Abstract
Background and purpose:
Mechanical thrombectomy (MT) has revolutionized the prognosis of acute ischemic stroke. Indications for MT are constantly expanding across countries, presenting healthcare professionals, and institutions with the challenge of offering MT to the entire population despite geographic constraints, while also training enough interventional neuroradiologists (INR) for this highly technical-level procedure. We aimed to provide an overview of current European MT practices and organizations in 2021.
Materials and methods:
Members of the European Society of Minimally Invasive Neurological Therapy (ESMINT) were invited to complete two different online surveys from March to November 2021 to collect data on MT practice.
Results:
A total of 240 individual responses from INR (from 33 European countries) were received. These included information from 56 thrombectomy-capable stroke centers (TCSC) data (across 26 European countries). The mean number of INR per center was 3.8 ± 1.43, median 4 (IQR, 3–4.5). Half of the centers (28/56, 50.00%) performed less than 150 MT per year. Most INR used a balloon guide catheter in less than 50% of cases (160/240, 66.67%), and limited the number of recanalization attempts to six passes to restore flow (209/240, 87.08%). Additionally, 37.92% of the respondents (91/240, 37.92%) indicated that they already performed MT for distal occlusions (M3, M4) as part of their routine practice. Other details of the MT procedure, anesthetic management, and patient selection are also presented and discussed.
Conclusions:
This European survey emphasizes the differences between TCSC and INR in modern thrombectomy practices. Even if most centers remain understaffed to meet current and future MT needs, most European TCSCs are actively training young INR.

Introduction
Mechanical thrombectomy (MT) has revolutionized the prognosis of acute ischemic stroke1,2 over the past decade, and indications for MT are constantly expanding across countries, presenting healthcare professionals, and healthcare institutions with the challenge of offering MT to their entire population despite demographic and geographic limitations. Training a sufficient number of interventional neuroradiologists (INR) for this high technical level procedure is also mandatory to handle the increasing number of MT procedures, particularly with the expansion of indications for distal thrombectomy 3 and the aging of the population. 4 Furthermore, INR professionals are becoming more and more involved and busy with new treatments/procedures.5–7 To reduce treatment times for these patients, other constraints must also be considered in all Thrombectomy-Capable Stroke Centers (TCSCs), such as individual practitioners’ approaches to patient selection, revascularization strategies, as well as periprocedural and anesthesia management. Regional stroke networks are another cornerstone of stroke care, which facilitate the opening of stroke centers to improve patient care. 4
To date, there has been no other such survey on MT practices in Europe. Therefore, it seems essential to gather data in order to compare patient care with current guidelines, and to assess inter-country/inter-center heterogeneity. Such data could advise authorities on the current needs and offer new insight into stroke management in Europe. Our aim was to provide an overview of current European MT practices and organizations in 2021.
Materials and methods
An online survey of 84 questions was designed using the SurveyMonkey website and was sent several times during March to November 2021 to members of the European Society of Minimally Invasive Neurological Therapy (ESMINT), inviting 1 INR per TCSC to respond to the survey. Furthermore, the content of the email was posted on ESMINT social media for ESMINT members. The survey was written in English and divided into two independent questionnaires:
- a questionnaire to collect responses about practitioner approaches available from March 2021 to July 2021;
- a second questionnaire to collect general center-based information available from August 2021 to November 2021;
Responses were collected anonymously and reminder emails were sent several times to all members prior to the survey deadline. Institutional review board approval was not required for this survey. No compensation was offered to the participants.
The survey was based on a previous report by Forestier et al., 8 including new questions to adjust to advances in stroke treatment and the increased number of centers. Most of the questions were multiple choice questions. SurveyMonkey records the IP address; therefore, respondents are limited to a single response. Standard descriptive statistics were performed.
Results
Between March 2021 and November 2021, 56 TCSC answered the questionnaire (representing 26 European countries), and 240 individual INR answered (representing 33 European countries). See Figure 1. All responses were complete for the individual questionnaire and some responses were incomplete for the TSCS questionnaire.
Representative maps of responses.
