Abstract
Background:
Acute intracranial occlusion of the internal carotid artery (ICA) can be distinguished into (a) occlusion of the terminal ICA, involving the proximal segments of the middle or anterior cerebral artery (ICA-L/-T) and (b) non-terminal intracranial occlusions of the ICA with patent circle of Willis (ICA-I). While patients with ICA-L/-T occlusion were included in all randomized controlled trials on endovascular therapy (EVT) in anterior large vessel occlusion, data on EVT in ICA-I occlusion is scarce. We thus aimed to evaluate effectiveness and safety of EVT in ICA-I occlusions in comparison to ICA-L/-T occlusions.
Methods:
A large international multicentre cohort was searched for patients with intracranial ICA occlusion treated with EVT between 2014 and 2023. Patients were stratified by ICA occlusion pattern, differentiating ICA-I and ICA-L/-T occlusions. Baseline factors, technical (modified thrombolysis in cerebral infarction (mTICI) scale) and functional outcomes (modified Rankin scale [mRS] at 3 months) as well as rates of (symptomatic) intracranial hemorrhage ([s]ICH) were analyzed.
Results:
Of 13,453 patients, 1825 (13.6%) had isolated ICA occlusion. ICA-occlusion pattern was ICA-I in 559 (4.2%) and ICA-L/-T in 1266 (9.4%) patients. Age (years: 74 vs 73), sex (female: 45.8% vs 49.0%) and pre-stroke functional independency (pre-mRS ⩽ 2: 89.9% vs 92.2%) did not differ between the groups. Stroke severity was lower in ICA-I patients (NIHSS at admission: 14 [7–19] vs 17 [13–21] points). EVT was similarly successful with respect to technical (mTICI2b/3: 76.1% (ICA-I) vs 76.6% (ICA-L/-T); aOR 1.01 [0.76–1.35]) and functional outcome (mRS ordinal shift cOR 1.01 [0.83–1.23] in adjusted analyses. Rates of ICH (18.9% vs 34.5%; aOR 0.47 [0.36–0.62] and sICH (4.7% vs 7.3%; aOR 0.58 [0.35–0.97] were lower in ICA-I patients.
Conclusion:
EVT might be performed safely and similarly successful in patients with ICA-I occlusions as in patients with ICA-L/-T occlusions.
Introduction
After the publication of five ground-breaking randomized controlled trials (RCTs) in 2015, endovascular therapy (EVT) became the guideline-recommended standard therapy for ischemic stroke patients with large vessel occlusion (LVO) of the anterior cerebral circulation.1–7 In the above-mentioned trials, LVO was defined as an occlusion of the intracranial or distal internal carotid artery (ICA) or the first two segments of the middle cerebral artery (MCA).1–5 A patient-level-meta-analysis of these trials (HERMES), reported about 20% of patients presenting with an ICA occlusion. 8 Intracranial ICA-occlusions, however, are not a homogenous group, but can be differentiated into at least two different occlusion patterns: (a) ICA-L-/-T occlusions, that is, thrombi in the terminal ICA segment, involving the proximal segments of the middle and/or anterior cerebral artery; (b) ICA-I occlusions, that is, thrombi located more proximally in the intracranial ICA segments with patent carotid-T, allowing collateral flow into the ipsilateral MCA.9,10 In the RCTs on EVT in the anterior circulation, the vast majority of ICA occlusion patients had a terminal ICA occlusion. Data on efficacy and safety of EVT in non-terminal intracranial ICA occlusion is scarce.1–5 We therefore analyzed technical and functional outcomes as well as safety of EVT in patients with acute ICA-I occlusion and compared them to patients with ICA-L/-T occlusion pattern.
Methods
Study population
For this study, we used prospectively collected data from 16 centers of the EndoVAscular treatment and ThRombolysis for Ischemic Stroke Patients (EVATRISP) collaboration. EVATRISP is an academic multicentre international collaboration with a defined structure, process and methodology to generate a pooled registry composed of individual patient data from each participating center.11,12
All patients treated for an intracranial ICA occlusion between 2014 and 2023 documented in EVATRISP were identified. Based on the occlusion pattern found in CT-/MR-angiography at admission, the cohort was divided into two groups: (a) ICA-L/-T occlusion (with or without occlusion of further proximal ICA segments); (b) ICA-I occlusion (intracranial ICA occlusion with patent carotid-T). Figure 1 illustrates the different ICA occlusion patterns. Patients with occlusion of the extracranial ICA or intracranial tandem occlusion (i.e. ICA-I occlusion with documented additional thrombi in any segment of the middle or anterior cerebral artery) were excluded from the analysis. We compared the occlusion patterns regarding baseline variables, treatment modality, procedure times, as well as technical and functional outcomes and rates of intracranial hemorrhage.
