Abstract
Introduction:
There are limited data regarding cerebrospinal fluid (CSF) and plasma biomarkers among patients with Cerebral Amyloid Angiopathy (CAA). We sought to investigate the levels of four biomarkers [β-amyloids (Aβ42 and Aβ40), total tau (tau) and phosphorylated tau (p-tau)] in CAA patients compared to healthy controls (HC) and patients with Alzheimer Disease (AD).
Patients and methods:
A systematic review and meta-analysis of published studies, including also a 5 year single-center cohort study, with available data on CSF and plasma biomarkers in symptomatic sporadic CAA versus HC and AD was conducted. Biomarkers’ comparisons were investigated using random-effects models based on the ratio of mean (RoM) biomarker concentrations. RoM < 1 and RoM > 1 indicate lower and higher biomarker concentration in CAA compared to another population, respectively.
Results:
We identified nine cohorts, comprising 327 CAA patients (mean age: 71 ± 5 years; women: 45%) versus 336 HC (mean age: 65 ± 5 years; women: 45%) and 384 AD patients (mean age: 68 ± 3 years; women: 53%) with available data on CSF biomarkers. CSF Aβ42 levels [RoM: 0.47; 95% CI: 0.36–0.62; p < 0.0001], Aβ40 levels [RoM: 0.70; 95% CI: 0.63–0.79; p < 0.0001] and the ratio Aβ42/Aβ40 [RoM: 0.62; 95% CI: 0.39–0.98; p = 0.0438] differentiated CAA from HC. CSF Aβ40 levels [RoM: 0.73; 95% CI: 0.64–0.83; p = 0.0003] differentiated CAA from AD. CSF tau and p-tau levels differentiated CAA from HC [RoM: 1.71; 95% CI: 1.41–2.09; p = 0.0002 and RoM: 1.44; 95% CI: 1.20–1.73; p = 0.0014, respectively] and from AD [RoM: 0.65; 95% CI: 0.58–0.72; p < 0.0001 and RoM: 0.64; 95% CI: 0.57–0.71; p < 0.0001, respectively]. Plasma Aβ42 [RoM: 1.14; 95% CI: 0.89–1.45; p = 0.2079] and Aβ40 [RoM: 1.07; 95% CI: 0.91–1.25; p = 0.3306] levels were comparable between CAA and HC.
Conclusions:
CAA is characterized by a distinct CSF biomarker pattern compared to HC and AD. CSF Aβ40 levels are lower in CAA compared to HC and AD, while tau and p-tau levels are higher in CAA compared to HC, but lower in comparison to AD patients.

Introduction
Sporadic Cerebral Amyloid Angiopathy (CAA) is a small vessel disease, characterized by the progressive deposition and accumulation of Aβ (amyloid-β) in the cortical and leptomeningeal vessel walls. 1 Its prevalence increases with age, and the Boston criteria were developed as a combination of clinico-radiological features to allow the diagnosis of CAA during life, in the absence of a neuropathological analysis from post-mortem examination or biopsy.2,3
CAA presents with various clinical and neuroimaging manifestations, including cognitive impairment or dementia, focal neurological signs due to spontaneous lobar intracerebral hemorrhage (ICH) and transient focal neurological episodes (TFNEs), attributed to either convexity subarachnoid hemorrhage (cSAH) or cortical superficial siderosis (cSS).4,5 The subclinical pathology appears to begin almost 30 years before the clinical manifestations of the hemorrhagic lesions. 6 In this framework, cognitive impairment represents a primary or prodromal clinical manifestation, even in the absence of the characteristic ICH. 7
Concomitant Alzheimer disease (AD) pathology has been estimated in almost 80%–90% of the CAA.1,8 Disentangling probable contributions to the cognitive impairment from the overlapping pathologies remains however a great challenge. Non-hemorrhagic lesions of CAA have been mostly associated with cognitive decline, which may progress, following an acute lobar ICH. 9
Cerebrospinal fluid (CSF) biomarkers may provide an opportunity for an earlier recognition of the disease or a key to differentiate the etiology of cognitive impairment.10,11 Despite that AD is characterized by decreased levels of CSF Aβ42 and not Aβ40, preliminary studies have demonstrated a distinct CSF pattern among patients with CAA, with decreased Aβ40 levels and marginally elevated tau protein, especially total tau (tau).12,13
Additionally, plasma biomarkers have emerged as an alternative tool in the investigation/diagnosis of CAA and the discrimination of CAA patients and healthy controls (HC).14,15 Plasma biomarker assessment is a less invasive and cost-effective alternative to the lumbar puncture (LP) and CSF analysis.
We performed an updated systematic review and meta-analysis of the available literature data regarding CSF and plasma biomarkers, while also incorporating data from our single-center cohort. We sought to investigate CSF and plasma biomarkers levels between either CAA patients and HC or CAA patients and AD patients. To the best of our knowledge this meta-analysis includes all the available published studies, with regard to the CSF biomarkers and is the first to discuss the available data with regard to the plasma biomarkers. The ratio of means was used as effect measure to assess differences of biomarker profiles from CAA patients compared to either HC or patients with AD.
Subjects and methods
Data availability statement
The datasets used and analyzed during the current study are included in this article and its Supplemental Material. More detailed datasets are available from the corresponding author on reasonable request.
Patient characteristics
Patients with CAA, AD, and HC, who presented either at our stroke service or at our outpatient stroke and dementia clinic (Second Department of Neurology, Attikon University Hospital, Athens, Greece) were prospectively recruited during a five-year period (2018–2023). Institutional review board approvals were obtained from the ethics committee of Attikon University Hospital (decision numbers: EBD 499/8-9-2020 and ΕΒD 157/16/03/2021)
Diagnosis of CAA and AD was based on the modified Boston criteria and diagnostic criteria of Alzheimer’s dementia according to the National Institute on Aging and Alzheimer’s Association (NIAAA), respectively.3,16 HC participants, recruited in our department, underwent a LP as a part of diagnostic work-up due to their presenting symptoms (e.g. headache, vomiting etc.). All patients underwent thorough diagnostic work-up, that excluded central nervous system disorder. Participants were selected in our cohort study as healthy controls, only if they fulfilled the following criteria: no history of neurological, and in particular degenerative disorders, no history of psychiatric disorder, no history of infectious, inflammatory or malignant diseases and normal cognitive evaluation using Mini-Mental State Examination (MMSE) testing.
