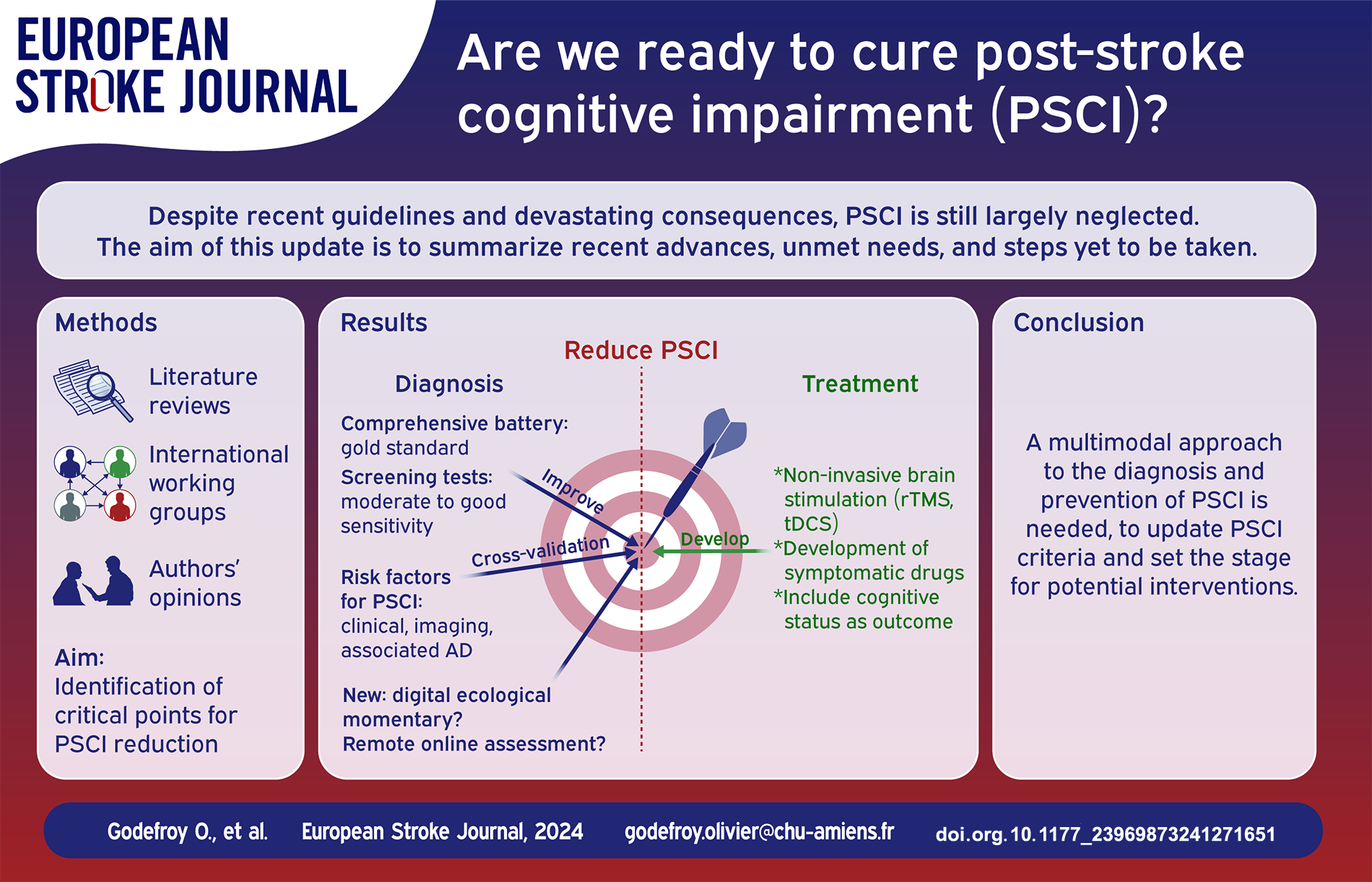
Abstract
Purpose:
Post-stroke (PS) cognitive impairment (CI) is frequent and its devastating functional and vital consequences are well known. Despite recent guidelines, they are still largely neglected. A large number of recent studies have re-examined the epidemiology, diagnosis, imaging determinants and management of PSCI. The aim of this update is to determine whether these new data answer the questions that are essential to reducing PSCI, the unmet needs, and steps still to be taken.
