Abstract
Background and Purpose:
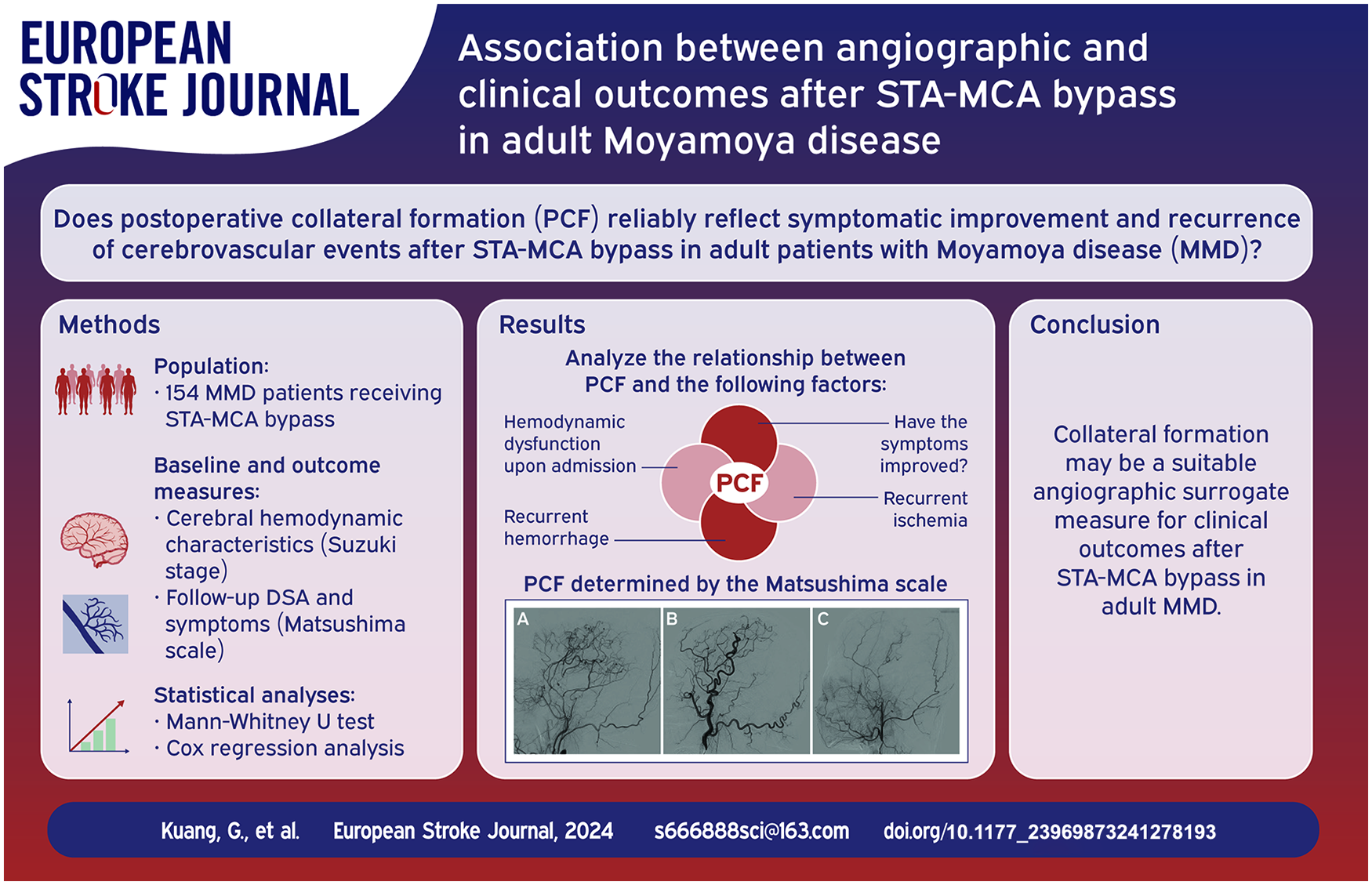
As an angiographic outcome, postoperative collateral formation (PCF) is commonly used to evaluate the effect of STA-MCA bypass in moyamoya disease (MMD), but whether it can reliably reflect clinical outcomes is still unclear. We investigated the association between PCF and clinical outcomes in adult MMD.
