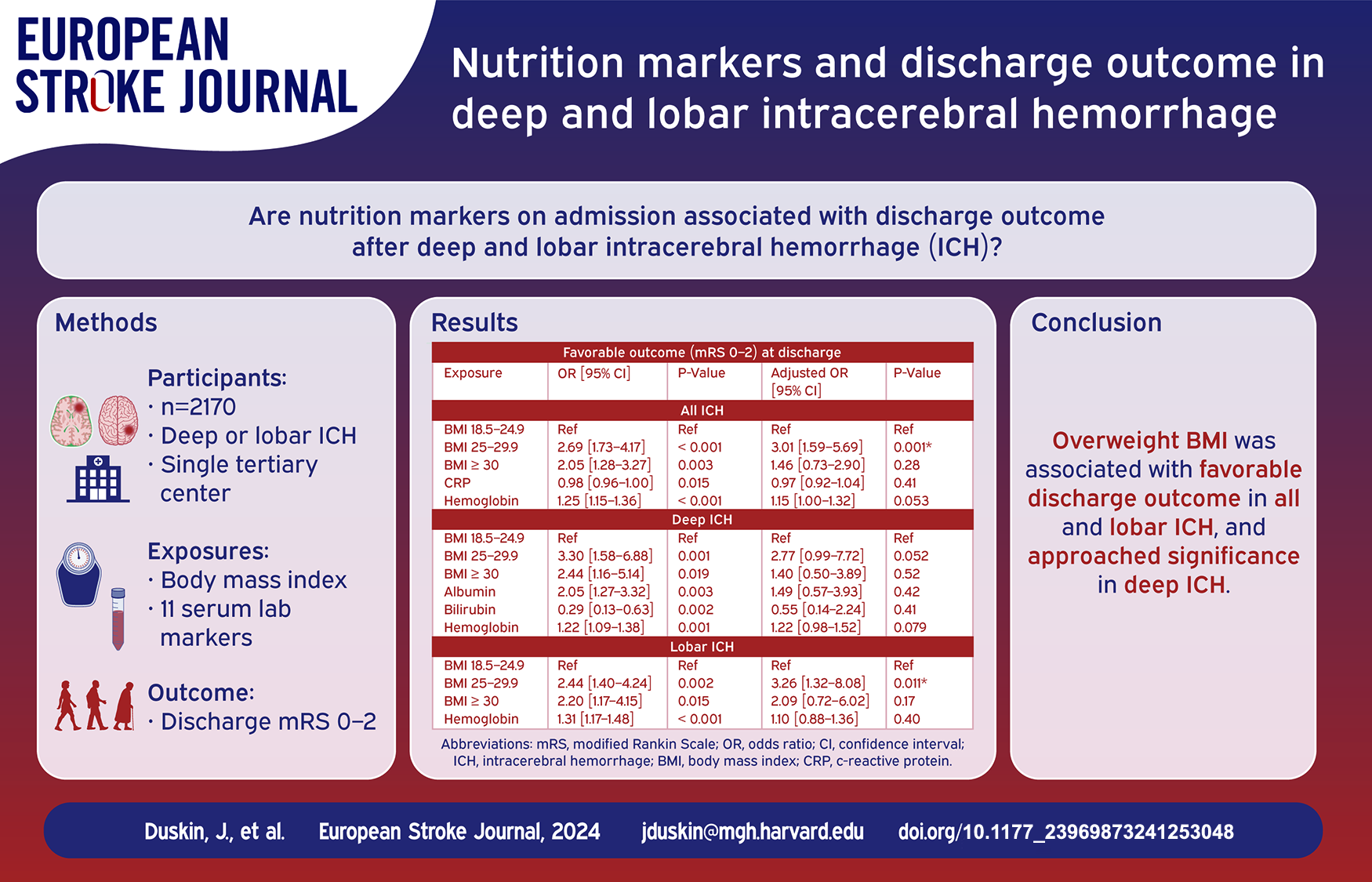
Abstract
Introduction:
Malnutrition is common in stroke patients and has been associated with poor functional outcomes and increased mortality after stroke. Previous research on nutrition status and post-intracerebral hemorrhage (ICH) outcomes, however, is limited and conflicting.
Patients and Methods:
Monocenter study of patients with spontaneous deep or lobar ICH from a longitudinal cohort enrolling consecutive patients between 1994 and 2022. Nutrition status was assessed using admission body mass index
Results:
Among 2170 patients, 1152 had deep and 1018 had lobar ICH. Overweight BMI was associated with higher odds of favorable discharge outcome in all (aOR = 3.01, 95% CI 1.59–5.69, p = 0.001) and lobar (aOR = 3.26, 95% CI 1.32–8.08, p = 0.011) ICH after adjustment for baseline differences. This association did not reach statistical significance in deep (aOR = 2.77, 95% CI 0.99–7.72, p = 0.052) ICH. No lab values were associated with functional outcome in all, deep, or lobar ICH after adjustment.
