Abstract
Introduction:
In intracranial medium-vessel occlusions (MeVOs), intravenous thrombolysis (IVT) shows inconsistent effectiveness and endovascular interventions remains unproven. We evaluated a new therapeutic strategy based on a second IVT using tenecteplase for MeVOs without early recanalization post-alteplase.
Patients and methods:
This retrospective, comparative study included consecutively low bleeding risk MeVO patients treated with alteplase 0.9 mg/kg at two stroke centers. One center used a conventional single-IVT approach; the other applied a dual-IVT strategy, incorporating a 1-h post-alteplase MRI and additional tenecteplase, 0.25 mg/kg, if occlusion persisted. Primary outcomes were 24-h successful recanalization for efficacy and symptomatic intracranial hemorrhage (sICH) for safety. Secondary outcomes included 3-month excellent outcomes (modified Rankin Scale score of 0–1). Comparisons were conducted in the overall cohort and a propensity score-matched subgroup.
Results:
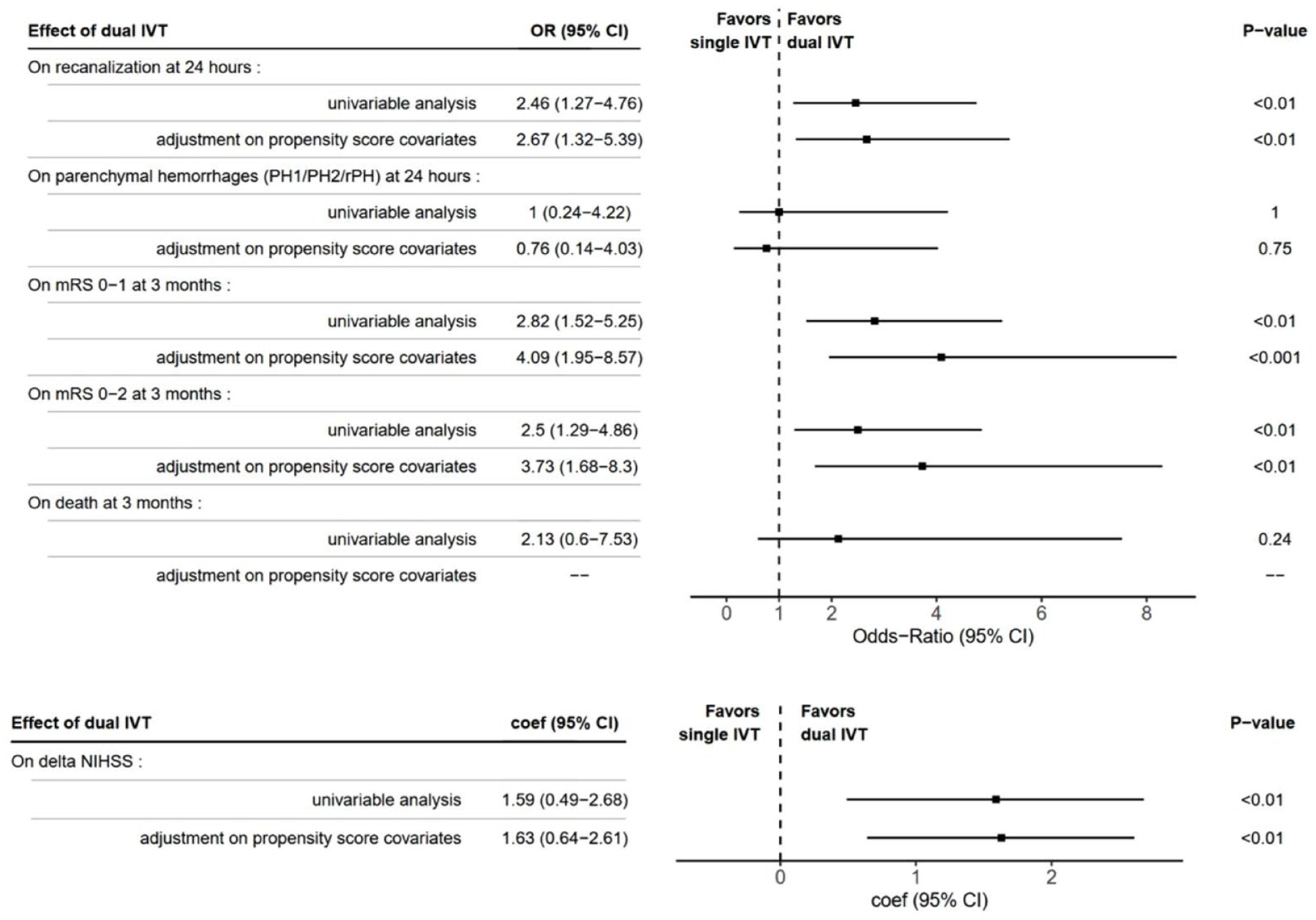
Among 146 patients in the dual-IVT group, 103 failed to achieve recanalization at 1 h and of these 96 met all eligible criteria and received additional tenecteplase. Successful recanalization at 24 h was higher in the 146 dual-IVT cohort patients than in the 148 single-IVT cohort patients (84% vs 61%, p < 0.0001), with similar sICH rate (3 vs 2, p = 0.68). Dual-IVT strategy was an independent predictor of 24-h successful recanalization (OR, 2.7 [95% CI, 1.52–4.88]; p < 0.001). Dual-IVT cohort patients achieved higher rates of excellent outcome (69% vs 44%, p < 0.0001). Propensity score matching analyses supported all these associations.
Conclusion:
In this retrospective study, a dual-IVT strategy in selected MeVO patients was associated with higher odds of 24-h recanalization, with no safety concerns. However, potential center-level confounding and biases seriously limit these findings’ interpretation.