Methods:
All STA-MCA bypass procedures performed from January 2013 to December 2019 were screened in this prospective cohort study. Patients who acquired presurgical and follow-up catheter angiography were included. The clinical outcomes consisted of symptom improvement and recurrent cerebrovascular events. Logistic and Cox regression and Kaplan–Meier analyses were performed to explore the association between PCF and clinical outcomes.
Results:
Of 165 included symptomatic hemispheres of 154 patients, 104 (63.0%) and 61 (37.0%) had good and poor PCF, respectively. The hemispheres with good PCF were younger (p = 0.004) and had a higher incidence of hemodynamic dysfunction on admission (p < 0.001) than those with poor PCF. Multivariate logistic regression analysis showed that the good PCF (odd ratio, 28.96; 95% confidence interval (CI), 9.12–91.98; p < 0.001) was associated with a higher incidence of symptom improvement. Multivariate Cox regression analysis showed that the poor PCF (hazard ratio, 3.77; 95% CI, 1.31–10.84; p = 0.014) was associated with a higher incidence of recurrent cerebrovascular events. In the hemorrhagic-onset hemispheres, good PCF group had a higher incidence of symptom improvement (p < 0.001) and a longer hemorrhage-free time (p = 0.031). In the ischemic-onset hemispheres, good PCF group also had a higher incidence of symptom improvement (p < 0.001) and a longer ischemia-free time (p = 0.028).
Conclusions:
As a angiographic outcome, collateral formation is a qualified surrogate measure for clinical outcomes after STA-MCA bypass in adult MMD.
Keywords
Introduction
Intracranial hemorrhage and cerebral ischemia are the main clinical symptoms of moyamoya disease (MMD), which seriously affect the quality of life and even endanger the life of patients.1–3 The superficial temporal artery (STA)–middle cerebral artery (MCA) bypass is a standard-of-care treatment for adult MMD and has been shown to prevent the recurrence of bleeding and ischemia.1,3–5 In previous studies, postoperative collateral formation (PCF) (assessed by Matsushima scale) as an angiographic outcome has generally been used to reflect the effect of STA-MCA bypass.6–8 Compared with clinical outcomes, PCF data are easier and less time-consuming to obtain; however, to date, it is unclear whether PCF is a qualified surrogate measure for clinical outcomes. Therefore, the aim of this study was to investigate the association between collateral formation and clinical outcomes after STA-MCA bypass in adult patients with MMD. Since the ideal treatment should be to effectively improve symptoms and prevent recurrence of stroke, 9 the clinical outcomes in this study were set as improvement of symptoms and recurrence of cerebrovascular events.
Methods
Data availability disclosure
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Case selection and clinical data collection
This is a single-center prospective cohort study. A consecutive series of adult MMD patients who underwent STA-MCA bypass at West China Hospital, Sichuan University from January 2013 to December 2019 were screened. Diagnosis of MMD by digital subtraction angiography according to the Japanese 2012 guidelines. 1 Exclusion criteria were: (1) Patients with asymptomatic MMD; (2) Patients with indistinguishable hemorrhagic hemispheres, such as extensive subarachnoid hemorrhage and bilateral intraventricular hemorrhage; (3) Patients with severe postoperative complications, such as cerebral hemorrhage and cerebral infarction; (4) Patients with other conditions that interfered with the study results, such as previous severe brain trauma, central nervous system tumors, previous craniotomy, etc.; (5) Patients who were lost to follow-up.
Demographic and history data such as age, gender, history of risk factors (hypertension, diabetes mellitus, hyperlipidemia, tobacco/alcohol abuse), onset type of MMD (hemorrhagic and ischemic onset), modified Rankin Scale (mRS) score on admission and discharge were collected. Two neurosurgeons worked independently to collect and review the data, and only findings recognized by consensus were considered for analysis. Informed consents were obtained from all the participants or their agents, and the protocol study was approved by the Institutional Review Board and Ethics Committee of West China Hospital, Sichuan University.