Teams, center, and thrombectomy volume
The average INR age was 44.2 years old ±9.47 (min 27, max 71, median 43 (IQR, 37–51)). The mean number of INR per center was 3.8 ± 1.43, median 4 (IQR, 3–4.5). Regarding the number of INR on the on-call team for MT, the mean number was 3.4 ± 1.52, median 3.5 (IQR, 2–4). 75.88% of INR (senior) had a radiological background (173/228, 75.88%), 14.91% had a neurological background (34/228, 14.91%), and 9.21% had a neurosurgical background (21/228, 9.21%). 54.85% of the INR were involved in the diagnostic of neuroradiology and 26.78% of them were also involved in other fields of diagnostic radiology.
37.50% of the centers (21/56, 37.50%) were concerned about at least one member approaching retirement (within 5 years). To ensure current and future needs for MT, 4.16 ± 1.92 INR per center were required and 96.43% of the centers (54/56, 96.43%) needed at least one more INR. Furthermore, 78.57% of the centers (44/56, 78.57%) trained at least one INR with an average number of 1.61 ± 1.32 INR in training per center.
During the day, procedures were performed routinely in duo with another senior/junior INR for 67.86% of the TCSC (38/56, 67.86%), and there was significant variability between the TCSC in terms of the composition of the team with the nurse(s) and the X-ray technician(s). See supplemental Figure 1.