Trial Registration
ClinicalTrials.gov Identifier:

Introduction
Intracranial medium-vessel occlusions (MeVOs) mainly affect the M2-middle cerebral artery (MCA), M3-MCA, A2/A3-anterior cerebral artery (ACA) and P2/P3 posterior cerebral artery (PCA).1–4 MeVO-associated acute ischemic strokes (AIS) can be disabling, justifying attempts to urgently recanalize the occluded artery, thereby improving the outcome. However, alteplase intravenous thrombolysis (IVT) fails to recanalize up to one-half of MeVOs.3,4 Endovascular treatment (EVT), possibly preceded by IVT, is nowadays unproven for MeVOs.1,2,4 Tenecteplase is a promising, genetically modified, more fibrin-specific variant of alteplase, with a longer half-life, allowing a single bolus administration.5,6 However, recent large randomized controlled trials (RCT) demonstrated non-inferiority but not superiority of tenecteplase over alteplase in AIS treatment, 7 especially in patients ineligible for EVT who included those with MeVOs. 8 As a result, it seems unlikely that tenecteplase can do much better than alteplase in MeVOs.
We aimed to assess a new therapeutic strategy for MeVO-AIS, based on a second sequential IVT with tenecteplase for persistent intracranial occlusion assessed 1 h after alteplase infusion, in this retrospective and comparative study.
Methods
The DIVA study (DIVA-retro, NCT05809921) was a bicenter, retrospective cohort study of consecutive patients aged ⩾18 years with MeVo-AIS treated by alteplase, from March, 2014 to November, 2018. Patients from the first center (Centre Hospitalo-Universitaire de Bordeaux = CHUB) had no additional treatment (single-IVT cohort), whereas those from the second center (Centre Hospitalier Sud Francilien = CHSF) were committed in the dual-IVT strategy (dual-IVT cohort), according with the local protocol of CHSF9 (see below).
Ethics
The present study was approved by the responsible Ethics committee (no. 02023) and obtained a “Commission Nationale de l’Informatique et des Libertés” authorization (no. 2219531). Patient written informed consent was waived due to this study’s retrospective and anonymized design. The study was investigator initiated, and was reported according to the STROBE guidelines.
Selection criteria and procedure assessment
All single and dual-IVT participants were selected based on the following criteria: pre-stroke modified Rankin Scale (mRS) <2; AIS diagnosis based on Magnetic Resonance Imaging (MRI) with clear MeVO identification; alteplase infusion within the 4.5-h therapeutic window or a negative Fluid Attenuated Inversion Recovery (FLAIR) sequence when time of onset was unknown 10 ; since 2015, EVT has to be considered unsuitable. To minimize the risk of bleeding, patients with >5 microbleeds, diffuse or focal cortical siderosis, 11 severe leukoaraiosis (Fazekas score 3/3), 12 any coagulopathy type, including a baseline international normalized ratio > 1.3 or dual antiplatelet therapy at admission, were not eligible for this study.
All patients from both cohorts received alteplase, 0.9 mg/kg, maximum 90 mg (IVT-1), but only dual-IVT patients had an MRI at 1-h post-alteplase, and those with persistent occlusion, as defined below, received tenecteplase bolus of 0.25 mg/kg, maximum 25 mg (IVT-2), in the absence of the following additional exclusion criteria on 1-h MRI: acute lesion visible on FLAIR sequence, new cerebral/subarachnoid hemorrhage of any type or size; significant extracerebral bleeding, such as abundant gingivorrhagia, epistaxis, diffuse ecchymoses or macroscopic hematuria. Patients in the dual-IVT group were included whether they received a second IVT dose or not.
All patients from both cohorts had an MRI at 24 h post-alteplase, or at any time if clinical status worsened, and were followed up to 3-month post-stroke.
Study definitions and outcomes
MeVO diagnosis was based on level of occlusion visible on MRI-angiography and/or location of visible thrombus on gradient echo-sequences (GE). More particularly, M2 segment was divided into M2 proximal (first half) and M2 distal (second half). Status of recanalization was assessed according with the Arterial Occlusion lesion (AOL) scale 13 (AOL 0/1: no recanalization; AOL 2/3: successful recanalization). However, to render patient information homogeneous for analysis, patients without visible occlusion on MRI-angiography but with visible thrombus on GE were also classified with the AOL scale, as follows: no recanalization, including AOL-0 (unchanged thrombus location) and AOL-1 (slight migration of the thrombus); successful recanalization, including AOL-2 (residual distal thrombus) and AOL-3 (complete thrombus disappearance) (Figure 1). Intracranial hemorrhage were categorized as hemorrhagic infarction type 1/2, parenchymal hematoma (PH) type 1/2 and remote PH (rPH), according to the Heidelberg Bleeding Classification. 14 Symptomatic intracranial hemorrhage (sICH) was defined according to the European Cooperative Acute Stroke Study II definition (blood at any site in the brain, in combination with worsening of NIHSS score by at least 4 points). 15 All MRI data sets were reanalyzed specifically for the study by paired stroke experts unblinded of the therapeutic strategy (NC, DS for the first center; SO, PR for second center), resolving any disagreements through consensus.
