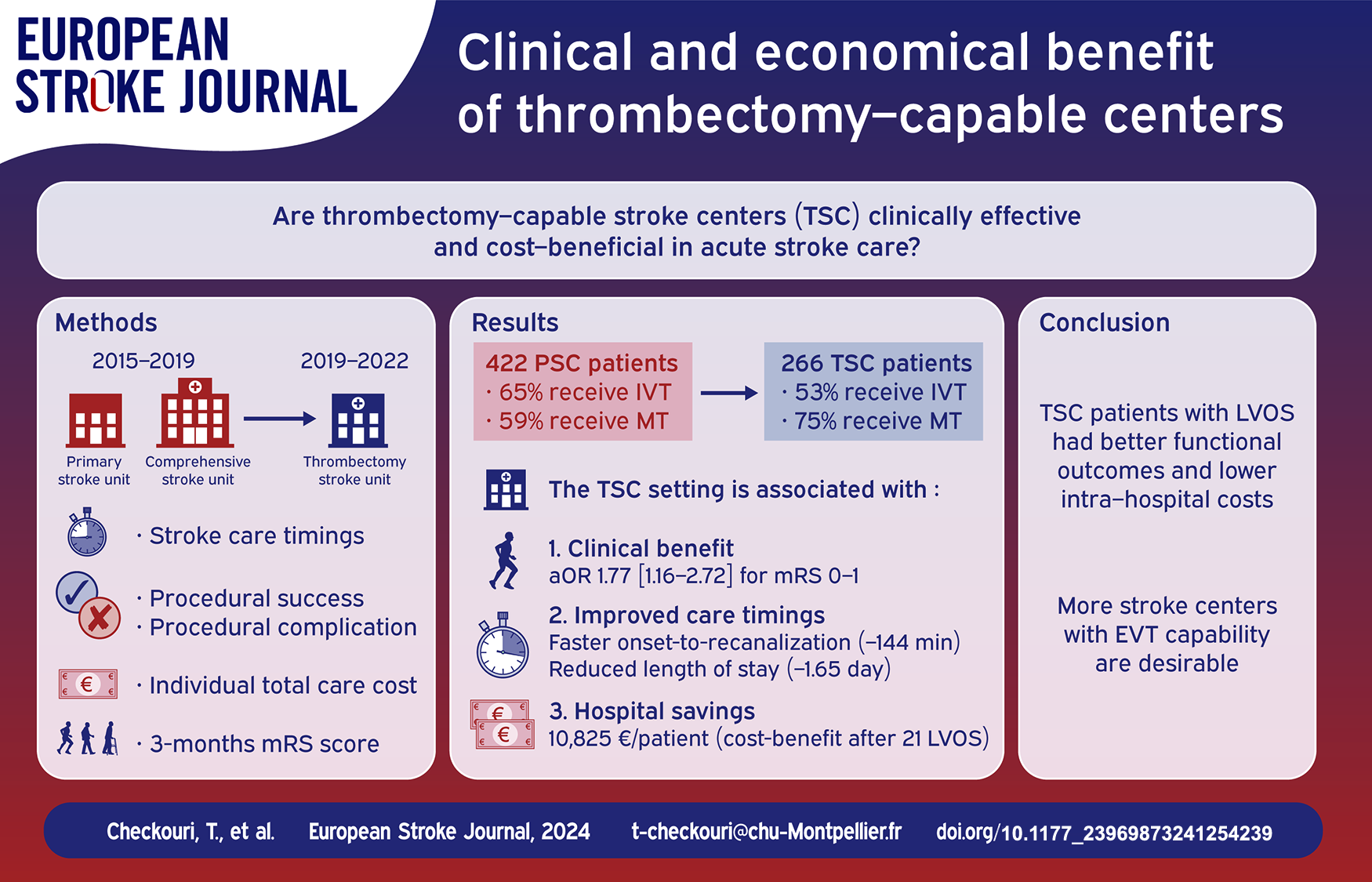
Abstract
Introduction:
Too few patients benefit from endovascular therapy (EVT) in large vessel occlusion acute stroke (LVOS), and various acute stroke care paradigms are currently investigated to reduce these inequalities in health access. We aimed to investigate whether newly set-up thrombectomy-capable stroke centers (TSC) offered a safe, effective and cost-effective procedure.