Collection and assessment of imaging data
The cerebrovascular morphological characteristics of MMD on admission were assessed by digital subtraction angiography. Based on Suzuki and Takaku stage, 10 each hemisphere included in this study was graded into 6 stages according to the stage. The anterior choroidal artery dilation was defined as this dilated artery with distal branching or abnormal branches. 6 The posterior cerebral artery involvement was defined as the presence of >50% occlusion or stenosis in the P1–P3 segments of this artery with reduced delineation of cortical arteries. 11 The cerebral hemodynamic characteristics of MMD on admission were evaluated by CT perfusion. The decreased regional cerebral blood flow and prolonged mean transit time suggests cerebral hemodynamic dysfunction. 2 The regions of interest were set in the cerebral cortex and basal ganglia regions and ischemic areas are visually identified in the pseudo-color images of CT perfusion. These images were independently interpreted by two trained neuroradiologists who were blinded to other results, and only findings recognized by consensus were considered for analysis.
Collection and assessment of follow-up data
In our center, the patient readmission was scheduled 1 year after STA-MCA bypass surgery, and the follow-up DSA and symptom data were obtained during this hospitalization. Based on the lateral view of DSA images, the PCF was determined according to the Matsushima et al. scale: Level A, more than 2/3 of the MCA distribution; Level B, between two-thirds and one-third of the MCA distribution; and Level C, slight or none (Figure 1). 12 As previously reported, Level A and B were set as “Good” PCF and Level C was set as “Poor” PCF.6,8 Follow-up DSA data were independently interpreted by two other neuroradiologists who were blinded to other results, and only findings recognized by consensus were considered for analysis. Follow-up symptom data were obtained to determine whether there was improvement compared with presurgical symptoms. Judgment of symptom improvement was performed by two independent neurosurgeons who were blinded to other information about these patients. Any disagreements were resolved by consensus.

Postoperative collateral formation was determined by the Matsushima et al. scale. 12 (a) Level A: more than 2/3 of the MCA distribution. (b) Level B: between two-thirds and one-third of the MCA distribution. (c) Level C: slight or none. MCA, middle cerebral artery.
Recurrent cerebrovascular events were the primary follow-up event, including recurrent intracranial hemorrhagic and cerebral ischemic events (cerebral infarction and transient ischemic attack). Follow-up cerebrovascular events were obtained by telephone interviews with these patients or their proxies annually after the STA-MCA bypass. The interview was performed by neurosurgeons in training who were blinded to other results. For the patients with suspected recurrence of vascular events, we encouraged them to come to our center for further diagnosis to clarify their condition.
Statistical analysis
Percentages and median values with interquartile ranges were used for summary statistics. We compared the baseline characteristics of hemispheres with good and poor PCF using the Chi-square test for categorical variables and Mann-Whitney U test for continuous variables. The associations between PCF and symptom improvement were assessed by univariate and multivariable logistic regression models. The associations between PCF and recurrent cerebrovascular events were assessed by univariate and multivariable cox regression models. We had tested for proportionality of the cox model and the time-dependent variable was not significant in the model (p = 0.186), which confirmed the validity of proportionality assumptions in the model. The hemorrhage and ischemia-free time from the recurrent cerebrovascular events was estimated using Kaplan-Meier curves with log-rank statistics. Odd ratios (ORs), hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated and a p value < 0.05 was considered statistically significant. We used SPSS (version 23.0) and R (version 4.1.1) statistical software.
Results
Clinical and imaging characteristics
From January 2013 to December 2019, 165 symptomatic hemispheres of 154 patients (age range 19–68 years) were included in this study. Figure 2 depicts the flow chart. Table 1 compares the clinical and imaging characteristics between the good PCF groups (n = 104) and poor PCF groups (n = 61). The hemispheres with good PCF were significantly younger (p = 0.004) and had a higher incidence of hemodynamic dysfunction on admission (p < 0.001) than those with poor PCF.

Flow diagram for study inclusion.
Clinical and imaging data obtained in the 165 MMD Hemispheres.