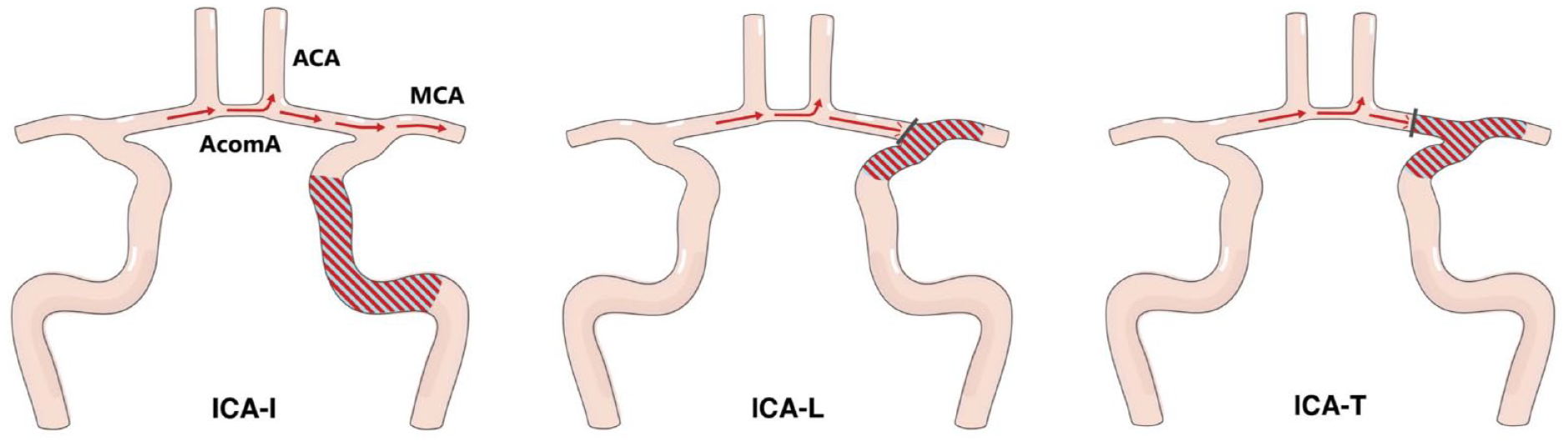
Occlusion patterns of the internal carotid artery.
Variables
Stroke severity at admission and 24 h after EVT was assessed using the National Institutes of Health Stroke Scale (NIHSS). 13 Early ischemic changes on imaging at admission were quantified with the Alberta Stroke Program Early CT Score (ASPECTS). 14 Intravenous Thrombolysis (IVT) was used as per local standard of care in the participating centers. All 16 centers performed IVT up to 4.5 h from symptom onset or last-seen-well. Twelve of 16 centers (75.0%) allowed IVT up to 9 h whenever perfusion based-imaging criteria were fulfilled and 2 of 16 centers (12.5%) applied IVT up to 24 h following a multimodal imaging approach. The modified Rankin Scale (mRS) 15 3 months after the index event was defined as the primary clinical outcome (shift analysis). As secondary clinical outcomes, we chose functional independence (mRS ⩽ 2) and capacity to walk without assistance of another person (mRS ⩽ 3) three months after stroke (binary outcomes). Furthermore, early neurological improvement (ENI, i.e. improvement of at least four points on the NIHSS or reaching a NIHSS of zero points 24 h after admission) and early neurological deterioration (END, worsening of at least four points on the NIHSS 24 h after admission) were compared. Technical outcome was assessed using the modified Thrombolysis in Cerebral Infarction (mTICI) scale 16 at the end of the procedure, defining successful recanalization as mTICI 2b/3. We analyzed clot migration/embolization to a primarily not affected vessel territory and perforation of cerebral vessels as treatment adverse events. As safety variables, we chose intracranial hemorrhage (ICH), symptomatic ICH as defined in ECASS-II 17 (sICH; any ICH with NIHSS worsening of four or more points), and mortality 3 months after stroke. We further assessed the occurrence of malignant infarction (defined as infarcted tissue in more than 2/3 of the MCA territory, significant midline shift in the absence of ICH and/or indication for decompressive hemicraniectomy (DHC)).
Statistical analysis
Continuous baseline variables and treatment times are presented as median (interquartile range; IQR), dichotomous variables as absolute numbers and percentage. Comparisons regarding distribution between groups were performed by the Kruskal-Wallis test for linear and ordinal variables, and Chi-Squared test for dichotomous variables. Binary logistic and ordinal regression (mRS shift) analyses were used to assess the impact of occlusion pattern on clinical and technical outcomes. For technical outcomes and treatment adverse events, adjustments were made for stroke etiology (large-artery-atherosclerosis (LAA) vs other etiologies) and thrombolysis. Odds Ratios (ORs) for functional and safety outcomes were adjusted for age, sex, stroke severity (NIHSS at admission), intravenous thrombolysis, pre-event dependency (mRS pre-stroke) and witnessed onset of stroke symptoms. Anticoagulation (use of Dabigatran, Rivaroxaban, Edoxaban, Apixaban, Heparin (therapeutic dose), Warfarin) was included in the model for ICH and sICH. Since ASPECTS was only available in about half of the patients, the variable was not included in the main adjusted analysis. However, considering ASPECTS being a well-established predictor of functional outcome and ICH rates, we conducted a subgroup analysis for all patients with available ASPECTS, including the variable into the models for clinical and safety outcomes. Since the presented study is based on observational data and thus only hypothesis-generating, we did not correct p-values for multiple testing. All analyses were conducted using IBM SPSS Statistics for Windows (Version 28.0.0.0, Armonk, NY: IBM Corp).