All patients included in this study underwent either a 1.5 Tesla (Philips Healthcare, Best, the Netherlands) or, preferably, a 3Tesla Magnetic Resonance Imaging (MRI) scan (Siemens Magnetom Prisma, Siemens Healthineers, Erlangen, Germany) with a standard comprehensive protocol, including a gradient-echo T2*-weighted (GRE-T2*) sequence or a susceptibility-weighted imaging (SWI) sequence, a T2-weighted sequence, a 3D Fluid attenuated inversion recovery (FLAIR) sequence, a diffusion-weighted imaging (DWI) sequence, and a post-contrast T1-weighted sequence. All participants underwent LP and CSF Aβ40, Aβ42, tau phosphorylated at threonine 181 (p-tau), and total tau levels were quantified. For patients with acute ICH the LP was performed at least 3 months after ICH onset. For all the other patients the LP was performed at the time of diagnosis/first presentation-assessment.
Exclusion criteria for the CAA group was any evidence of hereditary, inflammatory, or iatrogenic underlying cause of CAA17,18 and for the AD group any neuroimaging evidence of underlying probable CAA pathology according to the modified Boston criteria. Finally, patients who did not provide written informed consent to participate in the current study were also excluded. Details regarding the inclusion and exclusion criteria as well as the diagnostic work-up of our cohort have been previously published. 5
Cerebrospinal fluid biomarkers analysis
Lumbar Puncture was performed according to our department’s state-of-the-art protocol and CSF was collected in polypropylene tubes, centrifuged, aliquoted, and stored in polypropylene tubes at −80 °C. Samples with more than 500 red blood cells/μL were discarded. CSF Aβ40, Aβ42, tau, and p-tau levels were blindly quantified in duplicate using the Lumipulse chemiluminescent immunoassay (Fujirebio, Gent, Belgium). The inter-assay coefficients of variation were the following: Aβ42 − average CV: 2.9%, Aβ40 − average CV: 3.3%, tau − average CV: 5.1%, and p-tau − average CV: 4.2%. The samples were analyzed in different batches; however, we adhered to strict standard operating procedures and the manufacturer’s instructions.
Meta-analysis
Standard protocol approvals, registrations, and patient consents
The pre-specified protocol of the systematic review and meta-analysis has been registered in the International Prospective Register of Ongoing Systematic Reviews PROSPERO (CRD42023481511).
Our study adheres to the AHA Journals’ implementation of the transparency and Openness Promotion (TOP) Guidelines. The meta-analysis is reported according to the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 19 and was written according to the Meta Analysis of Observational Studies in Epidemiology (MOOSE) proposal. 20 The systematic review and meta-analysis did not require an ethical board approval according to the study design.
Data sources and database searches
A systematic literature search was conducted to identify eligible studies evaluating the CSF or plasma biomarkers among patients with CAA, and/or HC and/or AD patients.
The literature search was performed independently by three reviewers (A.T., I.T., and M.I.S.). We searched PUBMED and Scopus, using search strings that included the following terms: “Cerebral Amyloid Angiopathy,” “cerebrospinal fluid,” “plasma,” “biomarkers”; the complete search algorithm used in PUBMED and Scopus is described in the Supplemental Methods. The details on database search are included in the Methods section of the Supplemental Material. No language or other restrictions were applied. Our systematic literature search was conducted up to November 1, 2023 for each electronic database. Additional manual search included conference abstracts and bibliographies of candidate studies and recent systematic reviews for a comprehensive literature search.
Study selection
We included full-text, published studies and our 5 year single-center cohort study as well, involving: (1) patient cohorts diagnosed with symptomatic, sporadic CAAbased on original, modified (v1.5) and v2.0 Boston criteria and AD based on NIAAA or NINCDS-ADRDA2,3,14,21; (2) available data on the concentration of CSF or plasma biomarkers among patients with CAA and HC and/or AD patients; (3) adult patients (⩾18 years old). More specifically, the term “sporadic CAA” defines the spontaneous form of CAA, where no familial or iatrogenic causes are suspected or confirmed.
We excluded studies that did not report data with regard to CSF and/or plasma biomarkers among patients with CAA, studies with overlapping data and studies reporting on <7 patients. Editorials, commentaries, and narrative reviews were also excluded.
Data extraction
Three authors (A.T., L.P., C.Z.) independently reviewed the retrieved articles as summarized in the Supplemental Methods section of Supplemental Material and any disagreements were resolved after discussion with the senior author (G.T.).
The following information were extracted: name of the study, first author and year of the publication, study design and data collection interval, mean age, sex distribution, total number of study participants, and relevant method of CSF or plasma analysis.
Primary analyses
An aggregate data meta-analysis was performed with the inclusion of all the eligible cohort studies. We primarily assessed the ratio of means (RoM) as the effect measure, which was calculated as the CSF biomarker concentration ratio between two groups of interest. We sought to meta-analyze CSF and plasma biomarkers data between either CAA patients and HC or CAA patients and AD patients.
Study quality and assessment of publication bias
Eligible studies were subjected to quality control and bias assessment employing the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool for the observational studies. 22 The ROBINS-I tool assesses confounding, selection of participants, classification of intervention, deviations from intended intervention, missing data, measurement of outcomes, and selection of the reported result. Quality control and bias identification were performed independently by two authors (A.T. and M.I.S.) and any disagreements were resolved by a tie-breaking evaluator (G.T.).
Statistical approach
The publication bias across individual studies was evaluated graphically using funnel plots, whereas funnel plot asymmetry was assessed using Egger et al.’s linear regression test. The threshold of the statistical significance was set on p < 0.10.23,24
All statistical analyses were conducted using the R–software version 2023.06.0+421 (packages: meta and metafor).25,26 CSF biomarkers of our center participants were reported as mean ± standard deviation. These concentrations were also calculated in our meta-analysis.