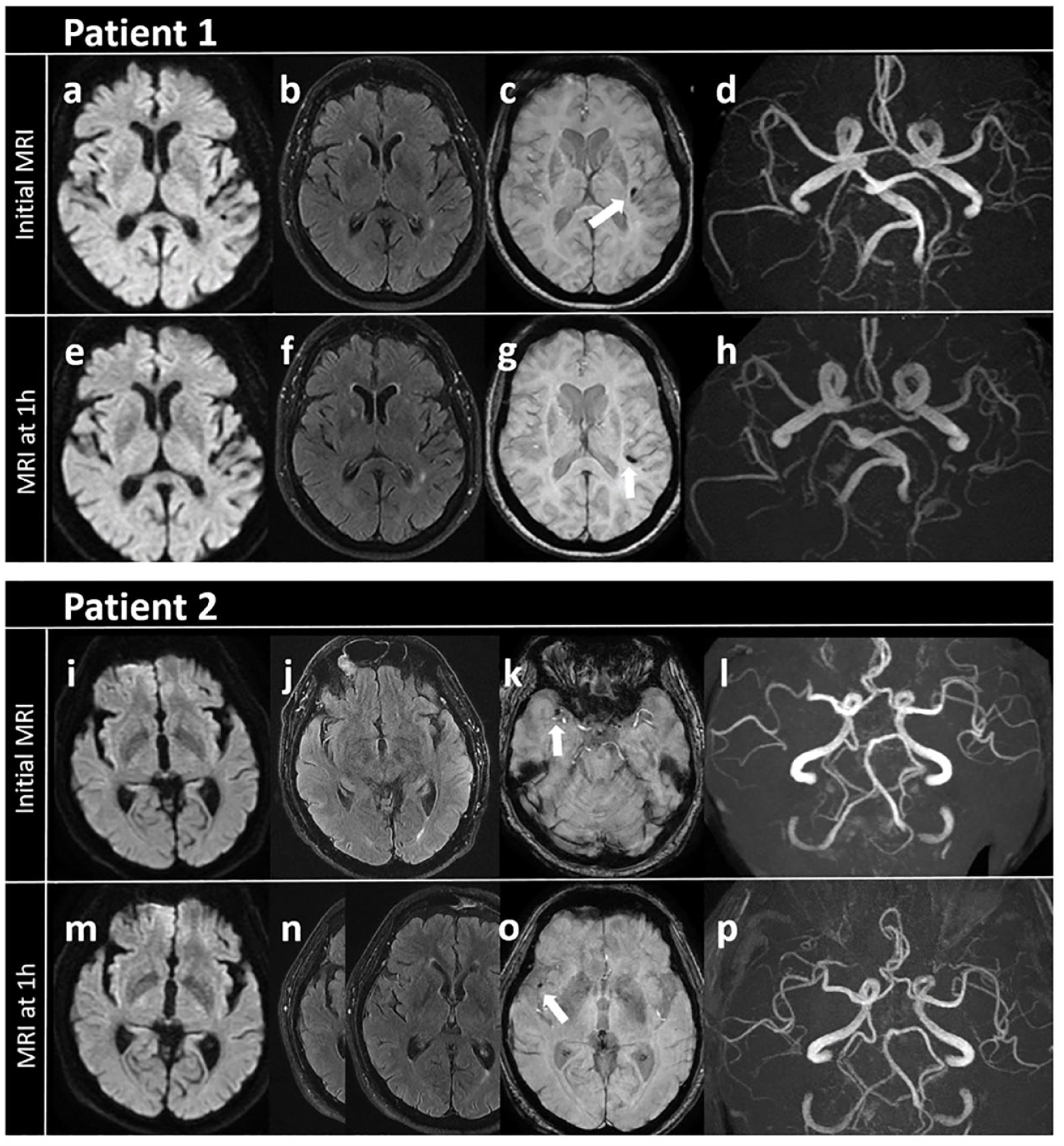
Two representative cases of arterial occlusive lesion (AOL) classification at 1-h, based on thrombus change on gradient echo (GE) sequence.
Major systemic bleeding was defined as hemoglobin decline ⩾2 g/dL post-treatment. Neurological outcomes were defined as follows: delta-National Institute of Health Stroke Scale (NIHSS) at 24-h as NIHSS admission score minus NIHSS 24-h score; early neurological improvement (ENI) as a NIHSS score decline of ⩾4 points or NIHSS = 0 at 24-h and early complete neurological recovery as NIHSS = 0 at 24-h; excellent and good functional outcomes as mRS 0_1 and mRS 0_2 at 3-month respectively. MRS was evaluated through structured interview by local trained unblinded physicians either in person or via phone call.
The primary efficacy endpoint was successful recanalization (AOL 2/3) on the 24-h MRI. The primary safety endpoint was sICH. Secondary endpoints were clinico-biological: ENI, early complete neurological recovery and delta-NIHSS score at 24-h, major systemic bleeding, excellent/good functional outcomes and death at 3-month, and radiological: any cerebral hemorrhage and PH/rPH at 24-h.
Statistical analyses
Comprehensive statistical analysis method is provided in Supplemental Data. Quantitative variables are expressed as median [interquartile range (IQR)] and qualitative parameters as numbers (percentages). Categorical variables were compared using chi-square or Fisher’s exact test and continuous variables with Wilcoxon’s rank-sum test, as appropriate.
Univariable and multivariable logistic-regression models were used across the whole cohort to identify associations between initial characteristics and successful recanalization at 24-h. All continuous variables were checked for log-linearity. Non-log-linear variables were discretized according to their inflection points or based on the literature. Non-collinear variables that achieved p < 0.2 in univariable analysis were included in the multivariable model, and no variable exclusion was done thereafter. Associations between variables and the main outcome measures are reported as crude and adjusted odds ratios (ORs) [95% confidence intervals (CIs)].
In order to make the two treatment groups comparable, we also used a propensity-score matching approach. This propensity score was derived from a logistic regression model using initial patient characteristics as covariates and the treatment group as the dependent variable. Covariates were selected after data inspection, including all baseline patient characteristics (age, sex, history of hypertension, hypercholesterolemia, diabetes mellitus, current smoking, atrial fibrillation) as well as stroke characteristics prior to alteplase (baseline NIHSS, occlusion site, etiologic classification and known onset to alteplase), and were all retained without a stepwise selection procedure.
Because proximal M2-MCA occlusions may be eligible for EVT since 2015, sensitivity analyses after exclusion of cases with proximal M2-MCA occlusion from both cohorts were conducted.
We also performed a statistical analysis using a propensity-score overlap weighting approach, using the entire patient population from both cohorts (See details for both propensity-score analyses, eMethod 1 and 2, Supplemental Data). 16
All tests were two-sided and p < 0.05 was considered significant. Analyses were computed using R statistical software version 4.0.2. F-XL and NC had full access to all the data in the study and take responsibility for its integrity and the data analysis.
Results
Patients’ characteristics and results in the whole cohort
Dual and single-IVT cohorts included 148 patients each. Two out of 148 dual-IVT patients wished to withdraw from the analysis (Table 1). Patients MeVO sites were M2-MCA for half of patients. The reasons for excluding M2-MCA occlusions from EVT was very similar between both cohorts (Supplemental Table 1). Five patients with P1-PCA occlusions, which are not strictly MeVOs, were nonetheless included. Both cohorts were very similar but differed slightly for smoking rate [22 (15) vs 38 (26); p < 0.03], median initial NIHSS score [5 (IQR, 2–9) vs 6 (IQR, 3–11); p < 0.01] and number of unclear stroke onsets [12/146 vs 3/148; p = 0.02].