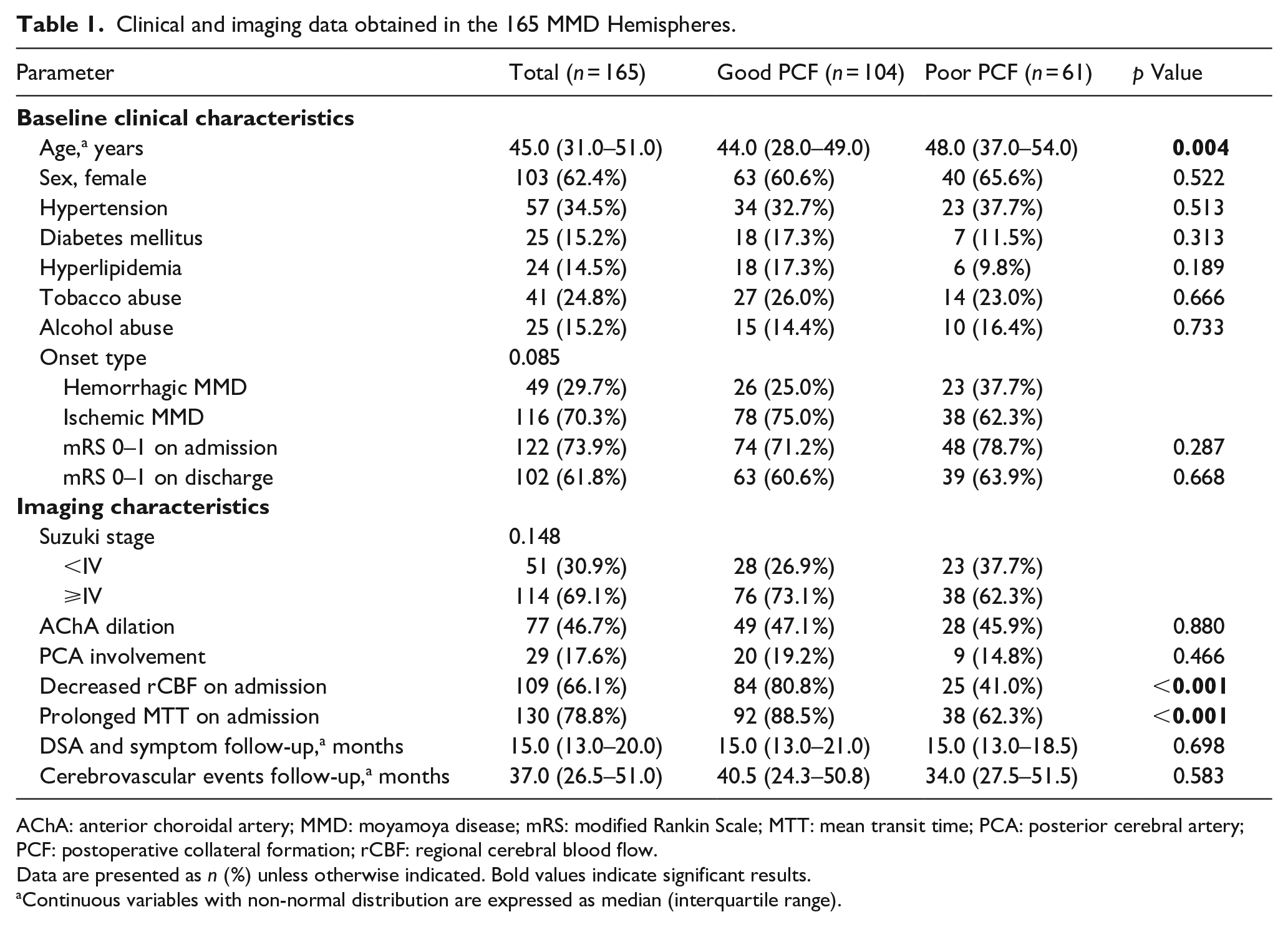
AChA: anterior choroidal artery; MMD: moyamoya disease; mRS: modified Rankin Scale; MTT: mean transit time; PCA: posterior cerebral artery; PCF: postoperative collateral formation; rCBF: regional cerebral blood flow.
Data are presented as n (%) unless otherwise indicated. Bold values indicate significant results.
Continuous variables with non-normal distribution are expressed as median (interquartile range).
