one-off costs that can benefit the patient for many years. Therefore, considering that our sample had an average age of 74 years, and survival data after stroke suggest 5–10 years as a critical survival period, 23 as well as the additional years of staying at home before requiring institutional care in older adults with some degree of physical disability is estimated to be 5–10 years, 24 a functional lifespan of 10 years was estimated for these modifications. The total cost was then divided by this period to obtain the annual cost. These costs were updated to 2023 using the Spanish CPI. 25
Patient and family costs
Patient and family costs included paid and unpaid patient productivity loss, paid and unpaid caregiver productivity loss and additional caregiving hours not considered productivity loss by the caregiver. These were collected retrospectively at 6 and 12 months, and we used the human capital approach to calculate productivity losses.6,26
Patients’ paid productivity loss was counted as the number of hours they reduced work or stopped working due to stroke. The patients’ unpaid productivity loss was counted as the number of hours the patient stopped performing other unpaid activities due to stroke, such as caring for children or grandchildren, doing household chores, shopping, tending to a garden or orchard, caring for another person, volunteering or, if unemployed, looking for a job, participating in continuing education courses or engaging in employment programmes. Patients’ productivity losses were set to a maximum of 8 h per day for 238 annual working days (5 days/week, minus 22 annual paid vacation days) up to 1904 h/year. Additionally, for patients who died during the study period at an age below the mean life expectancy of Spain in 2023 (men: 80.3 years; women: 85.8 years), 27 productivity losses were calculated depending on the age of the patient. For them, we considered productivity losses as paid hours for those patients who were of working age (<65) and unpaid hours for those who were retired (⩾65). In both cases, we used the mean working hours annually from the OECD for Spain in 2023, 28 adjusted for the number of days the patient was no longer alive during the study period.
The caregivers’ loss of paid and unpaid productivity was collected following the same procedure. Caregiving hours were collected as the number of hours per day spent caring for the patient post-stroke. Based on previous recommendations we set a maximum possible of 16 h a day, 1 however the actual reported range was 0–11.2 h/day. To avoid double counting, paid and unpaid productivity loss hours were subtracted from total reported caregiving hours, as there were hours the person spent caring but not considered lost productivity. Therefore, the variable of caregiving hours refers to additional hours dedicated to care, but not considered as lost productivity by the caregiver.