Baseline characteristics of the patients included in dual and single-IVT strategy cohorts in the whole cohort (n = 294) and after propensity-score matching (n = 212).
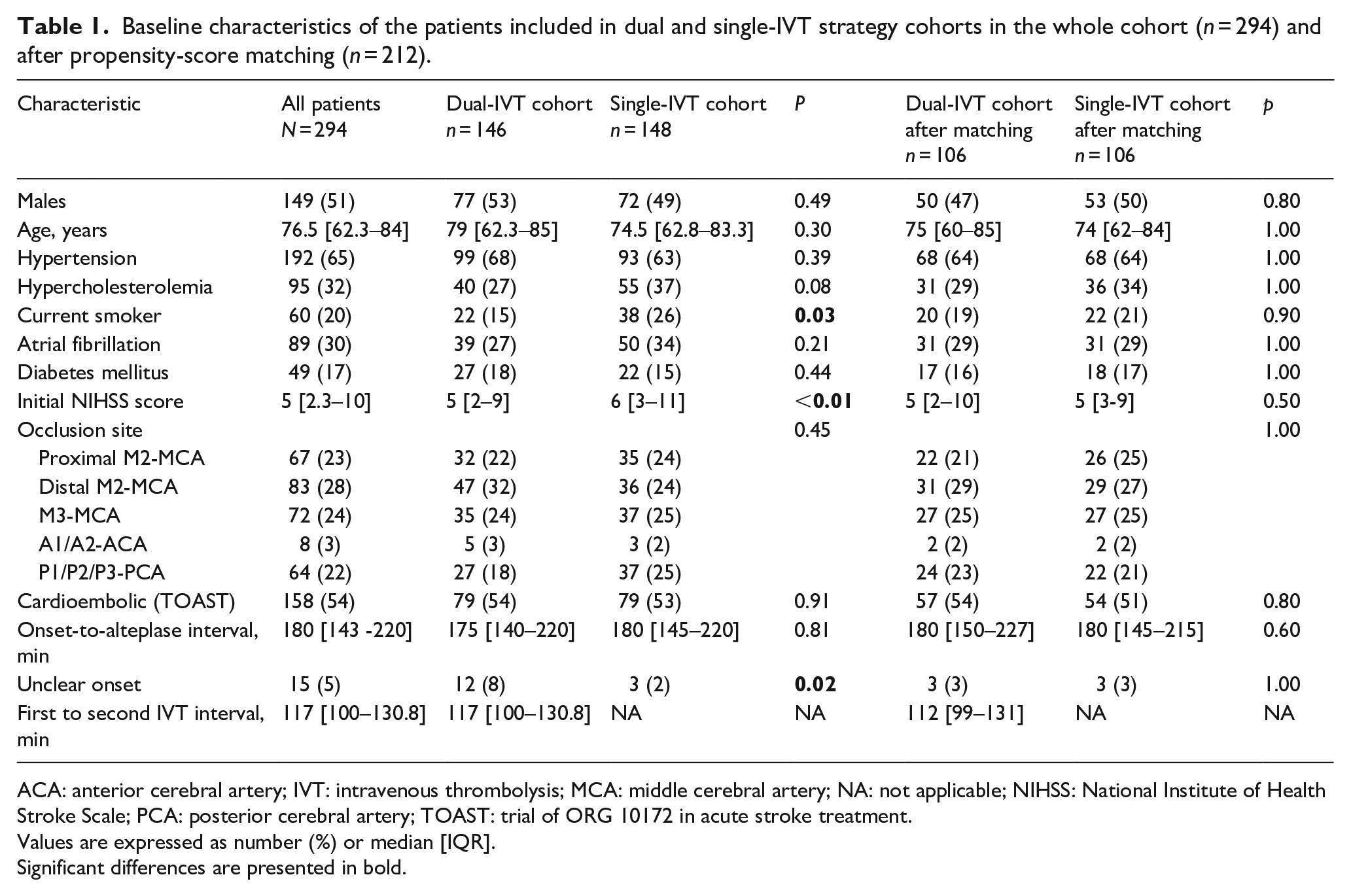
ACA: anterior cerebral artery; IVT: intravenous thrombolysis; MCA: middle cerebral artery; NA: not applicable; NIHSS: National Institute of Health Stroke Scale; PCA: posterior cerebral artery; TOAST: trial of ORG 10172 in acute stroke treatment.
Values are expressed as number (%) or median [IQR].
Significant differences are presented in bold.
Radiological and clinical outcomes are reported in Table 2 and in Figure 2. The 1-h MRI documented early successful recanalization in 43/146 (30%) dual-IVT patients. Among the 103 not-recanalized patients, 7 met exclusion criteria for IVT-2 and 96 received additional tenecteplase at a median time of 116.5 [100–130.8] min post-IVT-1. Of these, 74/96 (77%) achieved successful recanalization at 24-h. Overall, at 24-h, successful recanalization was achieved in 122/146 (84%) dual-IVT patients. By comparison, 91/148 (61%) single-IVT patients showed successful recanalization at 24-h (p < 0.0001) (Table 2 and Figure 2). The proportion of AOL-2 and 3 among recanalized patients at 24-h was the same between both cohorts (dual-IVT: AOL-2, 22 (18%)/AOL-3 102 (82%); single-IVT: AOL-2, 17 (19%)/AOL-3 74 (81%).
Radiological and neurological outcomes according to the dual or the single-IVT strategies, in the whole cohort (n = 294) and after propensity-score matching (n = 212).