MT was available around the clock for all centers except one (during office hours). Half of the centers (28/56, 50.00%) performed less than 150 MT per year and most of the TCSC (52/56, 92.86%) did not perform MT in patients under 5 years of age.
Forty-four TCSCs (44/56, 78.57%) worked with at least three Primary Stroke Centers, and the mean number of Primary Stroke Centers that refer to a TCSC was 5.34 ± 3.47.
TCSC had a mean number of 1.18 ± 0.73 angio-suites dedicated exclusively to neurointerventional procedures and shared 1.22 ± 0.96 angio-suites with other specialties during work hours.
Data related to the COVID-19 outbreak are expanded in the Supplementary Data.
Patients’ selection and imaging follow-up
For anterior or posterior strokes, CT was the most widely used imaging modality during the early time window (<6 h from symptom onset), while for the late time window (>6 h from symptom onset), most centers used MRI or CT or MRI (Figure 2). A quarter of TCSCs (14/56, 25.00%) have modified their workflow by establishing a direct to angio workflow considering specific inclusion criteria (e.g. NIHSS > 10). Workflow strategies for MT were different between TCSC: bridging therapy (MT directly after intravenous thrombolysis) and MT as first-line treatment in case of thrombolysis contraindication were the most common approaches (53/56, 94.64%), compared with MT alone as first-line treatment (3/56, 5.36%).