Informed consent and ethics approval
Each center has received necessary official approval from their respective local authorities and/or ethical committees according to national and local rules. 11
Results
Between 2014 and 2023, 13,453 patients from 16 centers in 8 different countries were included in the EVATRISP registry. Of these, 1825 (13.6%) presented with an intracranial ICA-occlusion. They constitute the population of this study and were differentiated into patients with acute ICA-I-occlusion (N = 559, 4.2%) and acute ICA-L/-T-occlusion (N = 1266, 9.4%; of the whole EVATRISP sample, respectively). With regard to the two groups of interest, the distribution of age (years: 74 [62–81] vs 73 [63–83], p = 0.60), sex (female: 45.8% vs 49.0%, p = 0.21) and functional independence before the index event (pre-stroke mRS ⩽ 2: 89.9% vs 92.2, p = 0.13) did not differ significantly (ICA-I vs ICA-L/-T, respectively). Patients with ICA-I occlusion presented with less severe stroke symptoms at admission than patients with ICA-L/-T occlusion (NIHSS: 14 [7–19] vs 17 [13–21] points, p < 0.001). In addition, the prevalence of diabetes (25.6% vs 16.8%, p < 0.001) and dyslipidemia (50.8% vs 41.4%, p < 0.001) were higher in patients with ICA-I occlusion. In concordance with this distribution of vascular risk factors, LAA was the most common stroke etiology in ICA-I occlusion patients (41.2%), while ICA-L-/-T occlusion patients were most likely to have a cardioembolic stroke (39.8%). More than one stroke etiology (atrial fibrillation and ICA-Stenosis > 50%) was present in 7.2% (ICA-I) and 6.4% (ICA-L/-T), respectively. The median systolic blood pressure at admission was higher in patients with ICA-I occlusion by 4 mmHg (154 [137–170] mmHg versus 150 [133–170] mmHg (ICA-L-/T), p = 0.03). All baseline demographics are presented in Table 1.
Baseline demographics, stroke etiology, imaging & treatment characteristics.
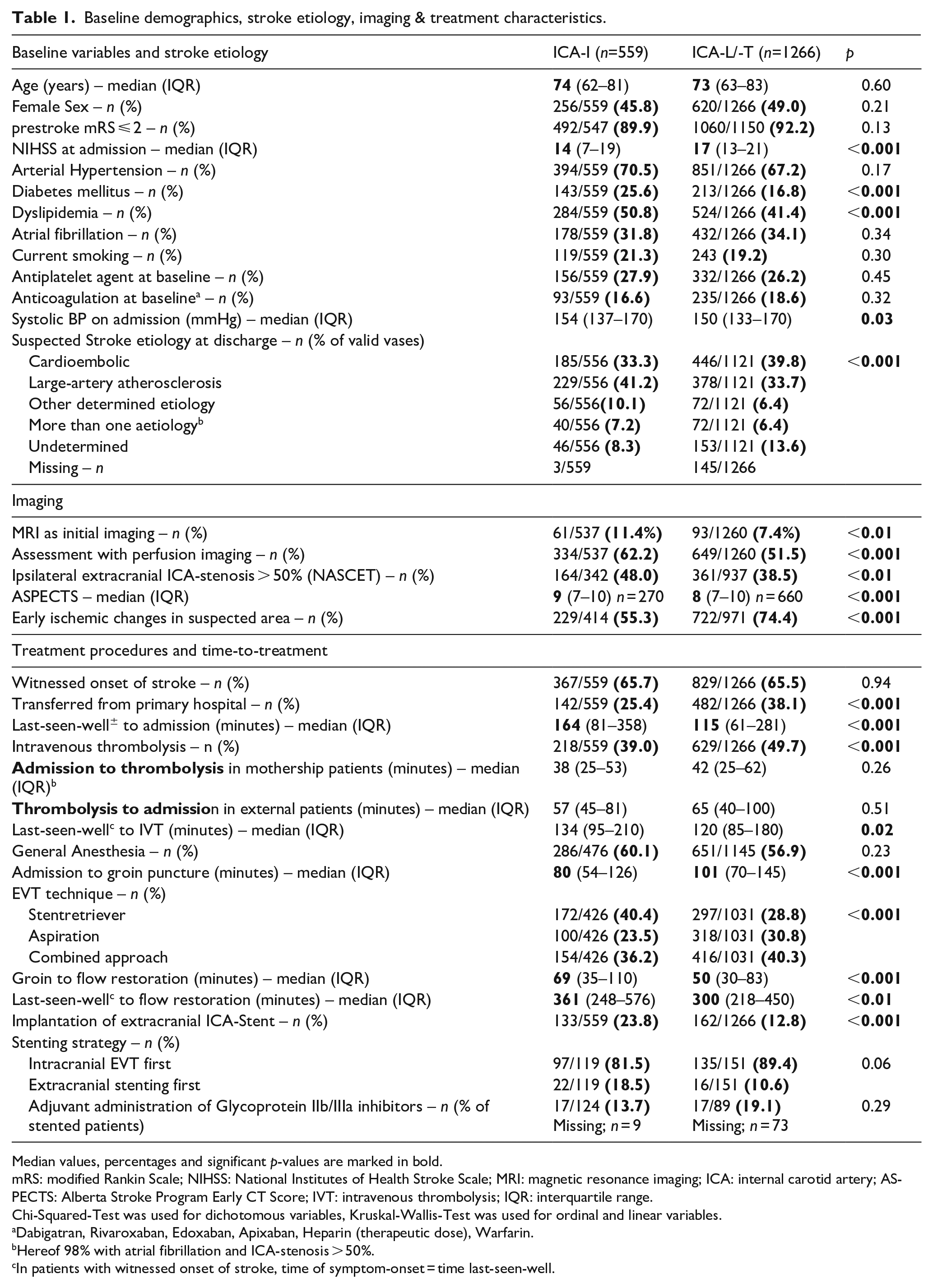
Median values, percentages and significant p-values are marked in bold.
mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; MRI: magnetic resonance imaging; ICA: internal carotid artery; ASPECTS: Alberta Stroke Program Early CT Score; IVT: intravenous thrombolysis; IQR: interquartile range.
Chi-Squared-Test was used for dichotomous variables, Kruskal-Wallis-Test was used for ordinal and linear variables.
