Abstract
Introduction:
The overestimation of ischemic core volume by CT perfusion (CTP) is a critical concern in the selection of candidates for reperfusion therapy. This phenomenon is termed a ghost infarct core (GIC). Core growth rate (CGR) is an indicator of ischemic severity. We aimed to elucidate the association between GIC and CGR.
Patients and methods:
Consecutive patients with acute ischemic stroke who underwent mechanical thrombectomy in our institute from March 2017 to July 2022 were enrolled. The initial ischemic core volume (IICV) was measured by pretreatment CTP, and the final infarct volume (FIV) was measured by diffusion-weighted imaging. A GIC was defined by IICV minus FIV > 10 ml. The CGR was calculated by dividing the IICV by the time from onset to CTP. Univariable analysis and a multivariable logistic regression model were used to evaluate the association between GIC-positive and CGR.
Results:
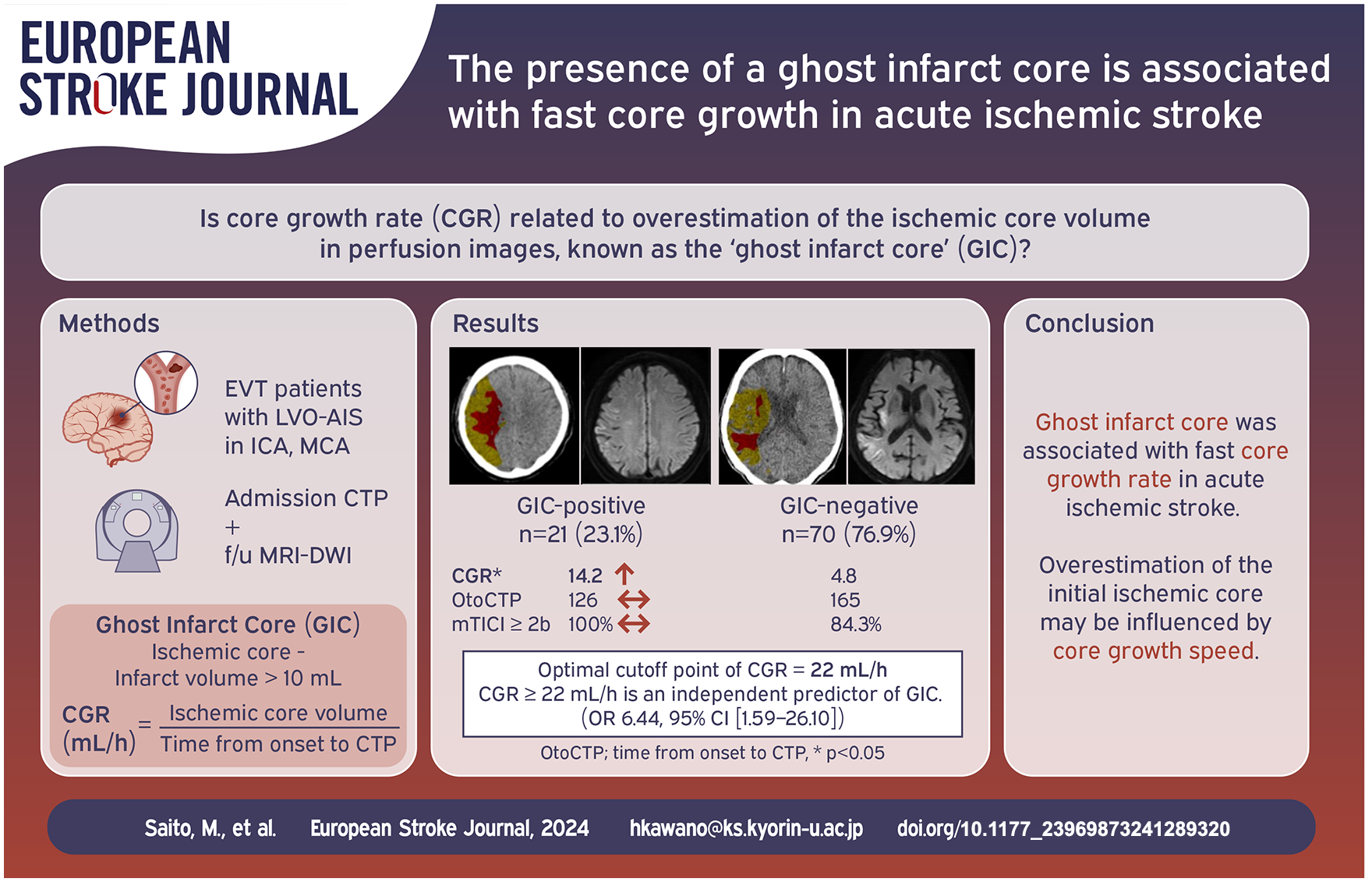
Of all 91 patients, 21 (23.1%) were GIC-positive. The GIC-positive group had higher CGR (14.2 [2.6–46.7] vs 4.8 [1.6–17.1] ml/h, p = 0.02) and complete recanalization (n = 15 (71.4%) vs 29 (41.4%), p = 0.02) compared to the GIC-negative group. On receiver-operating characteristic curve analysis, the optimal cutoff point of CGR to predict GIC-positive was 22 ml/h (sensitivity, 0.48; specificity, 0.85; AUC, 0.67). Multivariable logistic regression analysis showed that CGR ⩾ 22 ml/h (OR 6.44, 95% CI [1.59–26.10], p = 0.01) and complete recanalization (OR 3.72, 95% CI [1.14–12.08], p = 0.02) were independent predictors of GIC-positive.
Conclusions:
A GIC was associated with fast CGR in acute ischemic stroke. Overestimation of the initial ischemic core may be determined by core growth speed.
Introduction
Randomized clinical trials have shown the efficacy and safety of mechanical thrombectomy (MT) in patients with acute ischemic stroke (AIS) with large vessel occlusion (LVO).1–7 Ideal candidates for MT show a small ischemic core and a large volume of salvageable ischemic tissue (penumbral tissue). Computed tomography perfusion (CTP) with automated post-processing software has been implemented to identify the ischemic core and penumbra in AIS patients. Previous research has