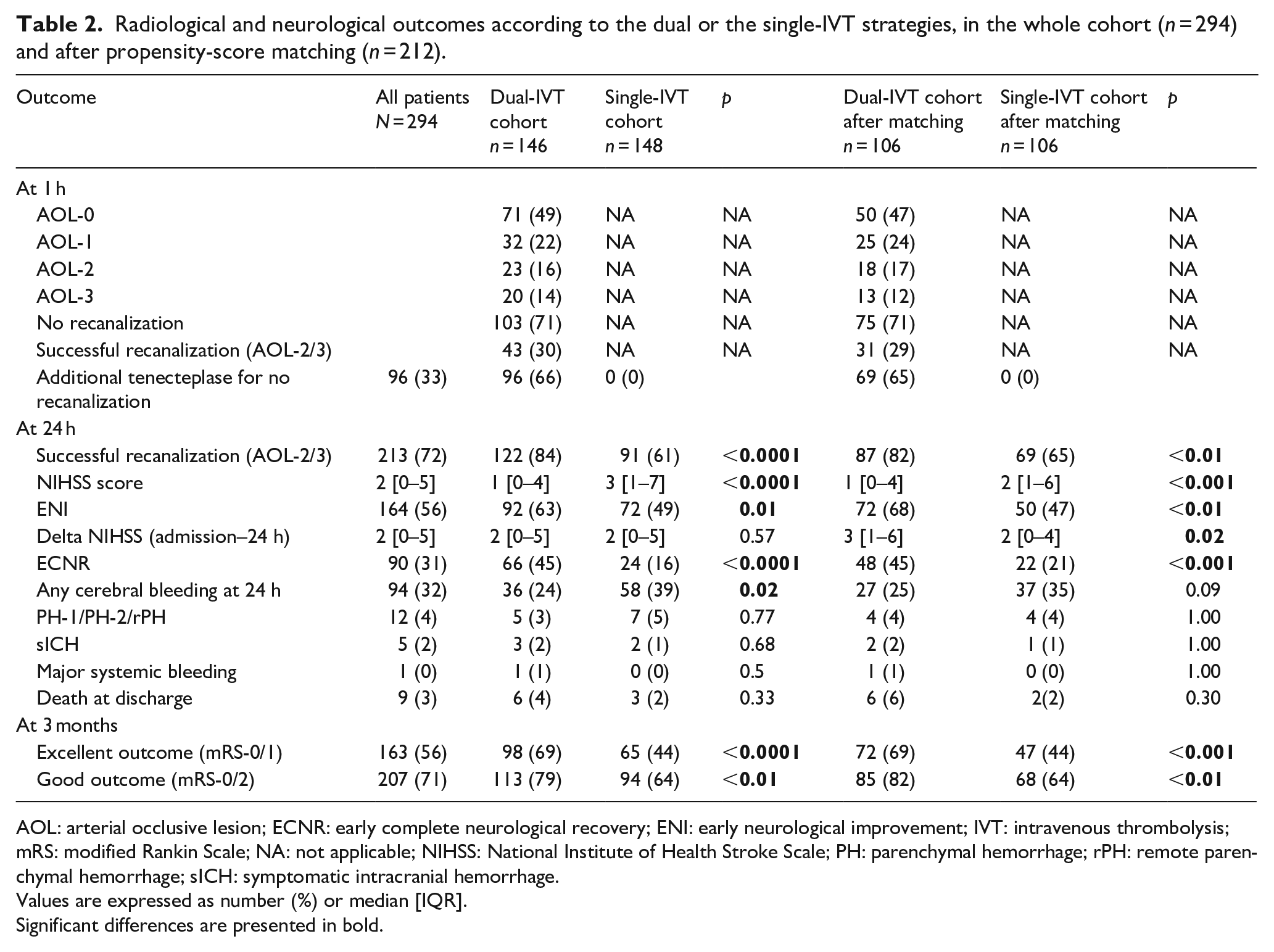
AOL: arterial occlusive lesion; ECNR: early complete neurological recovery; ENI: early neurological improvement; IVT: intravenous thrombolysis; mRS: modified Rankin Scale; NA: not applicable; NIHSS: National Institute of Health Stroke Scale; PH: parenchymal hemorrhage; rPH: remote parenchymal hemorrhage; sICH: symptomatic intracranial hemorrhage.
Values are expressed as number (%) or median [IQR].
Significant differences are presented in bold.
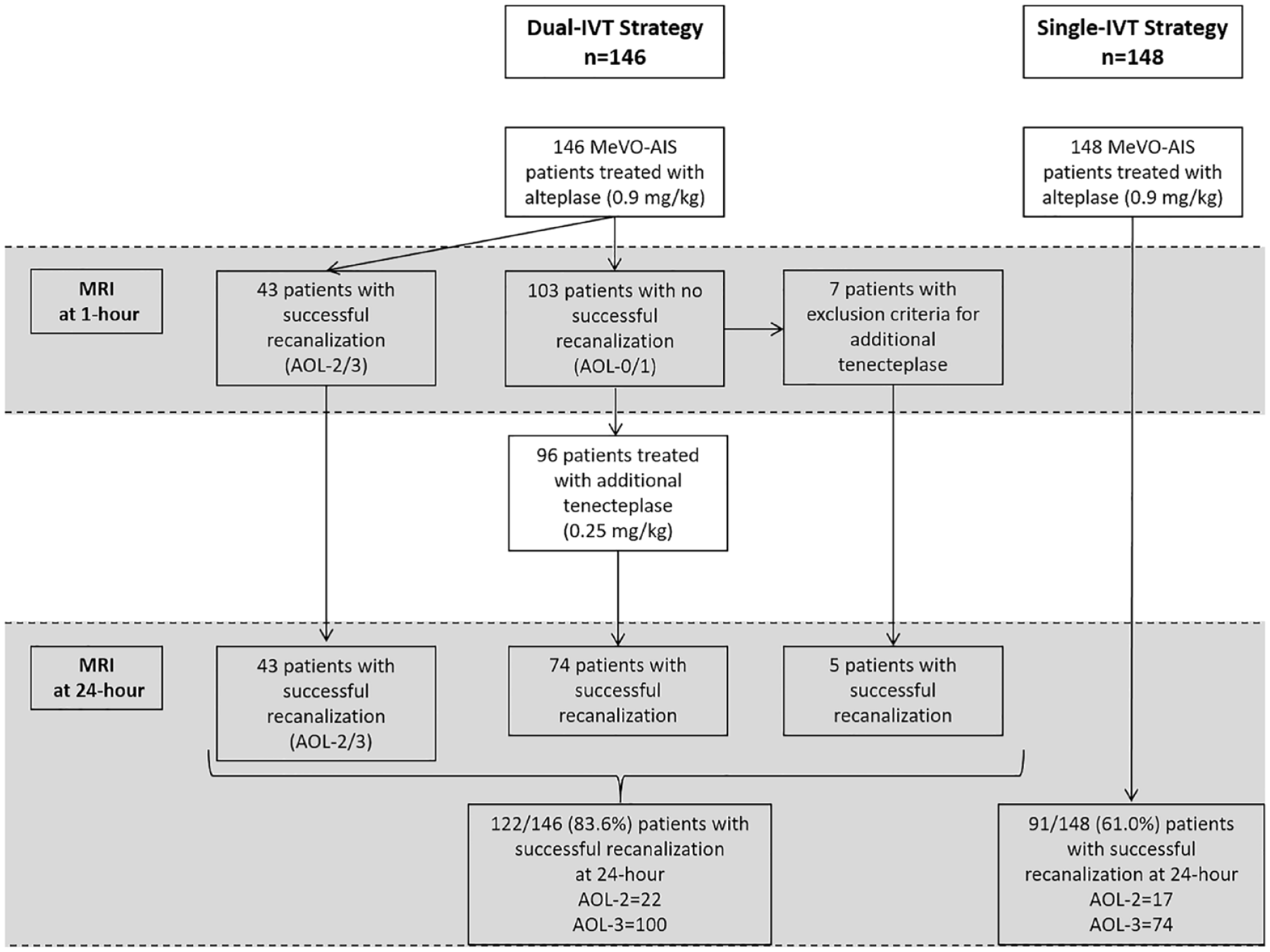
Flow diagram of the study.
PH1/PH2 and rPH rates for dual-IVT (5/146) and for single-IVT (7/148) cohorts were very similar (Table 2). Only 3/146 dual-IVT patients (two received the dual IVT regimen, one received only alteplase due to early successful recanalization) compared with 2/148 single-IVT patients, experienced sICH. One dual-IVT patient developed a psoas hematoma requiring blood transfusion, but no lasting damage ensued. Nine patients experienced minor bleedings (4 in the dual-IVT cohort vs 5 in the single-IVT cohort). There were 6 in-hospital deaths in the dual-IVT cohort versus 3 in the single-IVT cohort (p = 0.33). Only one death was related to sICH, in a patient from the dual-IVT cohort who received additional tenecteplase, but in whom a post-alteplase focal subarachnoid hemorrhage was unfortunately not seen on 1-h MRI. The 24-h NIHSS score was significantly lower in the dual-IVT cohort (1 [0–4]) than in the single-IVT one (3 [1–7], p < 0.0001), and dual-IVT patients showed more often ENI and early complete neurological recovery than single-IVT patients (63% vs 49%, p = 0.01 and 45% vs 16%, p < 0.0001 respectively). Dual-IVT compared with single-IVT patients achieved significantly better 3-month outcomes, defined as excellent (69% vs 44%, p < 0.0001) or good (79% vs 64%, p < 0.01).