Dabigatran, Rivaroxaban, Edoxaban, Apixaban, Heparin (therapeutic dose), Warfarin.
Hereof 98% with atrial fibrillation and ICA-stenosis > 50%.
In patients with witnessed onset of stroke, time of symptom-onset = time last-seen-well.
Regarding initial imaging modality, MRI was more frequently used in patients with ICA-I occlusion (11.4% vs 7.4%, p < 0.01). Furthermore, perfusion-based imaging (MR- or CT-perfusion) was more commonly applied in patients with ICA-I occlusion (62.2% vs 51.5%, p < 0.001). Individuals with ICA-I occlusion less often had early ischemic changes in the suspected area (ASPECTS < 10; 55.3% vs 74.4%, p < 0.001) and a higher median ASPECTS score (9 [7–10] vs 8 [7–10] points, p < 0.001). Ipsilateral extracranial ICA-stenosis > 50% (NASCET criteria) was detected in 48.0% of patients with ICA-I- and 38.5% of patients with ICA-L-/-T occlusion (p < 0.01).
In both groups, about two of three individuals presented with a witnessed onset of stroke. Primary admission at an external hospital occurred less often in patients with ICA-I- than ICA-L-/T-occlusion (25.4% vs 38.1%, p < 0.001), however, admission patterns in the participating centers were notably heterogeneous, with rates of secondary transport varying between 0% and 74%. Detailed information on admission patterns for each center can be found in Supplemental Table S2. Time between last-seen-well and arrival at the interventional hospital was longer in patients with ICA-I occlusion (median [IQR] 164 [81–358) vs 115 [61–281] minutes, p < 0.001). Correspondingly, rates of IVT were lower in individuals with ICA-I occlusion (39.0% vs 49.7%, p < 0.001). Time from hospital arrival to start of IVT did not differ between the two groups. Despite similar rates of general anesthesia (about 60%), the delay from hospital arrival to groin puncture was shorter in ICA-I occlusion patients (80 [54–126] vs 101 [70–145] minutes, p < 0.001). Focusing on the EVT itself, we found a longer time from groin puncture to flow restoration in ICA-I patients (69 [35–110] vs 50 [30–83] minutes, p < 0.001). The preferred strategy of the procedure itself was Stent Retriever (SR; 40.4%), followed by a combination of SR and Aspiration (36.2%) and Aspiration alone (23.5%) in patients with ICA-I occlusion. EVT for ICA-L/-T occlusion was most commonly performed as combined approach (40.3%), followed by Aspiration alone (30.8%) and SR alone (28.8%; p < 0.001; ICA-I vs ICA-L/-T). The rate of extracranial ICA-stenting was higher in ICA-I occlusion patients (23.8% vs 12.8%, p < 0.001). Regarding sequence of EVT in stenting, 18.5% of patients with ICA-I occlusion and 10.6% with ICA-L/-T occlusion were treated with extracranial stenting before intracranial thrombectomy (p = 0.06). Glycoprotein2b/3a inhibitors after stenting were used in 13.7% of ICA-I patients and 19.1% of ICA-L-/T patients (p = 0.29). All procedural variables and treatment times are depicted in Table 1.
In both ICA-I and ICA-L/-T occlusion patients, successful recanalization (mTICI 2b/3) was reached in about three of four patients (76.1% vs 76.6%, aOR 1.01 [0.76–1.35], p = 0.94). During the study period, rates of successful recanalization generally increased from 61.9% (2015) to 87.4% (2022). Regarding the clinical outcome, ICA-I-occlusion was associated with a better functional outcome in univariable analysis (mRS shift; common OR 1.26 [1.05–1.51], p < 0.01). However, after adjusting for stroke severity, age, sex, IVT, pre-event dependency and witnessed onset of stroke symptoms, functional outcome at 3 months did not differ between groups any more (adjusted cOR 1.01 (0.83–1.23), p = 0.92). Similarly, we found no difference regarding the adjusted binary mRS endpoints (mRS ⩽ 2: 38.4% vs 34.2%, aOR 0.95 [0.73–1.22], p = 0.67); mRS ⩽ 3: 55.6% versus 47.5%, aOR 1.13 (0.89–1.45), p = 0.32).
Figure 2 displays the distribution of functional outcomes across the whole mRS stratified for ICA occlusion pattern. We found numerically lower rates of early neurological improvement (37.2% vs 45.9%) and higher rates of early neurological deterioration (21.7% vs 14.9%) in ICA-I occlusion patients. This difference was even more pronounced in patients with less severe stroke (NIHSS at admission ⩽ median; cut-offs at 14 (ICA-I) and 17 (ICA-L/-T). However, these findings did not remain significant in adjusted analysis. For absolute numbers on ENI and END stratified by stroke severity, see Table 2. For safety outcomes, we observed a lower odds for any ICH (18.9% vs 34.5%, aOR 0.47 [0.36–0.62], p < 0.001) and for symptomatic ICH (4.7% vs 7.3%, aOR 0.58 [0.35–0.97], p = 0.04) in patients with ICA-I occlusion even in adjusted analyses. In univariable analysis, ICA-I patients had a lower odds of malignant infarction (12.3%) when compared to ICA-L/-T patients (17.8%), OR 0.65 (0.47–0.90), p < 0.01). However, this finding was not robust when adjusting for age, NIHSS and pre-stroke dependency. Decompressive hemicraniectomy was performed more frequently in ICA-L/-T (2.7%) than in ICA-I patients (1.7%), but the difference did not reach statistical significance. There was no association of ICA occlusion pattern with mortality 3 months after stroke. Rates of vessel perforation and clot migration / embolization to new territories during EVT did not differ between the groups. All clinical, technical and safety outcomes are displayed in Table 2.
