Abstract
Introduction:
Blood-brain barrier disruption in acute ischemic stroke is a well described phenomenon associated with hemorrhagic complications. The hyperintense acute reperfusion marker (HARM) represents gadolinium leakage in the cerebrospinal fluid. Gadolinium leakage into ocular structures (GLOS) might represent a dysfunction of the blood-ocular barrier. We aimed to investigate the presence of GLOS in the WAKE-UP trial and its association with HARM, hemorrhagic transformation (HT) and functional outcome.
Patients and methods:
Randomized patients from the WAKE-UP trial who underwent dynamic susceptibility contrast perfusion weighted imaging at baseline and fluid attenuated inversion recovery (FLAIR) imaging at both baseline and follow-up, were included. We studied the association between GLOS with baseline and outcome variables.
Results:
We were able to analyze the data of 192/503 (38%) randomized WAKE-UP patients. GLOS was detected in 56 (29%) cases, four times more frequently than HARM (7%). GLOS positive patients were older (73 vs 65 years; p < 0.01), had a lower glomerular infiltration rate (GFR 79.5 vs 87.2 mL/min; p = 0.01), higher white matter hyperintensity volume (15.0 vs 9.9 mL; p < 0.01), less often presented with a large vessel occlusion (27% vs 44%; p = 0.02) and HARM was more often observed (9/56 vs 4/136; p = 0.01). In multivariable analysis the presence of GLOS was not associated with HT nor poor functional outcome (modified Rankin scale 3–6).
Discussion and conclusion:
GLOS is a relatively common finding in acute ischemic stroke patients and is associated with older age, worse renal function, increased white matter hyperintensity burden and HARM but not with HT or functional outcome.
Introduction
Blood-brain barrier (BBB) disruption is a frequent finding in patients with an acute ischemic stroke and is associated with hemorrhagic complications. Dynamic contrast enhanced imaging metrics and the hyperintense acute reperfusion marker (HARM), are known imaging biomarkers of this BBB-disruption.1–4 HARM, which is visible on the post-contrast fluid attenuated inversion recovery (FLAIR) imaging, represents gadolinium leakage into the cerebrospinal fluid and is found to be a predictor of hemorrhage after reperfusion therapy. 5 Another closely related biomarker that has recently been described, is gadolinium leakage into ocular structures (GLOS). This peculiar phenomenon was noticed first in patients with central retinal artery occlusion and kidney disease,6,7 and was only later described in the context of an acute ischemic stroke. 8 The mechanism of GLOS is not yet completely understood. Much like HARM where gadolinium leaks into the cerebrospinal fluid (CSF) secondary to BBB-disruption, GLOS is thought to be caused by a disruption in the blood-ocular barrier (BOB).8–11 The visualization of both HARM and GLOS on FLAIR imaging results from a similar relative hyperintensity caused by leakage of gadolinium into respectively the CSF and the fluid inside the vitreous and aqueous ocular chamber. The reported prevalence of GLOS in ischemic stroke patients varies between 46% and 76%.8,9,11 GLOS has also been described in patients without clear brain or ocular abnormalities but with certain vascular risk factors, which rather suggests GLOS to be a marker of a physiological excretion of contrast into the eye or of underlying vascular risk factors.12,13
In this study, the presence of GLOS in acute stroke patients was investigated using data from the randomized Efficacy and Safety of MRI-Based Thrombolysis in Wake-Up Stroke (WAKE-UP) trial. 11 We analyzed the association of GLOS with baseline clinical and radiological characteristics, with HARM and hemorrhagic transformation at 24 h follow-up scan and with functional outcome at 90 days.
Methods
Study population
The data from patients included in the WAKE-UP trial were analyzed. 14 The WAKE-UP trial was an investigator-initiated, multicenter, randomized, double-blind, placebo-controlled clinical trial. In this study, acute stroke patients with unknown time since stroke onset and the presence of an hyperintense lesion on diffusion weighted imaging (DWI) coinciding with an absence of a FLAIR hyperintense lesion (i.e. DWI-FLAIR mismatch), were randomized to either alteplase (IV tPA) or placebo. 14 A standard imaging protocol at baseline, consisting of an MRI with at least DWI, FLAIR, and gradient echo (GE) or susceptibility weighted imaging (SWI), could optionally add perfusion weighted imaging (PWI). Follow-up imaging was done after 22–36 h following randomization. For this analysis we only included randomized patients who received gadolinium contrast at baseline for a dynamic susceptibility contrast PWI and who had baseline and follow-up FLAIR imaging of sufficient quality.