demonstrated a good correlation between the CTP-derived ischemic core and the final infarct volume (FIV) calculated by follow-up magnetic resonance imaging (MRI).8–10 CTP is superior in terms of its great precision in identifying and outlining the infarct core when compared to non-contrast CT (NCCT).11–13 In addition, CTP has greater accessibility, shorter imaging durations, and lower cost than MRI.14–16 Recently, overestimation of the initial ischemic core compared to FIV has been recognized.17–22 This phenomenon is termed the “ghost infarct core (GIC),” which implies that the estimated ischemic core may include salvageable tissue (Supplemental Material: Figure S1).17,18,21 Overestimation of the ischemic core is a critical concern, because patients who can benefit from MT may be inadequately excluded from reperfusion therapy. Several studies have suggested that the GIC represents a time-dependent phenomenon, in which a shorter time from stroke onset to imaging acquisition is associated with the GIC.17,18,21
It has been suggested that ischemic core overestimation may occur in patients with poor ischemic tolerance and fast core progression in the early stages of stroke. The concept of the core growth rate (CGR), calculated by dividing the time from stroke onset to image acquisition, has been reported as an indicator of ischemic severity.23–26 A high CGR indicates fast core progression and is associated with poor outcomes.23,25,26 However, the correlation between the CGR and the GIC has been unclear.
Aims and hypothesis
We hypothesized that a high CGR may be associated with the development of a GIC. The aim of the present study was to elucidate the association between the GIC and the CGR in AIS patients treated with MT.
Methods
Study design and patients
Consecutive patients with stroke due to internal carotid artery (ICA) and/or middle cerebral artery (MCA) occlusion who underwent multiparametric CT (NCCT, CTP, CT angiography (CTA)) at baseline and subsequent MT at the Stroke Center, Kyorin University Hospital (Tokyo, Japan) between March 2017 and July 2022, were retrospectively included in this study. This study protocol was reviewed and approved by the Faculty of Medicine of Ethics Committee, Kyorin University (R01-160-08).