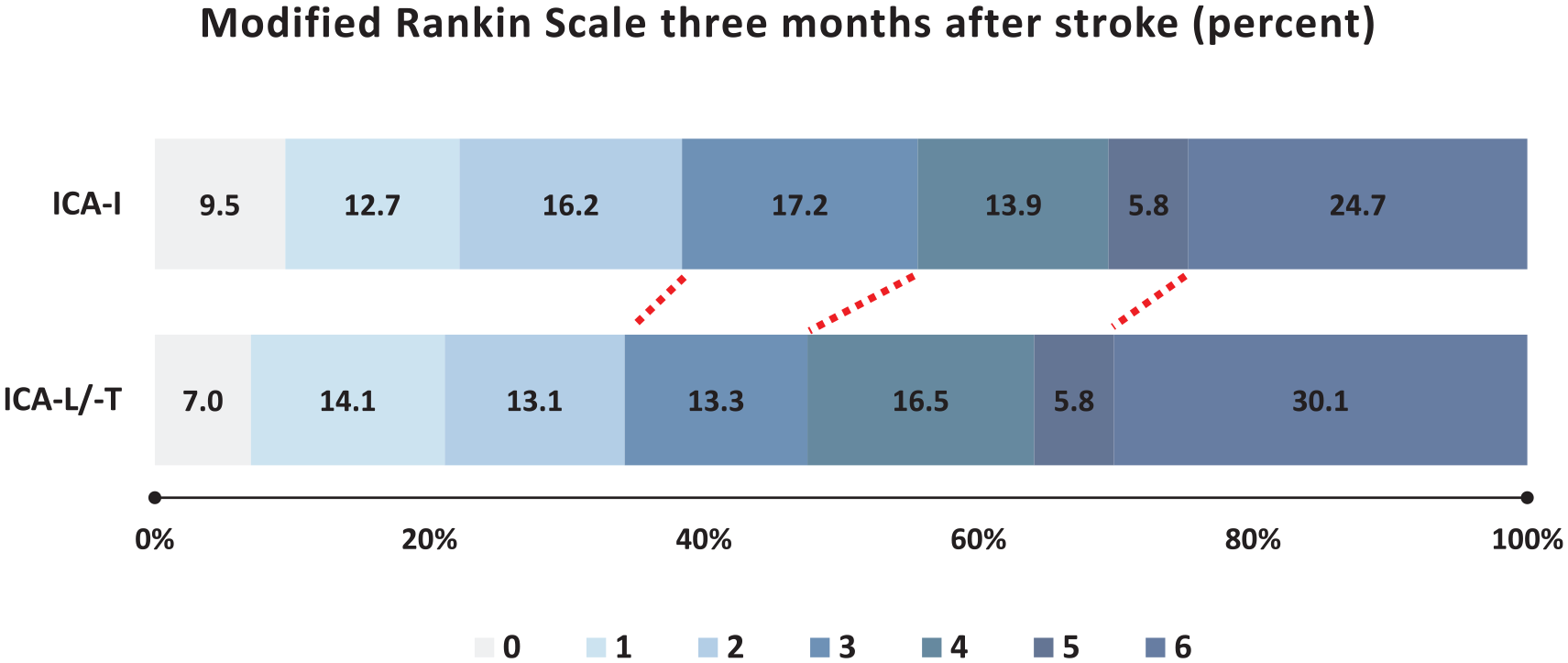
Distribution of clinical outcome (mRS) three months after stroke stratified by ICA occlusion pattern.
Clinical, technical and safety outcomes.
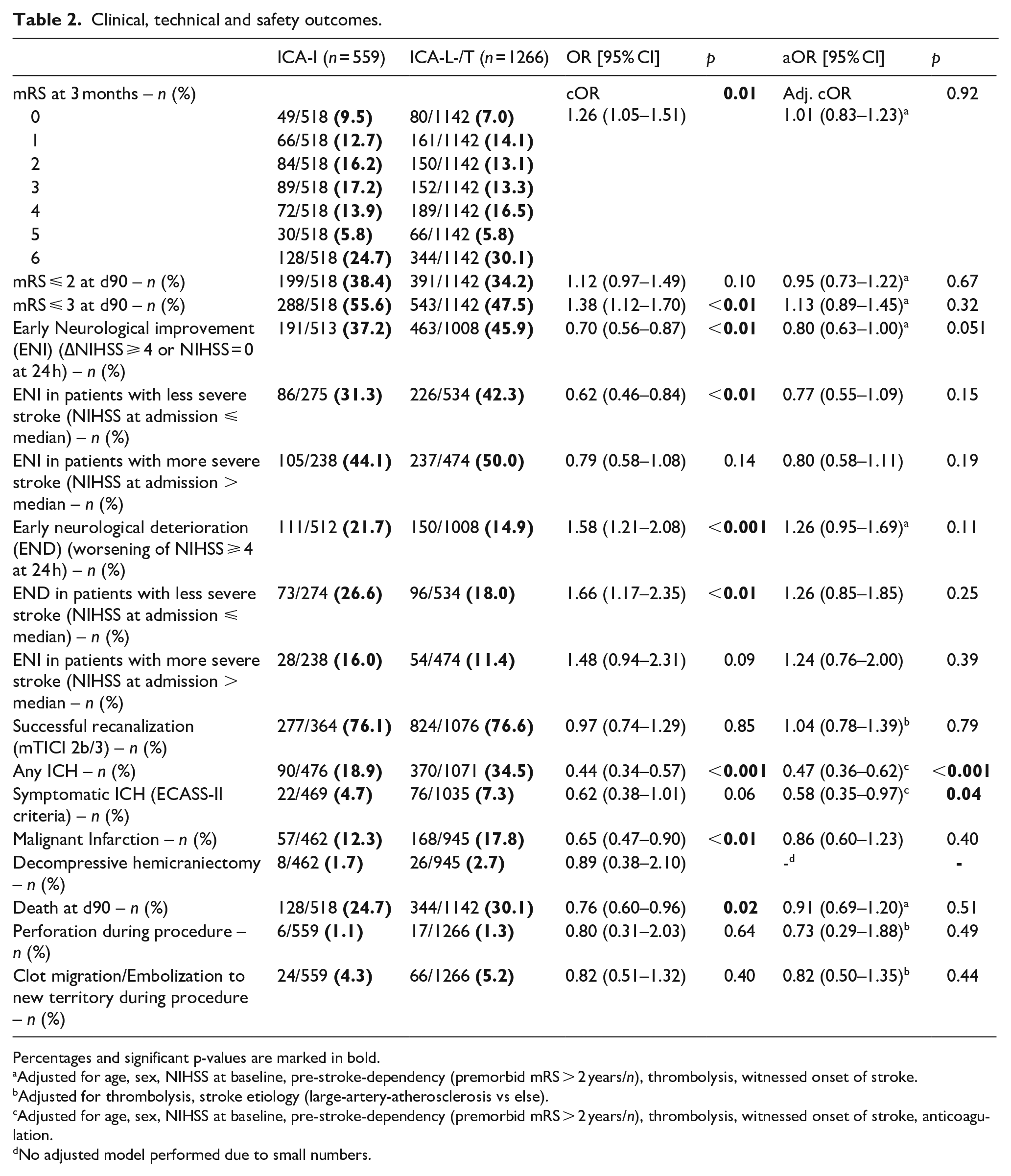
Percentages and significant p-values are marked in bold.
Adjusted for age, sex, NIHSS at baseline, pre-stroke-dependency (premorbid mRS > 2 years/n), thrombolysis, witnessed onset of stroke.
Adjusted for thrombolysis, stroke etiology (large-artery-atherosclerosis vs else).
Adjusted for age, sex, NIHSS at baseline, pre-stroke-dependency (premorbid mRS > 2 years/n), thrombolysis, witnessed onset of stroke, anticoagulation.
No adjusted model performed due to small numbers.
Due to the availability of ASPECTS score in only 970 of our study patients, we performed subgroup analysis including ASPECTS into the multivariable model, which did not reveal a relevant change in point estimates for functional outcome 3 months after stroke. In this subgroup, we found lower odds for ENI in individuals with ICA-I occlusion when compared with ICA-L-/-T occlusion patients (aOR 0.63 [0.46–0.86], p < 0.01. The above-mentioned association of ICA-I occlusion pattern and lower occurrence of sICH became non-significant, when taking ASPECTS score into account (aOR 0.58 [0.29–1.16], p = 0.13). All clinical outcomes for the subgroup with available ASPECTS can be found in the Supplemental Table S1.
Discussion
In this large observational, multicentre analysis of pooled data derived from prospectively ascertained local databases, we found that patients with ICA-I occlusion differ from patients with ICA-L/-T occlusion in several aspects. Differences are evident with respect to stroke severity, stroke etiology, procedure times and rates of intracranial hemorrhage: Patients with ICA-I occlusion were less severely affected and more frequently had large-artery-atherosclerosis as stroke etiology. Patients with ICA-I occlusion had longer prehospital delay and longer endovascular procedure time. Interestingly, patients with ICA-I occlusion had lower odds for intracranial hemorrhage after EVT but also lower rates of early neurological improvement. Functional outcome 3 months after stroke did not differ between patients with ICA-I and ICA-L/-T occlusion when adjusting for unequally distributed variables.
In the HERMES meta-analysis, 21% of patients suffered from an ICA-occlusion. 8 The prevalence of ICA-I occlusions, however, was very low in two of the contributing trials (0.8% in MR CLEAN, 0.5% in REVASCAT).1,2 Two other trials did not differentiate ICA-L/-T and ICA-I occlusion (SWIFT-PRIME, EXTEND-IA)4,5 and the fifth trial (ESCAPE) 3 did not include patients with an ICA-I occlusion pattern at all. 10 In contrast, we found a prevalence of 4.2% for ICA-I-occlusions in the EVATRISP registry, which represents real-life clinical practice in 16 academic tertiary stroke centers. This prevalence is higher than those reported in national EVT registries from the Netherlands (1.0%) 18 and Germany (2.4%), 10 yet still substantially lower than in an Italian multicentre registry study (7.8%). 