Discussion and Conclusion:
Overweight BMI was associated with favorable discharge status after ICH. These findings could inform future studies to determine whether overweight BMI has a protective effect in ICH patients.
Introduction
Malnutrition is common in stroke patients and has been associated with poor functional outcomes and increased mortality after stroke.1–4 Recent studies in patients with intracerebral hemorrhage (ICH) have found that malnutrition, defined by lower albumin, lymphocyte, and cholesterol levels, and underweight body mass index (BMI) were associated with poor functional outcome and mortality, respectively, while BMI ⩾ 25 has been associated with favorable outcome at 3 months.5–9 A conflicting study, however, found that compared to normal-weight patients, obese patients had higher risk of a discharge disposition of skilled nursing facility, long term acute care facility, or deceased. 10
One potential hypothesis for these inconsistent results is that although BMI has been shown to perform well in detecting malnutrition, it is relatively nonspecific. 11 Thus, additional use of previously studied biochemical markers of nutrition status, such as albumin, hemoglobin, folate, vitamin B12, homocysteine, iron, ferritin, c-reactive protein (CRP), total cholesterol, low-density lipoprotein, high-density lipoprotein, triglycerides, alanine transaminase, bilirubin, and uric acid could help to better identify malnutrition.7,12–16
Another hypothesis is that BMI could influence ICH severity, an important predictor of post-ICH outcomes. 17 Previous studies have had conflicting results with two showing no difference in ICH score 5 and ICH volume, 10 respectively, across BMI groups, while another showed increased ICH score and volume in underweight patients 6 on univariate analyses. Additionally, after adjusting for possible confounders, odds of hematoma expansion did not differ across BMI groups in one study, 10 but were increased in obese patients in another study. 8
A final hypothesis is that BMI may differentially affect outcomes in patients based on ICH location due to biological differences between deep hemorrhages, which are commonly due to hypertension, and lobar hemorrhages, where cerebral amyloid angiopathy can play a role. 18 A previous study showed that BMI was associated with risk of deep, but not lobar, ICH, 18 however research on how these biological differences influence outcomes is lacking.
The current study used data from the Massachusetts General Hospital ICH cohort to investigate the relationship between nutrition markers (i.e. BMI and relevant serum markers) and discharge outcome following deep or lobar ICH.
Methods
Patient recruitment and baseline data collection
We used data collected from the ongoing longitudinal ICH study at Massachusetts General Hospital (MGH). 19 This study enrolled consecutive patients 18 years or older admitted to MGH between 1994 and 2022 with spontaneous primary ICH confirmed on CT scan by stroke neurologists. At enrollment, information on demographics and socioeconomic status, personal and family medical history, and pre-ICH medication use were collected via dedicated research interview of patients and family, and supplemented with previously validated semi-automated review of electronic medical records, as previously described.20,21 ICH was classified as lobar if isolated to the cortex and as deep if selectively involving the internal capsule, thalamus, basal ganglia, or brainstem. 18 Given our aim to study differences in outcome based on ICH location, patients with multiple bleeds or cerebellar bleeds were excluded given evidence that these could be caused by hypertension and/or cerebral amyloid angiopathy, and given the strong influence of surgical intervention on cerebellar ICH survival22,23 (Figure 1). Primary intraventricular hemorrhages were additionally excluded.
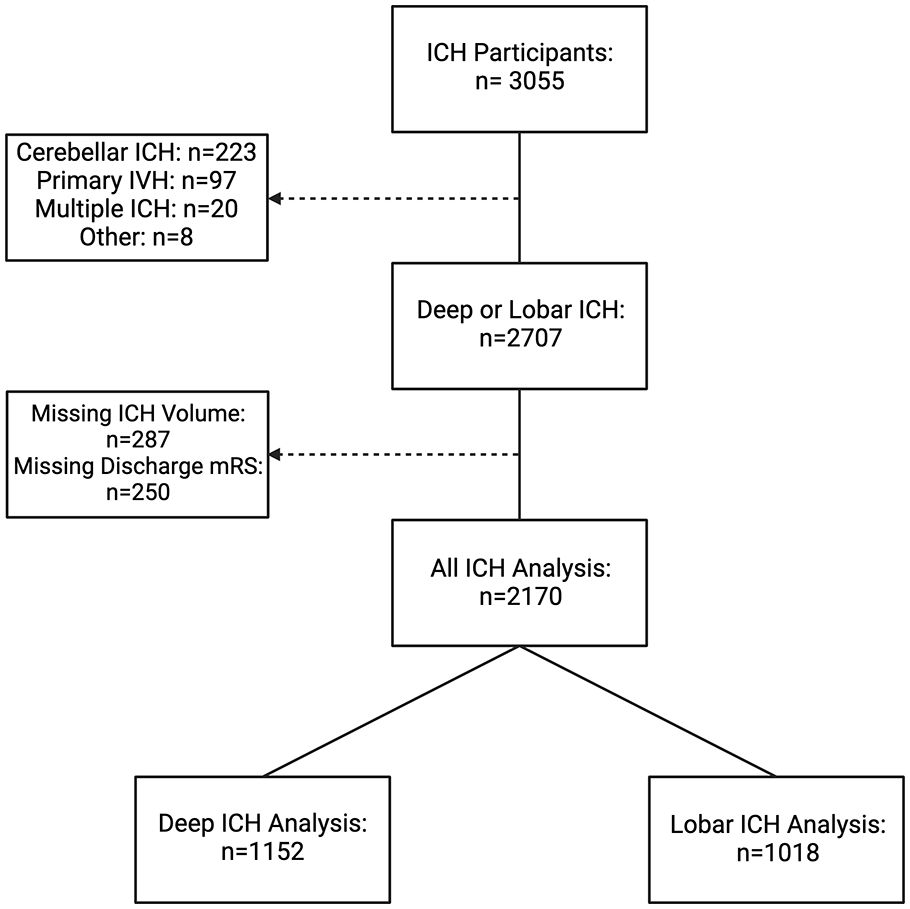
Sample size flow chart. ICH: intracerebral hemorrhage; IVH: intraventricular hemorrhage; mRS: modified Rankin Scale.