For lost paid hours in both patients and caregivers, the unit cost was the mean gross salary in Spain for the year 2022 by sex, 29 updated to 2023 with the Spanish CPI, 25 for unpaid lost hours we used the minimum gross wage 21 and for caregiving hours variable we used the public home-care workers tariff as unit cost. 20 Table 1 shows unit costs used in this study.
Unit costs.
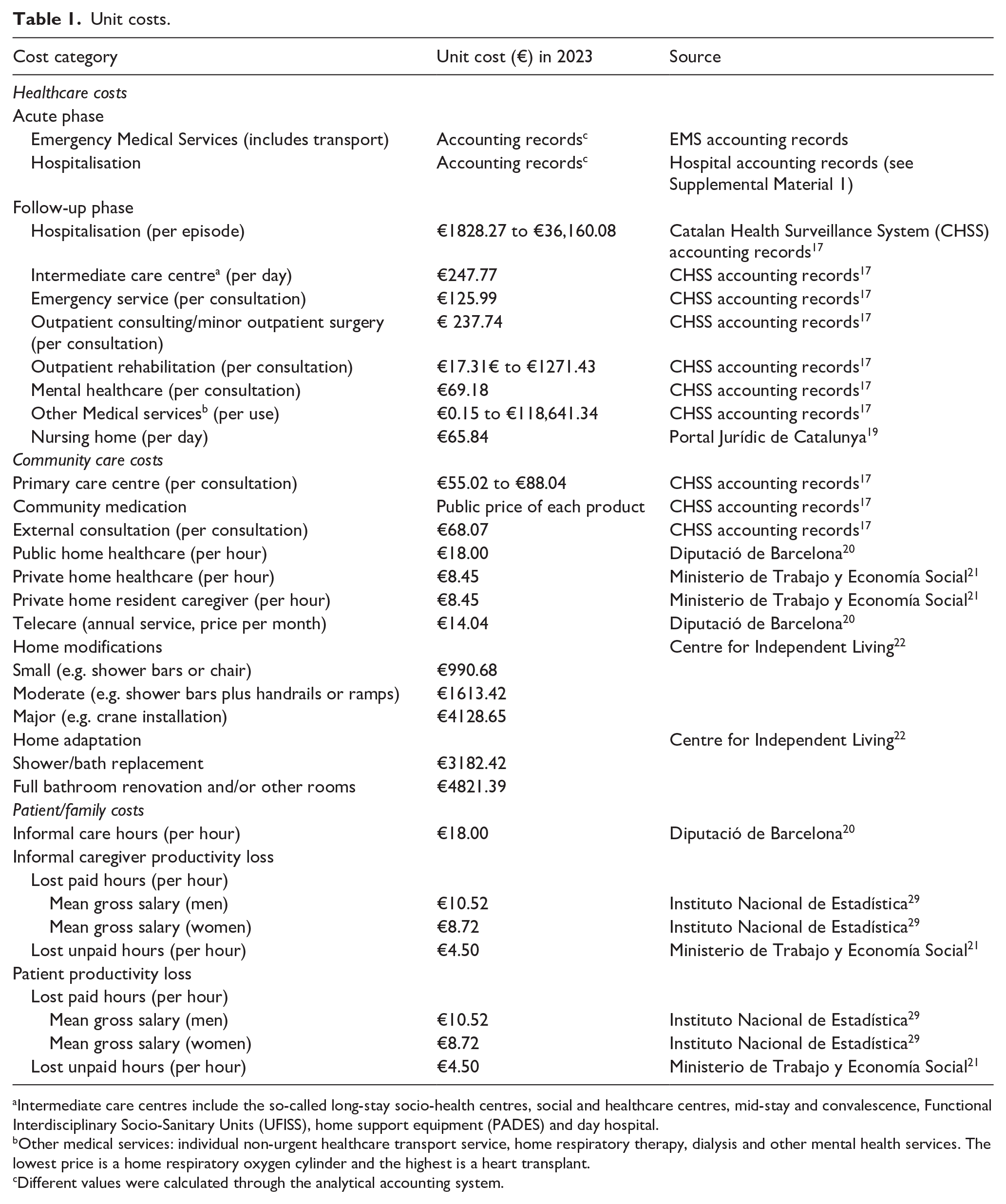
Intermediate care centres include the so-called long-stay socio-health centres, social and healthcare centres, mid-stay and convalescence, Functional Interdisciplinary Socio-Sanitary Units (UFISS), home support equipment (PADES) and day hospital.
Other medical services: individual non-urgent healthcare transport service, home respiratory therapy, dialysis and other mental health services. The lowest price is a home respiratory oxygen cylinder and the highest is a heart transplant.
Different values were calculated through the analytical accounting system.
Minor out-of-pocket expenses (e.g. hospital parking fees, fuel costs for hospital visits) were not included due to the risk of recall bias, as this information was gathered retrospectively during the 6- and 12-month follow-up interviews, covering the preceding 6 months in each interview and to avoid overburdening patients with an already lengthy questionnaire.
Statistical analysis
Baseline characteristics were described and compared by mRS using the chi-square test for categorical variables and ANOVA for continuous variables. To analyse resource utilisation, generalised linear models (GLMs) were applied. Continuous variables were modelled using a GLM with a gamma distribution and log link function, while categorical variables were analysed with a generalised logistic model using a binomial distribution and logit link. Cost data were also modelled using a GLM with a gamma distribution and log link. For both resource utilisation and cost variables with more than 10% zero values, two-part models with a gamma distribution and log link were employed, except for discrete variables such as telecare and nursing home utilisation, which were modelled using a Poisson distribution with a log link. Model fit was assessed using the Akaike and Bayesian Information Criterion (AIC and BIC).
Because there remains no consensus regarding the best calculation method for patient/family costs, 1 we performed three sensitivity analyses to address the potential variability in our assumptions: we estimated the total cost (1) considering the mean salary by sex in Spain; (2) the minimum wage in Spain; and (3) the private home healthcare minimum wage; as unit costs for informal care hours (not productivity losses).
All analyses were performed using Stata Statistical Software, version 17 (Stata Corp LLC, College Station, TX, USA).
Results
We included 567 patients in the study. The mean age was 74 years, with 59% being male. Among the patients, 53% had ischaemic LVO stroke, followed by 24% with intracranial haemorrhage and 23% with ischaemic non-LVO stroke (Table 2).
Baseline characteristics of patients and caregivers by 90 days mRS and overall.
