Abstract
Patent foramen ovale (PFO) is frequently identified in young patients with cryptogenic ischaemic stroke. Potential stroke mechanisms include paradoxical embolism from a venous clot which traverses the PFO, in situ clot formation within the PFO, and atrial arrhythmias due to electrical signalling disruption. The purpose of this guideline is to provide recommendations for diagnosing, treating, and long-term managing patients with ischaemic stroke and PFO. Conversely, Transient Ischaemic Attack (TIA) was not considered an index event in this context because only one RCT involved TIA patients. However, this subgroup analysis showed no significant differences between TIA and stroke outcomes. The working group identified questions and outcomes, graded evidence, and developed recommendations following the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach and the European Stroke Organisation (ESO) standard operating procedure for guideline development. This document underwent peer-review by independent experts and members of the ESO Guideline Board and Executive Committee. The working group acknowledges the current evidentiary gap in delineating an unequivocal diagnostic algorithm for the detection of PFO. Although transoesophageal echocardiography is conventionally held as the most accurate diagnostic tool for PFO identification, its status as the ‘gold standard’ remains unsubstantiated by rigorously validated evidence. We found high-quality evidence to recommend PFO closure plus antiplatelet therapy in selected patients aged 18–60 years in whom no other evident cause of stroke is found but a PFO (i.e. PFO-associated stroke). The PASCAL classification system can be used to select such candidates for PFO closure. Patients with both a large right-to-left shunt and an atrial septal aneurysm benefit most from PFO closure. There is insufficient evidence to make an evidence-based recommendation on PFO closure in patients older than 60 and younger than 18 years. We found low quality evidence to suggest against PFO closure in patients with unlikely PFO-related stroke according to the PASCAL classification, except in specific scenarios (Expert Consensus). We suggest against long-term anticoagulation in patients with PFO-associated stroke unless anticoagulation is indicated for other medical reasons. Regarding the long-term AF monitoring after PFO closure, the working group concluded that there remains significant uncertainty regarding the risks and benefits associated with the use of long-term cardiac monitoring, such as implantable loop recorders. This document provides additional guidance, in the form of evidence-based recommendations or expert consensus statements, on diagnostic methods for PFO detection, and medical management after PFO closure.

Introduction
One-third of ischaemic strokes are considered to be cryptogenic, indicating that no identifiable cause can be determined, and this tends to be more prevalent among younger patients. The standard evaluation of a symptomatic cerebral infarct relies on the patient’s history, physical examination and ancillary investigations.1,2 Typically, the work-up begins with brain imaging using MRI or CT to assess the extent and topography of ischaemia, and non-invasive assessment of intracranial and extracranial arteries using ultrasound, MR- or CT- angiography. Routine evaluation also includes assessment of cardiac rhythm and structure by 12-lead ECG, long-term monitoring or inpatient telemetry, transthoracic echocardiography (TTE) or transoesophageal echocardiography (TOE), and laboratory tests for haematology, coagulation, and blood chemistry. In cases where routine tests do not reveal common causes of stroke, such as dissection in younger individuals or significant supra-aortic atherosclerosis, atrial fibrillation (AF), or small vessel disease in older patients, it is advisable to broaden the diagnostic spectrum to include testing for a patent foramen ovale (PFO). Indeed, the persistence into adulthood of a PFO, a normal embryological communication between the cardiac atria, 3 has progressively emerged as being causally involved in 10% of all strokes in patients aged 18–60 years. 4 The involved pathophysiology can be paradoxical thromboembolism through the PFO and/or thrombus formation in the PFO leading to cerebral or systemic embolisation. 5 However, despite a prevalence of 25% in the general population, 6 a small minority of individuals with a PFO will develop a PFO related ischaemic stroke in their lifetime. In addition, the rate of recurrence while on medical treatment in patients with no other apparent cause of stroke other than a PFO is low, approximately one event per 100 person-years.7,8 This corresponds to a cumulative incidence of 4.6% after 3.8 years of follow-up in the available randomised trials. 8