The trial was approved by national or local ethics committees or institutional review boards. Written informed consent according to national and local regulations was given by all patients or their legal representatives.
Image analysis
We visually inspected the FLAIR images at follow-up for the presence of GLOS. GLOS was defined as an increase in signal intensity in the ocular chambers, either the aqueous or vitreous fluid, between baseline and follow-up FLAIR imaging, analogous to previous publications (Figure 1). 8 The rating was done by two different raters who were blinded for clinical and other imaging information. In case of disagreement, consensus reading was performed to define the final rating. The HARM rating was conducted as previously described. 5
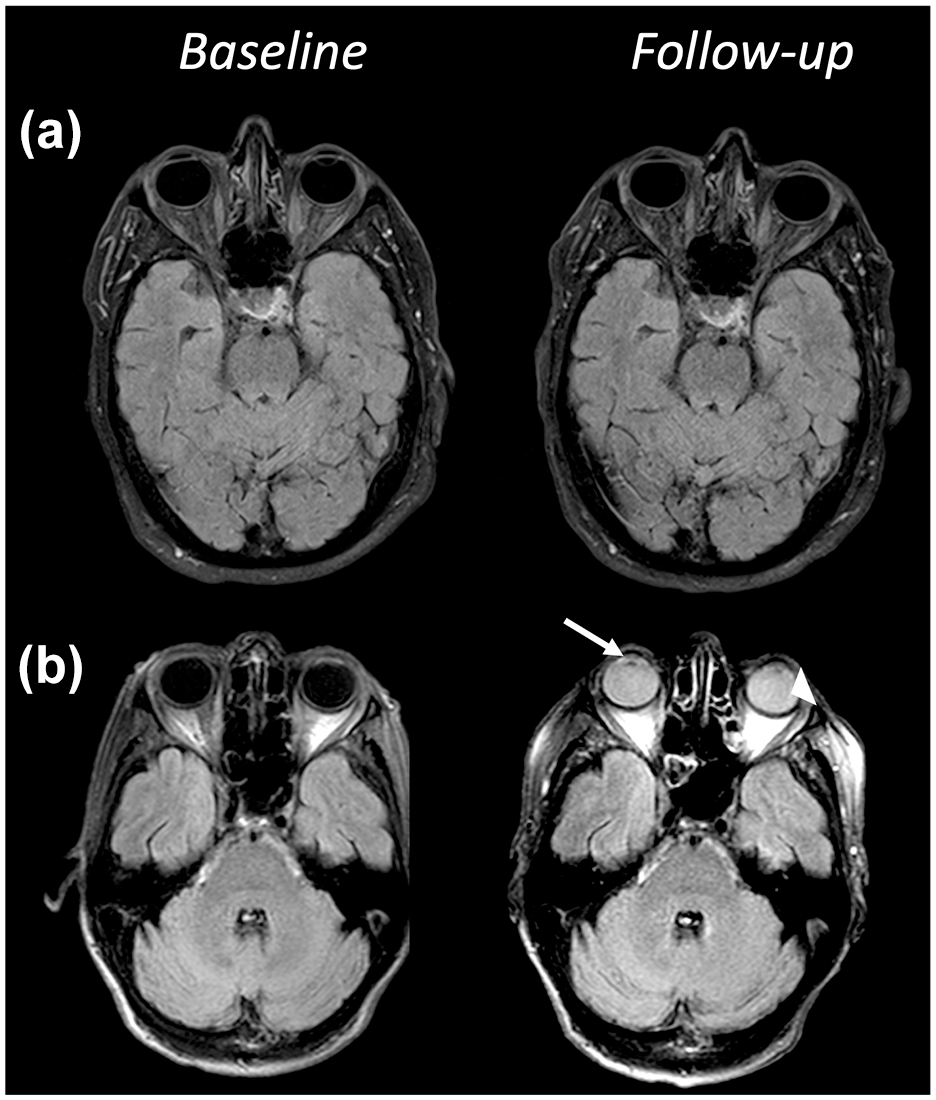
Examples of GLOS rating. This figure depicts the FLAIR images at baseline and follow-up of (a) a patient without GLOS and (b) a patient with GLOS in both the aqueous (arrow) and the vitreous (arrowhead) chamber.