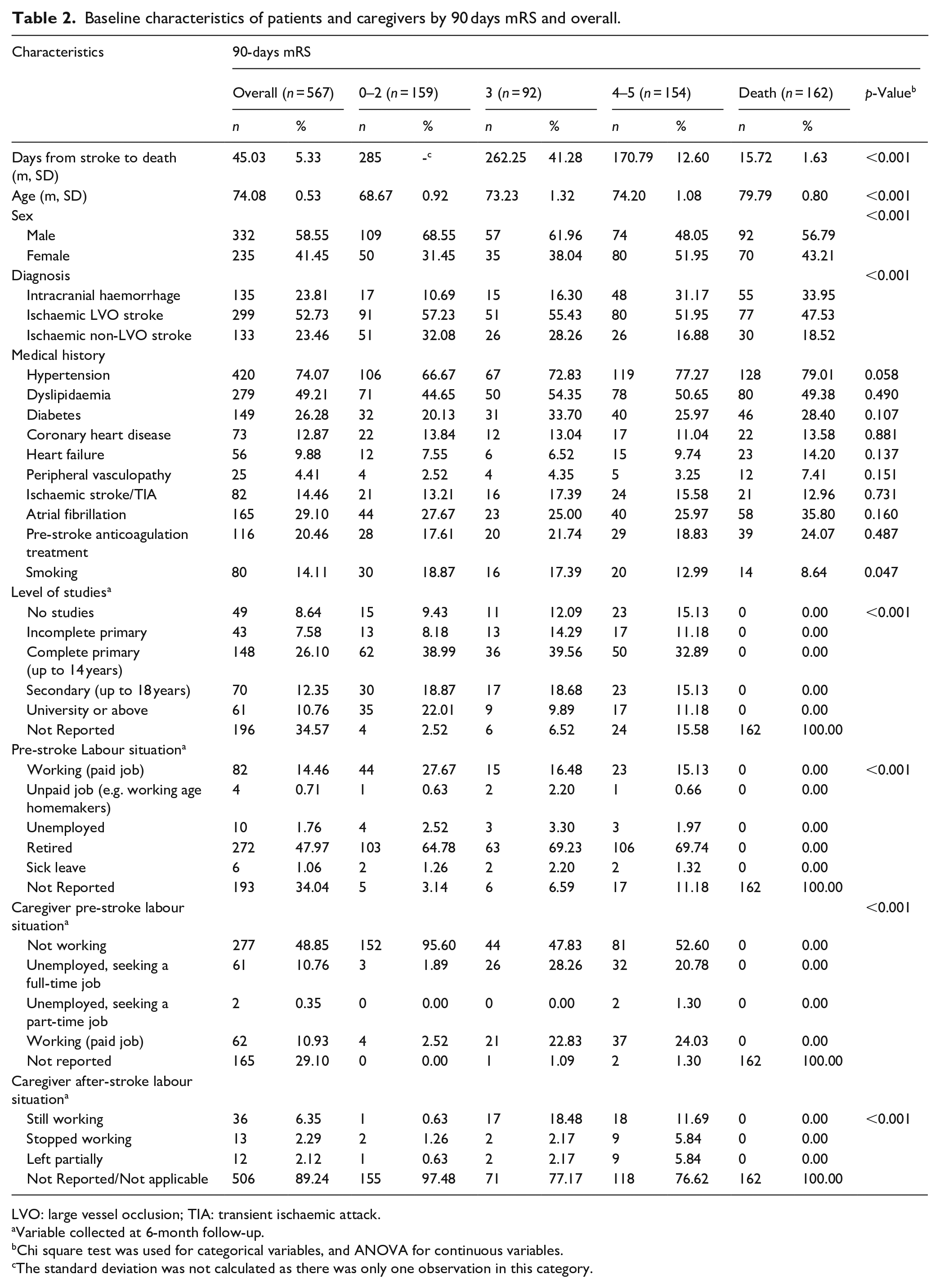
LVO: large vessel occlusion; TIA: transient ischaemic attack.
Variable collected at 6-month follow-up.
Chi square test was used for categorical variables, and ANOVA for continuous variables.
The standard deviation was not calculated as there was only one observation in this category.
Total costs per patient are described in Table 3. All costs increased with mRS score, with healthcare costs being higher than community and patient/family costs across all mRS groups (Figure 2).
Mean and median costs per patient during the first-year post-stroke by 90-days mRS.