All patients underwent CTA prior to MT to assess vessel occlusion. The Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 27 was calculated retrospectively. MT was performed by certified neurointerventionalists using commercially available stent retrievers and/or aspiration catheters. Intravenous recombinant tissue plasminogen activator (rt-PA) was also administered in accordance with Japanese guidelines. 28 The recanalization rate was assessed using the modified Thrombolysis In Cerebral Infarct (mTICI) grade 29 ; successful recanalization was defined as mTICI 2b-3, and complete recanalization was defined as mTICI 3. The duration of each component of workflow, including onset to CTP time, onset to recanalization time, and CTP to recanalization time, was documented. Time of stroke onset was defined as the last time patient was seen well. Final infarct volume (FIV) was manually assessed based on diffusion-weighted imaging at 24–72 h following MT. GIC was defined as the initial ischemic core volume (IICV) on baseline CTP minus FIV > 10 ml. 17 Symptomatic intracerebral hemorrhage (sICH) was defined as parenchymal hematoma type 2 within 36 h after treatment, combined with an increase from baseline in the National Institutes of Health Stroke Scale (NIHSS) score of ⩾4 points. 30 Early neurological improvement (ENI) was defined as a decrease of 6 or more on the NIHSS score in comparison to the baseline score at 24–48 h following MT. A good clinical outcome was defined as a modified Rankin Scale (mRS) score ranging from 0 to 2 at 90 days or clinical recovery to the status preceding the onset of stroke (i.e. the premorbid mRS score equaled the mRS score at 90 days).
Imaging Protocol
CTP was performed with 320-row CT scanners (Aquilion ONE ViSION-Edition, Canon Medical Systems, Tochigi, Japan). CTP data were processed using FDA- and CE- approved workstation (Vitrea version 7.8; Canon Medical Systems). Cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), time to peak, delay time, and time to maximum on residual function (Tmax) were retrospectively calculated using Bayesian methods. The detailed imaging acquisition and CTP analyses summarized in Supplemental File.
The regions affected by stroke were delineated using perfusion volumes. IICV was calculated as the volume with relative CBV (rCBV) < 40% and Tmax > 2.2 s, in comparison to the contralateral hemisphere. 31 Ischemic penumbra volume was defined as the volume with rCBV ⩾ 40% and Tmax > 2.2 s, compared to the contralateral side. 31 Perfusion lesion was defined as the volume with Tmax > 2.2 s, compared to the contralateral side. Follow-up imaging was conducted using MRI at 24–72 h after MT, performed on 1.5-T (Vantage Titan; Canon Medical Systems) or 3.0-T (Vantage Centurian 3 T; Canon Medical Systems) machines, including DWI, FLAIR, T2*WI, and MR angiography. The visually apparent DWI lesion on follow-up MRI was manually identified and delineated by consensus among two expert neurologists (MS, HK), who were blinded to the clinical data. The infarct lesion was outlined using the DWI b1000 image, with careful cross-referencing of the ADC image, utilizing SYNAPSE Enterprise-PACS (Fujifilm Medical Systems, Tokyo, Japan). The DWI b0 image was also examined for artifacts to ensure that the high signal intensity in the DWI b1000 image was not significantly influenced by T2 shine-through. Infarct volume was calculated by multiplying the cumulative DWI lesion area on each slice by the corresponding slice thickness.