Patients and methods:
This French retrospective study compared the outcomes of LVOS patients with an indication for EVT and treated at the Perpignan hospital before on-site thrombectomy was available (Primary stroke center), and after formation of local radiology team for neurointervention (TSC). Primary endpoints were 3-months functional outcomes, assessed by the modified Rankin scale. Various safety endpoints for ischemic and hemorragic procedural complications were assessed. We conducted a medico-economic analysis to estimate the cost-benefit of becoming a TSC for the hospital.
Results:
The differences between 422 patients in the PSC and 266 in the TSC were adjusted by the means of weighted logistic regression. Patients treated in the TSC had higher odds of excellent functional outcome (aOR 1.77 [1.16–2.72], p = 0.008), with no significant differences in the rates of procedural complications. The TSC setting shortened onset-to-reperfusion times by 144 min (95% CI [131–155]; p < 0.0001), and was cost-effective after 21 treated LVOS patients. On-site thrombectomy saves 10.825€ per patient for the hospital.
Discussion:
Our results demonstrate that the TSC setting improves functional outcomes and reduces intra-hospital costs in LVOS patients. TSCs could play a major public health role in acute stroke care and access to EVT.
Keywords
Introduction
Endovascular therapy (EVT) has revolutionized the treatment of large vessel occlusion stroke (LVOS), 1 with a steady increase in the number of procedures in most countries with organized stroke care.2–4 The number of eligible LVOS patients is expected to rise with the widening of EVT indications in late time windows5,6 and low ASPECT scores.7–9 In most countries, EVT is only available in comprehensive stroke centers (CSCs), with incomplete territorial coverage resulting in more than half of LVOS patients arriving at primary stroke centers (PSC) without EVT capacity.10,11 Presentation at a PSC leads to health disparities, with patients having substantially less chances to receive EVT.12,13 Thus, complete territorial coverage is a major public health issue and one of the overreaching targets of the European Stroke plan for 2018–2030. 14
In addition, patients transferred from a PSC for EVT have a longer time from onset-to-recanalization (OTR) compared to patients arriving in a CSC, with 20% lower odds of achieving functional independence for every 60 min lost. 15 The RACECAT randomized trial compared a bypass strategy for direct admission in a CSC and failed to show clinical efficacy, but these findings are likely to vary on local conditions, as OTR may be further reduced. 16 Other paradigms to reduce OTR for LVOS patients, such as direct admission to angiography suite 17 triage with mobile stroke units 18 or drip-and-drive setting 19 are currently being investigated.
It appears that the appropriate paradigm for acute stroke treatment might depend on local conditions.16,20 A recent survey showed that the main restrictions for access to EVT in France were long transfer times from the PSC and overworking of the CSC practitionners. 21 Compared with other European countries 10 and the United States, 22 France has a relatively low number of CSCs with respect to the number of addressing PSCs. EVT procedures performed by newly trained operators have been proven safe 23 and effective, with a learning curve plateauing after 20–40 thrombectomies performed. 24 Thus, becoming a Thrombectomy-capable stroke center (TSC) has emerged as a viable alternative for PSCs.21,25
EVT has been proven cost effective at a national level in various countries and indications,26,27 but no study has evaluated its local (intra-hospital) effectiveness, as the costs to become a TSC are mainly supported by the hospital.
Our study aims to investigate the clinical efficacy and safety of EVT performed in a newly opened center, and to evaluate its cost-effectiveness at the hospital level.
Methods
Geographical considerations and care organization
Perpignan Stroke center is located in southern France and serves a population of approximately 600.000 inhabitants. Decision about intravenous thrombolysis (IVT) and/or EVT is conjointly made by the neurologist and the neuroradiologist after CT perfusion or MRI. Perpignan PSC is located at 156 km (90 min) from the nearest CSC, at the Gui de Chauliac Hospital in Montpellier. From 1st January 2015, eligible patients were transferred to Montpellier CSC for EVT, and sent back to Perpignan within 24 h after the procedure (See Figure 1).