19 Based on these data, isolated intracranial ICA-I occlusion can be considered a rare, yet still relevant entity of LVO, with several patients to be expected in high volume EVT centers per year. It can be hypothesized, that the large variance of prevalence in different national registries mirrors the existing uncertainty regarding the optimal treatment for patients with ICA-I occlusion. Additionally, different definitions of ICA-I (strictly intracranial with patent carotid-T vs extracranial and/or intracranial with patent carotid-T) may account for the differences in prevalence. Based on the results of a previous observational study from our lab, a distinction of isolated intra- and extracranial ICA occlusion with patent carotid-T may be reasonable. In this study, isolated intracranial ICA occlusion had similar characteristics and outcomes as ICA-L/-T occlusions, while isolated extracranial occlusions differed in baseline variables and outcome. 10
Of note, the delay from last-seen-well to hospital arrival was about 50 min longer in patients with ICA-I occlusion (when compared to ICA-L/-T). This finding is consistent with recently published data from a national German EVT registry 10 and may be explained by anatomically intact but in the further course insufficient collateralisation in ICA-I patients. In our study, we found several differences regarding imaging diagnostics: First, MRI as initial imaging modality was found more often in patients with ICA-I occlusion. Second, advanced, perfusion-based imaging was more common in ICA-I occlusion patients and third, median ASPECTS Score was lower in individuals with ICA-L/-T occlusion. Both the finding of higher prevalence of MRI and advanced, perfusion-based imaging are corroborated by previous reports. 10 Possible reasons include the longer delay from last-seen-well / symptom onset to hospital arrival as well as a general uncertainty, whether EVT is beneficial in ICA-I occlusion patients. Regarding the actual endovascular procedure, we found that the time from groin to successful recanalization was about 20 min longer in patients with ICA-I occlusion (69 vs 50 min). We assume that this is attributed to the higher proportion of patients with LAA stroke etiology, making access-to-clot more difficult. The longer duration of the procedure may further be explained by the fact that stenting in the extracranial ICA was performed about twice as often in in ICA-I patients.