Methods:
Literature review of stroke unit-era studies examining key steps in the management of PSCI: epidemiology and risk factors, diagnosis (cognitive profile and assessments), imaging determinants (quantitative measures, voxelwise localization, the disconnectome and associated Alzheimer’s disease [AD]) and treatment (secondary prevention, symptomatic drugs, rehabilitation and noninvasive brain stimulation) of PSCI.
Findings:
(1) the prevalence of PSCI of approximately 50% is probably underestimated; (2) the sensitivity of screening tests should be improved to detect mild PSCI; (3) comprehensive assessment is now well-defined and should include apathy; (4) easily available factors can identify patients at high risk of PSCI; (5) key imaging determinants are the location and volume of the lesion and the resulting disconnection, associated AD and brain atrophy; WMH, ePVS, microhemorrhages, hemosiderosis, and cortical microinfarcts may contribute to cognitive impairment but are more likely to be markers of brain vulnerability or associated AD that reduce PS recovery; (6) remote and online assessment is a promising approach for selected patients; (7) secondary stroke prevention has not been proven to prevent PSCI; (8) symptomatic drugs are ineffective in treating PSCI and apathy; (9) in addition to cognitive rehabilitation, the benefits of training platforms and computerized training are yet to be documented; (10) the results and the magnitude of improvement of noninvasive brain stimulation, while very promising, need to be substantiated by large, high-quality, sham-controlled RCTs.
Discussion and conclusion:
These major advances pave the way for the reduction of PSCI. They include (1) the development of more sensitive screening tests applicable to all patients and (2) online remote assessment; crossvalidation of (3) clinical and (4) imaging factors to (5) identify patients at risk, as well as (6) factors that prompt a search for associated AD; (7) the inclusion of cognitive outcome as a secondary endpoint in acute and secondary stroke prevention trials; and (8) the validation of the benefit of noninvasive brain stimulation through high-quality, randomized, sham-controlled trials. Many of these objectives can be rapidly and easily attained.
Keywords
Introduction
Post-stroke (PS) cognitive impairment (CI) is frequent and its consequences well known, with an increased risk of disability, institutionalization,1–3 stroke recurrence and death.4,5 International guidelines emphasize the importance of identifying patients with PSCI, referring them for appropriate neuropsychological evaluations and initiating early treatment.6–8 However, they are still largely overlooked. 9 A large number of recent studies, pooled data analyses and meta-analyses have re-examined the epidemiology, diagnosis, imaging and management of PSCI. In this update based on literature review, our aim is to determine whether these new data answer the indispensable questions needed to define management that will reduce PSCI and its consequences in the near future. This update summarizes the recent advances, unmet needs, and steps yet to be taken before these disorders can be reduced.
Methods: Selection of critical points for PSCI reduction
Since the ultimate goal is to provide effective treatment/management for PSCI patients, recent advances in this area were reviewed. To achieve effective treatment/management, it is first necessary to diagnose patients with PSCI, so it is necessary to report on recent advances in diagnosis, that is, neuropsychological assessment (including screening tests). Since it is impossible (and probably inappropriate) to assess the cognitive and behavioral status of all stroke patients with sufficient accuracy, it is appropriate to review strategies for identifying patients at risk (i.e. patients who should be referred for neuropsychological assessment): we have focused on the two strategies that have been previously explored on the basis of the clinical and imaging characteristics of these patients. Despite their interest, new and interesting aspects such as vascular dysfunction and inflammation were not addressed because they do not currently have practical implications for patient management. The data used were derived from recent literature reviews, ongoing works by the authors, their opinions and discussions in various international working groups.