For the variables age and sex we calculated the means and the prevalence rates respectively and their corresponding 95% confidence intervals to measure the effect size. With regard to biomarker levels (continuous variable) we calculated the ratio of means (RoM), meta-analyzing patients with CAA to HC groups and patients with AD ratios of CSF biomarker or plasma concentrations (Aβ42, Aβ40, tau, and p-tau). RoM exhibits comparable performance characteristics to other methods, including mean difference, controls for the variability in concentration ranges between studies and simplifies the clinical interpretation of the results. 10 The variance of this ratio was assessed using the delta method and we used inverse variance weighting to pool ratios. RoM < 1 and RoM > 1 indicates lower and higher biomarker concentration in CAA compared to another population respectively. In studies with more than one CAA patient group based on various clinical or radiological presentations, separate CSF biomarker ratios per CAA subgroups compared to HC and patients with AD were created. Heterogeneity between studies was assessed with the Cochran Q and I2 statistics. A p-value, resulting from the Cochran’s Q test, and less than 0.1 indicated heterogeneity. Moreover and for the qualitative interpretation of the heterogeneity, I2 > 50% and I2 > 75% indicated substantial and considerable heterogeneity, respectively. 27 Random-effects models (DerSimonian and Laird) were used, incorporating between-study heterogeneity. 28 A p-value of 0.05 or less was considered significant.
Results
Patient characteristics
In the CAA group we recruited 31 patients with probable CAA, according to the modified Boston criteria (no patient underwent a brain biopsy) with a mean age of 71 ± 7 years and a female prevalence of 51.6% (n = 16). A total of 26 HC participants with a mean age of 70 ± 11 and a female prevalence of 26.9% (n = 7) were also recruited. In the AD group 37 patients were included, with a mean age of 68 ± 9 and 72.9% (n = 27) females. LP was performed in all cases and the results of CSF analysis for the CAA-group, the HC and AD-group are displayed in Table 1. There was no missing data in the baseline characteristics and biomarkers of our cohort. These data as pooled estimates were subsequently included in the aggregate meta-analysis as reported in the following sections.
Cerebrospinal fluid biomarker analysis in our cohort participants, * .
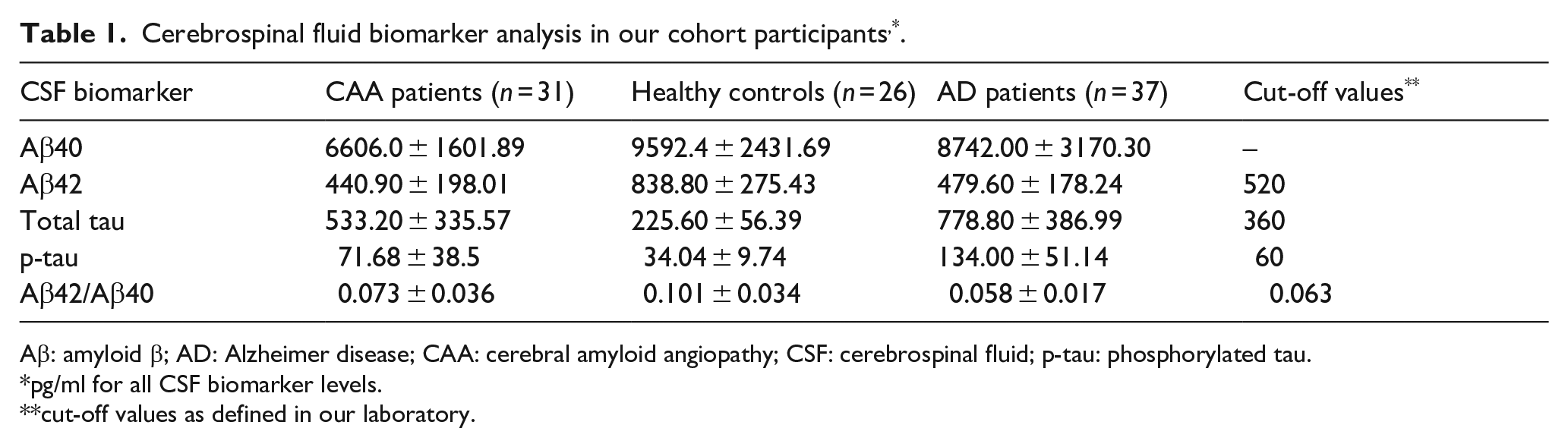
Aβ: amyloid β; AD: Alzheimer disease; CAA: cerebral amyloid angiopathy; CSF: cerebrospinal fluid; p-tau: phosphorylated tau.
pg/ml for all CSF biomarker levels.
cut-off values as defined in our laboratory.
Meta-analysis
Study selection and study characteristics
We screened 612 titles and abstracts from which 167 eligible studies were retained for full text evaluation. After careful evaluation of the available literature, 154 studies were excluded (Supplemental Table S1) resulting in selection of eight studies that met the inclusion criteria for the CSF biomarkers meta-analysis11,13,29–34 and four studies that met the inclusion criteria for the plasma biomarkers meta-analysis35–38 (PRISMA; Supplemental Figure S1 and Supplemental Table S2).
Study quality and publication bias
The risk of bias in the included observational studies was assessed by the Risk of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool 22 and is presented in the Supplemental Figures S2 and S3. Significant confounding and selection bias was observed, since the study design in the majority of the included reports was retrospective. Also, due to the limited number of participants, control for confounders was restricted. However, bias in classification of intervention, bias due to deviations from intended interventions and information/reporting biases were moderate.
We inspected funnel plot asymmetry and Egger et al. 24 statistical test for outcomes involving ⩾4 cohorts (Supplemental Figures S4–S13). Funnel plot inspection revealed no evidence of asymmetry in included studies.
Overall analysis for CSF biomarkers
The mean age of the patients in the included studies was 71 ± 5 (95% CI: 68.8–73.3; I2 = 82%; Supplemental Figures S14–S16) for the CAA-group, 66 ± 5 (95% CI: 62.2–68.7; I2 = 92%; Supplemental Figures S15–S16) for HC and 68 ± 3 (95% CI: 65.3–69.9; I2 = 87%; Supplemental Figure S17) for the AD-group. The prevalence of female-sex was 45% (95% CI: 38%−52%; p for Cochran Q statistic = 0.14; I2 = 32%; Supplemental Figure S18) for the CAA-group, 45% (95% CI: 37%−53%; p for Cochran Q statistic = 0.05; I2 = 49%; Supplemental Figure S19) for HC and 53% (95% CI: 44%−61%; p for Cochran Q statistic = 0.01; I2 = 61%; Figure S20) for the AD-group.