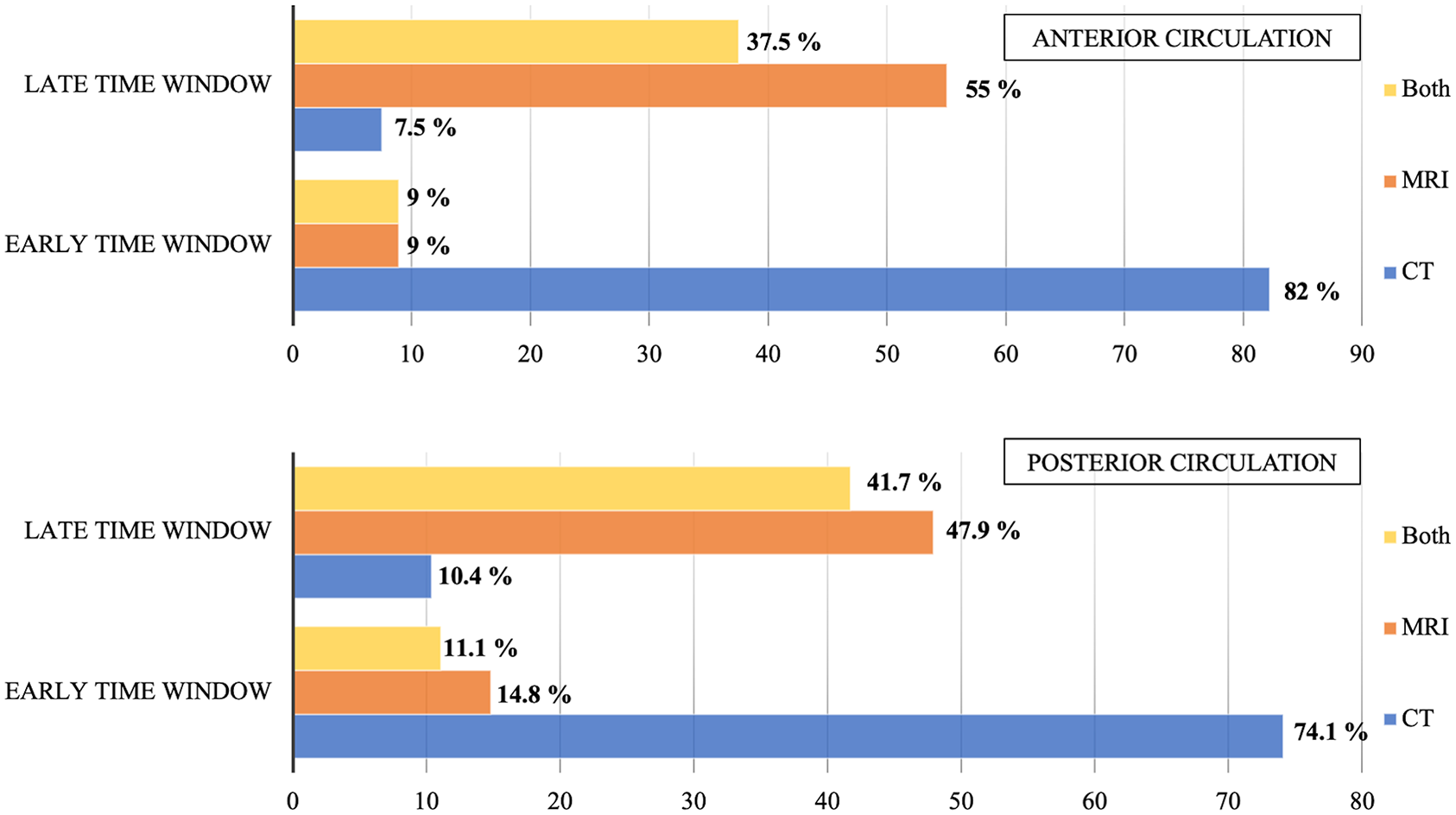
Patient selection according to imaging modalities.
INR were asked what conditions led them to rule out MT among different proposals (see Table 1).
Propositions leading to the rejection of MT according to INR.
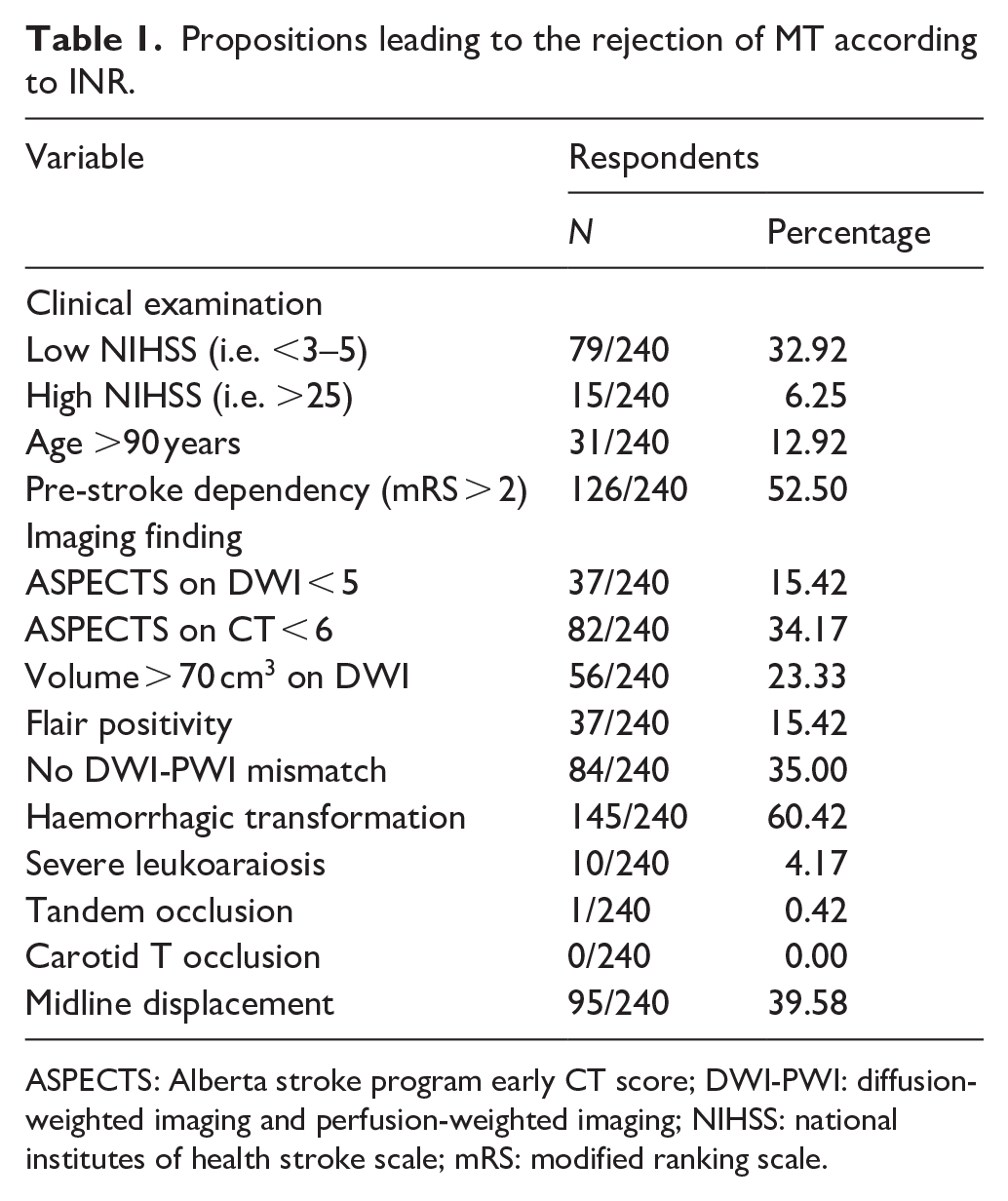
ASPECTS: Alberta stroke program early CT score; DWI-PWI: diffusion-weighted imaging and perfusion-weighted imaging; NIHSS: national institutes of health stroke scale; mRS: modified ranking scale.
Pre-stroke dependency (mRS > 2) and haemorrhagic transformation were the only two proposals that exceeded the average with 52.50% (126/240) and 60.42% (145/240) respectively. Most INR declared performing MT regarding anterior (138/240, 57.50%) or posterior (147/240, 61.25%) circulation regardless of the delay, as long as there was a diffusion/clinical or diffusion/perfusion mismatch (See supplemental Figure 2). Despite significant clinical improvement or low NIHSS (NIHSS < 3), most INR performed MT for proximal occlusions involving M1 (188/240, 78.33%) or the superior part of the basilar artery (166/240, 69.16%) but only half of them for occlusion of the inferior part of the basilar artery (120/240, 50.00%), and very few for other more distal occlusions (Figure 3). A significant part of INR (91/240, 37.92%) performed MT for distal occlusions (M3, M4) as part of their routine practice.