Multivariable analyses (Supplemental Table 2) indicated that the dual-IVT strategy was significantly and independently associated with successful recanalization at 24-h (OR 2.78 [1.73–4.52], p < 0.0001). Factors independently associated with excellent and good outcomes at 3 months, PH1/PH2/rPH at 24-h and death at 3-month are presented in the supplementary data (Supplemental Tables 3–6).
After exclusion of proximal M2-MCA occlusions, successful recanalization at 24 h was still significantly more frequent in dual-IVT compared with single-IVT patients (85% vs 61% respectively, p < 0.01) as well as excellent outcome at 3-month (72% vs 50%, respectively, p < 0.01) (Supplemental Table 7).
Primary and secondary outcomes after matching
Propensity score matching (106 patients selected in each cohort) revealed roughly the same results. Successful recanalization at 24-h was significantly associated with dual-IVT strategy (OR = 2.67 [1.32–5.39], p < 0.01). Excellent and good outcomes were also closely associated with dual-IVT strategy (OR = 4.09 [1.95–8.57], p < 0.001; OR = 3.73 [1.68–8.3], p < 0.01, respectively) with a better delta-NIHSS at 24-h (Coefficient = 1.63 [−0.64–2.61], p < 0.01). Conversely, PH1/PH2/rPH at 24-h and death at 3-month were not associated with dual-IVT strategy (Table 3).
Efficacy and safety outcomes of patients according to the dual-IVT strategy compared with the single-IVT strategy after propensity score matching analysis (n = 212).