Improvement of symptoms
One year after STA-MCA bypass surgery, 129 (78.2%) of 165 hemispheres had symptomatic improvement and 36 (21.8%) had no improvement. Univariate logistic regression analysis showed that the good PCF (odd ratio (OR), 20.46; 95% confidence interval (CI), 7.31–57.26; p < 0.001) was associated with a higher incidence of symptom improvement, and no significant associations were found for other factors (Table 2). Multivariate logistic regression analysis also showed that the good PCF (OR, 28.96; 95% CI, 9.12–91.98; p < 0.001) was associated with a higher incidence of symptom improvement (Table 2).
Logistic regression analysis exploring the association between postoperative collateral formation and symptom improvement.
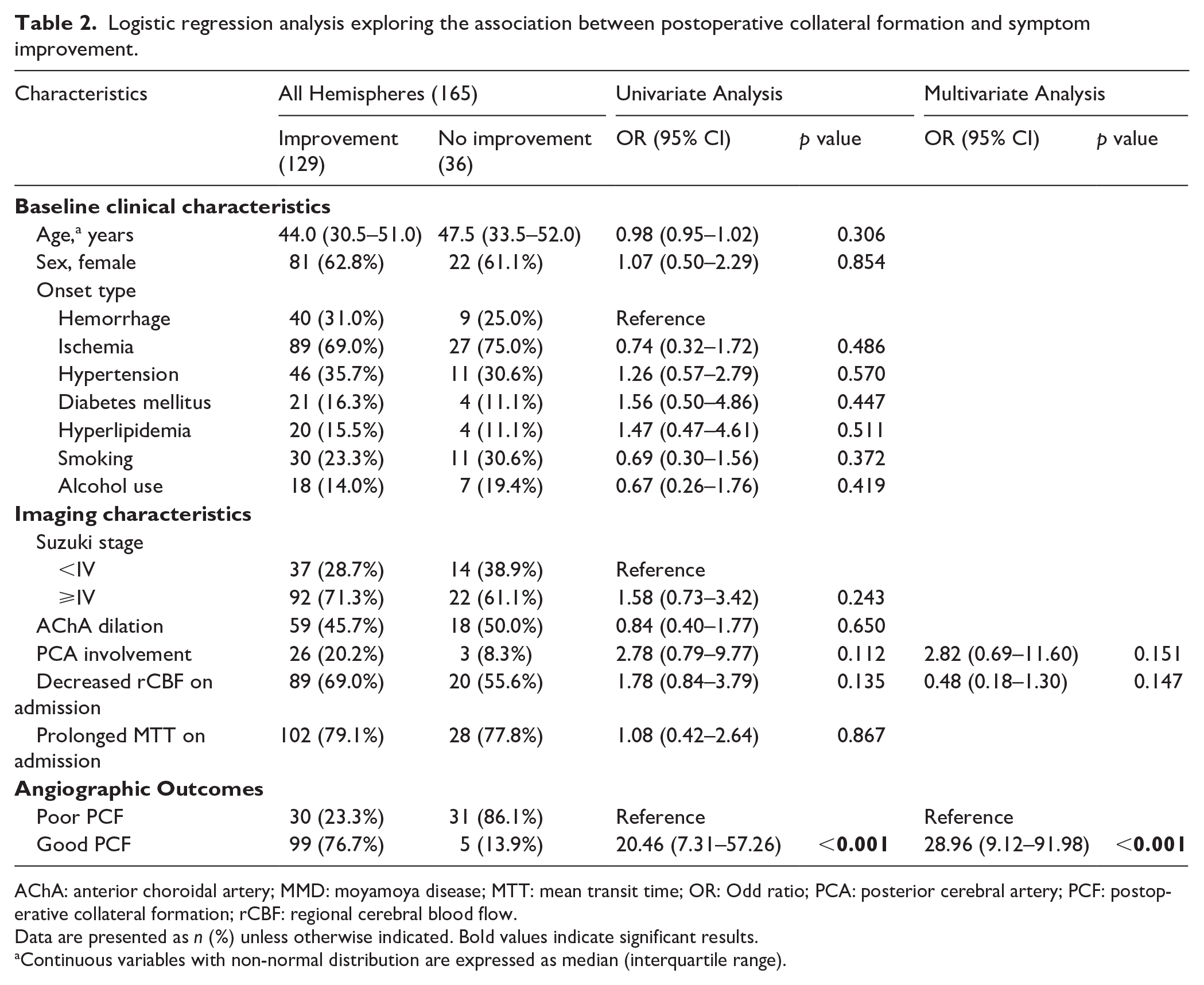
AChA: anterior choroidal artery; MMD: moyamoya disease; MTT: mean transit time; OR: Odd ratio; PCA: posterior cerebral artery; PCF: postoperative collateral formation; rCBF: regional cerebral blood flow.