This study was approved by the institutional review board of Massachusetts General Hospital under the Biorepository for Neurological Injury (2013P000494) and ICH Database (2006P000570) protocols. Written informed consent was obtained from all participants or authorized surrogates.
Assessment of body mass index
On admission, weight was determined by bed scale and height was determined through interview of patient or next of kin. BMI was calculated by dividing admission weight (kg) by admission height squared (m2). Patients were then classified into four groups based on WHO classifications: <18.5 kg/m2 for underweight, 18.5–24.9 kg/m2 for normal weight, 25.0–29.9 kg/m2 for overweight and ⩾30.0 kg/m2 for obese. 24 BMI was analyzed as a categorical variable with the normal weight group used as the reference group for analyses.
Assessment of lab markers
Values for the following lab markers within ±7 days of admission were abstracted using Mass General Brigham Enterprise Data Warehouse and Research Patient Data Registry: albumin, vitamin B12, total bilirubin, cholesterol, c-reactive protein (CRP), ferritin, iron, folate, hemoglobin A1c, high-density lipoprotein, low-density lipoprotein, hemoglobin, mean corpuscular volume, alanine transaminase, triglycerides, uric acid, and vitamin D. In cases of multiple measurements, the lab value taken closest to admission time was used in analyses.
Statistical methods
Variable definitions
Analysis was conducted on available data. Age and all lab values were analyzed as continuous variables. ICH volume was both dichotomized into <30 and ⩾30 mL based on previous research, 25 and used as a continuous variable. Modified Rankin Scale (mRS) was assessed on discharge and dichotomized into favorable (0–2) and unfavorable (3–6) outcome.26,27
Alcohol was divided into never/rare, moderate intake (<6 drinks/week), and high intake (>6 drinks/week). Tobacco use was divided into never, previous, and current use. Education status was divided into none, 1–6, 7−9, 10−13, and ⩾14 years. Favorable pre-stroke outcome was defined as mRS 0–2 prior to ICH.
Statistical models
Initial analyses were performed in the overall cohort of patients with deep or lobar ICH (“all ICH”). Patients were then stratified by ICH location and each analysis was performed separately in the “deep ICH” and “lobar ICH” group. All continuous numerical variables were nonparametric and expressed as median and interquartile range (IQR). Baseline characteristics between favorable and unfavorable discharge outcome were compared in univariate analyses using logistic regression and chi square. Multiple testing burden was addressed using the Benjamini-Hochberg adjustment for false discovery rate (FDR) set at 0.05. There were 38 tests carried out in the all, deep, and lobar groups to assess the association between the different variables and functional outcome.
Multivariable logistic regression was then conducted separately for each nutritional marker significantly associated with discharge outcome on univariate analysis. The models of each marker and functional outcome were adjusted for covariates with p < 0.05 after correction for FDR from corresponding univariate analysis, with continuous ICH volume used in place of dichotomized volume given 0 patients with ICH ⩾ 30 mL had favorable outcome in the deep group.
To account for the potential influence of withdrawal of care on discharge outcome, we repeated multivariable analyses after excluding subjects who were transitioned to comfort care. We performed an additional analysis of the relationship between BMI and discharge outcome adjusted for FUNC score, a validated tool for predicting post-ICH functional independence at 90 days based on ICH volume, patient age, ICH location, Glasgow Coma Scale (GCS) score, and presence of pre-ICH cognitive impairment.17,28 We also performed secondary analysis to assess whether BMI was associated with dichotomized ICH volume.
All analyses were performed using Stata, version 18 (StataCorp). Missing data was not imputed.
Results
Baseline characteristics
Following exclusion of subjects with missing ICH volume or discharge mRS, 2170 patients with deep or lobar ICH were available for overall analysis (Figure 1). The lab markers vitamin D, vitamin B12, ferritin, iron, folate, and uric acid were excluded from analysis given small sample size.