RAPID software was used to analyze baseline DWI and PWI images (IschemaView, Menlo Park, version 4.9 and 5.0). This software calculates the ischemic core lesion based on an apparent diffusion coefficient (ADC) value <620 × 106 mm2/s and the critical hypoperfusion area with a time to the maximum of the residue function (Tmax) with a threshold of >6 s. 15
White matter hyperintensity (WMH) volume was quantified semi-automatically on FLAIR images using FSLs Brain Intensity AbNormality Classification Algorithm (BIANCA) with a training dataset as described previously.16–18 Due to the variable quality of the clinical scans in an acute setting, all resulting masks were reviewed twice by two trained raters and false-positive voxels were excluded manually.
Outcome measures
The Heidelberg criteria for hemorrhagic transformation (HT) of infarcted brain tissue were used to define any hemorrhagic transformation: hemorrhagic infarction type 1 and 2 (Class 1a and 1b), parenchymal hematoma (PH) type 1 (Class 1c) or type 2 (Class 2). 19
We defined functional outcome based on the modified Rankin Scale (mRS) as scored 90 days after symptom onset with poor functional outcome meaning an mRS score of 3–6.
Statistical analysis
Baseline characteristics were compared using the Chi-square test for categorical variables and the Mann–Whitney-U test for continuous variables. The interrater agreement was assessed with Cohen’s kappa analysis. Multivariable models were constructed for poor outcome, any HT and any PH with backward logistic regression based on the Akaike information criterion (AIC). Explanatory variables that were introduced into the model were age, the baseline National Institutes of Health Stroke Scale (NIHSS), GLOS, large vessel occlusion (LVO), DWI-volume, PWI-volume, and treatment with IV tPA. R-software was used for all statistical analyses.
Results
Baseline characteristics
From the 503 patients randomized in the WAKE-UP trial, 213 (42%) patients had PWI at baseline and FLAIR imaging at both baseline and follow-up. Due to low imaging quality and artifacts hampering the interpretation of ocular structures, another 21 patients were excluded. Hence, we analyzed imaging data for a total of 192 (38%) patients (Figure 2).