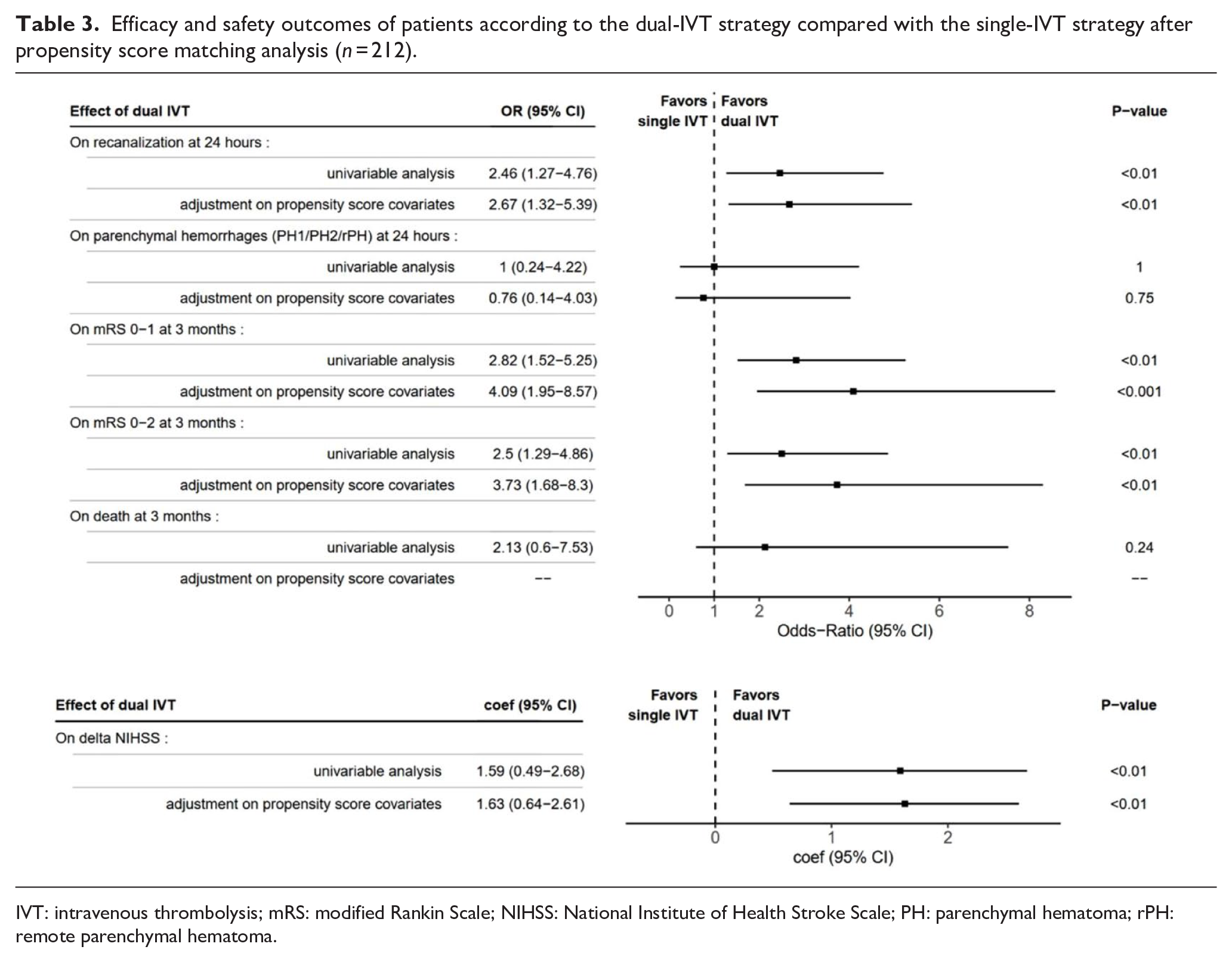
IVT: intravenous thrombolysis; mRS: modified Rankin Scale; NIHSS: National Institute of Health Stroke Scale; PH: parenchymal hematoma; rPH: remote parenchymal hematoma.
After exclusion of proximal M2-MCA occlusions, successful recanalization at 24 h remained significantly more frequent in dual-IVT compared with single-IVT patients (83% vs 66% respectively, p = 0.03) as well as excellent 3-month outcome (73% vs 54%, respectively, p = 0.01) (Supplemental Table 7).
Results of the sensitivity analysis on the whole cohort using the propensity score with the overlap weighting method were very similar (Supplemental Table 8).
Discussion
Our results showed that a dual sequential IVT strategy (alteplase 0.9 mg/kg ± tenecteplase 0.25 mg/kg) in patients with MeVOs resistant to alteplase can lead to an approximately 20% higher absolute rate of successful recanalization at 24-h. Exclusion from the analysis of proximal M2-MCA occlusions, which could be eligible for EVT, did not alter significantly the benefits of dual sequential IVT strategy. Our findings also confirmed that a better recanalization rate in MeVO-AIS could yield clinical benefits at 24-h and at 3-month. Our results provide a new argument in favor of a dose-effect relationship of IVT on recanalization at 24-hour, with a good safety of a sequential regimen as opposed to a single higher dose. 17 In addition to a higher 24-hour recanalization rate, dual sequential thrombolytic administration could also be associated with earlier artery reperfusion compared with treatment with alteplase alone. Thus, earlier and higher recanalization rate could explain the early (24-hour) and late (3-month) clinical benefits associated with dual sequential IVT in our study.
MeVO-AIS account for 25%–40% of all AIS. Based on a series of 258 patients with MeVO-AIS, 3 less than 1/3 had a “minor stroke” (NIHSS score < 6), thereby emphasizing that strategically located MeVO-AIS can be debilitating.2,3 Consistent with prior studies,3,18,19 we observed that alteplase alone achieved MeVO recanalization rates of 30% at 1-h and 64% at 24-h. Consequently, only 44% of our patients treated by alteplase alone had excellent outcome, and 37% were functionally dependent at 3 months (mRS > 2), all results which illustrate the need to develop new therapeutic strategies for MeVOs. Based on current knowledge, two options for improving the results have to be considered. The first could be to use tenecteplase rather than alteplase as a thrombolytic. In LVO, the EXTEND-IA trial showed a very early recanalization rate of 22% with tenecteplase, compared with 10% with alteplase, giving hope for better effectiveness. 20 However, our own real-life results suggest, rather than a superior effect, only a faster effect of tenecteplase, 6 while in trials comparing tenecteplase with alteplase with a clinical endpoint, no clear superiority of tenecteplase was shown.7,8 The other option could be a bridging therapy IVT/EVT. However, EVT has not yet demonstrated its relevance in MeVOs.1,2,4 Owing to the fragility of the small intracranial arteries, new EVT techniques are being developed to improve procedure performance and safety for MeVOs.21,22 A recent meta-analysis of EVT in MeVOs showed promising efficacy benefit without safety concerns, 23 and several RCT assessing EVT in MeVOs are currently ongoing.