Characteristics of all ICH patients with favorable (mRS 0–2) and unfavorable (mRS 3–6) outcome are shown in Table 1. Patients with favorable outcome had higher rates of overweight (47% vs 32%, p < 0.001) and obese (32% vs 29%, p = 0.003) BMI, lower median CRP (3.3 mg/dL [IQR = 7.7 mg/dL] vs 6.5 mg/dL [IQR = 20.5 mg/dL], p = 0.015), higher median hemoglobin (13.9 g/L [IQR = 2.2 g/L] vs 13.2 g/L [IQR = 2.3 g/L], p < 0.001), and lower median ICH volume (5.0 mL [IQR = 9.8 mL] vs 22.3 mL [IQR = 49.7 mL], p < 0.001) after adjustment for FDR.
Nutrition markers and baseline characteristics by functional outcome in all ICH patients.
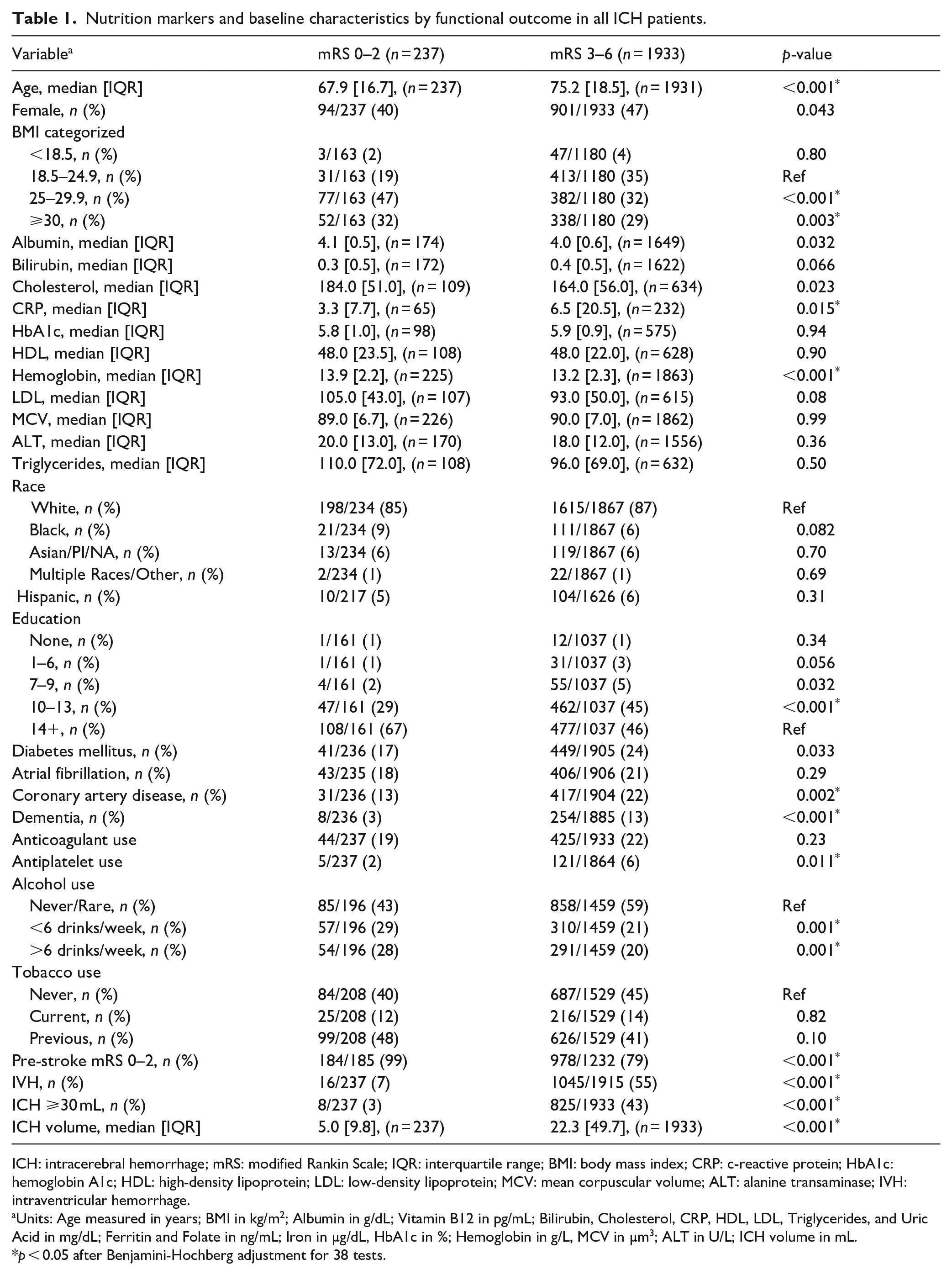
ICH: intracerebral hemorrhage; mRS: modified Rankin Scale; IQR: interquartile range; BMI: body mass index; CRP: c-reactive protein; HbA1c: hemoglobin A1c; HDL: high-density lipoprotein; LDL: low-density lipoprotein; MCV: mean corpuscular volume; ALT: alanine transaminase; IVH: intraventricular hemorrhage.