Figure 1 summarizes the mean levels of Aβ42, Aβ40, tau, and p-tau among patients with CAA, HC, and patients with AD and Table 2 displays the pooled RoM for CSF and plasma biomarkers in CAA patients in comparison to HC and AD patients. In brief, CSF Aβ42 RoM and Aβ40 RoM (Figure 2(a) and (b)) were <1 in CAA patients compared to HC while tau RoM and p-tau RoM (Figure 2(d) and (e)) were higher in CAA patients compared to HC. In the case of Ab42/Ab40, comparing CAA patients to HC, the analysis resulted in a pooled RoM of 0.62 (95% CI, 0.39–0.98; p for Cochran Q statistic = 0.0438; I2 = 75%; Figure 2(c)). Furthermore, CAA patients had CSF RoM < 1 for Aβ40, tau and p-tau (Figure 3(b), (d) and (e)) compared to AD patients. Statistically non-significant were the RoM for Aβ42 and for the ratio Aβ42/Aβ40 in CAA patients compared to AD patients (Figure 3(a) and (c)).
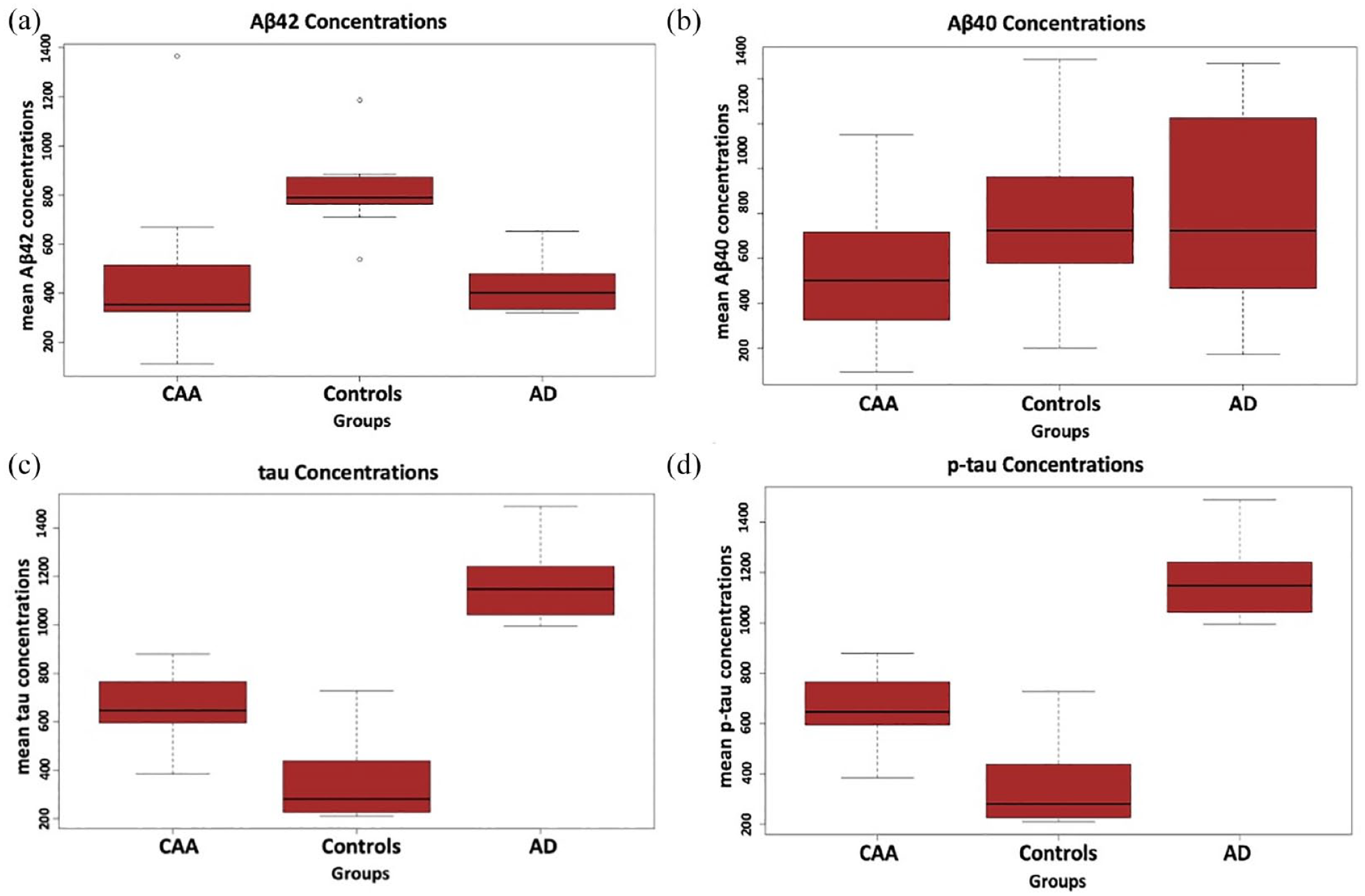
Mean levels of Aβ42, Aβ40, tau, and p-tau among patients with CAA, HC, and patients with AD in the overall meta-analysis.
Ratio of mean (RoM) biomarker concentrations comparing CAA patients versus HC and AD patients in the overall meta-analysis.