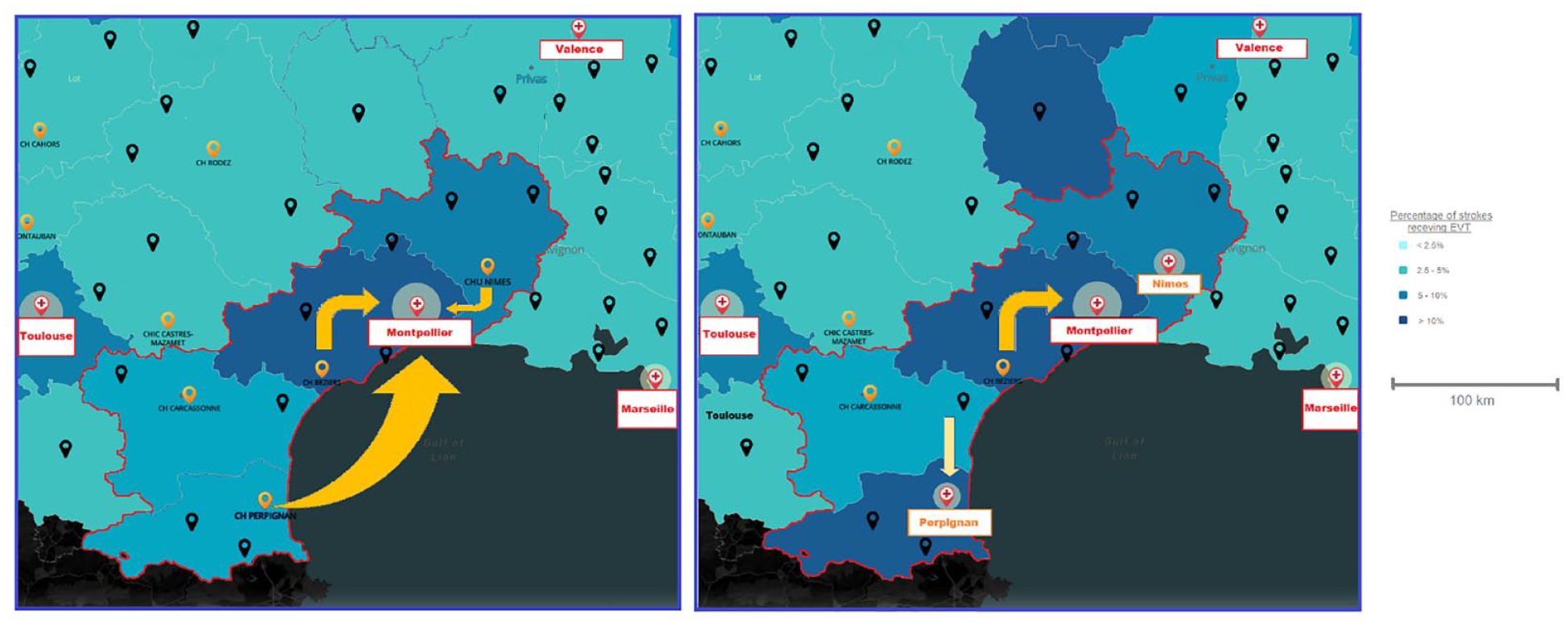
Evolution of EVT centers in the south of France, adapted from SliceMap. 13
Training and qualification of interventional radiologists
Two interventional radiologists underwent additional theoretical and practical training from 2017 to 2019, and were authorized to perform EVT after having carried out more than 30 thrombectomy procedures as principal operators, and having inserted at least five carotid stents.28,29 Perpignan became a TSC with on-site EVT from 5th December 2019, initially monday to Friday, 8am to 6pm. Patients admitted outside these hours were transferred to Montpellier’s CSC. On February 2022, the thrombectomy team was completed by another four radiologists after the same training procedure, and EVT was performed on site to all eligible patients (24 h/7 days).
EVT procedures were performed after femoral access using a “maximalist” approach using a large-bore balloon catheter, deployment of a stentretriever device and retrieval inside an aspiration catheter, with cervical balloon inflation.
Study design
We retrospectively collected clinical, demographical and stroke processing data from all patients with LVOS admitted within 24 h of symptoms onset at Perpignan Stroke center, from 1st January 2015 to 23 May 2022. We included all patients with an indication for EVT, defined by:
- Evidence of LVOS in the proximal anterior circulation (internal carotid artery, middle cerebral artery) or the basilar artery, and a National Institutes of Health Stroke Scale (NIHSS) score ⩾ 6.
- Time from symptom onset to imaging <6 h (or <24 h in patients with the mismatch criteria from the DAWN or DEFUSE-3 trials).5,6
Patients enrolled in randomized controlled trials, treated for non classical occlusion sites (vertebral artery, distal occlusions) or with significant pre-stroke disability (modified Rankin scale > 3) were excluded from this study, as they do not constitute the target population for TSCs, mainly dedicated to acute stroke treatment outside of clinical trials.