Units: Age measured in years; BMI in kg/m2; Albumin in g/dL; Vitamin B12 in pg/mL; Bilirubin, Cholesterol, CRP, HDL, LDL, Triglycerides, and Uric Acid in mg/dL; Ferritin and Folate in ng/mL; Iron in µg/dL, HbA1c in %; Hemoglobin in g/L, MCV in µm3; ALT in U/L; ICH volume in mL.
p < 0.05 after Benjamini-Hochberg adjustment for 38 tests.
A total of 1152 had a deep hemorrhage and 1018 had a lobar hemorrhage. Patients with lobar ICH had higher median age (76.9 [IQR = 15.0] vs 71.2 [IQR = 20.9], p < 0.001) and ICH volume (33.1 mL [IQR = 56.3 mL] vs 10.5 mL [IQR = 25.3 mL], p < 0.001) compared to those with deep ICH. Additionally, compared to deep, lobar ICH patients had a higher rate of favorable outcome (13% vs 9%, p = 0.006).
Characteristics of patients with favorable (mRS 0–2) and unfavorable (mRS 3–6) outcome stratified by ICH location are shown in Table 2. Patients with favorable outcome had higher rates of overweight (46% vs 30%, p = 0.001 in deep; 48% vs 35%, p = 0.002 in lobar) BMI, and lower rates of large ICH (0% vs 27%, p < 0.001 in deep; 6% vs 61%, p < 0.001 in lobar) in both deep and lobar ICH. Favorable outcome was additionally associated with higher median hemoglobin (13.9 [2.1] g/dL vs 13.4 [2.2] g/dL, p = 0.001 in deep; 13.8 [2.4] g/dL vs 13.1 [2.2] g/dL, p < 0.001 in lobar) in both the deep and lobar groups. Lower bilirubin and higher albumin were associated with favorable outcome in deep, but not lobar, ICH patients.
Nutrition markers and baseline characteristics by functional outcome in deep and lobar ICH patients.
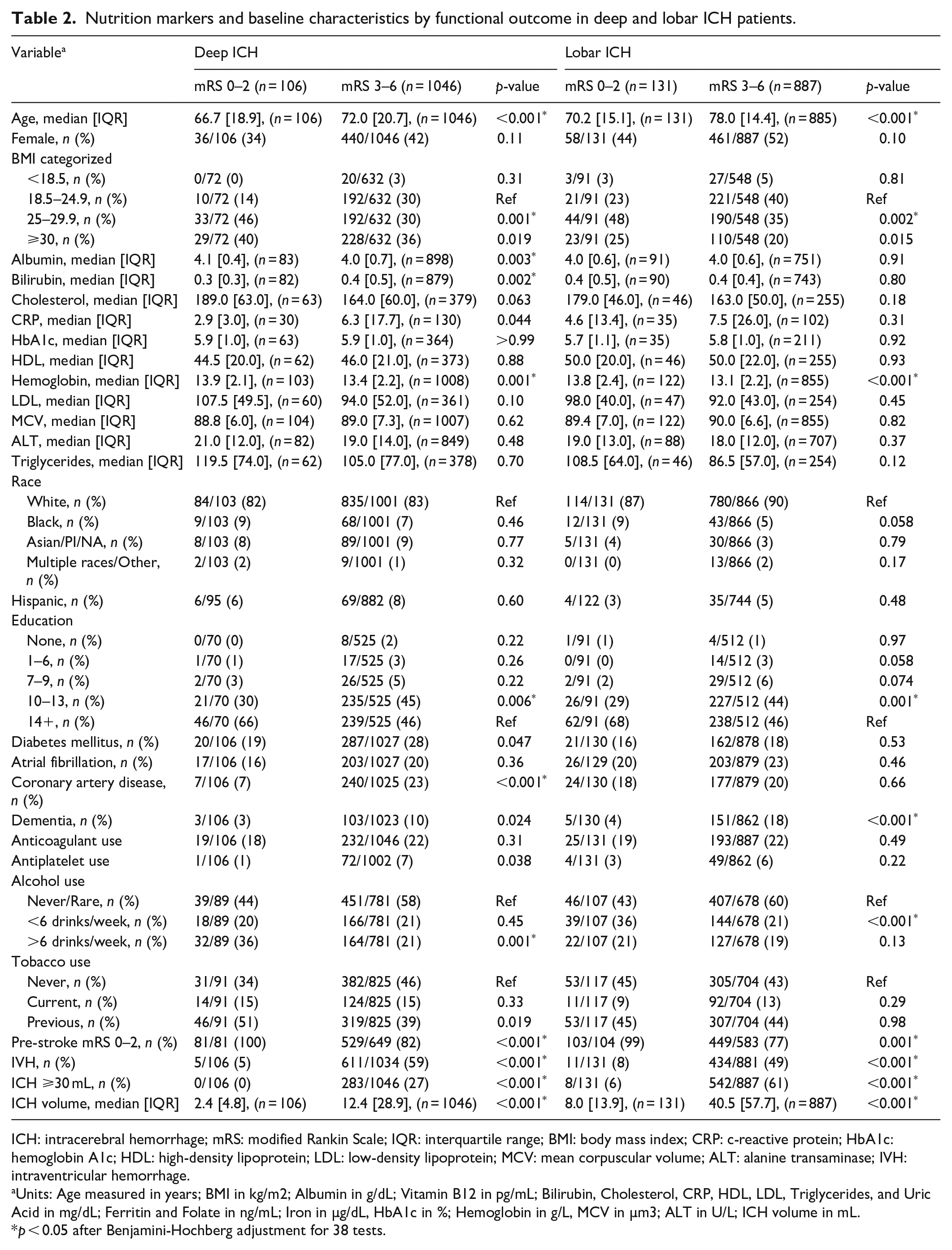
ICH: intracerebral hemorrhage; mRS: modified Rankin Scale; IQR: interquartile range; BMI: body mass index; CRP: c-reactive protein; HbA1c: hemoglobin A1c; HDL: high-density lipoprotein; LDL: low-density lipoprotein; MCV: mean corpuscular volume; ALT: alanine transaminase; IVH: intraventricular hemorrhage.