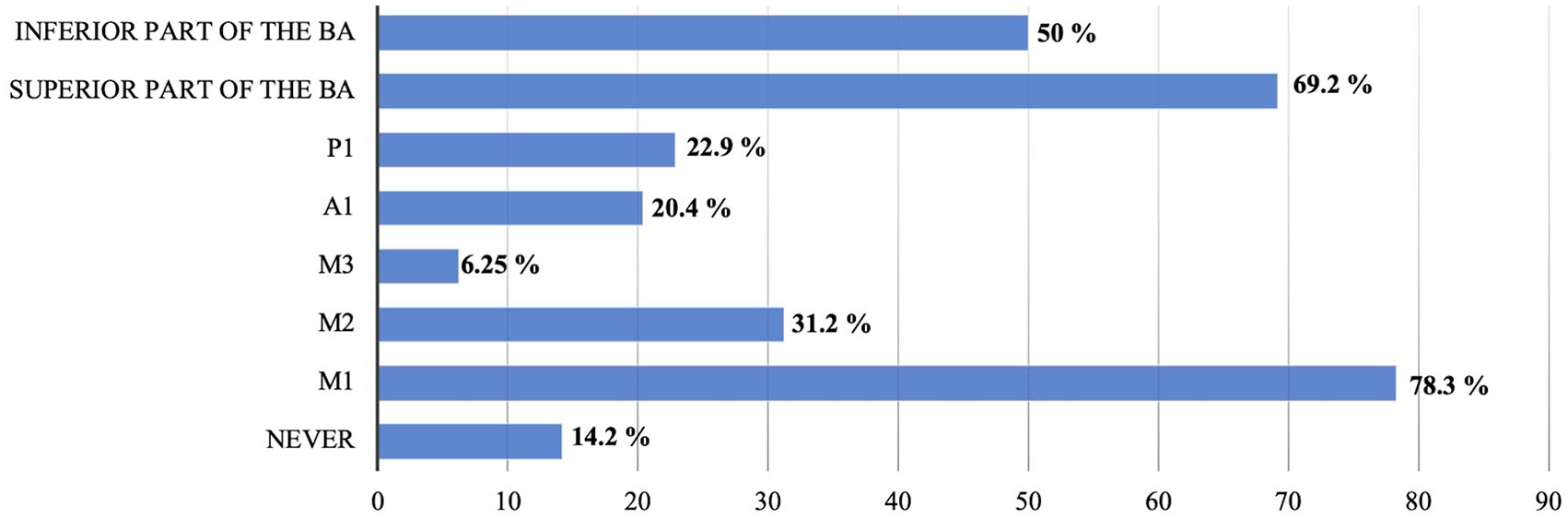
Thrombectomy despite significant clinical improvement or low NIHSS (NIHSS < 3).
Twenty-eight percent of INR (68/240, 28.33%) systematically performed a head flat panel CT in the angio room at the end of the procedure or only in case of complication during MT for 71.67% of them (172/240, 71.67%).
The follow-up imaging was mostly 24-h CT (see details of follow-up imaging modalities in Supplemental Figure 3).
Endovascular procedure and intravenous thrombolysis
In case of IVT, most centers used Alteplase (52/56, 92.86%). Arterial puncture was performed with palpation and anatomical landmarks for 78.75% of the INR (189/240) and with ultrasound guidance for 21.25% (51/240). The first-line arterial access was femoral for most of them (233/240, 97.08%).
When starting the MT procedure, most of the INR (206/240, 85.83%) explored only the occluded vessel, while 34 INR (34/240, 14.17%) evaluated the whole intracranial circulation. Sixty INR (60/240, 25.00%) used a balloon guide catheter very often (75.00% of cases), and 61 of them (61/240, 25.42%) never used it.
The three most used stent retrievers were Solitaire / Solitaire X (Medtronic, Irvine, CA, USA) (171/240, 71.25%), Trevo/Trevo NXT (Stryker Neurovascular, Fremont, CA, USA) (150/240, 62.50%) and Embotrap (Cerenovus, Johnson & Johnson Medical Devices, Irvine, CA, USA) (132/240, 55.00%). See Supplemental Figure 4.
The three most widely used aspiration catheters were Sofia / Sofia plus (MicroVention Terumo, Tustin, California, USA) (190/240, 79.17%), the ACE 60/64/68 (Penumbra, Alameda, CA, USA) (114/240, 47.50%), and Catalyst (Stryker Neurovascular, Fremont, CA, USA) (92/240, 38.33%). See Supplemental Figure 5.
The use of an aspiration system during clot retrieval differed according to the INR responses: 40.00% of them (96/240, 40.00%) used both the aspiration device and a syringe, 27.50% (66/240, 27.50%) of them used a dedicated aspiration device with an aspiration pump, and 32.50% of them (78/240, 32.50%) used only a syringe. The mean number of maximum deployments of the device to restore flow was 4.73 ± 2.30. Most INR (209/240, 87.08%) limited the number of recanalization attempts to six passes to restore flow. When MT with a first stent retriever failed, most INR (159/240, 66.25%) used a second stent retriever. Seventy-two percent of INR (174/240, 72.50%) decided to change the technique after two or more failed attempts, while 20.42% of them (20.42%, 49/240) when the first attempt failed or almost never (7.08%, 17/240).
Thirty-eight percent of INR (91/240, 37.92%) used intraarterial fibrinolysis during MT, mainly to treat distal fragmentation (76.92%, 70/91), in the case of failure of MT (18.68%, 17/91), or systematically (4.40%, 4/91).