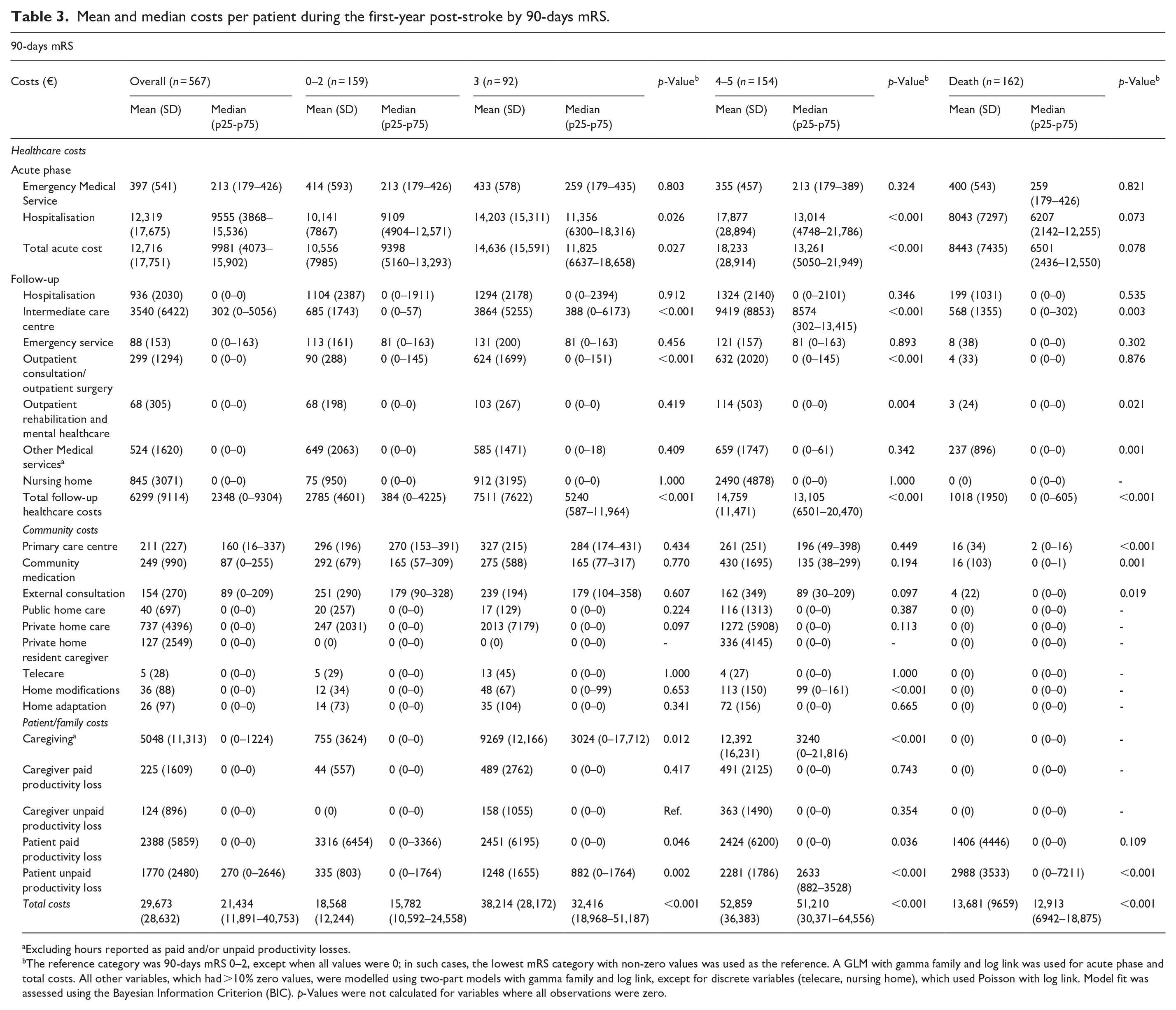
Excluding hours reported as paid and/or unpaid productivity losses.
The reference category was 90-days mRS 0–2, except when all values were 0; in such cases, the lowest mRS category with non-zero values was used as the reference. A GLM with gamma family and log link was used for acute phase and total costs. All other variables, which had >10% zero values, were modelled using two-part models with gamma family and log link, except for discrete variables (telecare, nursing home), which used Poisson with log link. Model fit was assessed using the Bayesian Information Criterion (BIC). p-Values were not calculated for variables where all observations were zero.