Units: Age measured in years; BMI in kg/m2; Albumin in g/dL; Vitamin B12 in pg/mL; Bilirubin, Cholesterol, CRP, HDL, LDL, Triglycerides, and Uric Acid in mg/dL; Ferritin and Folate in ng/mL; Iron in µg/dL, HbA1c in %; Hemoglobin in g/L, MCV in µm3; ALT in U/L; ICH volume in mL.
p < 0.05 after Benjamini-Hochberg adjustment for 38 tests.
Primary outcome analyses
BMI and lab markers with at least one statistically significant association (p < 0.05 after FDR correction) on univariate analysis underwent multivariable analysis to adjust for baseline differences (Table 3). Obese BMI was tested in deep ICH and lobar ICH, in addition to all ICH, given prior literature5,6,9 and significance prior to correction. Overweight BMI was associated with higher odds of favorable discharge outcome in all (aOR = 3.01 [1.59–5.69], p = 0.001) and lobar (aOR = 3.26 [CI 1.32–8.08], p = 0.011) ICH. This association did not reach statistical significance in deep (aOR = 2.77 [CI 0.99–7.72], p = 0.052) ICH. There were no associations between obese BMI or any lab marker and outcome. Increased ICH volume was associated with lower odds of favorable outcome in all (aOR = 0.93 [CI 0.91–0.95], p < 0.001), deep (aOR = 0.85 [CI 0.78–0.92], p < 0.001), and lobar (aOR = 0.91 [CI 0.88–0.94], p < 0.001) ICH.
Exposures predicting favorable outcome (mRS 0–2) at discharge on multivariable analysis in all, deep, and lobar ICH.
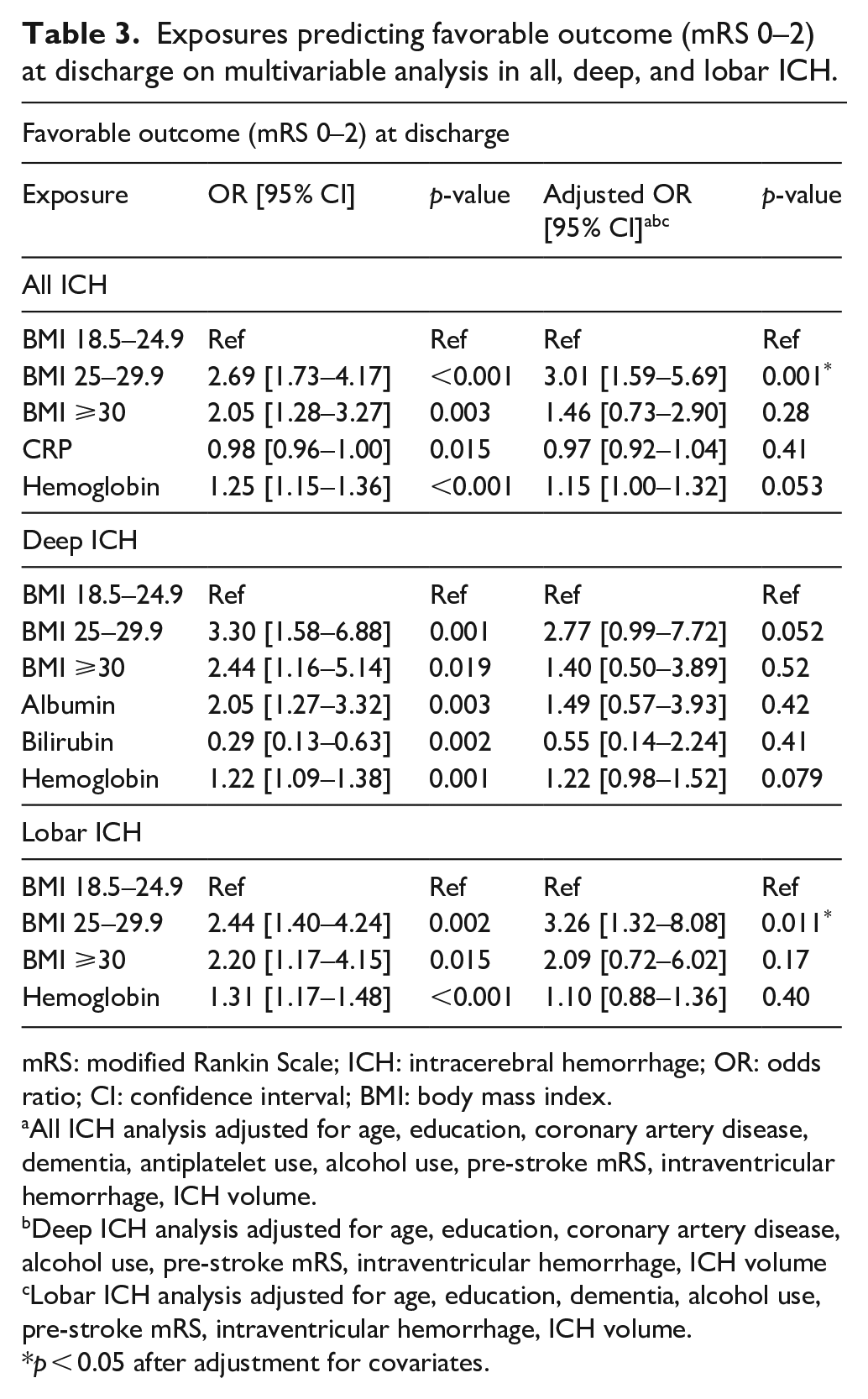
mRS: modified Rankin Scale; ICH: intracerebral hemorrhage; OR: odds ratio; CI: confidence interval; BMI: body mass index.
All ICH analysis adjusted for age, education, coronary artery disease, dementia, antiplatelet use, alcohol use, pre-stroke mRS, intraventricular hemorrhage, ICH volume.
Deep ICH analysis adjusted for age, education, coronary artery disease, alcohol use, pre-stroke mRS, intraventricular hemorrhage, ICH volume
Lobar ICH analysis adjusted for age, education, dementia, alcohol use, pre-stroke mRS, intraventricular hemorrhage, ICH volume.
p < 0.05 after adjustment for covariates.
Secondary analyses
Multivariable analysis was repeated after excluding patients who underwent withdrawal of care (n = 667). The association between overweight BMI and favorable discharge outcome remained significant in all (aOR = 3.05 [CI 1.61–5.76], p = 0.001) and lobar (aOR = 3.27 [CI 1.31–8.14], p = 0.011) ICH, and became significant in deep (aOR = 2.80 [CI 1.00–7.80], p = 0.049) ICH. There remained no association between obese BMI or any lab marker and outcome. Additional analysis of BMI and discharge outcome adjusted for FUNC score yielded similar associations between overweight BMI and favorable outcome in all (aOR = 2.24 [CI 1.38–3.62], p = 0.001), lobar (aOR = 2.40 [CI 1.28–4.50], p = 0.006), and deep (aOR = 2.14 [CI 0.99–4.64], p = 0.054) ICH (Supplemental Table 1). Finally we assessed the relationship between overweight BMI and ICH volume and found that overweight BMI was not associated with dichotomized ICH volume in all (OR = 1.02 [CI 0.77–1.35], p = 0.88), deep (OR = 1.05 [CI = 0.62-1.77], p = 0.85), or lobar (OR = 1.10 [CI = 0.77-1.58], p = 0.59) ICH.