During MT, most INR (196/240, 81.67%) did not use heparin, while 30 INR (12.50%, 30/240) used a single bolus and 14 of them (14/240, 5.83%) used a bolus with a maintained dose.
Most TCSCs (49/56, 87.50%) had an anesthetic team available at all times. Taking into account the occlusion of the middle cerebral artery occlusion or the carotid occlusion, TCSC used almost the same anesthetic procedures, mainly general anesthesia (22/56, 39.29% and 25/56, 44.64% respectively) or initially conscious sedation switched to general anesthesia if necessary (16/56, 28.57% and 13/56, 23.21% respectively), and for basilar artery occlusion most of them (34/56, 60.71%) started procedures directly with general anesthesia detailed in Figure 4.
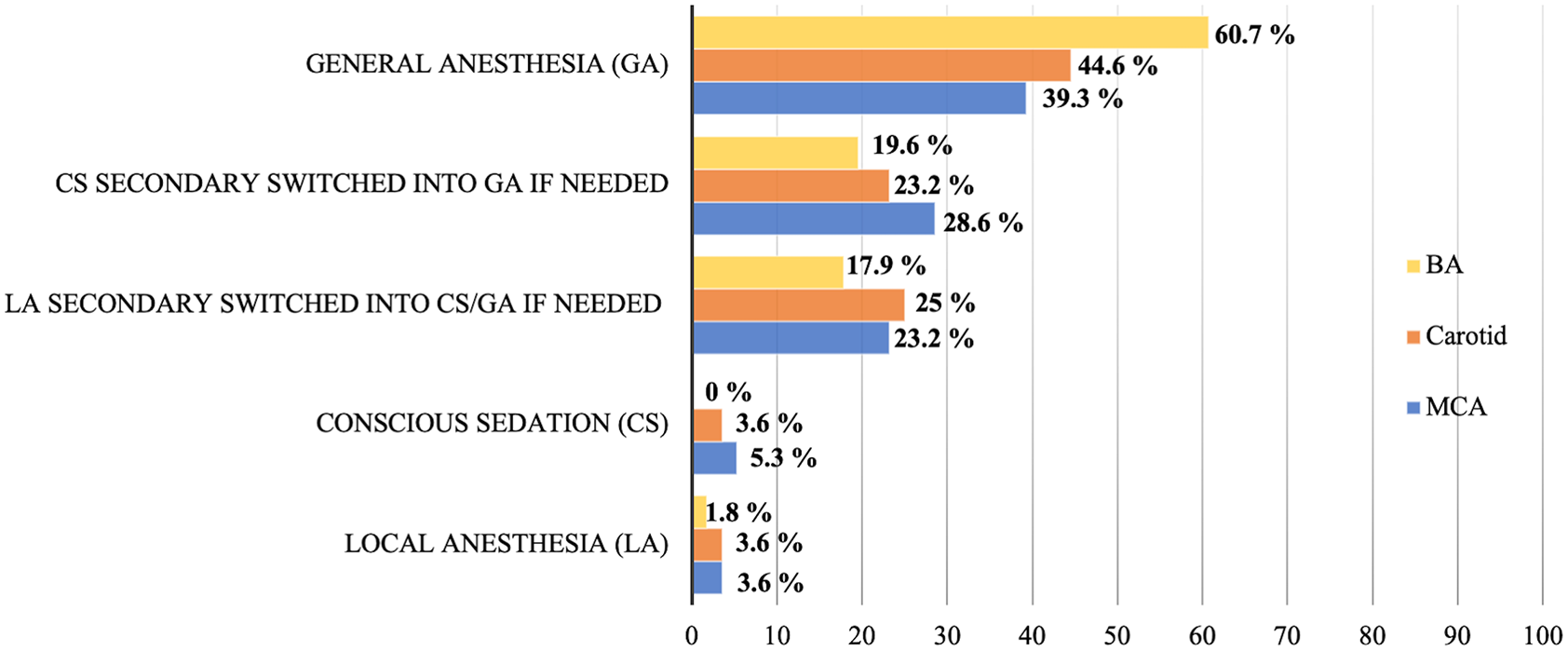
Anesthetic considerations according to the location of the occlusion.