The primary efficacy endpoints were the proportion of patients achieving excellent outcome, defined by modified Rankin scale (mRS) of [0-1], and functional independence (mRS 0–2) at 3 months, after a neurological consultation. Secondary efficacy endpoints were rates of arterial recanalization before and after the procedure (Thrombolysis in Cerebral Infarction (TICI) scores of 2B or 3). Safety endpoints were clinical changes during transfer, rates of hemorragic (symptomatic intracranial hemorage, subarachnoidal hemorrage) and ischemic (arterial dissection, stent thrombosis) procedural complications, and death by any cause.
Cost-benefit analysis
The average gross loaded costs for each member of the on-site staff (interventional radiologist, anesthesiologist, radiology technician, stroke and anesthesiology nurses), stroke imaging, use of IVT, costs of transfer (air or ground transportation) and costs for the thrombectomy material were obtained by the financial department and the pharmacy of the hospital. These were added to the yearly running costs for the angiosuites and represented the hospital budget to maintain thrombectomy capability.
We then calculated the patient-centered benefit gained from thrombectomy, by obtaining the cost of hospitalization for each patient. Briefly, the daily cost of hospitalization in the neurological intensive care unit and neurology cards were obtained in the French reference manual of Etude Nationale des Couts (based on a representative sample of public and private healthcare facilities monitored over 3 years, with an extensive evaluation of clinical, monitoring, intensive care, imaging machines, logistics and medico-technical expenses, as well as non-medical expenses – e.g. laundry). We also stratified the costs using using the Groupements Homogènes de Malades method, a reference manual estimating daily added costs for each stroke patients according to the level of residual disability. These costs were multiplied by the length of hospital stay to constitute an individual, patient-dependant variable.
Statistical analysis
Quantitative variables were described as mean ± standard deviation (SD) or median (interquartile range (IQR)), as appropriate, and qualitative variables as counts and percentages. A bidirectional stepwise method was used for the selection of variables to be included in the logistic model. Specific variables of interest for stroke treatment (namely age, initial NIHSS score and pre-stroke disability) were forced into the model. The association between treatment setting and outcome was estimated through odds ratios (ORs) and their 95% confidence intervals (95% CIs), calculated in multivariate logistic regressions. For the cost-benefit analysis, multiple regression was performed at the individual level after the same variable selection procedure. All tests were two-sided and the significance level was set at p < 0.05. Analyses were performed using MedCalc (MedCalc Software Ltd, Belgium).