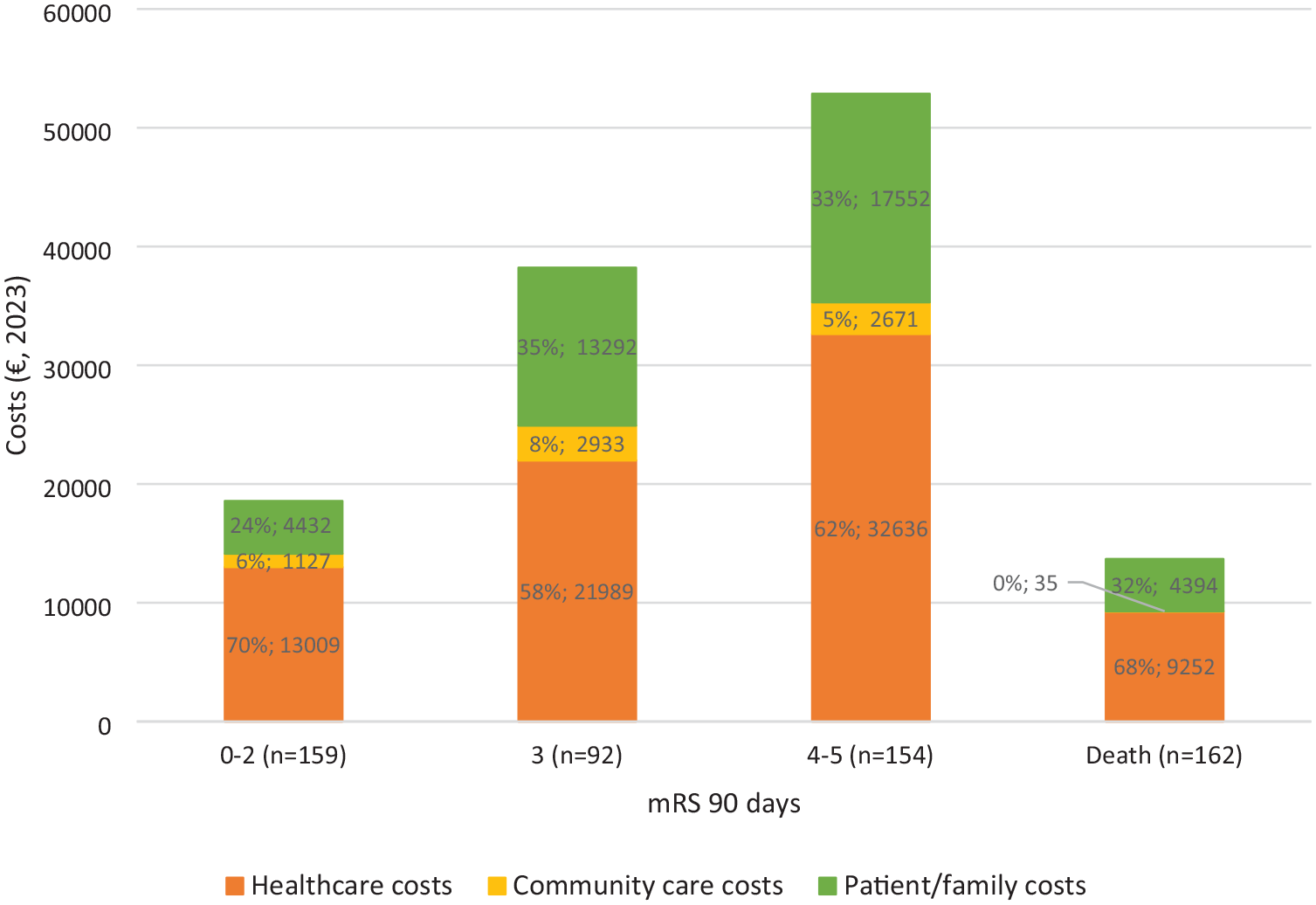
Mean costs per patient by type of resource and 90-day mRS group.
Costs by diagnosis, use of resources and costs by mRS categories are shown in Supplemental Tables 2–4. In all sensitivity scenarios (Supplemental Table 5), estimated costs were similar.
Discussion
Our results demonstrate a strong relationship between functional disability measured with mRS and costs after acute stroke, in line with previous literature.7,30,31 Each mRS category was associated with a different average length of hospital stay in the acute phase of stroke recovery, 31 and with different rehabilitation needs and nursing home care in the chronic phase, 30 increasing as mRS increased.