Core growth rate (CGR)
The CGR was calculated by dividing IICV by the time from stroke onset to CTP acquisition.23,25,26 The equation for the core growth rate is as follows: CGR (ml/h) = IICV on CTP/time from stroke onset to CTP acquisition. Essentially, this calculation assumes that core volume is zero immediately before symptom onset, and that it grows linearly within 24 h of stroke onset.32,33
Statistical analysis
Categorical variables are presented as absolute values and percentages, and continuous variables are presented as medians with interquartile range (IQR) for non-normally distributed data. The significance of intergroup differences was assessed by Pearson’s χ 2 test for categorical variables and the Wilcoxon rank-sum test for continuous variables. Correlation analysis was performed using Spearman’s rank correlation coefficient to compare the ischemic core on baseline CTP and FIV on follow-up DWI. Volumetric agreement between the ischemic core and FIV was summarized using Bland-Altman plots. To predict GIC, receiver-operating characteristic (ROC) curve analysis was used to determine the optimal cut-off point for the CGR. In addition, multivariable logistic regression models were used to evaluate the association between the CGR and GIC. Complete recanalization, onset to imaging time, and imaging to recanalization time were included as variables in the multivariable logistic regression models, because they have been reported to be associated with GIC and core overestimation.17,22,34 Significance was defined at p < 0.05. The statistical analysis was performed using JMP 17.0.0 (SAS Institute Inc., Cary, NC, USA).
Results
Of the 148 patients who underwent MT, 41 were excluded due to insufficient imaging data (29 lacked baseline CTP, 12 lacked follow-up MRI), and 17 patients with posterior circulation occlusion were also excluded. Therefore, 91 patients were analyzed (Supplemental Material: Figure S2). The median baseline NIHSS score was 21 (IQR, 14–25), and the median time from stroke onset to CTP was 157 (87–514) min. The rate of successful recanalization was 87.9%. The median time from CTP to recanalization was 88 (63–116) min. Forty-three patients (47.2%) achieved good clinical outcomes at 90 days. The median IICV and FIV were 20.1 and 25.9 ml, respectively. A moderate correlation between IICV and FIV was observed (r = 0.69, p < 0.01) (Figure 1(a)). The mean volumetric difference between the IICV and FIV was −14.1 ml (LoA, −106.1 to 77.9 ml) (Figure 1(b)). The Bland-Altman plot showed negative bias, indicating that FIV was generally larger than the ischemic core on baseline CTP. The plot also showed a tendency toward greater bias with increasing average lesion volume.

Correlation between ischemic core volume on baseline computed tomography perfusion (CTP) and final infarct volume: (a) scatter-plot of the correlation between ischemic core volume on baseline CTP and final infarct volume shows a moderate correlation (r = 0.69, p < 0.01) and (b) Bland-Altman plot demonstrating agreement between ischemic core volume on baseline CTP and final infarct volume on follow-up DWI. The figure shows the negative bias indicating that final infarct volume is generally larger than ischemic core volume on baseline CTP.
Twenty-one patients (23.1%) showed a GIC, and the median GIC volume (FIV – IICV) was 19.8 ml. A GIC was associated with larger IICV (27.9 ml [18.6–69.6] vs 17.3 ml [9.3–38.6], p = 0.01) and higher CGR (14.2 ml/h [2.6–46.7] vs 4.8 ml/h [1.6–17.1], p = 0.02) (Table 1). All patients in the GIC-positive group had successful recanalization, and the GIC-positive group showed a higher rate of complete recanalization (n = 15 [71.4%] vs 29 [41.4%], p = 0.02) than the GIC-negative group. There was no significant difference between the two groups in time from stroke onset to CTP (126 min [58–499] vs 165 min [101–525], p = 0.16), whereas time from CTP to recanalization tended to be shorter in the GIC-positive group than in the GIC-negative group (69 min [51–108] vs 92 [63–122] min, p = 0.07) (Table 1). GIC-positive patients tended to have a higher proportion of ENI than the GIC-negative group (18 [85.7%] vs 46 [65.7%], p = 0.08), although there were no differences in good clinical outcomes and mortality at 90 days.
Baseline characteristics and outcome measures for ghost infarct core (GIC)-positive and GIC-negative patients.

ASPECTS: Alberta Stroke Program Early CT Score; CGR: core growth rate; CTP: computed tomography perfusion; FIV: final infarct volume; GIC: ghost infarct core; ICA: internal carotid artery; IICV: initial ischemic core volume; IQR: interquartile rage; IV: intravenous; mRS: modified Rankin Scale; mTICI: modified Thrombolysis In Cerebral Ischemia; M1: M1 portion of middle cerebral artery; M2: M2 portion of middle cerebral artery; and NIHSS: National Institutes of Health Stroke Scale.