Data are presented as n (%) unless otherwise indicated. Bold values indicate significant results.
Continuous variables with non-normal distribution are expressed as median (interquartile range).
In the hemorrhagic-onset cohort, 40 (81.6%) of 49 hemispheres had symptomatic improvement and 9 (18.4%) had no improvement. All 26 (100.0%) hemispheres with good PCF had symptomatic improvement, but only 14 (60.9%) of 23 hemispheres with poor PCF had (p < 0.001). In the ischemic-onset cohort, 89 (76.7%) of 116 hemispheres had symptomatic improvement and 27 (23.3%) had no improvement. 73 (93.6%) of 78 hemispheres with good PCF had symptomatic improvement, but only 16 (42.1%) of 38 hemispheres with poor PCF had (p < 0.001).
Recurrence of cerebrovascular events
During the 37-month median follow-up, recurrence cerebrovascular events were observed in 17 (10.3%) hemispheres, including 5 (3.0%) hemispheres with hemorrhage, 12 (7.3%) hemispheres with ischemia (transient ischemic attack in 9 hemispheres and infarction in 3 hemispheres). Among all 165 patients, Kaplan-Meier analysis showed that the hemorrhage and ischemia-free time was significantly longer in hemispheres with good PCF than in those with poor PCF (p = 0.013) (Figure 3(a)). This was also demonstrated by multivariate Cox regression analysis (hazard ratio, 3.77; 95% CI, 1.31–10.84; p = 0.014), and the other factors were not significant (Table 3).
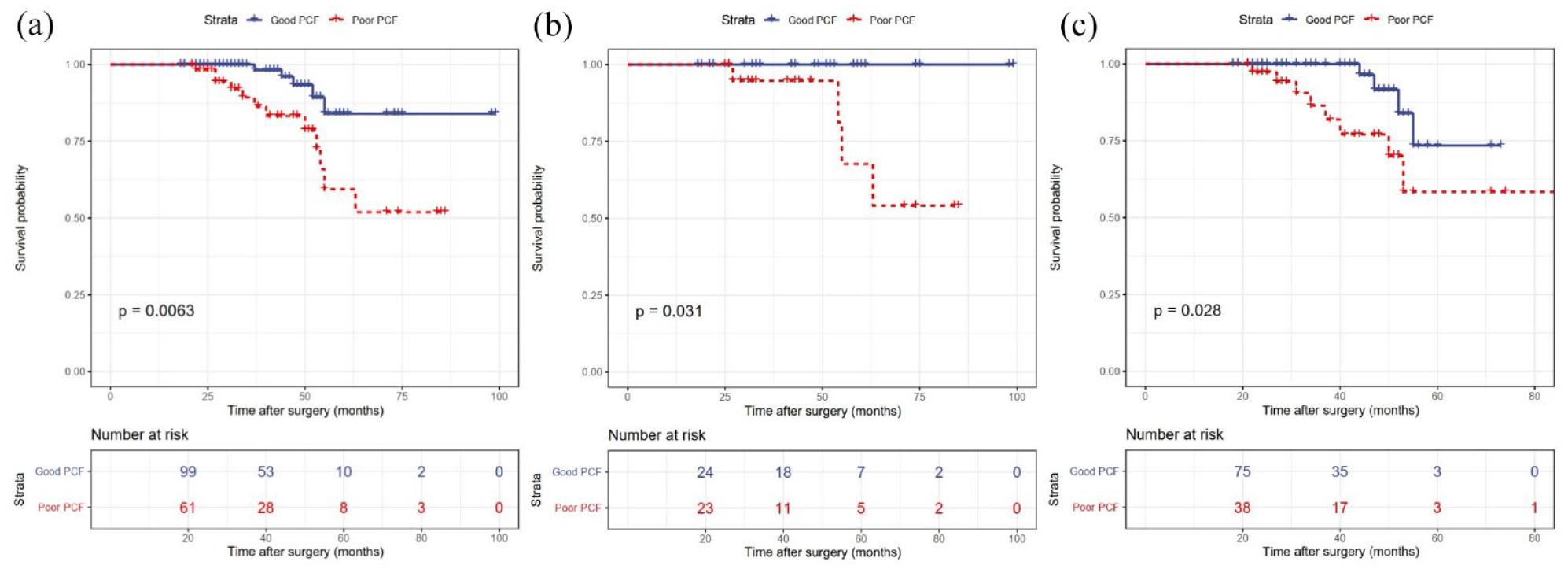
Kaplan-Meier plot for hemorrhage and ischemia-free survival after STA-MCA bypass. (a) Hemorrhage and ischemia-free time for all hemispheres for different PCF. (b) Hemorrhage-free time for the hemorrhagic-onset hemispheres for different PCF. (c) Ischemia-free time for the ischemic-onset hemispheres for different PCF. MCA, middle cerebral artery; PCF, postoperative collateral formation; STA, superficial temporal artery.