Results
From 1 January 2015 to 23 May 2022, we included 688 patients with an indication for EVT, among whom 422 treated in the PSC setting and 266 in the TSC setting. Baseline patient characteristics (age, sex ratio, vascular risk factors, previous disability and NIHSS score) were similar in the two cohorts. Compared with patients treated in the PSC setting, patients treated in the TSC had longer onset-to-door and door-to-needle times when IVT was used. Patients treated in the TSC were more frequently imaged with CT perfusion, more frequently had tandem occlusions, and less frequently received IVT. Baseline characteristics and univariate comparisons are shown in Table 1.
Baseline characteristics of the two treatment groups.
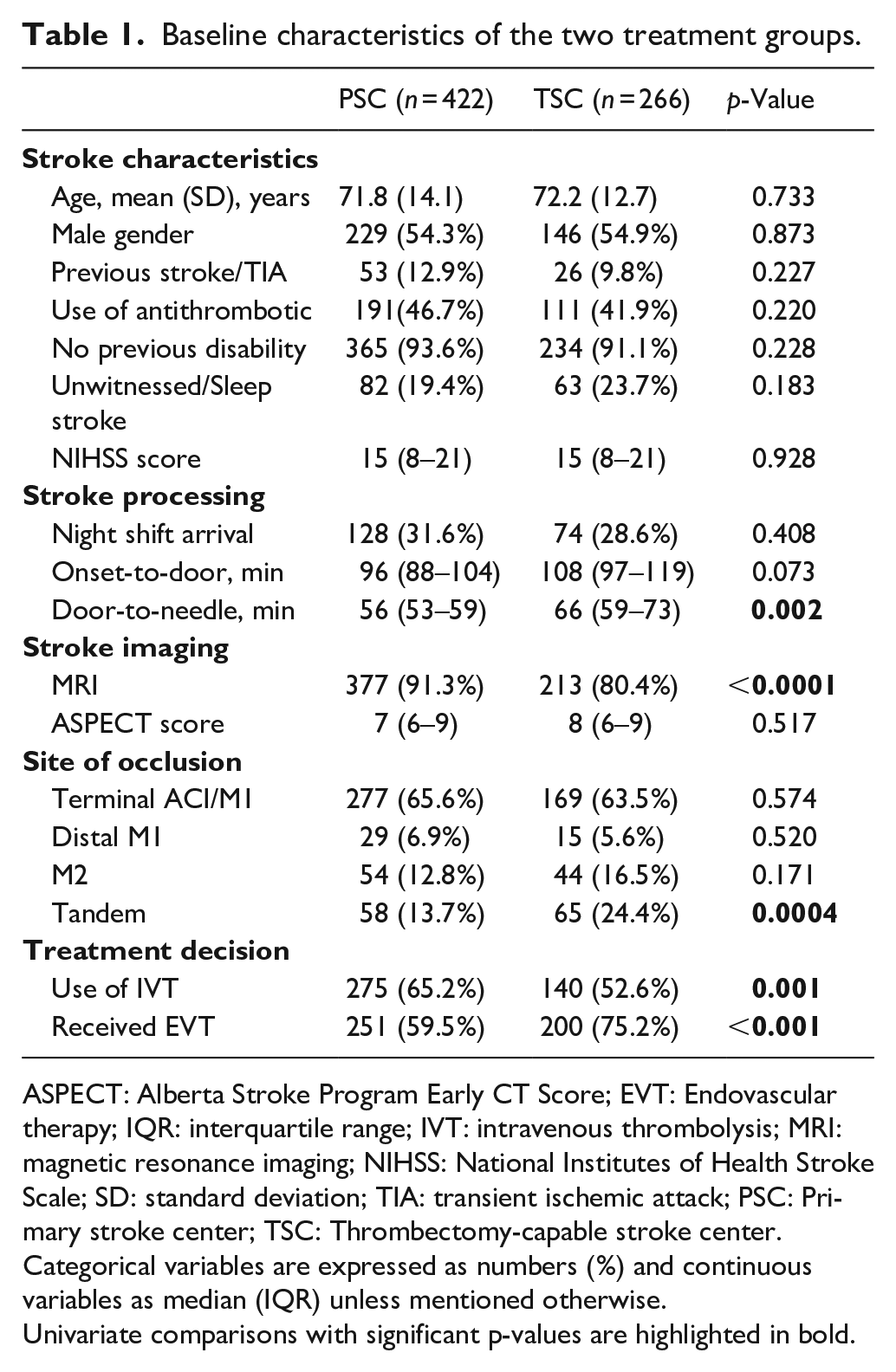
ASPECT: Alberta Stroke Program Early CT Score; EVT: Endovascular therapy; IQR: interquartile range; IVT: intravenous thrombolysis; MRI: magnetic resonance imaging; NIHSS: National Institutes of Health Stroke Scale; SD: standard deviation; TIA: transient ischemic attack; PSC: Primary stroke center; TSC: Thrombectomy-capable stroke center.
Categorical variables are expressed as numbers (%) and continuous variables as median (IQR) unless mentioned otherwise.Univariate comparisons with significant p-values are highlighted in bold.
Safety and efficacy of EVT
The following confounders were included in adjusted logistic regression: age, initial NIHSS score, pre-stroke disability, door-to-imaging and onset-to-needle times, imaging modality for assessing LVOS, presence of a tandem cervical lesion, use of bridging IVT. The TSC setting in Perpignan was significantly associated with higher odds of excellent functional outcome (mRS 0–1; aOR 1.77, 95% CI 1.16–2.72, p = 0.008), and functional independence (mRS 0–2; aOR 1.62, 95% CI [1.07–2.46], p = 0.024). Onset-to-recanalization time was 144 min shorter (95% CI 131–155 min; p < 0.0001) with the TSC setting. Patients treated in the TSC setting were less likely to receive IVT (Table 1) and had lower odds of early recanalization before EVT (3.6% in the TSC vs 12.6% in the PSC, aOR 0.29, 95% CI 0.11–0.83, p = 0.009). There were no differences in post-procedural recanalization comparing procedures conducted in Perpignan versus Montpellier (aOR 2.34 [0.82–6.60], p = 0.109, after adjustment for the same variables used in the principal analysis).
Safety outcomes were reassuring with no significant increase in procedure-associated ischemic (5.1% vs 3.7%) or hemorragic complications (3.5% vs 3.7%) nor in death by any cause (20.4% vs 16.9%) between TSC and PSC-treated patients, respectively. The results of the safety and efficacy analysis are presented in Table 2.
Results of the logistic regression for the safety and efficacy analysis.