After a brief epidemiological review highlighting the limitations of current estimates of the prevalence of PSCI, the study was organized around the following questions: (1) can we improve diagnosis both through the use of screening tests and with (2) comprehensive batteries, (3) can we identify patients at risk by both clinical (4) and imaging factors, analyzing features that account for the emergence of PSCI such as (5) post-stroke cavity characteristics, (6) white matter hyperintensities (WMH), (7) markers of small vessel pathology, (8) the contribution of Alzheimer’s disease (AD) and Cerebral amyloid angiopathy (CAA), and (9) structural and functional disconnection. Finally, we reviewed (10) more recent and promising approaches, including (11) remote cognitive assessment and (12) PSCI management, including (13) non-invasive stimulation.
Epidemiology: The prevalence of PSCI
PCSI has been assessed in numerous studies, most focusing on major CI (i.e. dementia), and reported varying prevalence10–12 (Table 1). A meta-analysis reported a PSCI prevalence of 53%, two-thirds corresponding to mild CI and one-third to dementia. 11 Heterogeneity across studies is due to several factors (Table 1) the most important of which is probably the applicability of cognitive assessment, as 4%–25% of patients are non-assessable and are at high risk of CI.13–15
Epidemiology of poststroke cognitive impairment (PSCI): Facts and knowledge taken for granted versus yet to be clarified-unmet needs.

WMH: white matter hyperintensities.
This indicates that the prevalence of PSCI of approximately 50% among initially hospitalized patients is probably underestimated.
The diagnosis of PSCI
The diagnosis of PSCI relies on the accuracy of cognitive tools, which have evolved. The aim of this section is to examine whether we can improve the diagnosis of PSCI.
Screening tests
The use of cognitive-screening tests (Table 2) is part of the clinical assessment at the poststroke visit and sometimes before discharge from the acute stroke unit. The MiniMental State Examination 40 and Montreal Cognitive Assessment (MoCA) 41 are the most frequently used but their sensitivity for PSCI is only moderate.21,22 A meta-analysis 22 suggests that acute testing yields higher sensitivity (and lower specificity), although this is still too low to use it as a substitute for comprehensive assessment. Other screening tests are being developed (e.g.,42,43) but their superiority over existing tools still requires validation.
Diagnosis of poststroke cognitive impairment: Facts and knowledge taken for granted versus yet to be clarified-unmet needs.

MMSE: MiniMental State Examination; MoCA: Montreal Cognitive Assessment; EAN: European Academy of Neurology; PSCI: post-stroke cognitive impairment; VCI: vascular cognitive impairment; WMH: white matter hyperintensities; PET: positron emission tomography.
This indicates that the sensitivity of screening tests should be improved to detect mild PSCI in routine practice, or that an alternative strategy should be developed to select at-risk patients.
Comprehensive assessment
The comprehensive assessment19,24,31,44 (Table 2) is the gold standard, appears to be very sensitive19,24,27,45,57,65 and is independently related to disability.3,46 Assessment should include apathy because it is observed in approximately one-third of patients27,51,52 and is independently associated with disability.3,27 The comprehensive battery needs to be supplemented in specific situations: certain executive tests need to be added for certain causes of stroke, 27 as well as language assessment in aphasics 66 and hemineglect assessment in certain right hemisphere stroke patients using recent recommendations 48 and a parsimonious combination of tests 50 (Table 1).
Given that it is impossible and unnecessary to carry out a comprehensive assessment of all stroke patients, and that the sensitivity of screening tests is moderate, we suggest developing alternative strategies based on the identification of at-risk patients.
Identification of patients at risk for PSCI on the basis of clinical factors
Although CI can be observed in all patients, clinical and gross imaging risk factors have been identified. CI is more frequent in cerebral hemorrhage and infarct11,24–26 than in ruptured aneurysm and cerebral venous thrombosis where the cerebral lesion is inconstant.27–30 Reviews have identified a long list (Table 1) of clinical factors10,24,45 and gross imaging markers.6,10,35,67 Other poststroke adverse outcomes, such as fatigue 68 or obstructive sleep apnea 69 may also be associated with PSCI.
The predictive value of these factors has been examined in a few studies using initial data to determine the six-month risk. Models mainly based on vascular risk factors showed low predictive accuracy in crossvalidation studies. 39 The combination of five to six clinical factors (Table 1) can provide good to excellent sensitivity to predict six-month PSCI defined based on a comprehensive battery 35 or MoCA, 36 although crossvalidation is required.