In case a second patient also requiring MT during night procedures (when the first MT is not yet over) was treated differently according to the TCSC: Most of them (39/55, 70.91%) waited until the end of the first MT, 20.00% of the TCSC (11/55, 20.00%) called for a second INR, and 9.09% of them (5/55, 9.09%) started the second MT by the trainee INR; one center could not provide an answer.
Discussion
The main findings of our study indicate that even if most centers remain understaffed to ensure current and future MT needs, most European TCSCs are actively training young INR. In Europe, stroke is responsible for approximately 1.1 million deaths 5 and is a leading cause of disability and mortality worldwide, with a significant burden on healthcare systems. Stroke care in Europe has seen great improvements in recent years,9,10 leading to better outcomes and a reduced burden on healthcare systems. In recent years, there has been a significant increase in the use of MT to treat patients with AIS in Europe, with evidence of its effectiveness 11 This approach involves collaboration between primary care physicians, radiologists, neurologists, emergency services, rehabilitation teams and INR. Timely and well-coordinated care across the continuum of care is crucial to reducing the morbidity and mortality associated with stroke. To this end, it is essential to have an overview of current practices in order to standardize treatments and care. It is also necessary to evaluate compliance with current guidelines, so that future improvements can be assessed through similar surveys. To date, the present study is the first to report on MT practices at the European level. Here, we focus on key aspects of the real-world response to the challenge posed by MT, as well as the evolution of practices and organizations at TCSC, and present an overview of MT practices for the year 2021 at European level.
Second, European healthcare systems have invested in the implementation of stroke units and the use of evidence-based clinical guidelines. ESO-ESMINT contribute to maintaining guidelines for indications, techniques, or devices, which can serve as advice for our practices. 12 For that, analysis of current evidence and evidence-based recommendations through guidelines helps INR and encourage the use and appropriate implementation of tools in their clinical decisions to help physicians treat patients with AIS. 6 In this study, the selection of patients shows new information on the various responses to the different imaging and/or clinical criteria that lead to the dismissal of MT for the patient. In the real world, for example, patients with pre-stroke dependency are less likely to benefit from revascularization according to INR surveyed. But such patients should not be systematically excluded from MT because a significant proportion of these patients can recover after surgery. 7 Surprisingly, we also learn that three-quarters of physicians already routinely perform distal MT in the sylvian artery, even though there are as yet no guidelines on these indications and no results from randomized clinical trials.
In this survey, trainees-INR are substantial in the composition of the team during day and night, as well as X-ray technicians, especially since they also participate in performing the imaging to diagnose the stroke. A significant number of TCSC also involve nurses, which also appears to be a solution for understaffed X-ray technician teams. Indeed, thrombectomy is a complex procedure that often requires having a second person to help the main operator, the latter, if not a doctor or a medical student, may be a nurse or a X-ray technician. On a positive note, 78.57% of TCSC (44/56, 78.57%) trained at least one INR, but the number of INR qualified for MT in the on-call team remains low in most centers (3.4 ± 1.52, median 3.5 (IQR, 2–4)). This is even more true given that the number of thrombectomies is set to increase 13 for many reasons as indications multiply and the population ages. Furthermore, only 3.57% of the TCSC (2/56) declared themselves to be adequately staffed (as perceived by practitioners) at the time of the survey, and 37.50% of the TCSC (21/56, 37.50%) were concerned that at least one staff member was approaching retirement, while MT remains a complex procedure. There is probably a lack of sufficient increase in the number of trained INR, especially if we look at new indications for intracranial hypertension and hypotension, or embolization for chronic subdural hematoma.8,9,14,15 A solution could also come with the training and participation of interventional radiologists to perform thrombectomies, as is currently the case in France.4,11,12 This survey showed that a significant part of INR are involved in diagnostic neuroradiology and in other fields of radiology. Similarly, pooling angio-suites with other radiology specialties or cardiology can help meet the urgent needs related to MT at a lower cost.