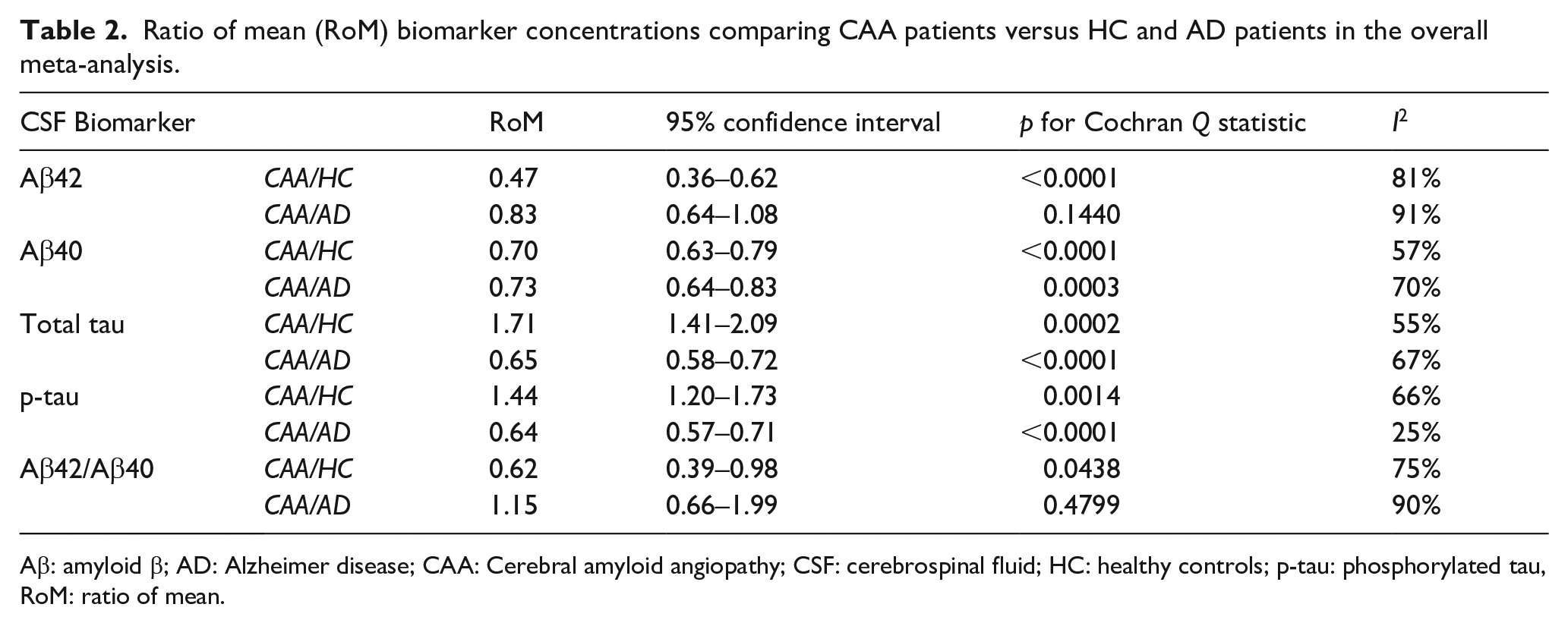
Aβ: amyloid β; AD: Alzheimer disease; CAA: Cerebral amyloid angiopathy; CSF: cerebrospinal fluid; HC: healthy controls; p-tau: phosphorylated tau, RoM: ratio of mean.
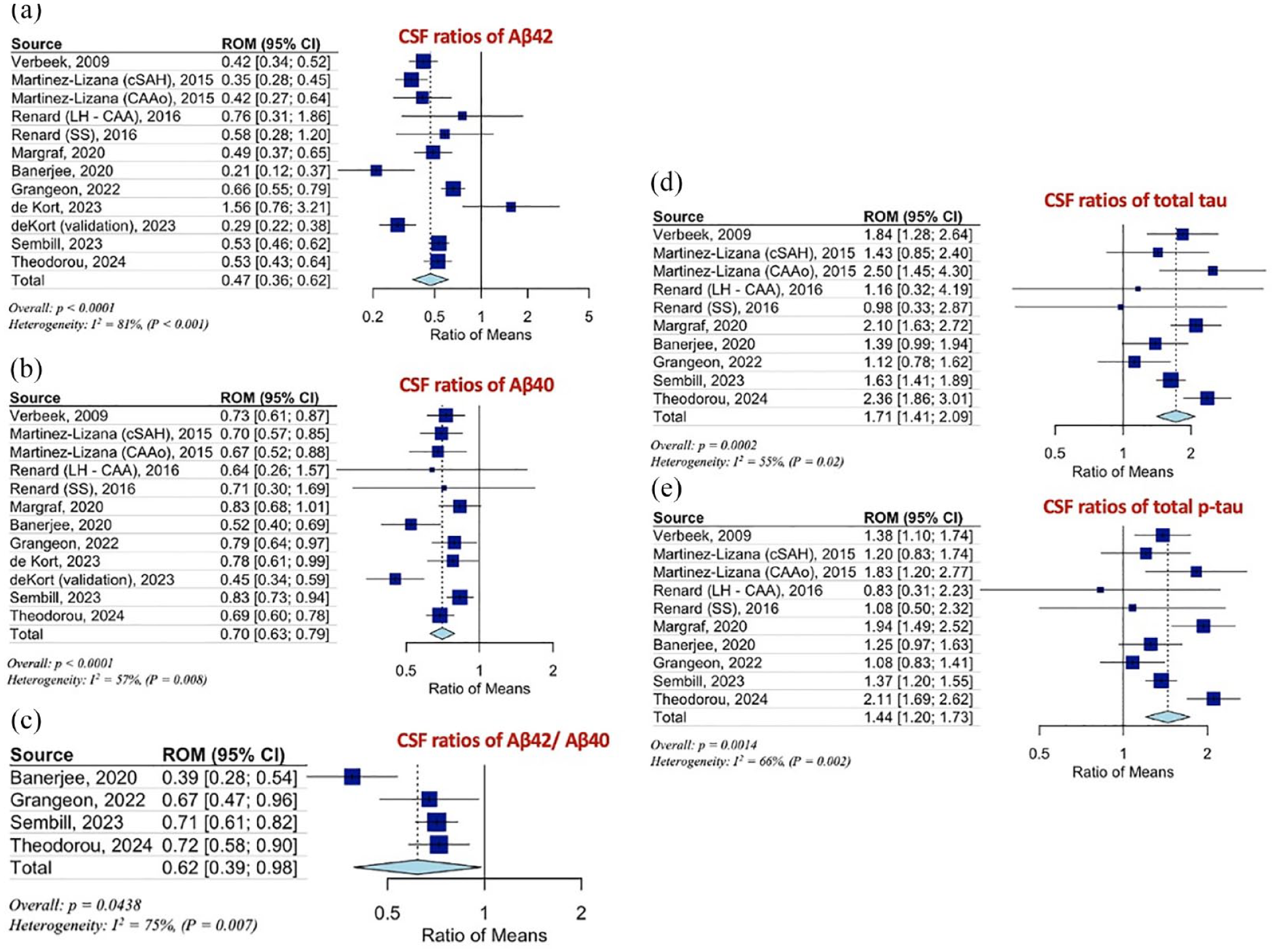
(a) Ratios for CSF Aβ42, (b) Aβ40, (c) Aβ42/Aβ40, (d) total tau (t-tau), and (e) phosphorylated tau (p-tau) levels, of CAA patients to HC.