Studies examining stroke costs often focus on ischaemic stroke.1,18,32 In contrast, our study provides a comprehensive analysis encompassing various stroke subtypes, including LVO ischaemic stroke (53%), non-LVO ischaemic stroke (23%), and haemorrhagic stroke (24%). Compared to the recent Spanish study by Lucas-Noll et al., 33 which reported a mean cost of €22,605.66, our study found higher overall mean costs. This discrepancy is likely attributable to our cohort’s composition, characterised by a high proportion of severe LVO strokes, in contrast to their inclusion of TIAs, which generally incur lower costs. 33
Although 60% of costs across all mRS categories were healthcare costs, the highest proportion being index hospitalisation (M = €12,319), 18 patient/family costs represented a considerable burden, ranging from €4394 to €17,552, including caregiving (from €755 to 12,392€)1,6 and productivity losses. Patients’ mean paid productivity loss was €2388, being higher for mRS 0–2 (M = €3,316/patient), probably because there were more active workers pre-stroke in this group with lower mean age.
Limitations
Our sample represented a more severe illness profile compared to the general population, which limits the generalisability of overall results. Data obtained from the Catalan Health Surveillance System were provided in aggregated form, thus it was not possible to directly separate stroke-related and unrelated costs. To address this, we attributed a percentage of each cost category to stroke based on previous studies 18 and expert consensus. Estimations of productivity losses for deceased patients were limited due to missing follow-up questionnaire data, therefore assumptions were made based on age and life expectancy. Minor family/patient out-of-pocket expenses were not included due to the potential recall bias and the length of the questionnaire, which could slightly underestimate the costs. Moreover, as loss of leisure time of patients or caregivers was not directly collected, those costs could be underestimated. Finally, as some data were self-reported by patients, recall and/or comprehension bias cannot be excluded (e.g. unpaid productivity loss could be underestimated, since it can be difficult for caregivers to report this).
Implications for practice and research
Calculating stroke costs by incorporating healthcare, community and patient/family expenses enhances disease cost estimation, and facilitates the formulation of targeted measures and policies. These data indicate the need for healthcare and social policies aimed at caregiver support. Data on stroke costs also aids in refining future economic modelling frameworks, such as Markov or microsimulation models, thus facilitating more precise long-term economic forecasting and evaluation of intervention cost-effectiveness.
Conclusion
Our findings underscore the established association between functional disability and increased costs, with our sample exhibiting an overrepresentation of LVO strokes. Nonetheless, by quantifying the economic impact of severe strokes and taking into account healthcare, community, and patient/family expenses, we provide policymakers and healthcare providers valuable insights to improve resource allocation and service delivery. Moreover, investing in the acute treatment of patients with suspected LVO not only improves clinical outcomes but also reduces overall societal costs in the longer term.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241301904 – Supplemental material for Costs during the first year after stroke by degree of functional disability: A societal perspective
Supplemental material, sj-docx-1-eso-10.1177_23969873241301904 for Costs during the first year after stroke by degree of functional disability: A societal perspective by Mercè Soler-Font, Aida Ribera, Ignacio Aznar-Lou, Alba Sánchez-Viñas, John Slof, Emili Vela, Mercè Salvat-Plana, Lorena Villa-García, Antoni Serrano-Blanco, Natàlia Pérez de la Osa, Marc Ribó and Sònia Abilleira in European Stroke Journal
Footnotes
Acknowledgements
We acknowledge Laura del Carpio, the Centre de Vida Independent (Sandra Millet, Lluisa Pla), EMS (Francesc Xavier Jiménez), University Hospital Vall d’Hebron (Carme Díaz, Mar Vila), Arnau de Vilanova University Hospital (M Antònia Torres, Francisco Mercader) and all the investigators and participants of the RACECAT trial.
Abbreviations
Modified Rankin scale (mRS), Large Vessel Occlusion (LVO), Transient Ischaemic Attack (TIA), Emergency Medical Services (EMS), Generalised Linear Model (GLM).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fundació Marató de TV3 (ref.19/U/2017). IAL had a Miguel Servet research contract (CP22/00029).
Ethical approval
Ethical approval for this study was obtained from the Ethical Committee for Clinical Research with Medications of the Vall d’Hebron University Hospital (PR(AG)229/2017).
Informed consent
Written informed consent was obtained from all patients or surrogates before the study.
Guarantor
AR
Contributorship
MSF wrote the first draft of the manuscript. AR, MSP, LVG, ASB, NP, MR and SA were involved in the study conception. AR obtained fuding. MSF, ARS, IAL, ASV, JS, EV were involved in the data analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request.
Trial registration
RACECAT trial was registered at ClinicalTrials.gob: NCT02795962.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