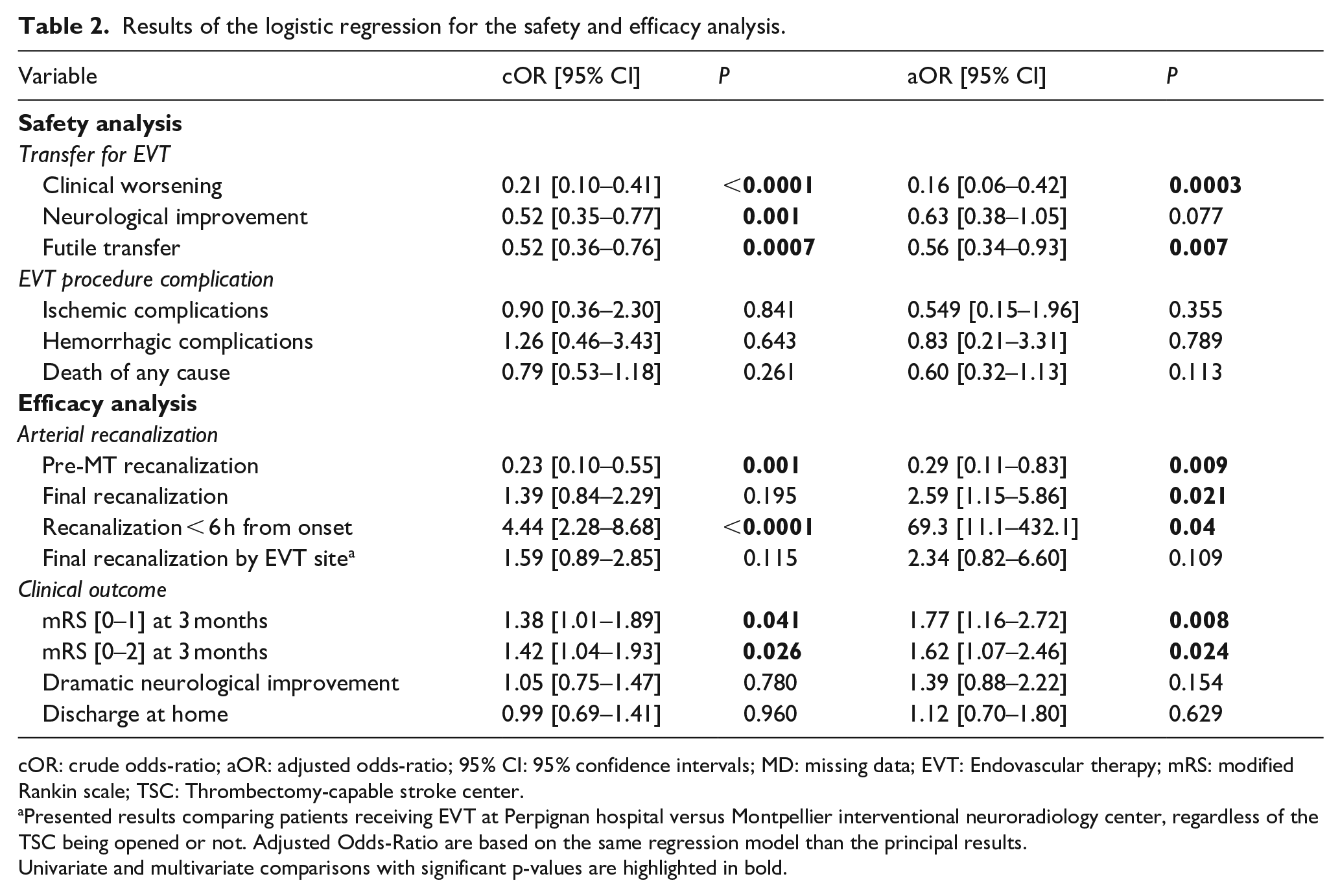
cOR: crude odds-ratio; aOR: adjusted odds-ratio; 95% CI: 95% confidence intervals; MD: missing data; EVT: Endovascular therapy; mRS: modified Rankin scale; TSC: Thrombectomy-capable stroke center.
Presented results comparing patients receiving EVT at Perpignan hospital versus Montpellier interventional neuroradiology center, regardless of the TSC being opened or not. Adjusted Odds-Ratio are based on the same regression model than the principal results.Univariate and multivariate comparisons with significant p-values are highlighted in bold.
Cost-benefit analysis
The yearly cost of maintaining the EVT capability was estimated at 157.830 € per year, and each additional EVT procedure brought a mean cost of 5.835 € for the hospital (personel salary, material expenses and EVT-associated imaging). Median reduction of transfer expenses was 3.573€ per patient.