Therefore, the major challenge is the identification of patients in whom a PFO can be considered to be the cause of stroke, as inappropriate treatment may be potentially harmful. 4 To achieve this aim, an accurate diagnosis of PFO is the first step. Contrast-enhanced transcranial Doppler (c-TCD), TTE, and/or TOE represent established methods for detecting a right-to-left shunt (RLS) and/or a PFO. However, which of these yields the best diagnostic accuracy is still unclear. 9 After the diagnosis of PFO, structural features can help defining PFO with a causal role in stroke, defined as PFO-related stroke. The first is the presence of an atrial septal aneurysm (ASA), a bulging of the atrial septum into the left and/or right atria, which can facilitate a large inter-atrial shunt which is the second highest-risk feature. 10
Two risk scores have been proposed to assist clinicians in assessing the likelihood of stroke causality from a PFO in patients with cryptogenic stroke, in conjunction with a comprehensive clinical assessment. 7 The first one, Risk of Paradoxical Embolism (RoPE), mainly includes clinical features 11 while the second, PFO-Associated Stroke Causal Likelihood (PASCAL), is a classification system that includes both the RoPE score and the anatomical features mentioned above. 7
After a PFO-associated stroke, secondary prevention is required, with options including antiplatelet therapy, anticoagulation, and percutaneous closure of the PFO. 8 The latter option is based on mechanical prevention of paradoxical embolism and/or local thrombus formation. However, PFO percutaneous closure has been a controversial therapy for most of the past three decades. In 2017, randomised clinical trials (RCT) have demonstrated the superiority of device closure plus medical therapy over medical therapy alone in reducing stroke recurrence in adults up to 60 years of age with no identified alternative cause of stroke. 12 A recent individual patient data meta-analysis (IPDMA) of available RCTs showed a 60% relative risk reduction of recurrent stroke with PFO closure in these patients, although the absolute risk reduction was small (2-year ARR: 1.7%). 7 Conversely, it is worth noting that there have been more frequent reports of AF following PFO closure. However, it is important to highlight that this increase in AF occurrence was primarily observed within 45 days following the closure procedure. 13
The aim of this guideline document is to provide recommendations for physicians on the diagnosis and management of PFO-associated stroke.
Methods
This guideline was prepared according to the ESO standard operating procedure (SOP) for guideline development, 14 which is based on the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) framework. 15 The ESO Guideline Board invited two chairs, one neurologist (VC) and one cardiologist (CP), who established a Module Working Group (MWG) consisting of 7 stroke specialists (AHAR, HM, EK, SW, DT, PC, GT), 2 cardiologists (LS, CP) and 2 methodologists (SH, AL). The Guideline Board and the ESO Executive Committee approved the composition of the working group. The full disclosures of all MWG members are listed in Supplemental Table 1.
The MWG developed a list of topics and corresponding questions of greatest clinical interest. Three overarching themes were identified: PFO diagnosis, treatment of PFO-associated stroke and post-procedural management. Questions to be addressed by the MWG were formatted using the Population, Intervention, Comparator, and Outcome (PICO) approach and reviewed by two external reviewers and members of the ESO Guideline Board and Executive Committee. PICO questions on the same subjects were grouped under overarching themes to improve readability. Members of the MWG rated the outcomes as critical, important, or of limited importance according to GRADE criteria. The final decision on outcomes used a Delphi approach. Results of the outcome ratings are presented below. The experts of the MWG discussed and decided by consensus on the PICO questions to be addressed and on the outcomes of interest during virtual meetings. No face-to-face meetings were organised due to the COVID-19 pandemic.