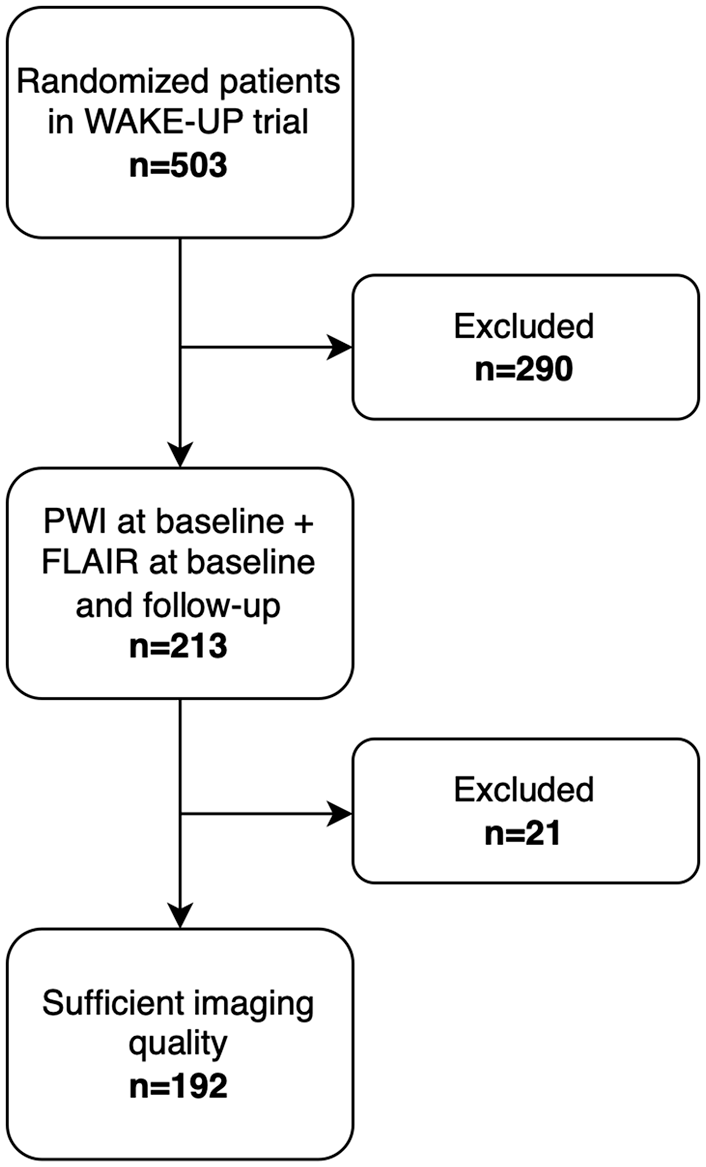
Patient selection flowchart.
The interrater agreement for the presence of GLOS was substantial (kappa = 0.70). After consensus reading GLOS was detected in 56 (29%) of the included patients.
The baseline characteristics in patients with and without GLOS are shown in Table 1. Patients who developed GLOS at follow-up were older, 73 (IQR 65–76) versus 65 (IQR 54–72) year; p < 0.01, had a worse kidney function with a lower glomerular filtration rate (GFR), 79.5 (IQR 60.9–93.3) versus 87.2 (IQR 75.4–103.0) mL/min; p = 0.01 and higher serum creatinine 1.0 (IQR 0.8–1.1) versus 0.9 (IQR 0.8–1.0) mg/dL; p = 0.02, and a higher WMH-volume, 15.0 (IQR 9.6–22.6) versus 9.9 (IQR 5.1–20.2) mL; p = 0.01. LVO was present less often (27% vs 44%; p = 0.04) in patients who developed GLOS.
Baseline characteristics.

Data are n (%) or median (IQR).
aHT: arterial hypertension; DWI: diffusion weighted imaging; GFR: Glomerular filtration rate (calculated from baseline creatinine with the Modification of Diet in Renal Disease Study (MDRD) equation); NIHSS D0: National Institute of Health Stroke Scale at admission; PWI: perfusion weighted imaging; rtPA: recombinant tissue plasminogen activator; WMH: white matter hyperintensity.
Data were missing for 1 patients.
Data were missing for 2 patients.
Data were missing for 13 patients.
Data were missing for 3 patients.
Data were missing for 14 patients.
Characteristics of GLOS and association with HARM
GLOS was more common in the vitreous fluid (n = 53/56, 95%) compared to the aqueous fluid (n = 37/56, 66%; p < 0.01). GLOS involved only the aqueous in 3/56 (5%) patients, only the vitreous in 19/56 (34%) patients and both chambers in 34/56 (61%) patients. GLOS presented strictly unilateral in five patients, in all other cases, GLOS was found bilaterally. In the unilateral cases, GLOS was found contralateral to the infarct-side in 4/5 (80%) patients.
In our population, 13 (7%) patients presented with HARM at follow-up FLAIR imaging. HARM was seen more frequently in patients with GLOS (n = 9, 16%) versus those without GLOS (n = 4, 3%; p < 0.01; Table 2).
HARM and outcome characteristics in patients with versus without GLOS.