ROC curve analysis showed that the optimal cutoff point of the CGR to predict GIC was 22 ml/h (sensitivity, 0.48; specificity, 0.85; area under the curve, 0.67). Multivariable logistic regression analysis showed that CGR ⩾ 22 ml/h (OR 6.44, 95% CI [1.59–26.10], p = 0.01) and complete recanalization (OR 3.72, 95% CI [1.14–12.08], p = 0.02) were independent predictors of GIC (Table 2). Similar results were obtained in the analysis, including only those patients with successful recanalization (Supplemental Material: Tables S1 and S2). A GIC occurred in approximately half of the patients with CGR ⩾ 22 ml/h, whereas a GIC occurred in only 16% of the patients with CGR < 22 ml/h (Figure 2). Furthermore, in patients with CGR ⩾ 22 ml/h, the GIC-positive group showed significantly higher rates of time from stroke onset to CTP < 60 min and of complete reperfusion than the GIC-negative group (Supplemental Material: Figure S3).
Multivariable logistic regression analysis for independently predicting ghost infarct cores.

CI: confidence interval; CTP: computed tomography perfusion; CGR: core growth rate; and mTICI: modified Thrombolysis In Cerebral Ischemia.
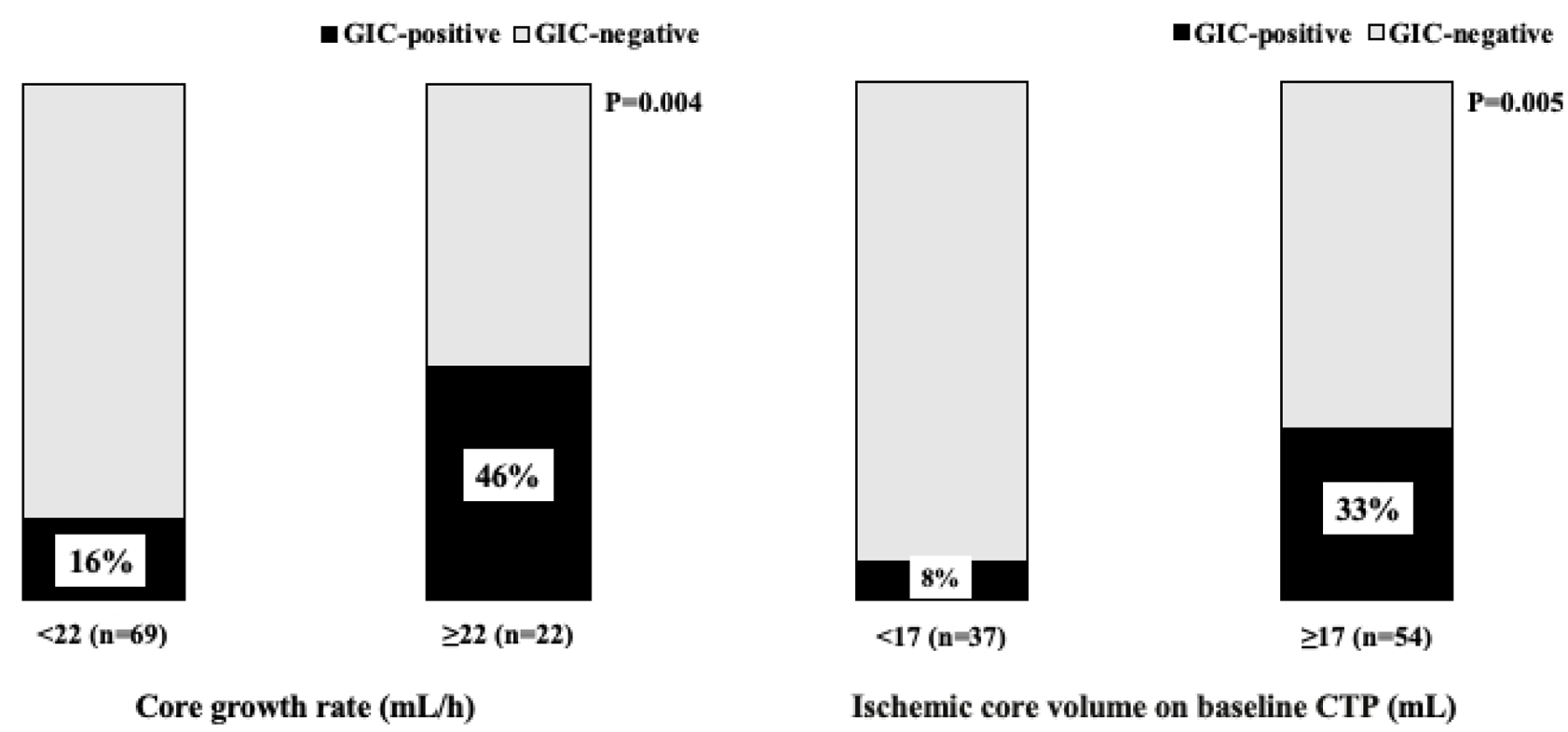
Bar graphs showing the proportion of patients with a ghost infarct core: Bar graph showing the proportion of patients with a ghost infarct core by core growth rate (a) and ischemic core volume on CTP (b).
Discussion
The present study had the following two major findings. First, a higher core growth rate was a significant predictor of GIC. Second, in patients with CGR less than 20mL, because of a high specificity, CTP is less likely to be overestimated. These findings suggest that an ischemic core can be salvaged by complete recanalization even in a patient with fast core progression.
The incidence of a GIC in the present study was consistent with previous studies, which showed that a GIC occurred in between 6% and 38% of anterior circulation LVO cases treated with MT.17,18,21,22 The core overestimation of CTP may be associated with discrepancies between the ischemic core identified on perfusion imaging and the infarct lesion delineated by MRI. CTP assesses the hemodynamic status of the brain at the time of acquisition and not tissue viability. CTP estimates the ischemic core based on the severity of