Literature search
For each question, systematic searches of the MEDLINE, EMBASE, CINAHL and SCOPUS databases, covering the period from the inception of each database to June 2023 were conducted by the ESO Guidelines methodologists. We conducted the research with Medical Subject Headings (MeSH) terms (‘Foramen Ovale, Patent’ OR ‘Patent Oval Foramen ‘ OR ‘Oval Foramen, Patent’ OR ‘Patent Foramen Ovale’) AND (‘Stroke’ OR ‘Cerebrovascular Accident’ OR ‘Cerebrovascular Accidents’ OR ‘CVA’ OR ‘CVAs’ OR ‘Cerebrovascular Apoplexy’ OR ‘Apoplexy, Cerebrovascular’ OR ‘Vascular Accident, Brain’ OR ‘Brain Vascular Accident ‘ OR ‘Brain Vascular Accidents’ OR ‘Vascular Accidents, Brain ‘ OR ‘Cerebrovascular Stroke’ OR ‘Cerebrovascular Strokes’ OR ‘Stroke, Cerebrovascular’ OR ‘Strokes, Cerebrovascular’ OR ‘Apoplexy ‘ OR ‘Cerebral Stroke’ OR ‘Cerebral Strokes’ OR ‘Stroke, Cerebral’ OR ‘Strokes, Cerebral’ OR ‘Stroke, Acute’ OR ‘Acute Stroke’ OR ‘Acute Strokes’ OR ‘Strokes, Acute’ OR ‘Cerebrovascular Accident, Acute’ OR ‘Acute Cerebrovascular Accident’ OR ‘Acute Cerebrovascular Accidents’ OR ‘Cerebrovascular Accidents, Acute’) AND Closure’.
Clinicians of the MWG independently screened titles, abstracts, and full texts for potentially relevant studies for each PICO question. We prioritised RCTs, but where data were limited, we also considered observational studies, and systematic reviews or meta-analyses of observational studies. We excluded publications with only conference abstracts available, narrative reviews, single case reports, or comments.
Data extraction, risk of bias assessment of individual studies and meta-analysis
The ESO methodologists performed data extraction and analysis. The corresponding authors of eligible studies were contacted if relevant data were not reported in original publications. In case of no response, the co-authors of the study were also contacted. If no answer was received, data were considered missing.
The risk of bias of each included RCT was assessed with the Cochrane risk of bias (RoB-2) tool. The ROBINS-I tool was used to assess the risk of bias for observational studies. 16 The risk of bias for studies included in the analysis for the diagnostic PICOs was assessed with the Quality Assessment of Diagnostic Accuracy Studies (QUADAS)-2 tool. 17 Where appropriate, fixed or random-effects meta-analyses were conducted using Review Manager (RevMan) or Stata softwares. The MetaDTA ShinyR app was used for bivariate meta-analyses of diagnostic test accuracy.
All results were presented as estimates of effect with associated 95% confidence intervals (95% CIs). Statistical heterogeneity across studies was assessed using the I2 statistic, and classified as moderate (⩾30%), substantial (⩾50%), or considerable (⩾75%). 18 The I2 statistic typically quantifies heterogeneity in meta-analyses, but it was not employed for the diagnostic test accuracy meta-analyses. This is because a certain degree of heterogeneity is anticipated in these studies, and I2 or similar metrics are rarely used in such contexts.19,20
Evaluation of the quality of evidence and formulation of evidence-based recommendations
Two or three MWG members graded the evidence and wrote the recommendations for each PICO question. As recommended, the evidence synthesis did not use a quality ‘score’ threshold but classified the overall risk of bias at the study level and then in aggregate. 21 The results of meta-analyses were imported into the GRADE Pro Guideline Development Tool (McMaster University, 2015; developed by Evidence Prime, Inc.) For each PICO question and each outcome, the following were considered: risk of bias based on the type of available evidence (randomised or observational studies); considerations on the inconsistency of results; indirectness of evidence, imprecision of results, and other possible bias. GRADE evidence profiles/summary of findings tables were generated and used to prepare recommendations. The quality of evidence was graded as high, moderate, low, and very low. Strengths of recommendations were graded as strong when the desirable effect of an intervention clearly outweighed the undesirable effects or clearly did not, or weak when the trade-off was less certain, either because of low-quality evidence or because the evidence suggested that desirable and undesirable effects were more closely balanced.22,23
‘Evidence-based recommendations’ were based on the GRADE methodology. The recommendations’ direction, strength, and formulation were determined according to the GRADE evidence profiles and the ESO-SOP. 24 All working group members critically revised all recommendations, and discrepancies in grading and recommendations were discussed during regular virtual conferences.