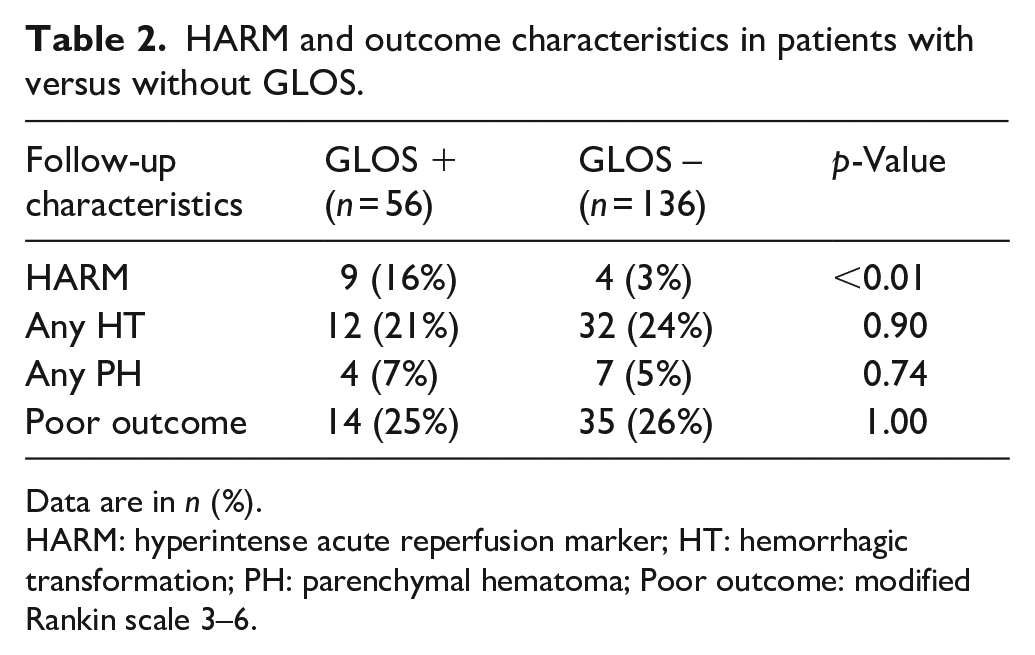
Data are in n (%).
HARM: hyperintense acute reperfusion marker; HT: hemorrhagic transformation; PH: parenchymal hematoma; Poor outcome: modified Rankin scale 3–6.
Association between GLOS and outcome parameters
Any HT was detected at follow-up imaging in 44 out of 192 patients (23%) and any PH in 11 (6%) patients. The occurrence of any HT and PH was similar in patients with GLOS versus in patients without GLOS (21% vs 24%; p = 0.90 and 7% vs 5%; p = 0.74; Table 2). In multivariable analysis, GLOS was not independently associated with HT (aOR 1.29, 95%CI 0.48, 3.45).
We documented poor outcome in 49 (26%) patients at 90 days. The rate of poor functional outcome did not differ between patients with (n = 14, 25%) and without GLOS (n = 35, 26%; p = 0.89; Table 2). In multivariable analysis, GLOS was not associated with poor outcome (aOR 1.93, 95%CI 0.70, 5.46).
Discussion
In this retrospective analysis of patients with a DWI-FLAIR mismatch randomized in the WAKE-UP trial, the GLOS imaging marker was present in 29% of patients with acute ischemic stroke. GLOS was associated with increasing age, worse renal function, increased WMH-volume and HARM. We found no association of GLOS with hemorrhagic transformation, parenchymal hematoma or with functional outcome.