Discussion
We utilized a large cohort of patients with spontaneous ICH to study the associations of nutrition markers and ICH discharge outcomes in deep and lobar ICH. Only overweight BMI emerged as a predictor of favorable outcome in all ICH patients. The association between overweight BMI and favorable discharge outcome remained significant in lobar ICH patients, and did not reach statistical significance in those with deep ICH. Overweight BMI was not, however, associated with ICH volume in all, deep or lobar ICH.
The association between overweight BMI and favorable outcome is consistent with two previous studies, although these studies grouped all patients with BMI ⩾ 25 together and did not separate based on ICH location.5,9 Another more recent study also found that patients with BMI ⩾ 25 had better mRS, however overweight BMI was not associated with mRS 0–2, 0–3, or overall mRS. 6 One possible reason why we observed better functional outcomes in overweight patients is that early on after ICH, patients may have both higher resting energy expenditure3,29,30 and high rates of dysphagia, 3 placing them at high risk for malnutrition. Indeed, a previous study found that baseline undernutrition predicted undernutrition at 1-week post-stroke, which was associated with poor functional outcome at 3 months. 31 Thus, overweight BMI may be protective against malnutrition without predisposing patients to the adverse events that obese patients are at increased risk for, such as deep vein thrombosis, gastrointestinal bleeding, and hematoma expansion. 8
We assessed whether the relationship between BMI and discharge outcome was influenced by withdrawal of care or by a patient’s FUNC score, which is comprised of established predictors of functional independence after spontaneous ICH (i.e. ICH volume, patient age, ICH location, GCS score, and pre-ICH cognitive impairment).17,28 Overweight BMI, however, remained a significant predictor of favorable outcome after exclusion of patients who underwent withdrawal of care and after adjustment for FUNC score. Smaller ICH volume was associated with favorable outcome, consistent with previous studies.17,25 Thus, to further understand the relationship between overweight BMI and favorable outcome, we assessed whether overweight BMI was associated with smaller ICH volume, but found no association.