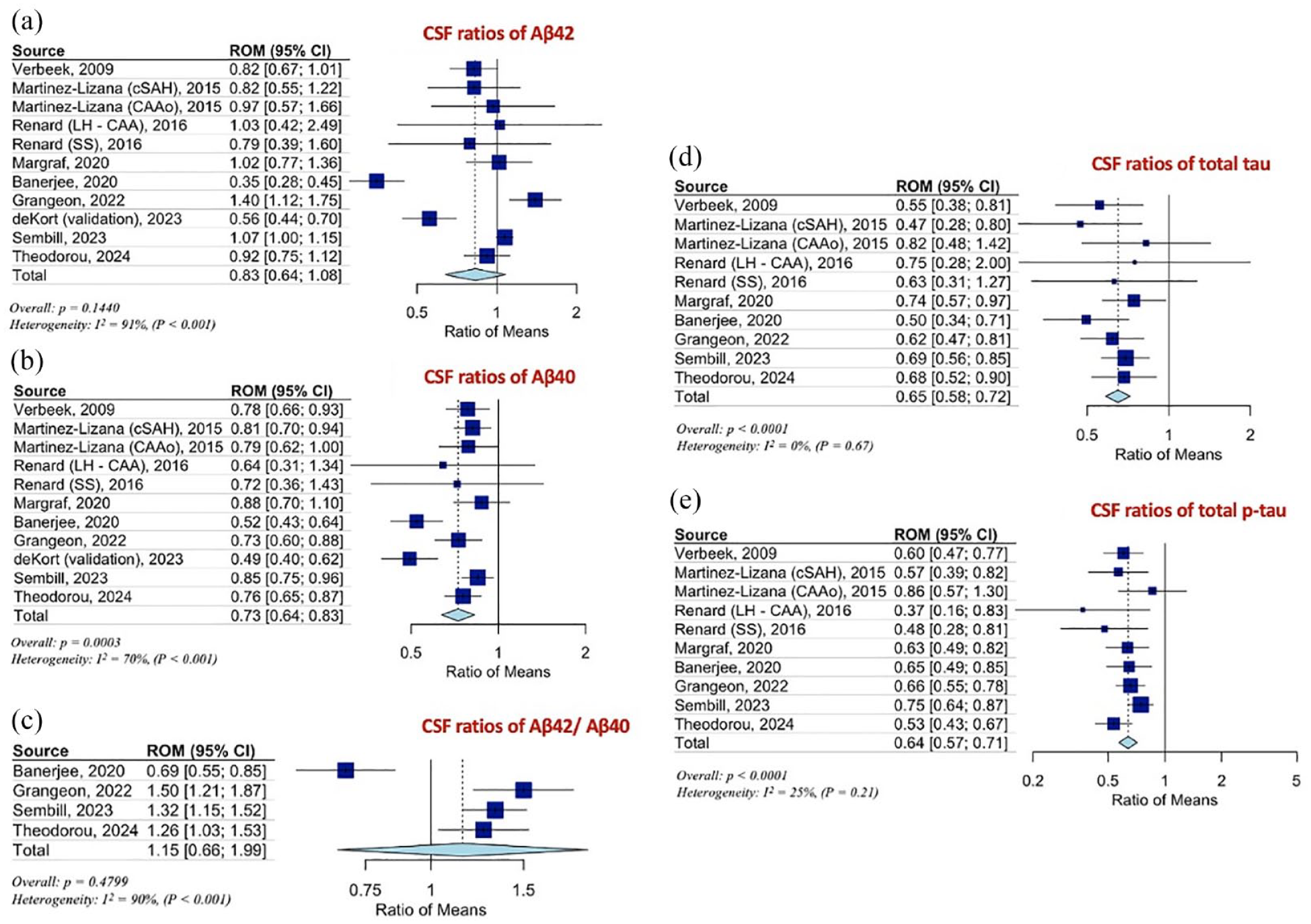
(a) Ratios for CSF Aβ42, (b) Aβ40, (c) Aβ42/Aβ40, (d) total tau (t-tau), and (e) phosphorylated tau (p-tau) levels, of CAA patients to AD patients.
Overall analysis for plasma biomarkers
Five cohorts from four studies were included in this meta-analysis with regard to plasma biomarkers.36–39 There was no difference in plasma Aβ42 and Aβ40 levels in CAA patients compared to HC (Figure 4(a) and (b)). Additional comparisons with regard to plasma biomarkers could not be performed due to data unavailability.