Noticeably, no excess bleeding risk was observed in our dual-IVT cohort, despite addition of full-dose tenecteplase. Tenecteplase could be associated with lower odds of sICH than alteplase.24,25 Also, MeVO-AIS carries a lower hemorrhagic risk compared with LVO-AIS due to smaller infarcts which, moreover, spare the basal ganglia, a critical site for massive hemorrhagic transformation. 26 Moreover, dual-IVT patients were very carefully selected to be at low bleeding risk. Finally, sequential thrombolytic administration is expected to carry less risk profile than a high single dose. 17
The safety of a therapeutic strategy using a dual sequential thrombolysis has already been assessed, but with a second infusion by intra-arterial (IA) route. A meta-analysis that compared low-dose to full-dose IV alteplase followed by IA thrombolysis reported similar rates of sICH in both groups. 27 Another systematic review involving 517 patients from 16 studies found comparable incidence of death or sICH in patients treated with combined IV (0.6 or 0.9 mg/kg) + IA approaches and those treated by IV or IA thrombolysis alone. 28 More recently, the randomized CHOICE trial, which assessed the efficacy and safety of adjunctive IA alteplase (0.225 mg/kg) after successful EVT, did not find any case of sICH within 24-h, even among the 38/62 patients who previously received IVT at full dose. 29 In the CHOICE trial, IA alteplase was injected distally to the lenticulostriate branches point of origin, thus bypassing the basal ganglia, a major site of sICH following IVT and/or EVT. 29
We acknowledge several noteworthy limitations in our comparative study. Study’s retrospective design, absence of randomization and blinded radiological and clinical assessments may introduce potential major bias, possibly favoring our DIS-cohort patients.
Notably, since patients were treated in two different centers not using the same protocols, a high likelihood of center-level confounding and ascertainment bias limit the interpretation of our findings. Propensity-score matching approach partially correct selection bias, but only accounting for measurable confounders. Unmeasured differences between the two centers cannot be addressed through this method. Another difficulty in interpreting our results comes from the fact that a substantial proportion of people in both arms were actually treated exactly the same. Moreover, the relatively small sample size seriously limits the scope of our results. The size of clots was not assessed in our study, and we cannot exclude an imbalance in clot size between our two patient cohorts favoring the dual-IVT cohort. Our protocol also has potential limitations for widespread implementation. In particular, control MRI at 1 h is mandatory to identify exclusion criteria that are difficult to detect on computed tomography, and could be a limiting factor in number of hospitals. Finally, because tenecteplase is easier to use than alteplase, probably works faster 6 and has a potentially more favorable risk profile,24,25 forthcoming studies assessing this new dual-IVT strategy should probably only use tenecteplase, including for the first run of IVT.
Conclusions
Although subject to probable multiple biases, our results suggest that a strategy of dual sequential IVT in carefully selected cases of MeVo-AIS could be safe and associated with a higher rate of successful recanalization at 24 h, prompting randomized trials to test these hypotheses.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241254936 – Supplemental material for Second-dose intravenous thrombolysis with tenecteplase in alteplase-resistant medium-vessel-occlusion strokes: A retrospective and comparative study
Supplemental material, sj-docx-1-eso-10.1177_23969873241254936 for Second-dose intravenous thrombolysis with tenecteplase in alteplase-resistant medium-vessel-occlusion strokes: A retrospective and comparative study by Nicolas Chausson, Stéphane Olindo, François-Xavier Laborne, Manvel Aghasaryan, Pauline Renou, Djibril Soumah, Sabrina Debruxelles, Tony Altarcha, Mathilde Poli, Yann L’Hermitte, Sharmila Sagnier, Moussa Toudou-Daouda, Nana Rahamatou Aminou-Tassiou, Leila Bentamra, Narimane Benmoussa, Cosmin Alecu, Carole Imbernon, Léonard Smadja, Gary Ouanounou, François Rouanet, Igor Sibon and Didier Smadja in European Stroke Journal
Supplemental Material
sj-xlsx-2-eso-10.1177_23969873241254936 – Supplemental material for Second-dose intravenous thrombolysis with tenecteplase in alteplase-resistant medium-vessel-occlusion strokes: A retrospective and comparative study
Supplemental material, sj-xlsx-2-eso-10.1177_23969873241254936 for Second-dose intravenous thrombolysis with tenecteplase in alteplase-resistant medium-vessel-occlusion strokes: A retrospective and comparative study by Nicolas Chausson, Stéphane Olindo, François-Xavier Laborne, Manvel Aghasaryan, Pauline Renou, Djibril Soumah, Sabrina Debruxelles, Tony Altarcha, Mathilde Poli, Yann L’Hermitte, Sharmila Sagnier, Moussa Toudou-Daouda, Nana Rahamatou Aminou-Tassiou, Leila Bentamra, Narimane Benmoussa, Cosmin Alecu, Carole Imbernon, Léonard Smadja, Gary Ouanounou, François Rouanet, Igor Sibon and Didier Smadja in European Stroke Journal
Footnotes
Acknowledgements
We thank Ms. Edwige Lescieux for her help in collecting data.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I.S reports consultant fees for Bayer, BMS/Pfizer, Boehringer Ingelheim outside the submitted work, Medtronic, Novartis, Novo Nordisk, Bioprojet, Astra-Zeneca, Alexion and speaker activities for AstraZeneca, Bayer, BMS/Pfizer, Boehringer Ingelheim, Medtronic, Novartis, Novo Nordisk; DS reports consultant fees for BMS/Pfizer, Boehringer-Ingelheim outside the submitted work, Novartis and speaker activities for BMS/Pfizer, Boehringer-Ingelheim, Novo Nordisk, Servier, Sanofi; Other authors do not report any conflicting interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from local Ethics Committee of Centre Hospitalier Sud Francilien, no. 02023 (February 14, 2023).
Informed consent
Informed consent was not sought for the present study because of its retrospective design. However, patients had the possibility to withdraw from analyses.
Guarantor
The guarantor is Pr Didier SMADJA.
Contributorship
NC, SO, IS, and DS researched literature and conceived the study. PR, MA, DSo, SD, TA, MP, YLH, SS, MTD, NRAT, LB, NB, CA, CI, LS, GO, and FR gained data for the study. NC, FXL were involved in data analysis. NC and DS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Trial registration
ClinicalTrials no. NCT05809921 (April 12, 2023): “Dual IntraVenous Thrombolysis Approach (DIVA) in Patients With Medium-vessel-occlusion Strokes: a Retrospective Study.”
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.