This indicates that easily available factors can identify patients at high risk of PSCI, although crossvalidation studies on large cohorts are warranted.
Imaging determinants of PSCI and their use to identify patients at risk
In addition to the vascular lesions themselves, associated diseases such as Alzheimer’s disease may contribute to PSCI.1,70–72 In terms of imaging, several features are associated with CI (Table 3): WMH, microhemorrhages and hemosiderosis, microinfarcts, enlarged perivascular spaces (ePVS), and cerebral and hippocampal atrophy.10,25,32,56,57,73 Quantitative measures have recently refined these imaging features (e.g. lesion volume, voxelwise analysis of lesion, brain connectivity).
Imaging characteristics of white matter hyperintensities and markers of small vessel disease (adapted from Strive 2 74 ).

FLAIR: fluid-attenuated inversion recovery.
Imaging characteristics responsible for PSCI occurrence
Lesion location and volume
Voxelwise lesion symptom mapping analysis showed the prominent role of left frontotemporal, thalamus, and right parietal lesions. 31 The “strategic stroke” classically encompasses a large number of locations 45 that have been variably defined. Five strategic locations were identified using a strict definition, involving the left middle frontal gyrus, the temporoparietal junction, antero-middle thalamus, and both pyramidal tracts. 32 The contribution of cerebellar lesions, especially in the crus VIII, warrants further studies.31–33
This indicates that the location of lesions within the hemisphere should be used to identify patients at risk. A risk score based on lesion location 31 has been developed and requires crossvalidation.
White matter hyperintensity and features of small vessel disease: Simple markers or key-players?
White matter hyperintensity
Confluent WMH promote the emergence of cognitive decline when a focal ischemic or hemorrhagic lesion has occurred24,78–80 (Table 3). However, the underlying mechanisms remain unclear. WMH may reduce the brain resources needed for efficient recovery and promote persistent cognitive manifestations associated with a sudden ischemic or hemorrhagic lesion.81,82 Furthermore, they may themselves contribute to a greater cognitive deficit. When extensive, they are associated with psychomotor slowing. 83 Finally, WMH may increase the risk of recurrent stroke that will finally result in a larger number of lesions, with subsequent cognitive decline.83,84 In clinical practice, the degree of CI specifically associated with WMH in stroke patients remains difficult to estimate. Accumulating data in the literature suggest that the impact of WMH may largely vary depending on (1) their extent within the cerebral connectivity network, 85 (2) their exact location with a larger impact of lesions affecting anterior thalamic radiations or the forceps minor, 86 (3) the severity of the underlying tissue lesions, varying from simple water accumulation to severe neuronal and axonal loss,87–89 (4) the age and clinical status prior the stroke event, with protective factors, such as cognitive reserve, 90 and (5) the previous cerebral status, with the presence or absence of lacunes, neurodegenerative lesions, and cerebral atrophy. 91 Overall, these multiple factors explain why the exact impact of WMH is ultimately highly variable after stroke at the individual level, despite their generally pejorative predictive value.
Markers of small-vessel disease
Cerebral microbleeds, hemosiderosis, cortical microinfarcts, and ePVS constitute MRI manifestations of small-vessel disease (Table 3). There is an expanding literature on microbleeds, ePVS, and microinfarcts as predictors of PSCI.56,92–94 Some studies found a modest association between these MRI markers and the occurrence of PSCI, whereas others did not (systematic review 92 ), likely reflecting small effect sizes. Indeed, a recent study that included a head to head comparison of these various imaging markers reported much smaller effect sizes for microbleeds and ePVS in relation to PSCI than for WMH. 56
Overall, the available data suggest that the observed associations with PSCI do not so much reflect a causative effect of these tiny lesions themselves on cognitive performance, but rather their presence indicates much more widespread microvascular disease in the brain, with a global impact on brain tissue and its resilience, thus predisposing affected individuals to a poor cognitive outcome in the event of a stroke.