Despite longer EVT time, good technical outcome (mTICI 2b/3) in ICA-I was achieved as frequently as in ICA-L/-T occlusion. Seventy-six percent of patients reached successful recanalization in both groups of our study and these results are consistent with data on ICA-I occlusions from the Italian IRETAS registry and the German Stroke Registry.10,19 When compared with the MR CLEAN registry from the Netherlands, which reports data on patients receiving EVT for an ICA-I occlusion between 2014 and 2017, our rates of successful recanalization are notably higher (76.1% vs 56.0%), potentially reflecting technical advances in recent years. 18 While 76.1% recanalization rate might still seem low from current day clinical perspective, it has to be noted that our study included patients from the early years of thrombectomy, when both neurointerventional techniques and clinical experience in EVT were inferior. Improved technical devices and increased clinical experience are mirrored in the higher recanalization rate over time we report in our study sample (61.9% in 2015; 87.4% in 2022). In univariable analysis, ICA-I occlusion pattern was associated with a better functional outcome three months after stroke. However, this finding is probably due to better collateral flow, causing the lower NIHSS at baseline which translates to better outcomes. After adjustment for stroke severity and other factors, the finding was not robust. Thus, occlusion site cannot be seen as independent predictor of the clinical outcome. Corroborating our results, a post-hoc analysis of the MERCI and MULTI-MERCI trials and a large German multicentre registry report numerically better clinical outcomes for ICA-I patients (when compared to ICA-L-/-T) – however, likewise not significant in adjusted analysis.9,10 ICA-I occlusion patients appeared to have a lower rate of ENI than ICA-L-/T patients (aOR 0.80, 95% CI 0.63–1.00, p = 0.051) and numerically higher rates of END. As possible explanation, we hypothesize that the usually better collateralisation of intracranial arteries in ICA-I occlusions – possibly supported by the higher systolic blood pressure at admission – may be wearing off over time, leading to progression of stroke symptoms. Lower rates of ENI (and vice versa high rates of END) have been reported for isolated extracranial ICA occlusion, in the literature, in which a similar pathophysiology can be expected.10,20 In our study, rates of ENI were even lower and rates of END even higher in ICA-I patients with less severe stroke symptoms at admission. This finding is consistent with the hypothesis that patients might have sufficient penumbral perfusion (and thus lower NIHSS) at admission, but experience deterioration in the course of the disease facing hemodynamic compromise. Blood pressure (BP) might play a crucial role in patients with non-terminal ICA occlusion since interhemispheric cross-flow over the circle of Willis naturally depends on hemodynamic premises. Given that BP is a modifiable factor and evidence on the optimal BP management in LVO patients is still scarce, 21 ICA-I occlusions might be seen as an eminently suitable model to study the impact of blood pressure in EVT patients, since BP drops might have immediate clinical response. Consequently, we strongly recommend further research in this field.
Regarding safety outcomes, we noticed a lower odds for asymptomatic and symptomatic ICH in patients with ICA-I occlusion. Lower infarct volumes due to better collateralisation in ICA-I patients might explain this finding. Consistently, lower infarct volumes most probably explain the difference in univariable analyses with regard to malignant infarction and hemicraniectomy. The above-mentioned association between sICH and ICA-occlusion site was not robust in the smaller population adjusted for ASPECTS, however, this was due to widening of the confidence interval whereas the point estimate did not change remarkably. Therefore, loss of significance may be attributed to a possible type-II error. While a high rate of intraprocedural clot migration and embolization has been reported for isolated extracranial ICA occlusions,10,20 our study does not show any signals for a similar phenomenon in ICA-I occlusion patients.
Our study has several limitations: First, there is selection bias, because the EVATRISP registry includes only patients with ICA-I occlusion who received EVT. Therefore, patients with low NIHSS as well as stable and persistent collateralization are underrepresented in our sample. Second, our study does not include a control group of ICA-I occlusion patients without EVT, therefore decisions on whether or under which circumstances EVT should be recommended in ICA-I occlusions cannot be deduced from our results. Third, the EVATRISP registry does not hold a central imaging database and anatomical configuration of the circle of Willis relevant to collateral capacity is not regularly assessed in the individual centers. Fourth, collateral scores, data on intracranial ICA-stenosis and quantitative results of perfusion imaging were not recorded by the majority of contributing centers. Consequently, we were not able to adjust for the impact of these variables. Fifth, ASPECTS was not available in all study patients. Therefore, we were able to adjust for this confounder only for a subgroup of patients.