Additional factors related to ICH location may influence the relationship between overweight BMI and favorable discharge status. For example, the underlying ICH etiology may play an important role in lobar ICH, as cerebral amyloid angiopathy is a common cause of lobar hemorrhages, particularly in older populations, and has been found to interact and co-occur with Alzheimer’s Disease (AD) pathology.18,32 Previous research showed that lower late-life BMI predicted increased odds of AD neuropathology in subjects without dementia, with average BMIs of 24.9 and 26.6 in the positive and negative neuropathology groups, respectively. 33 The decreased BMI was hypothesized to be secondary to pathology in brain regions modulating appetite, olfaction, taste, and weight regulation. 33
Separate factors may influence this relationship in deep ICH, which is often secondary to hypertension, leading to co-morbidity with cardiovascular diseases such as coronary artery disease, where hypertension is also a risk factor.18,34 Indeed, we found that the proportion of coronary artery disease was higher in deep ICH patients with unfavorable outcome, but did not differ in those with lobar ICH. These patients may thus be susceptible to the obesity paradox observed in cardiovascular diseases where overweight and class 1 obese patients have been found to have better prognosis than underweight or normal weight patients after incident disease, possibly due to underweight and normal weight patients experiencing a catabolic state and lean mass loss. 35 The association between overweight BMI and outcome may, however, have been weakened in these patients given that deep ICH itself is a risk factor for poor outcome, likely due to its predilection for motor fibers, suggesting a strong influence of the initial ICH injury on discharge outcome.17,36,37
Our study has limitations. While BMI has the benefit of being a widely available and commonly used tool that correlates with validated nutritional assessments, 11 it is a nonspecific marker of nutrition status affected by several additional factors including age-related loss of lean body mass and effects of hormonal concentrations. 38 Similarly, the lab markers used have the benefit of being commonly found in electronic health records, but can also be influenced by non-nutritional factors. Although we adjusted our analyses for false discovery rate, the exploratory nature of this study necessitated multiple statistical tests, which increases the risk of false positives. Thus, the results should be viewed as hypothesis generating. Last, we were only able to assess functional ability on discharge and were thus unable to assess associations with long term outcomes. Future studies utilizing more specific nutrient biomarkers and longer term outcomes are needed to better understand the relationship between nutrition and post-ICH outcomes and to assess whether targeted nutrition interventions can improve outcomes.
Conclusion
In a large cohort of ICH patients, overweight BMI was associated with favorable discharge status. These findings could inform future studies to determine whether overweight BMI has a protective effect in ICH patients.
Supplemental Material
sj-xlsx-1-eso-10.1177_23969873241253048 – Supplemental material for Nutrition markers and discharge outcome in deep and lobar intracerebral hemorrhage
Supplemental material, sj-xlsx-1-eso-10.1177_23969873241253048 for Nutrition markers and discharge outcome in deep and lobar intracerebral hemorrhage by Jonathan Duskin, Nirupama Yechoor, Sanjula Singh, Samantha Mora, Jasper Senff, Christina Kourkoulis, Christopher D Anderson and Jonathan Rosand in European Stroke Journal
Footnotes
Acknowledgements
No additional acknowledgments outside of contributing authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JD reported funding from National Institutes of Health/National Institute of Neurological Disorders and Stroke during the conduct of the study (R25 NS065743). NY reported funding from the American Academy of Neurology (project 3005). CA reported receiving grants from the NIH, the American Heart Association, Massachusetts General Hospital, and Bayer AG, personal fees from ApoPharma, and nonfinancial support from Invitae outside the submitted work. JR receives sponsored research support from the US National Institutes of Health and the American Heart Association, receives payments for consulting and expert testimony from the National Football League, consulting for Eli Lilly, and has a leadership or fiduciary role at Columbia University and Lancet Neurology. No other disclosures were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors’ work on this study was supported by funding from the National Institutes of Health (grants R25 NS065743 and R01NS093870) and American Heart Association-Bugher Foundation (814722). The funding source had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethical approval
This study was approved by the institutional review board of Massachusetts General Hospital under the Biorepository for Neurological Injury (2013P000494) and ICH Database (2006P000570) protocols.
Informed consent
Written informed consent was obtained from all participants or authorized surrogates. The data supporting this study’s findings are available from the corresponding author upon reasonable request.
Guarantor
JD
Contributorship
JD, NY, and JR were involved in study concept and design; JD, NY, SS, SM, CK, CA, and JR were involved in acquisition, analysis, or interpretation of data; JD drafted the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