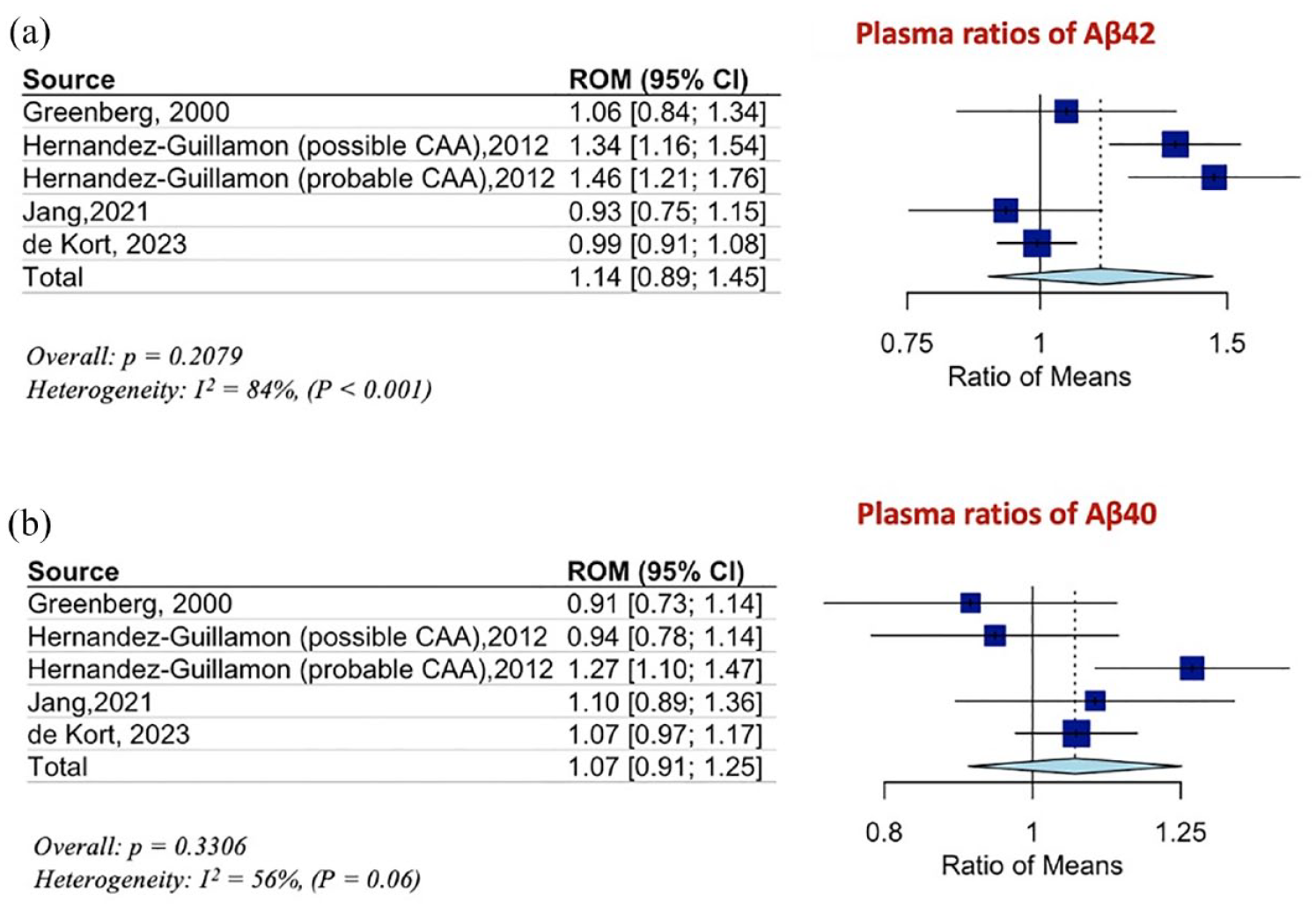
(a) Ratios for plasma Aβ42, and (b) and Aβ40 levels, of CAA patients to HC.
Discussion
The present systematic review and meta-analysis demonstrate a distinct pattern of CSF biomarkers in CAA, characterized by decreased levels of Aβ40 compared to HC and patients with AD, and decreased levels of Aβ42 compared to HC but not compared to AD (Figure 1). The CSF ratio Aβ42/Aβ40 was also decreased in CAA compared to HC, but no significant difference was detected between CAA and AD (Figure 2). Notably, total tau and p-tau CSF levels were significantly higher in CAA compared to HC, but lower compared to AD (Figure 1). This specific pattern of CSF biomarkers was also disclosed in the results of our single center cohort study. Additionally, plasma Aβ42 and Aβ40 levels were comparable between CAA and HC, although further comparisons regarding plasma biomarkers could not be performed due to lack of data.
The present findings on CSF biomarkers are concordant with results of previous smaller-scale meta-analyses and observational studies.10,11,29,33,39 Compared to previous works, the present updated meta-analysis has included data from eight observational studies and previously unpublished data from our single-center experience, enhancing the precision and robustness of our estimates.11,13,29–34 Notably, a previous meta-analysis on this topic has included only 59 patients (compared to 327 patients included in the current meta-analysis). 10
The emerging evidence for the role of CSF biomarkers highlights the potential for CAA detection at earlier disease stages. Established use of CSF biomarkers could contribute to a multiparametric work-up for the diagnosis of CAA, including clinical characteristic and neuroimaging/genetic findings as well. This approach may lead to a safer and more reliable management, avoiding delays and misdiagnoses in cases of CAA at earlier stages and avoiding also the initiation of anti-amyloid β monoclonal antibodies, in CAA patients misdiagnosed as AD. Of special interest is the possible role of blood biomarkers, which represent a cost-effective and easily accessible diagnostic method that may complement MRI-based diagnosis and aid in monitoring of disease progression.35–38 Our meta-analysis also contributes to the potential differentiation of CAA and AD based on the plasma biomarkers’ profile.