Additional information paragraphs and expert consensus statements
For each PICO question, ‘Additional information’ paragraphs could be added to provide more details on RCTs mentioned in the ‘Analysis of current evidence’ paragraph, summarise observational study results, or provide information on ongoing or future trials. Finally, Expert Consensus Statements were added where the MWG considered that there was insufficient evidence to make Evidence-based Recommendations and where practical guidance for routine clinical practice was needed. The Expert Consensus Statements were based on a Delphi approach and voting by all clinicians of the MWG. Importantly, these Expert Consensus Statements should not be regarded as evidence-based recommendations since they only reflect the opinion of the writing group.14,25
Results
Part 1: PFO DIAGNOSIS
In this section, we address the following
Which diagnostic test should be used to detect PFO-related right-to-left shunt in patients with cryptogenic stroke?
Accurate diagnosis of PFO-related RLS and PFO characteristics in cryptogenic stroke patients is of paramount importance for risk stratification and therapeutic decision-making. The most commonly used techniques for the detection and evaluation of RLS are contrast-enhanced (c-) transcranial doppler (c-TCD), transthoracic echocardiogram (c-TTE) and transoesophageal echocardiogram (c-TOE).
A large shunt is usually defined by the appearance of >20 microbubbles in the left atrium within 3 cardiac cycles after opacification of the right atrium at c-TTE, >20 bubbles at c-TOE, and >10 high-intensity transient signals (HITS) at c-TCD.26–29 During c-TCD, the numbers of hits can counted using bilateral or unilateral imaging by monitoring either the right or left MCA. 30
c-TOE allows for unparallelled visualisation of the interatrial septum and measurement of the size of the PFO. However, TOE renders Valsalva manoeuvre difficult with sub-optimal accuracy and problematic reproducibility. Nevertheless, because of its ability to directly visualise the PFO, TOE has been considered the conventional reference for any PFO-related cardiac assessment and has been used as such in the following analyses.
c-TCD allows for a comfortable procedure for the patient with an easily reproducible Valsalva manoeuvre but is limited by the availability of a good cranial window for ultrasounds and by the impossibility to determine the RLS anatomical location (i.e. cardiac or extracardiac shunting). c-TTE can provide a widely available and easily accessible technique for the detection of PFO-related RLS. ASA is defined as a septum primum excursion of at least 10 mm from the plane of the atrial septum into the right atrium and/or left atrium and is a critical piece of information for therapeutic decisions. 28
Analysis of current evidence
The literature search identified 34 observational studies evaluating the diagnostic accuracy of
Twenty-three studies included patients after acute ischaemic stroke and/or transient ischaemic attack (TIA).38,47,51 The remaining studies also included patients with various other medical conditions such as migraine, vertigo, syncope, myocardial infarction, and peripheral embolism.34,38,43–45,47,49,53,59,60
Acute ischaemic stroke patients included in this analysis presented with suspected cardioembolic stroke38,47,51 or cryptogenic stroke.32,35,39,41,42,48,52,59,60,65 Cryptogenic stroke was diagnosed when no underlying stroke cause (other than a PFO) could be identified during acute stroke assessment.
Nine studies included patients with various stroke aetiologies.33,34,44,46,54–56,64
Thirteen studies did not describe any aetiological workup and classification.30,31,36,37,40,43,45,49,50,53,57,58,63
Most studies (N = 16) were conducted before 2000, another seven were conducted before 2010, and only 11 after 2010 (Supplemental Table 2).
The studies used to address this PICO question comprised a total of 3133 patients with a mean age of 48 ± 9 years. Among the analysed patients, 1372 (44%) were women; 2280 (73%) had acute ischaemic stroke, 329 (11%) had TIA, and 50 (2%) had clinically silent cerebral infarcts; 199 (6%) patients had migraine and 17 (0.5%) had vertigo (Supplemental Table 2). In this population, PFOs were detected in 1513 (48%) patients using the conventional standard method of TOE (Table 2).