CBF or CBV reduction. However, neuronal tissue death occurs when reductions in CBF or CBV persist continuously and severely enough to induce irreversible damage. Therefore, if recanalization of the occluded vessel shortens the duration of ischemia, irreversible injury may not occur, unless the ischemic severity is profound 35 Because of the recent development of rapid and effective MT, it may be necessary to re-define the CTP thresholds used to predict the true ischemic core, taking into account the success or failure of recanalization and timing of recanalization. Bivard et al. demonstrated that ischemic thresholds for predicting FIV were different for patients treated with MT versus rt-PA alone, and that stricter thresholds were more suitable for predicting FIV in patients who achieved complete recanalization with MT than with intravenous thrombolysis alone. 34 Even in patients with rapid core progression based on the traditional ischemic thresholds, early recanalization can reduce FIV compared to IICV based on CTP.
The present results show a significant association between the CGR and the presence of a GIC in AIS patients. The CGR is considered an indicator of ischemic severity.24,26 The extent of core growth varies among AIS patients; in short, a higher CGR leads to infarct expansion within a short period, whereas a lower CGR does not lead to infarct expansion despite longer time intervals from stroke onset to imaging acquisition. Patients with a high CGR can be presumed to have rapid conversion from reversible to irreversible tissue without early reperfusion. The present study showed that, especially in patients with a CGR ⩾ 22 ml/h, the GIC-positive group showed higher rates of time from onset to CTP < 60 min and of complete recanalization than the GIC-negative group (Supplemental Material: Figure S2). We believe that, even if a patient has fast core progression, early complete recanalization can partially salvage the pretreatment ischemic core based on CTP.
There were no differences in the interval between stroke onset and CTP acquisition, although previous studies reported that a GIC was associated with a shorter interval between stroke onset and imaging.17,18,21 The present study suggests that core growth speed rather than chronological time determines GIC development.