The synergistic contribution of multiple imaging features
The imaging features that contribute to the assessment of cognitive performance are usually examined separately, whereas they contribute together. The few studies that have used statistical modeling of multiple imaging features suggest a dominant role for lesion location, followed by stroke volume, hippocampal and cerebral atrophy, and WMH volume, whereas the contribution of ePVS and microhemorrhages appears to be minor.32,95–98
Pending crossvalidation studies, this indicates that, in addition to lesion location, multiple imaging features (including stroke volume, hippocampal and cerebral atrophy, and WMH volume), should be used to refine the identification of at-risk patients.
The contribution of Alzheimer disease to PSCI
Although PSCI is generally attributed solely to the vascular lesions, pioneering studies suggested that a third of PS dementia is due to associated AD.1,70–72 The contribution of AD has been refined following amyloid positron emission tomography (PET) studies. First, PET studies refuted the promotion of amyloid deposition by stroke lesions.99–101 Second, they showed a prevalence of amyloid positivity of approximately 15%–20% depending on the age and frequency of CI in the sample.99,100,102–104 Third, the largest studies showed that the amyloid burden is associated with a more severe cognitive status at baseline99,102 and a high risk of developing severe CI at follow-up.102,105,106 Accordingly, PET positivity was found in 30% to 38%99,104 of PS dementia cases, thus supporting the pioneering studies.1,70,72 This converges with the additive effect of vascular and neurodegenerative lesions on cognitive outcome.107–109 Fourth, a high burden of posterior WMH, cortical microbleeds, centrum semiovale ePVS, and hippocampal atrophy has been shown to be suggestive of amyloïdopathy, 99 although this requires replication. Fifth, the use of cognitive profiles (based on the executive/memory contrast) to orient the cause of PSCI (i.e. pure vascular vs CI associated with AD) has been called into question by the finding of overlapping cognitive profiles in the two diseases47,99 due to a high rate of dysexecutive disorders in AD 110 and the predominance of the encoding-storage (also known as hippocampal) profile of vascular memory deficit. 65 Thus, only severe memory deficits (relative to executive disorders) that are not explained by stroke location should be considered an indication of associated AD. In addition to documenting the highly deleterious effect of associated AD, these data offer imaging possibilities for identifying associated AD in PSCI patients.
CAA can also be positive on amyloid PET111,112 and is a factor associated with cognitive impairment independent of associated AD. 113 Accordingly, the presence of MRI markers of CAA is associated with an increased risk of CI. 25 This suggests that CAA may represent a therapeutic target: a phase 2 trial targeting Aβ1-40, the major Aβ species deposited in the arterial wall in CAA, did not show the expected result. 114
Pending more precise predictors, this indicates that association with AD should be sought, given its frequency and major prognostic impact, in at-risk patients which include age, severe memory deficits (vs executive disorders) not explained by stroke location, high posterior WMH load, cortical microbleeds, centrum semiovale ePVS, and hippocampal atrophy. Diagnostic approach (annual cognitive and imaging follow-up, CSF biomarkers, or amyloid PET) depends on patient status, the evolving availability of amyloid PET, the development of AD-modifying drugs, and the demonstration of their benefit in patients with mild to moderate stroke.
Is disconnection the key player that determines the occurrence of PSCI?
Current predictions for PSCI based on lesion location are satisfying, although limited by the accuracy of the current model of brain function. 115 Recent evidence suggests that brain function relies more on connections within the brain than on the local contribution of regions to functions.116–119 This can be explained by the importance of the interaction between brain regions in the achievement of brain functions, as well rapid plasticity, allowing for other connected regions to take over the additional workload.120,121 In the context of brain damage, the contribution of disconnections to behavior has frequently shown higher statistical and explanatory power for the symptoms observed in patients.95,97,122–127 Hence, disconnection could be a key player in determining the occurrence of PSCI. 128 Despite the improvement provided by disconnexion, the amount of variance explained by these models is still limited.95,123
To sum up, these data suggest that the key players are the location and volume of the lesion and the resulting disconnection, associated AD and brain atrophy. WMH, ePVS, microhemorrhages, hemosiderosis, and cortical microinfarcts may contribute to cognitive impairment but are more likely to be markers of brain vulnerability or associated AD that reduce PS recovery. Further studies are warranted to accurately determine the minimal lesion load (or disconnection) that induces PSCI and refine the modeling of imaging determinants of PSCI. Thus, additional improvement is needed before using such modeling to accurately predict patients at risk of PSCI.