The underlying frequent neuropathologic overlap between CAA and AD is well established, so that the discrimination of these two entities remains challenging.40–42 With respect to AD or even mild cognitive impairment due to AD, predominant deposition of Aβ42 in diffuse senile plaques constitutes the neuropathological hallmark of disease, leading to selective reduction of CSF Aβ42 levels. 43 Moreover, alterations in the dynamic equilibrium of production, clearance and accumulation in combination with a selective trapping of Aβ40 and Aβ42 isoforms in the cerebral vasculature are the major causes of CAA underlying pathology, that typically lead to decreased levels of both Aβ40 and Aβ42 among patients with CAA.29,44 Our meta-analysis strengthens this hypothesis, demonstrating statistically significant decreases in levels of Aβ40 in CAA compared to AD, but similar Aβ42 levels between the two groups. In addition, the ratio of Aβ42/Aβ40 has been proven to discriminate reliably AD from HC. Notably, in our meta-analysis this ratio was statistically different between CAA patients and HC (significantly lower in CAA patients), but did not differ between CAA and AD patients. 11
The role of tau proteins was also investigated in the current meta-analysis. Pathological tau aggregation plays an important role in AD pathology, leading to synaptic dysfunction and neuronal loss, while tau pathology has been mostly associated with cognitive impairment among AD patients.40,45–47 In accordance with our findings, previous observational studies and meta-analyses have also documented intermediate tau levels in CAA patients compared to HC (RoM > 1) and AD patients (RoM < 1).10,48 It is known that, in contrast to AD, formation of neurofibrillary tangles is not considered a typical pathological feature of CAA. 49 However, the intermediate tau and p-tau levels in CAA could be explained by an accompanying AD pathology, even in non-demented CAA patients. 29
In addition to CSF biomarkers, the current meta-analysis assessed levels of plasma biomarkers in CAA. To the best of our knowledge, this is the first systematic review and meta-analysis involving 127 CAA patients with available plasma biomarkers, suggesting that plasma Aβ40 and Aβ42 levels cannot sufficiently differentiate CAA from HC. This finding is in accordance with two previous studies reporting non-significant differences of plasma Aβ40 and Aβ42 levels between patients with CAA and HC,40,42 but stands in contrast to the findings of another study that demonstrated higher Aβ40 and Aβ42 levels in CAA patients compared to HC. 37 Our experience lends support to the notion that these biomarkers are unreliable in discriminating sporadic CAA from HC, however data from larger cohort studies are required prior to making this determination.
Our study has certain limitations that warrant further consideration. First, the main limitation is the moderate sample size, due to the limited data availability with regard to CSF and especially plasma biomarkers. Second, many of the included studies were observational with retrospective study design, that predispose to selection biases. Third, no study relied on biopsy confirmation for CAA diagnosis, instead using the proposed Boston criteria for probable/possible CAA diagnosis. Fourth, lack of consensus on laboratory methods used for biomarkers measurement may have contributed to heterogenous measurements. This heterogeneity may explain the asymmetry in funnel plots. Fifth, significant biases due to confounding and participant selection, as they were identified in the studies included in the present meta-analysis, should be acknowledged as a limitation of this meta-analysis as well. Timing of screening for CAA represents another limitation of our study, since the potential influence of the disease stage on the biomarker levels and on the discrimination from AD, remains controversial. Moreover, we did not control for multiple testing based on the statistical design of the observational cohort study. In addition, there was no formal adjustment for age between CAA and HC. However, CAA patients were significantly older than HC (on average 6 years) and this needs to be taken into account when interpreting our study findings. Last, there were no available data with regard to novel biomarkers (Aβ38/Aβ43) or cognitive assessment scores, which could be implemented for earlier recognition and discrimination of CAA disease. 33
In conclusion, this systematic review and meta-analysis of available data provide evidence that CSF biomarker panel, consisting of Aβ40, Αβ42, tau and p-tau, may be able to distinguish CAA from AD and HC. These findings are salient for the design and conduct of future randomized controlled clinical trials, aiming to treat CAA at preclinical stages.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241260538 – Supplemental material for CSF and plasma biomarkers in cerebral amyloid angiopathy: A single-center study and a systematic review/meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873241260538 for CSF and plasma biomarkers in cerebral amyloid angiopathy: A single-center study and a systematic review/meta-analysis by Aikaterini Theodorou, Ioanna Tsantzali, Maria-Ioanna Stefanou, Simona Sacco, Aristeidis H Katsanos, Ashkan Shoamanesh, Theodoros Karapanayiotides, Ioanna Koutroulou, Polyxeni Stamati, David J Werring, Charlotte Cordonnier, Lina Palaiodimou, Christina Zompola, Efstathios Boviatsis, Lampis Stavrinou, Frantzeska Frantzeskaki, Thorsten Steiner, Andrei V Alexandrov, Georgios P Paraskevas and Georgios Tsivgoulis in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241260538 – Supplemental material for CSF and plasma biomarkers in cerebral amyloid angiopathy: A single-center study and a systematic review/meta-analysis
Supplemental material, sj-docx-2-eso-10.1177_23969873241260538 for CSF and plasma biomarkers in cerebral amyloid angiopathy: A single-center study and a systematic review/meta-analysis by Aikaterini Theodorou, Ioanna Tsantzali, Maria-Ioanna Stefanou, Simona Sacco, Aristeidis H Katsanos, Ashkan Shoamanesh, Theodoros Karapanayiotides, Ioanna Koutroulou, Polyxeni Stamati, David J Werring, Charlotte Cordonnier, Lina Palaiodimou, Christina Zompola, Efstathios Boviatsis, Lampis Stavrinou, Frantzeska Frantzeskaki, Thorsten Steiner, Andrei V Alexandrov, Georgios P Paraskevas and Georgios Tsivgoulis in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Katsanos is supported by the New Investigator Award from the Heart and Stroke Foundation Canada.
Informed consent − Ethical approval
This study did not require an ethical board approval or written informed consent by the patients according to the study design (systematic review and meta-analysis).
Guarantor
GT
Contributorship
Dr Theodorou participated in study concept and design, acquisition of data, analysis and interpretation, critical revision of the article for important intellectual content. Drs Tsantzali and Stefanou participated in acquisition of data, critical revision of the article for important intellectual content. Drs Sacco, Katsanos and Shoamanesh participated in acquisition of data, critical revision of the article for important intellectual content. Drs Karapanayiotides, Koutroulou and Stamati participated in acquisition of data, critical revision of the article for important intellectual content. Drs Palaiodimou, Zompola, Stavrinou and Frantzeskaki performed analysis, critical revision of the article for important intellectual content. Drs Werring, Cordonnier, Boviatsis, Steiner, Alexandrov, and Paraskevas participated in critical revision of the article for important intellectual content. Dr. Tsivgoulis was responsible for study concept, analysis and interpretation, critical revision of the article for important intellectual content.
Data availability
All data needed to evaluate the conclusions in the paper are present in the main manuscript and in the supplemental material. Additional data related to this paper may be requested from the corresponding author, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