Conclusion
Our study suggests that EVT might be performed safely and similarly successful in patients with ICA-I occlusions as in patients with ICA-L/-T occlusions.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241278948 – Supplemental material for Endovascular therapy in patients with acute intracranial non-terminal internal carotid artery occlusion (ICA-I)
Supplemental material, sj-docx-1-eso-10.1177_23969873241278948 for Endovascular therapy in patients with acute intracranial non-terminal internal carotid artery occlusion (ICA-I) by Christoph Riegler, Regina von Rennenberg, Kerstin Bollweg, Eberhard Siebert, Gian Marco de Marchis, Georg Kägi, Pasquale Mordasini, Mirjam R Heldner, Mauro Magoni, Alessandro Pezzini, Alexander Salerno, Patrik Michel, Christoph Globas, Susanne Wegener, Nicolas Martinez-Majander, Sami Curtze, Maria Luisa Dell’Acqua, Guido Bigliardi, Nabila Wali, Paul J Nederkoorn, Dejana R Jovanovic, Visnja Padjen, Issa Metanis, Ronen R. Leker, Giovanni Bianco, Carlo W Cereda, Rosario Pascarella, Marialuisa Zedde, Maria Maddalena Viola, Andrea Zini, João Nuno Ramos, João Pedro Marto, Heinrich J Audebert, Simon Trüssel, Henrik Gensicke, Stefan T Engelter and Christian H Nolte in European Stroke Journal
Footnotes
Acknowledgements
We would like to thank all participating centers of the EVATRISP collaboration and the respective patients for contributing. Furthermore, we thank Carlotta Törnqvist for the graphical illustrations and Pimrapat Gebert for statistical support.
List of abbreviations
aOR – Adjusted Odds Ratio
ASPECTS – Alberta Stroke Programme Early CT Score
BP – Blood Pressure
cOR – common Odds Ratio
END – Early Neurologic Deterioration
ENI – Early Neurologic Improvement
EVT - Endovascular therapy
ICA – Internal Carotid Artery
ICH – Intracranial Hemorrhage
IQR – Interquartile Range
IVT – Intravenous Thrombolysis
LAA – Large Artery Atherosclerosis
LVO – Large Vessel Occlusion
MCA – Middle Cerebral Artery
mRS – Modified Rankin Scale
mTICI – Modified Thrombolysis in Cerebral Infarction Scale
NIHSS – National Institutes of Health Stroke Scale
OR – Odds Ratio
RCTs – Randomized Controlled Trials
sICH – Symptomatic Intracranial Hemorrhage
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RRL received speaker honoraria from IscemaView, Boehringer Ingelheim, Pfizer, Jansen, Biogen, Medtronic and Abott and advisory board honoraria from Jansen and Bayer.
STE has received funding for travel or speaker honoraria from Bayer, Boehringer Ingelheim and Daiichi-Sankyo. He has served on scientific advisory boards for Bayer, Boehringer Ingelheim, BMS/Pfizer, and MindMaze and on the editorial board of Stroke. His institutions have received an educational grant from Pfizer, compensation from Stago for educational efforts and research support from Daiichi-Sankyo, the Science Funds [Wissenschaftsfonds] of the University Hospital Basel, the University Basel, from the “Wissenschaftsfonds Rehabilitation” of the University Hospital for Geriatric Medicine Felix Platter, the “Freiwillige Akademische Gesellschaft Basel,” the Swiss Heart Foundation, and the Swiss National Science Foundation.
HG has received research support from the Swiss National Science Foundation, advisory board honoraria from Daiichi Sankyo and funding for travel from BMS/Pfizer.
AZ received speaker honoraria from CSL Behring, Boehringer-Ingelheim, Alexion-Astra Zeneca and Daiichi Sankyo and advisory board honoraria from Bayer, Astra Zeneca and Daiichi Sankyo.
DRJ received speaker honoraria from Medtronic and Boehringer Ingelheim.
VP received speaker honoraria from Medtronic and Boehringer Ingelheim.
H.J.A. reports receiving personal fees from Astra Zeneca, Boehringer Ingelheim, Novo Nordisk, and Roche that all produce products for hyperacute stroke care.
MRH reports grants from SITEM Research Support Funds and Swiss National Science Foundation, Swiss Heart Foundation, not directly related to this manuscript.
AS received travel grants from NovoNordisk
PM received grants from the Swiss National Science Foundation, the Swiss Heart Foundation, and Faculty of Biology and Medicine of the Lausanne University
CHN reports receiving speaker honoraria from Abbot, Alexion, AstraZeneca, Bristol-Meyers Squibb, Pfizer and Takeda, all outside the submitted work.
All further authors declare no conflicts of interest related to the presented study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Each center has received necessary official approval from their respective local authorities and/or ethical committees according to national and local rules.
Informed consent
Informed consent was waived due to the retrospective design of the study.
Guarantor
CR accepts official responsibility for the overall integrity of the manuscript (including ethics, data handling, reporting of results, and study conduct)
Contributorship
CR and CHN researched literature, conceived the study and were responsible for the central data clearance and management. CR was responsible for statistical analysis. CR and CHN wrote the first draft of the manuscript. RvR, KB, ES, GMM, GK, PM, MRH, MM, AP, AS, PM, CG, SW, NMM, SC, MLD, GB, NW, PJN, DRJ, VP, IM, RRL, GB, CWC, RP, MZ, MMV, AZ, JNR, JPM, HJA, ST, HG, STE were responsible for the acquisition of data in their respective centers and critically reviewed the manuscript for important intellectual content. All authors approved the final version of the manuscript.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