Recent and promising approaches
Improving the diagnosis: Remote online cognitive assessment
Pending the development of more sensitive screening tests, comprehensive cognitive assessment is needed for patients at risk of PSCI based on clinical and imaging risk factors8,83 (see above). The recent development of remote and online assessment may offer such an opportunity for selected patients. 23
Tools such as the Telephone Interview for Cognitive Status and telephone short MoCA 41 were the first available methods for remotely measuring cognitive performance in stroke patients. 129 Recent results, obtained on a small stroke sample, showed that a self-administered tablet-based neurocognitive platform is widely acceptable and has good convergent validity. 130 The organization of a remotely accessible detailed cognitive assessment on an internet platform that can be controlled under professional supervision also appears promising. The feasibility of this procedure has already been established using a flexible, integrated system. 131 Other organizational (links with professionals) and practical factors (financing of equipment, cost) must also be taken into consideration. The development of digital ecological momentary assessment presents a promising approach to monitor at-risk patients outside the hospital environment and provide access to digital therapeutics. 132 This approach needs to be validated on a large control population before standards can be established for their clinical use. Finally, remote cognitive assessment will necessarily remain limited to patients who have the necessary computer skills, are free of major deficit and who can access a high-performance internet communication network. Professional supervision will remain crucial to ensure reproducible and high-quality data. Several studies are currently underway to document these critical points.
Another advantage of online remote assessment is that it facilitates continuous assessment. PSCI is a dynamic process that can be exacerbated by mood or sleep disturbances and external factors such as social support and environment. Therefore, continuous follow-up is needed to capture potential changes in cognition and to assess the impact of these symptoms on daily life, which is not addressed in the guidelines. The current development of digital ecological momentary assessment is a promising approach to monitor patients outside the hospital setting and provide access to digital therapeutics.
Finally, such an online assessment can feed a collaborative digital platform that offers the opportunity to optimize collaboration between physicians, including neurologists, gerontologists, and primary care physicians, speech-language pathologists, occupational therapists, neuropsychologists, nurses, and allied health professionals for optimal identification and management of cognitive problems after stroke. This indicates that remote and online assessment is a promising approach for selected patients, and their value should be clarified by ongoing studies.
Management of PSCI
Interventions (Table 4) for secondary stroke prevention including vascular risk factors (hypertension, smoking, diabetes, hyperlipidemia, obesity, obstructive sleep apnea and physical activity) are mandatory but their contribution to prevent CI remains to be proven.6,7,133 The cognitive benefit of lacunar infarction treatment demonstrated in a recent small phase 2 trial requires validation in further studies. 134