The present study also showed the association of the GIC-positive group with ENI. However, there was no significant difference in good clinical outcomes with or without a GIC, which was consistent with previous studies.18,19 In the present study, the high recanalization rate in the GIC-negative group (84.3%) may have led to the relatively high rate of good clinical outcomes, resulting in no significant difference. Another consideration is that the GIC regions may have sustained histological damage. In the present study, the FIV of the GIC-positive group was notably diminished compared to that of the GIC-negative group. On the other hand, the IICV in the GIC-positive group is comparable to the FIV in the GIC-negative group. The GIC regions were identified as severely hypoperfused on perfusion imaging but did not exhibit diffusion restriction on DWI. In preclinical models, regions demonstrating recovery from diffusion abnormalities on DWI have been reported to exhibit neuronal damage upon histopathological examination. 36 This dissociation highlights the fact that normalization of imaging findings does not necessarily correspond with tissue recovery. Thus, the GIC regions may still sustain significant tissue damage due to severe hypoperfusion. On the other hand, an association between the presence of a GIC and good functional outcomes has been reported. 17 This may be because of the accuracy of FIV based on NCCT. 17
The present study has several limitations. First, the relatively small sample size and the low incidence of GIC may make our results to be generalized. Therefore, these results require validation in studies with larger cohorts to draw more definitive conclusions. Furthermore, while our findings align with previous research suggesting a higher prevalence of GIC in cases with poor collateral, it is noteworthy that most of the patients with CGR less than 22 ml/h did not have GIC.21,22 This observation implies that multiple factors, including the rate of recanalization, the time to CTP imaging, and CGR, may play a role in the occurrence of GIC. Second, because the findings of the present study were obtained using a single software package (Vitrea), the results may not be generalized to other software. On the other hand, previous research has shown that Vitrea is comparable to RAPID, with similar mean differences and correlations between the ischemic core and FIV in patients who underwent MT.31,37 Moreover, our analysis revealed that the Bayesian-based Vitrea correlated better with FIV than the SVD method in large ischemic lesions, showing less influence from time to imaging and pre-treatment core volume. 38 These findings support the validity of Vitrea’s ischemic core assessment. Additionally, prior studies reported GICs and core overestimation with other CTP auto-processing software, such as RAPID and MIStar.18,19,21,34 In studies of GIC using RAPID, the incidence of GIC has been reported to range from 6.0% to 20.0%, which is similar to the frequencies observed in our research.18,20–22 Therefore, this phenomenon may be observed regardless of software. Third, the formula for the CGR was predicated on the assumption that the core grows linearly within 24 h following stroke onset.32,33 Finally, the present study included 52% of patients who lacked precise awareness of the time of stroke onset. For these patients, the CGR was calculated based on the last time known well, which could potentially underestimate the actual rate of core growth.
Conclusion
A GIC was associated with a higher CGR in patients with acute ischemic stroke. Overestimation of the initial ischemic core may be determined by core growth speed and complete recanalization, not clock time.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241289320 – Supplemental material for The presence of a ghost infarct core is associated with fast core growth in acute ischemic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873241289320 for The presence of a ghost infarct core is associated with fast core growth in acute ischemic stroke by Mikito Saito, Hiroyuki Kawano, Takuya Adachi, Miho Gomyo, Kenichi Yokoyama, Yoshiaki Shiokawa and Teruyuki Hirano in European Stroke Journal
Footnotes
Acknowledgements
Canon Medical Systems Corporation (Tochigi, Japan) provided Vitrea version 7.8 free of charge for this study. Canon Medical Systems Corporation had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from JSPS KAKENHI (Grant Number 21K07468).
Ethical approval
This study protocol was reviewed and approved by the Faculty of Medicine of Ethics Committee, Kyorin University (R01-160-08).
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Guarantor
The guarantor of our paper is Dr. Hiroyuki Kawano.
Contributorship
M.S. contributed to the conception or design of the work, analysis and interpretation of data, and writing the draft of the manuscript. H.K. contributed to the conception or design of the work and interpretation of data, wrote the draft of the manuscript, and acquired the funding. T.A. contributed to the collection and interpretation of the data. M.G., K.Y., Y.S., and T.H. contributed to the interpretation of the data. All authors critically reviewed the manuscript and approved the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