After multiple regression, the TSC setting was associated with a mean reduction in the length of stay of −1.65 day per patient. Using individual costs-analysis, becoming a TSC was associated with a reduction of 10.825€ in hospital expenses per patient. Thus, having on-site thrombectomy was cost-effective after treating 21 LVOS patients a year. The results of the cost-benefit analysis are presented in Table 3.
Results of the cost-benefit analysis.
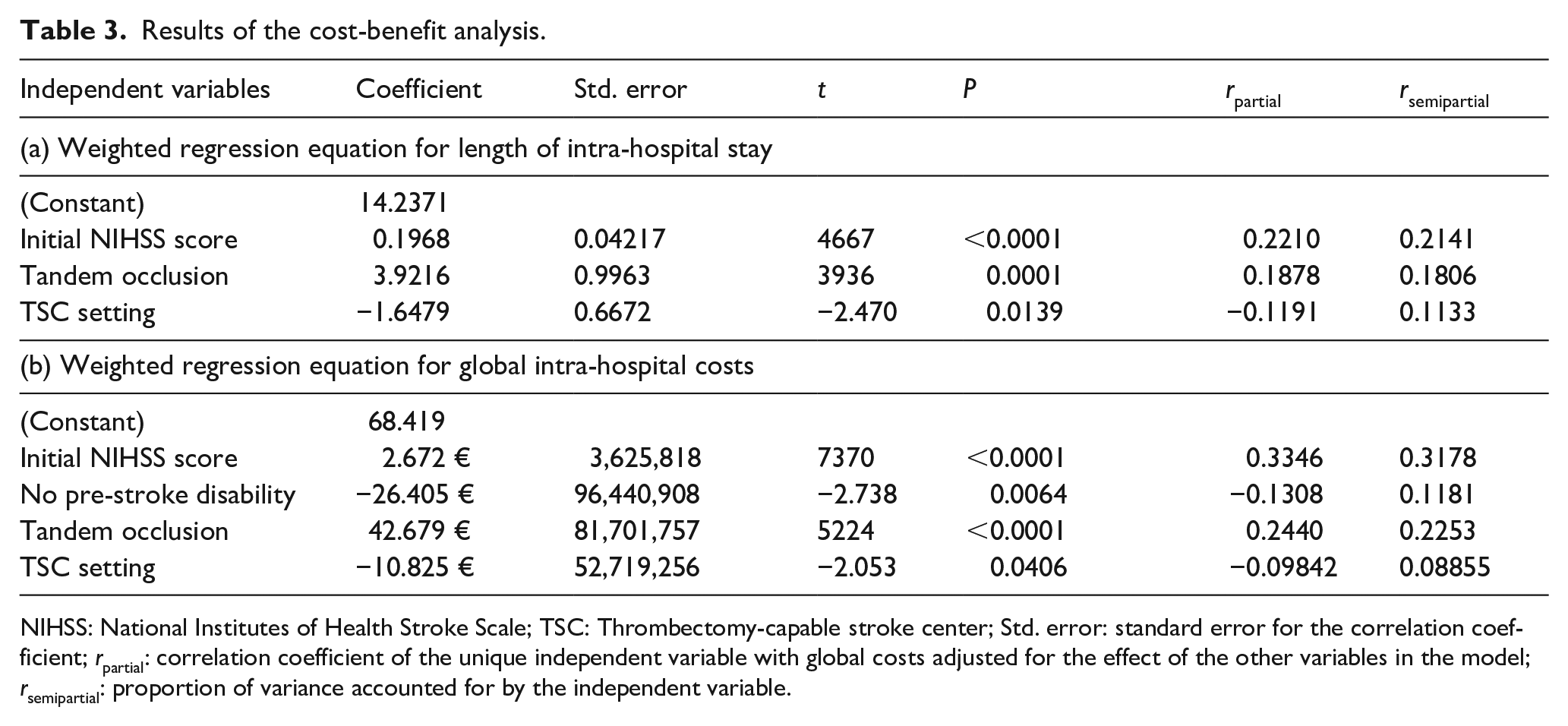
NIHSS: National Institutes of Health Stroke Scale; TSC: Thrombectomy-capable stroke center; Std. error: standard error for the correlation coefficient; rpartial: correlation coefficient of the unique independent variable with global costs adjusted for the effect of the other variables in the model; rsemipartial: proportion of variance accounted for by the independent variable.
Discussion
Our study suggests that the transformation from PSC to TSC offered LVOS patients timely access to a safe and effective procedure. Becoming a TSC greatly decreased intra-hospital costs, mainly by reducing length of stay and level of care associated with residual disability.