Classical and promising approaches in management and brain stimulation.
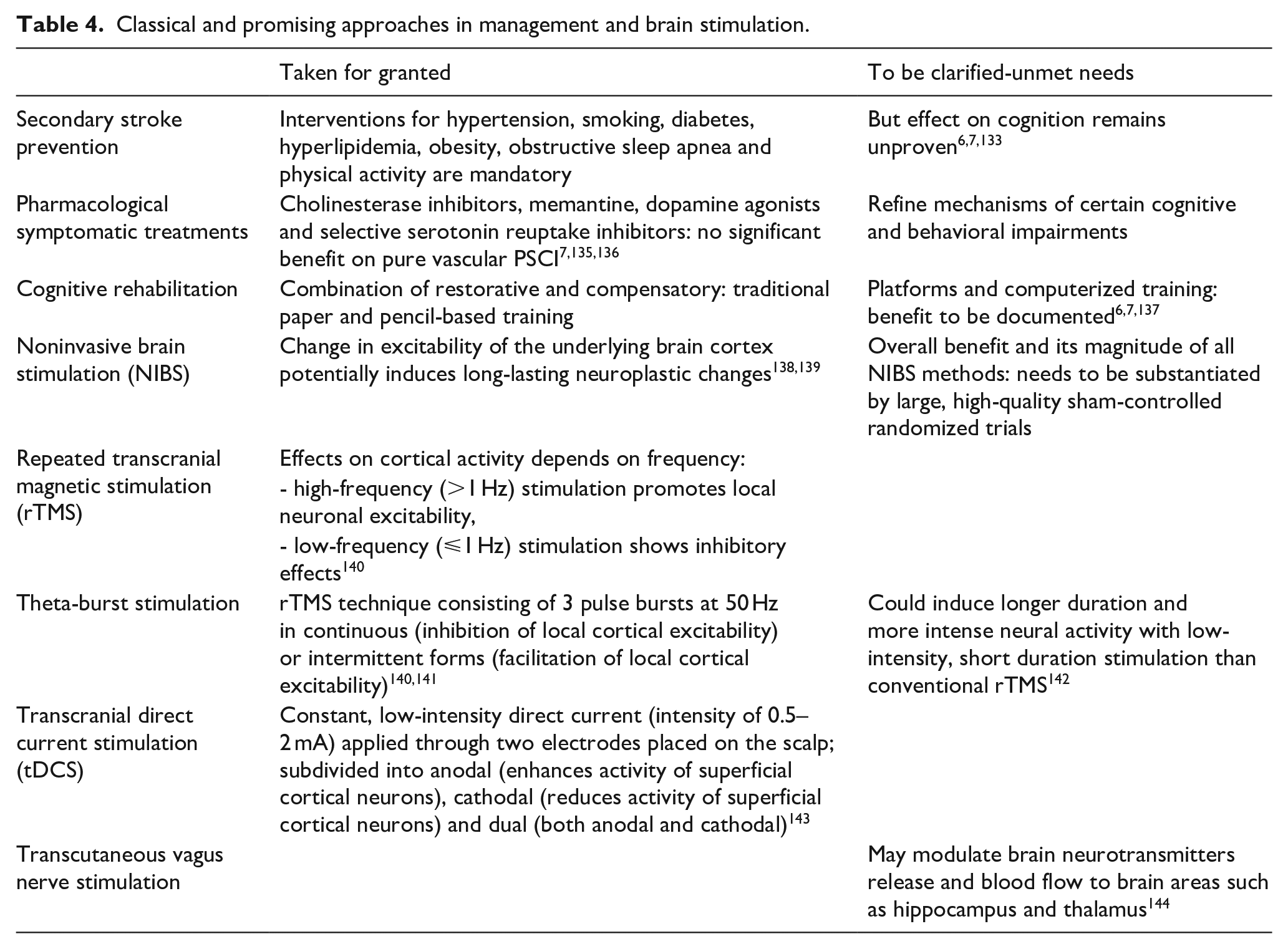
In addition, there are no standard pharmacological treatments for the treatment of PSCI as cholinesterase inhibitors, glutamate N-Methyl-D-aspartate receptor antagonist, dopamine agonists and selective serotonin reuptake inhibitors have failed to demonstrate a significant benefit on global cognitive function after stroke.7,135,136,145 The lack of efficacy of symptomatic drugs on PSCI (Table 4) may also be due to their multiple mechanisms, as recently suggested for two leading impairments in PSCI, psychomotor slowing45,54,57–62 and apathy51,52,63,64,146 (Table 1). To date, the management of PSCI mainly relies on cognitive rehabilitation, including a combination of restorative and compensatory approaches, both using traditional paper-and-pencil tasks. Recent approaches offer new opportunities to improve cognitive rehabilitation. The benefits of training platforms and computerized training are yet to be documented.6,7,137
Noninvasive brain stimulation
New hope has arisen from the promising results of noninvasive brain stimulation (Table 4), including repeated transcranial magnetic stimulation (rTMS), theta-burst stimulation (TBS), and transcranial direct current stimulation (tDCS). 140 These approaches assume that, under normal circumstances, the left and right hemispheres are in a balanced state of mutual inhibition. After a stroke, the lack of inhibitory effect of the damaged hemisphere on the undamaged hemisphere causes a relative increase in excitability of the intact hemisphere, ultimately resulting in an increase in inhibition of the damaged hemisphere. In addition noninvasive brain stimulation can induce changes in the excitability of the underlying cortex and potentially induce long-lasting neuroplastic changes by promoting neurogenesis, angiogenesis, anti-inflammatory, antioxidant, and anti-apoptosis effects. 138 Different stimulation frequencies have different effects on cortical activity, with high frequency (>1 Hz) stimulation (HF-rTMS) promoting local neuronal excitability and low frequency (⩽1 Hz) stimulation (LF-rTMS) showing inhibitory effects. 140
Given the small size of these studies, meta-analyses have been invaluable in assessing their interest. Concerning rTMS, systematic reviews and meta-analyses of randomized controlled trials (RCT) indicate a positive effect on cognitive outcome. 138 In a systematic review of 12 RCTs involving 497 patients with PSCI, Gong et al. 147 reported that rTMS had a positive effect on cognitive rehabilitation. Another study 148 pooling data from 8 studies and 336 participants found a large effect of rTMS combined with cognitive training on global cognition, executive function, and working memory, but no effect on memory. With regards to aphasia, a large number of studies converge on its benefits, 149 and it is now recommended in some countries. 150
TBS is a novel rTMS consisting of three pulse bursts at 50 Hz. Compared with conventional rTMS, TBS can induce longer and more intense neural activity with low-intensity, short-duration stimulation. Intermittent TBS (which plays a facilitating role in local cortical excitability 140 ) applied to left dorsolateral prefrontal cortex has been reported to improve executive function and semantic comprehension.151,152