Some points have not been evaluated and should be considered in future surveys related to training through simulation. Trainee-INR should never try the first time in humans, and should benefit from extensive simulator training, as well as when INR tests a new device.16,17 There are no guidelines on this topic, but the relationship between institutions and industry could fully benefit from a legal framework governing these obligations to improve INR training. Finally, the time spent in the angio room for training as well as the number of procedures performed should also be appraised and assessed. Indeed, we know that in trained INR, increasing experience in MT is associated with shorter procedural duration and better reperfusion rates, with a theoretical ceiling effect observed after around 100 procedures. 18 Formal statistical analyses cannot be performed when comparing self-reported data from individual INR regarding MT practices, or general data on routine selection and organization at the TCSC level. Another limitation of this work is that INR declarations cannot be controlled, and a single person per center was designated to complete the survey to collect general center-based information. This potential bias is limited by the use of protocols and standardization of MT at most TCSC, and the recording of IP addresss of the responders, limiting respondents to a single response.
To our knowledge, no such data has been published before except in 2015, with the study by Fargen et al. in the US, which concluded in a modest increase in thrombectomy procedural volumes since the publication of the five major stroke trials in the same year. 19 However, this survey was probably carried out too early in relation to the publication of the trials. Nevertheless, the global challenge of stroke care reorganization likely means that there are similar disparities and shortages in trained physicians worldwide. Furthermore, the results of this survey present a snapshot of MT practices in Europe at a specific time point and could be different at the time of the publication of the results, as, for example, there has been new data on MT outcomes for stroke patients with low ASPECT10,20–23 and new insights for basilar artery occlusion. 24 Future studies on MT in Europe could also be based on the national registers of the countries, such as the ETIS register, to provide additional information, such as treatment times.
Conclusions
This survey provides an overview of MT practices and compliance with INR guidelines in Europe. Additionally, it establishes a framework for evaluating organizational, training, and staffing needs at TCSC. Future efforts should focus on adequately training INR to ensure long-term sustainability, and on sufficient staffing without restricting MT indications, which are rapidly expanding over time.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241286000 – Supplemental material for Mechanical thrombectomy practices in Europe: Insights from a survey of European neuroradiologists from the ESMINT
Supplemental material, sj-docx-1-eso-10.1177_23969873241286000 for Mechanical thrombectomy practices in Europe: Insights from a survey of European neuroradiologists from the ESMINT by Géraud Forestier, Uta Hanning, Johannes Kaesmacher, Grégoire Boulouis, Kamil Zeleňák, Anne-Christine Januel, Zsolt Kulcsár, Jens Fiehler and Aymeric Rouchaud in European Stroke Journal
Supplemental Material
sj-pdf-2-eso-10.1177_23969873241286000 – Supplemental material for Mechanical thrombectomy practices in Europe: Insights from a survey of European neuroradiologists from the ESMINT
Supplemental material, sj-pdf-2-eso-10.1177_23969873241286000 for Mechanical thrombectomy practices in Europe: Insights from a survey of European neuroradiologists from the ESMINT by Géraud Forestier, Uta Hanning, Johannes Kaesmacher, Grégoire Boulouis, Kamil Zeleňák, Anne-Christine Januel, Zsolt Kulcsár, Jens Fiehler and Aymeric Rouchaud in European Stroke Journal
Footnotes
Acknowledgements
The authors thank all their ESMINT colleagues who agreed to participate in this survey and both ESMINT and EYMINT.
Abbreviations
AIS: Acute Ischemic Stroke
ASL: Arterial Spin Labeling
ASPECTS: Alberta Stroke Program Early CT score
A1: Pre-communicating segment of the anterior cerebral artery
BA: Basilar Artery
CS: Conscious Sedation
CT: Computed Tomography
DWI: Diffusion Weighted Imaging
ESMINT: European Society of Minimally Invasive Neurological Therapy
GA: General Anesthesia
INR: Interventional Neuroradiologist
LA: Local Anesthesia
LVO: Large Vessel Occlusion;
mRS: Modified Rankin Scale
MT: Mechanical Thrombectomy
M1: Horizontal segment of the middle cerebral artery
M2: Insular segment of the middle cerebral artery
M3: Opercular segment of the middle cerebral artery
NIHSS: National Institutes of Health Stroke Scale
PWI: Perfusion Weighted Imaging
P1: Pre-communicating segment of the posterior cerebral artery
TCSC: Thrombectomy-capable stroke center
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Informed consent
Not applicable
Guarantor
GF
Contributorship
All authors conceived the study. GF, AR, JF were involved in protocol development. GF was involved in the data analysis, and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