Cox regression analysis exploring the association between postoperative collateral formation and recurrent cerebrovascular events.

AChA: anterior choroidal artery; HR: hazard ratio; MMD: moyamoya disease; MTT: mean transit time; PCA: posterior cerebral artery; PCF: postoperative collateral formation; rCBF: regional cerebral blood flow.
Data are presented as n (%) unless otherwise indicated. Bold values indicate significant results.
Continuous variables with non-normal distribution are expressed as median (interquartile range).
In the hemorrhagic-onset cohort, recurrent hemorrhage occurred in 4 (8.2%) hemispheres and ischemia occurred in none. None of the hemispheres with good PCF experienced rebleeding, while 4 (17.4%) of 23 hemispheres with poor PCF did (p = 0.042). Kaplan-Meier analysis showed that the hemorrhage-free time was significantly longer in hemispheres with good PCF than in those with poor PCF (p = 0.031; Figure 3(b)).
In the ischemic-onset cohort, recurrent ischemia occurred in 12 (10.3%) hemispheres and hemorrhage occurred in 1 (0.9%). 4 (5.1%) of 78 hemispheres with good PCF experienced recurrent ischemia, while 8 (21.1%) of 38 hemispheres with poor PCF did (p = 0.018). Kaplan-Meier analysis also showed that the ischemia-free time was significantly longer in hemispheres with good PCF than in those with poor PCF (p = 0.028; Figure 3(c)).
Discussion
The Matsushima et al. scale was originally developed for the pediatric population with MMD after revascularization procedures. 12 For ease of use in clinical practice, the Matsushima scale is often designed as a dichotomous scale (i.e. Level A and B are set as “Good” collateral formation and Level C is set as “Poor” collateral formation) to evaluate the angiographic outcome of the entire MMD population, whether after direct or indirect bypass.6–8,13,14 Ge et al. 6 studied 188 MMD patients who had undergone direct and combined bypass surgery. After an 18-month median follow-up, 125 (63.2%) hemispheres had good PCF and 85 (36.8%) had poor PCF. 6 Their team also analyzed 64 hemorrhagic MMD patients who had undergone indirect bypass surgery. 8 After a median 8.5 months DSA follow-up, 27 (42.2%) hemispheres had good PCF and 37 (57.8%) had poor PCF. 8 Zhao et al. 13 retrospectively included 209 MMD patients who had undergone indirect bypass surgery for analysis. After a mean 9 months DSA follow-up, 146 (63.2%) hemispheres had good PCF and 85 (36.8%) had poor PCF. 13
The STA–MCA bypass is a standard-of-care treatment for adult MMD and PCF data can be obtained by DSA at 6–12 months after the bypass.8,13 Therefore, this angiographic outcome is often used to evaluate the effect of the bypass.2,6,15 However, until now, it has not been clear whether the postoperative angiographic outcome is really a reliable reflection of clinical outcomes. This study was the first to investigate the association between collateral formation and clinical outcomes after STA-MCA bypass in adult patients with MMD. The results showed that the good PCF was associated with a higher incidence of symptom improvement and the poor PCF was associated with a higher incidence of recurrent cerebrovascular events. In subgroup analysis, due to the limited sample size, only inter-group comparison and Kaplan-Meier analysis were performed. In the hemorrhagic-onset hemispheres, good PCF group had a higher incidence of symptom improvement and a longer hemorrhage-free time. In the ischemic-onset hemispheres, good PCF group also had a higher incidence of symptom improvement and a longer ischemia-free time. Therefore, PCF can replace clinical outcomes as a more manageable outcome measure after STA-MCA bypass in adult MMD. In addition to DSA, there are currently studies using less invasive MR angiography and MR fusion16,17 or even non-invasive bidirectional ultrasound6,7 for research, And some progress has been made for example, studies have evaluated the postoperative efficacy of Moyamoya disease through MRI combined with dual function ultrasound. 17 Whether these non-invasive evaluation methods are truly related to long-term clinical prognosis remains to be studied. This is also a direction worthy of attention and in-depth exploration in the future.