Research findings on tDCS show contradictory effects153–155 with a slight improvement in general cognitive performance and attention provided by anodal tDCS. 155 In a meta-analysis 155 of 15 studies (N = 820 participants) of tDCS compared with sham tDCS or control, anodal tDCS was associated with small improvements in general cognitive and attention performance, but not in memory. However, most of these studies lacked sham tDCS and safety data.
A network meta-analysis 140 of RCTs comparing any active noninvasive brain stimulation with sham stimulation in stroke survivors showed that high-frequency rTMS improved global cognitive function while dual-tDCS improved memory performance.
Although promising, these results and the magnitude of improvement still need to be substantiated by large, high-quality, sham-controlled RCT. They should examine the influence of the timing of stimulation, treatment frequency and duration, and stimulation parameters, particularly the stimulation site. 156 Indeed, the location of the lesion and its effects on connectivity can help to select the most appropriate stimulation parameters to individualize the location of the stimulation.149,157,158 Finally, the potential benefit of combining noninvasive brain stimulation methods in the same patient should be examined, as recently suggested. 159
Conclusions
This update shows the major advances and provides guidance on the main issues to be resolved before reducing PSCI. This mainly includes (1) crossvalidation of factors that identify PS patients at risk of CI, (2) identification of the minimal vascular lesion that induces PSCI and (3) factors to prompt the search for associated AD; (4) development and validation of screening tests to improve the sensitivity and applicability to all patients, as well as (5) digital ecological momentary assessment and (6) cognitive assessment that are remotely administered online, and (7) used to monitor the course PSCI and (8) to provide access to digital therapeutics; (9) validation of the benefit of noninvasive brain stimulation by high-quality, individually-based, sham-controlled RCT; (10) systematic inclusion of cognitive outcome as a secondary endpoint in both acute and secondary prevention stroke trials, and (11) development of symptomatic drug trials targeting behavioral and cognitive disorders of pre-defined mechanisms. A significant number of these objectives can be easily and rapidly attained. Their findings may be incorporated into a revised version of the VASCOG criteria for vascular cognitive impairment. 160
Footnotes
Acknowledgements
Acknowledgments none related to this study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for this article because it is based on literature review.
Informed consent
Informed consent was not sought for this article because it is based on literature review.
Guarantor
OG
Contributorship
OG conceived the review and wrote the first draft of the manuscript; YB researched literature on epidemiology and wrote §; AA, GJB, HC, MTdS researched literature and wrote § on imaging; OG and VTM researched literature and wrote § on Alzheimer disease; MR and OG researched literature and wrote § on cognitive assessment; HC and IS researched literature and wrote § on remote and online assessment; OG and BG researched literature and wrote § on rehabilitation and non-invasive brain stimulation. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