GLOS has only recently been described as a marker in acute stroke patients. 8 The presence of GLOS in the setting of an acute ischemic stroke was similar in our population (29%) when compared to a recently published study, 13 but lower than earlier studies where prevalence ranged from 46% to 76%.9–11 This wide range in the reported prevalence of GLOS could be due to differences in scanners and imaging protocols or to a high interrater variability. Previous GLOS studies typically were small single-center studies with only one scan protocol, whereas the WAKE-UP trial was a large randomized multicenter study. Despite the reported difficulties in the visual assessment of GLOS we found a substantial interrater agreement (kappa = 0.70) making our findings on prevalence rather robust. Differences in imaging timing, patient characteristics and dosage of gadolinium might be other factors contributing to the GLOS prevalence variability between the studies. 12 An independent association between GLOS and renal function, as well as gadolinium dosage, has been described. 13 The prevalence of GLOS on a very early MRI appeared to be slightly lower compared to a 24-h MRI. 8
When present at 24–36 h, GLOS was most often found in both ocular chambers (61%), followed by the vitreous (34%) and aqueous chamber (5%). The predominance of GLOS appearing in the vitreous fluid is in line with previous findings,8,12 but in our population the aqueous fluid was more frequently involved. We could not evaluate the evolution of GLOS over time as we only had one time-point after contrast injection in contrast to a previous study. 8
In our study, GLOS was associated with HARM, increasing age, worse renal function and a higher burden of WMH. The associations with increasing age6,8,10–13 and worse renal function6,12,13 have most consistently been described. The association between GLOS and HARM was previously described in only a small cohort of patients (n = 51) with stroke due to an internal carotid artery stenosis. 11 Here we report this relationship in a much larger cohort of stroke patients with different stroke etiologies. Presumably, this association is a result of the similarities in the vascularization of the brain and the eye. Both organs possess a barrier between the bloodstream and the organ itself, the blood-brain barrier (BBB) and the blood-ocular barrier (BOB) respectively. It is likely that both barriers are vulnerable to similar conditions and are thus often damaged in a similar manner, allowing gadolinium to leak from the bloodstream. Theoretically, it is probable that there are also distinct pathophysiological differences. HARM could reflect the extent of damage to BBB near the infarcted region, while GLOS might be a marker of the general endothelial integrity not necessarily influenced by an acute ischemic event located in the brain. This is reflected in the fact that HARM is a rather rare finding, more specific to stroke patients and is associated with hemorrhagic complications. 5 GLOS has a much higher prevalence and can be found in patients without brain abnormalities.12,13,20 Furthermore, in our study we confirmed the association between GLOS and older age and a higher burden of WMH, which can reflect an increased blood-retinal barrier permeability due to pre-existing vascular risk factors as a possible pathophysiological mechanism associated with GLOS. 13 The association between small vessel disease and a subtle increase of the BBB permeability has been described previously.21,22 In the future more advanced imaging techniques for BBB and BOB might provide more insight in the exact pathophysiological meaning of GLOS.
One of the major strengths of this study is the large cohort of stroke patients from a multicenter randomized controlled trial. This allowed the assessment of an association of two markers representing barriers between blood vessels and the central nervous system, in contrast to previous studies in stroke patients that were either small9,11 or didn’t include HARM assessment.8,13 Our findings suggest GLOS and HARM to partially identify similar changes associated with age, renal function and gadolinium dosage. However, HARM is less frequently present and is more specific for acute ischemic injury in the brain. Our previously reported association between HARM and HT was not present for GLOS arguing for distinct pathophysiological bases for these two neuroimaging markers. 5 On the other hand, the association between HARM and HT has not been consistently found in other studies.23,24 Another strength was the rating of GLOS in our study followed by a consensus read which is in contrast with earlier publications. As we noticed discrepancies between two readers, differences between previously reported prevalence could at least partially be due to interrater variability.
GLOS has some inherent limitations, such as its inability to be assessed at baseline, which prevents it from influencing acute therapy. Additionally, imaging artifacts can hamper the evaluation of its presence, and only centers that perform MRI in acute stroke therapy can apply GLOS. Our study design also has certain limitations. First, since we only included patients with an acute ischemic stroke, we could not investigate the prevalence of GLOS in non-stroke patients. Second, we cannot rule out potential selection bias as it relates to patients included in the WAKE-up trial who were predominantly patients with less severe strokes reflected in the relatively small infarct volumes. For this subanalysis we could only include the proportion of patients for whom baseline PWI was available introducing an extra form of selection bias. Next, we could not study the time-course of GLOS, since we only had one post-contrast MRI available. Also, there was no standard MRI protocol implemented in this multi-center trial. Although this could have introduced some variability in the GLOS-scoring, it makes the results more generalizable. Next, in our study we included patients in the presumably early time window after stroke onset (i.e. patients with DWI-FLAIR mismatch). It might be possible that GLOS may be more frequent when contrast is injected at later time-points after stroke onset or in patients without DWI-FLAIR mismatch. Furthermore, due to the low incidence of HT, our findings are only exploratory and as no remote PH occurred in this cohort, the association between remote PH and GLOS could not be assessed. Finally, we didn’t have information about ocular diseases available in the patients’ medical histories and GLOS has been described in patients with specific ocular conditions.20,25
To conclude, GLOS was relatively common in acute ischemic stroke patients with a DWI-FLAIR mismatch and was associated with increasing age, worse renal function, higher WMH-volume and the presence of HARM. GLOS was four times more frequent in comparison to HARM. We found no association of GLOS with hemorrhagic transformation or functional outcome.