The initial workflow of LVOS patients assessed by mean onset-to-groin puncture time of 343 min, was markedly slower than other PSCs (260 min in the French ETIS registry, 300 min in the German NEUROSQUAD registry and 275 min in the international SWIFT-PRIME trial).15,19,30 After transformation into a TSC, Perpignan did not achieve the onset-to-groin puncture times recorded in other CSCs, but the average reduction of the OTR of 144 min is likely to be the main driver of our positive results. It favorably compares with reductions achieved by other strategies evaluated in randomized controlled trials, such as the 56-min reduction in the RACECAT trial and the 37-min reduction observed in the DIRECT-ANGIO trial.17,31
In our study, fewer patients were thrombolyzed in the TSC setting (52% of EVT-eligible patients), compared to 65% in the PSC setting. This is in line with results of the RACECAT study, with 47% of patients receiving IVT when admitted in a thrombectomy facility, compared to 60% of those admitted in a PSC. 31 Consequently, there were fewer cases of pre-EVT recanalization in the TSC setting, and a recent meta-analysis of the 6 IVT “bypass” randomized controlled trials suggested an added clinical benefit of IVT. 32 Lower use of IVT has been reported in observational studies, and though it may be partially explained by selection bias, further work is needed to understand why easier EVT access lowers the use of IVT. 33 Conversely, more patients were eligible for thrombectomy with the TCS setting (75.2% vs 59.5%) which could be partly explained by selection bias, or a widening of EVT indication due to the availability of on-site thrombectomy. Either way, this is likely to be an an important driver of our results.
In France, the number of INR centers in France was particularly low (37 EVT-capable centers for 135 referring stroke units in 2018, compared to 107 EVT-capable for 279 stroke units in Germany and 713 for 1941 in the USA),34,35 leading to a “ ceiling effect” limiting access to stroke care for LVOS patients. 21 This led to the creation of 10 new French stroke centers with various systems (drip-and-drive, TSC. . .), as well as a 30% increase in INR trainees volume. 21 In other studies, operators training for EVT procedures showed a learning curve effect, with necessity of a certain case volume to maintain efficacy.24,36,37 A recent optimization model-based study in the US have shown that transforming the top 10% referring PSCs into TSCs would lead to 7%–13% more LVOS patients having timely EVT access. 34 This is in line with the number of stroke patients receiving EVT in the Pyrénées-Orientales department, rising from 5 to 6% before the opening of Perpignan TSC to more than 10.3% thereafter. 13 Encouraging though this figure is, it needs to be strenghtened by other studies in different settings.
Our study is limited by its retrospective nature, and other possible confounding factors remain, despite our strict adjustement method. Potential bias from temporal trends due to technical improvements or better post-acute stroke care could have favored the TSC setting. In addition, the assessment of the secondary efficacy criteria (recanalization via the TICI score) was not centralized, preventing any conclusion about the learning curve of operators, or subtle differences in efficacy stratified along the TICI score. Finally, our study may be subject to indication bias, even though the absence of significant differences in night shift arrivals, NIHSS scores, occlusion sites and ASPECTS scores diminish this risk. Even so, the use of EVT only in standard thrombectomy indications could have favored a better outcome for the Perpignan hospital. The strengths of our study include the relatively large and exhaustive collection of LVOS patient data, and the low number of missing data. Our results are encouraging, mainly over the period of partial opening of the EVT center. These results are likely to be further strengthened once the Perpignan center is fully open 24 h a day.
Conclusion
Setting up a new TSC after a 2-year training has offered patients a fast, safe and effective EVT procedure leading to a significant improvement in clinical outcome, as well as a direct financial benefit for the local hospital after 21 EVT-treated patients. These positive results should encourage the opening of new EVT centers in high-volume PSCs distant from their reference INR center. TSCs could be an effective addition to the public health arsenal for equitable and widespread access to stroke care.
Footnotes
Acknowledgements
Study data are available from the corresponding author upon reasonable request
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Patients were informed they could oppose the use of their health-related data for research purposes. Ethical approval was not sought for the present study because it used only retrospectively collected anonymized data. This study was completed in accordance with the Helsinki Declaration as revised in 2013.
Informed consent
According to French legislation, as a non-interventional retrospective study of routinely acquired data, the need for written informed consent for this study was waived.
Guarantor
TC
Contributorship
TC and DS researched literature and conceived the study. DS, GF and VC were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. TC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data sharing statement
Study data are available from the corresponding author upon reasonable request