In this study, 104 (63.0%) and 61 (37.0%) hemispheres had good and poor PCF, respectively. The hemispheres with good PCF were younger and had a higher incidence of hemodynamic dysfunction on admission than those with poor PCF. Ge et al. 6 studied 188 MMD patients who had undergone direct and combined bypass and 85 (36.8%) hemispheres had poor PCF. They found that poor PCF was related to older age at operation, presence of hemorrhage and dilated anterior choroidal artery. In summary, PCF is not always ideal, and poor PCF occurs in about one-third of the hemispheres. In addition, severe postoperative complications, including postoperative ischemic/hemorrhagic stroke and cerebral hyperperfusion syndrome, can increase patients’ pain and economic burden.16,18–21 Therefore, presurgical prediction of PCF is particularly important, and the prediction model needs to be developed in future study.
This study had a few limitations. First, the study was conducted in a single center and there may be selection bias. Although the clinical and pathological manifestations of patients with Moyamoya disease are similar among different ethnicities,22,23 large-scale multicenter clinical studies across ethnicities can still be conducted to further clarify. Second, patients with asymptomatic moyamoya disease were not included in this study due to uncertain surgical indications and inability to determine symptom improvement. Third, the incidence of recurrent cerebrovascular events in MMD patients after STA-MCA bypass is low, so it is necessary to further increase the sample size and extend the follow-up time in future studies, especially for some subgroup analysis.
Conclusions
As an angiographic outcome, collateral formation can reliably reflect the improvement of symptom and recurrent of cerebrovascular events after STA-MCA bypass in adult patients with MMD. These findings provide a clinical evidence for the angiographic outcome to replace the clinical outcomes as a more manageable outcome measure.
Glossary
CI = confidence interval; HR = hazard ratio; MCA = middle cerebral artery; MMD = moyamoya disease; mRS = modified Rankin Scale; OR = odd ratio; PCF = postoperative collateral formation; STA = superficial temporal artery.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241278193 – Supplemental material for Association between angiographic and clinical outcomes after STA-MCA bypass in adult moyamoya disease
Supplemental material, sj-docx-1-eso-10.1177_23969873241278193 for Association between angiographic and clinical outcomes after STA-MCA bypass in adult moyamoya disease by Guicheng Kuang, Hang Ji, Jixuan Zheng, Xinchen Li, Kejin Luo, Yajun Hu, Zheyuan Zhang and Haogeng Sun in European Stroke Journal
Footnotes
Acknowledgements
We sincerely thank Rui Guo and Zhiyuan Yu for they have provided general advice and guidance on statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the National Natural Science Foundation of China (No. 82301480); the Sichuan Provincial Science and Technology Department (No. 24NSFSC1634); and the 1·3·5 project for disciplines of excellence–Clinical Research Fund, West China Hospital, Sichuan University (No. 2024HXFH024).
Informed consents and ethical approval
Informed consents were obtained from all the participants or their agents, and the protocol study was approved by the Institutional Review Board and Ethics Committee of West China Hospital, Sichuan University.
Guarantor
Haogeng Sun
Contributorship
Guicheng Kuang and Hang Ji researched literature and conceived the study. Jixuan Zheng, Xinchen Li and Kejin Luo was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. Guicheng Kuang and Haoge Sun wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