Footnotes
Acknowledgements
None.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JBF reports consulting and advisory board fees from AbbVie, AC Immune, Alzheon, Artemida, BioClinica/Clario, Biogen, Bristol Myers Squibb, Brainomix, Cerevast, C2N Diagnostics, Daiichi-Sankyo, EISAI, Eli Lilly, F. Hoffmann-LaRoche AG, GlaxoSmithKline, Guerbet, Ionis Pharmaceuticals, IQVIA, Janssen, Julius Clinical, jung diagnostics, Lantheus Medical Imaging, Merck, Novo Nordisk, Octapharma AG, Premier Research, ProPharma Group, Prothena Biosciences, Regeneron Pharmaceuticals, Roche, Syneos, Tau Rx, Vertex Pharmaceuticals, and Worldwide Clinical Trials outside the submitted work.
GT reports personal consulting fees from Acandis, personal consulting fees from Astra Zeneca, personal consulting fees from Bayer, personal consulting fees from Boehringer Ingelheim, personal consulting fees from Stryker, personal payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from: Acandis, Alexion, Amarin, Bayer, Boehringer Ingelheim, BristolMyersSquibb/Pfizer, Daiichi Sankyo, Stryker, outside the submitted work.
CZS is supported by a research grant from Health Research Foundation of Central Denmark Region. Also reports speaker fee from Pfizer.
VT reports consulting fees from Bayer, Boehringer Ingelheim, Medtronic.
CG declares, independent of the presented study, grants from Deutsche Forschungsgemeinschaft (DFG), Deutsches Zentrum f. Luft- und Raumfahrt (DLR), Hertie Foundation, Wegener Foundation, Schilling Foundation, Werner Otto Foundation, Merz Pharmaceuticals, Allergan, European Union; CG declares consulting fees from AlphaSights Ltd. and Life Science Praxis S.L., honoraria (for lectures, presentations) from AstraZeneca GmbH, Elements Communications Ltd., Boehringer Ingelheim, Streamedup GmbH, Abbott Medical, Bayer AG; CG declares participation in the DSMB of RESSTORE1, work as an editor of INFO Neurologie & Psychiatrie, Therapie und Verlauf neurologischer Erkrankungen (Textbook), and membership of the presidium of the German Neurological Society (DGN).
KWM reports consultancy fees from Boehringer Ingelheim, Abbvie, Lumosa, Hyperfine; Lecture Fees – Boehringer Ingelheim, Brainomix, IschemaView; Trial Support – Boehringer Ingelheim (drug supply for ATTEST-2).
ME reports grants from Bayer and fees paid to the Charité from Amgen, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, BMS, Daiichi Sankyo, Sanofi, Pfizer, all outside the submitted work.
RL reports compensation from iSchemaView for other services.
All other authors report nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: WAKE-UP received funding from the European Union Seventh Framework Programme [FP7/2007-2013] under grant agreement n°278276 (WAKE-UP).
Dr Lemmens is senior investigator (1841923N) at the Flanders Research Fund (fonds Wetenschappelijk Onderzoek, FWO) funded project G049620N.
Ethical approval
The trial was approved by national or local ethics committees or institutional review boards.
Informed consent
Written informed consent was obtained from all patients or their relatives before inclusion in the WAKE-UP trial.
Guarantor
AW
Contributorship
AW, LS, TV, and BS researched literature and conceived the study. TV wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