The RLS was graded following three different criteria: the International Consensus Criteria 29 in 11 studies,33,38,47,48,51,53,56,59,60,63,64 the Spencer et al. 66 logarithmic scale in five studies.42,51,52,55,58 Twelve studies used 1–5 HITS as a threshold to diagnose RLS.35–37,40,41,43–46,49,50,54 These studies were performed before the publication of the consensus recommendations on RLS determination by TCD. 29 Two studies used more than 5 HITS as the threshold,32,43 two other studies31,39 used at least 10 HITS to confirm RLS. Two studies did not describe their criteria.30,57
The Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) of the studies included in the analysis is shown in Figures 1 and 2. Fourteen (41%) of the 34 studies reported including consecutive patients.32–35,41–44,47,48,51,54,59,65 No inappropriate exclusions after recruitment were reported, and no risk of bias in patient selection could be detected.

Risk of bias of individual studies included in the analysis of PICO 1.1 using the QUADAS-2 tool.
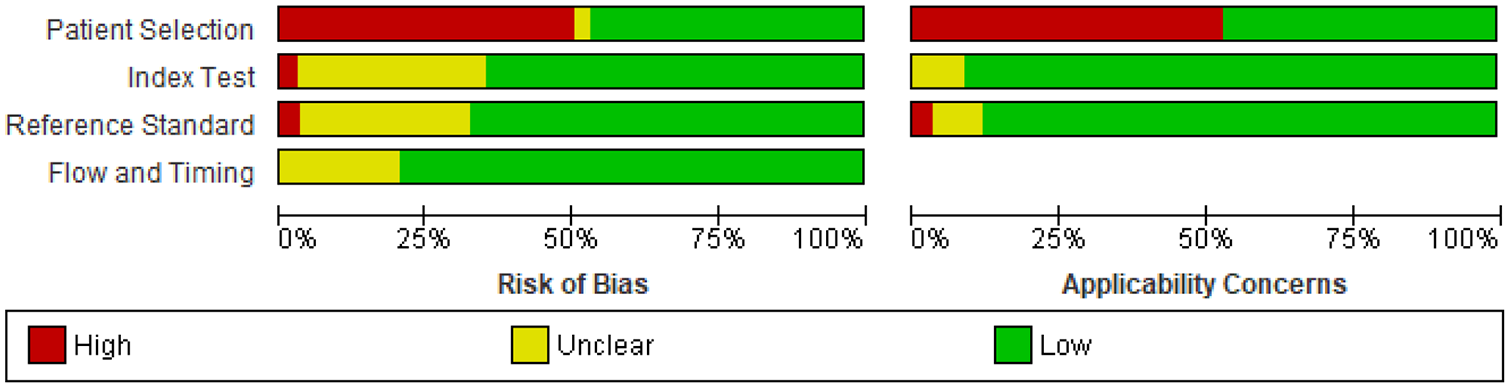
Risk of bias summary for PICO 1.1.
In 20 (59%) studies, clinicians who performed the c-TCD examinations as index test, were blinded to the results of the c-TOE (reference test) results.31–35,38,40,44–48,50,52–54,56,59,63,64 Four of these studies (12% of all) used a double-blind approach, with TOE examiners also blinded to the results of the index test.47,48,52,59
Valsalva manoeuvre was performed before contrast injection in 12 studies.30,31,33,35,41,42,45,51,54,57,60,63 In 16 studies, Valsalva manoeuvre followed contrast injections either immediately,38,47,53 or 5 s later,32,36,37,44,46,48,51,52,56,59,64 or when right atrium opacity was detected.39,43 The timing between contrast injection and Valsalva manoeuvre was not reported in six studies.35,40,50,55,58
The contrast medium consisted of air-agitated saline (AS),30,32,35,47,53–57 a mixture of blood and saline agitated with air (BAS),42,48,49,51,52,59,60,65 a specific echo contrast compound,33,36,37,39–41,43–46,50,63,64 a combination of these31,34 or was not clearly reported. 52 When reported, the duration of microbubble detection ranged from 5 to 40 s.
The risk of bias in flow and timing between index tests (TCD) and reference tests (TOE) was unclear in 16 (47%) of the 34 studies.31–34,36,37,40,54,56,60,63–65,67 The other studies reported times ranging from simultaneous assessment,30,35,38,39,43,44,46,49,50,53,57,58 to the same day45,60 within 48 h or the same week42,48 or with a median delay of 17 days. 50 In all studies, all analysed patients received the index and reference tests (Supplemental Table 2).
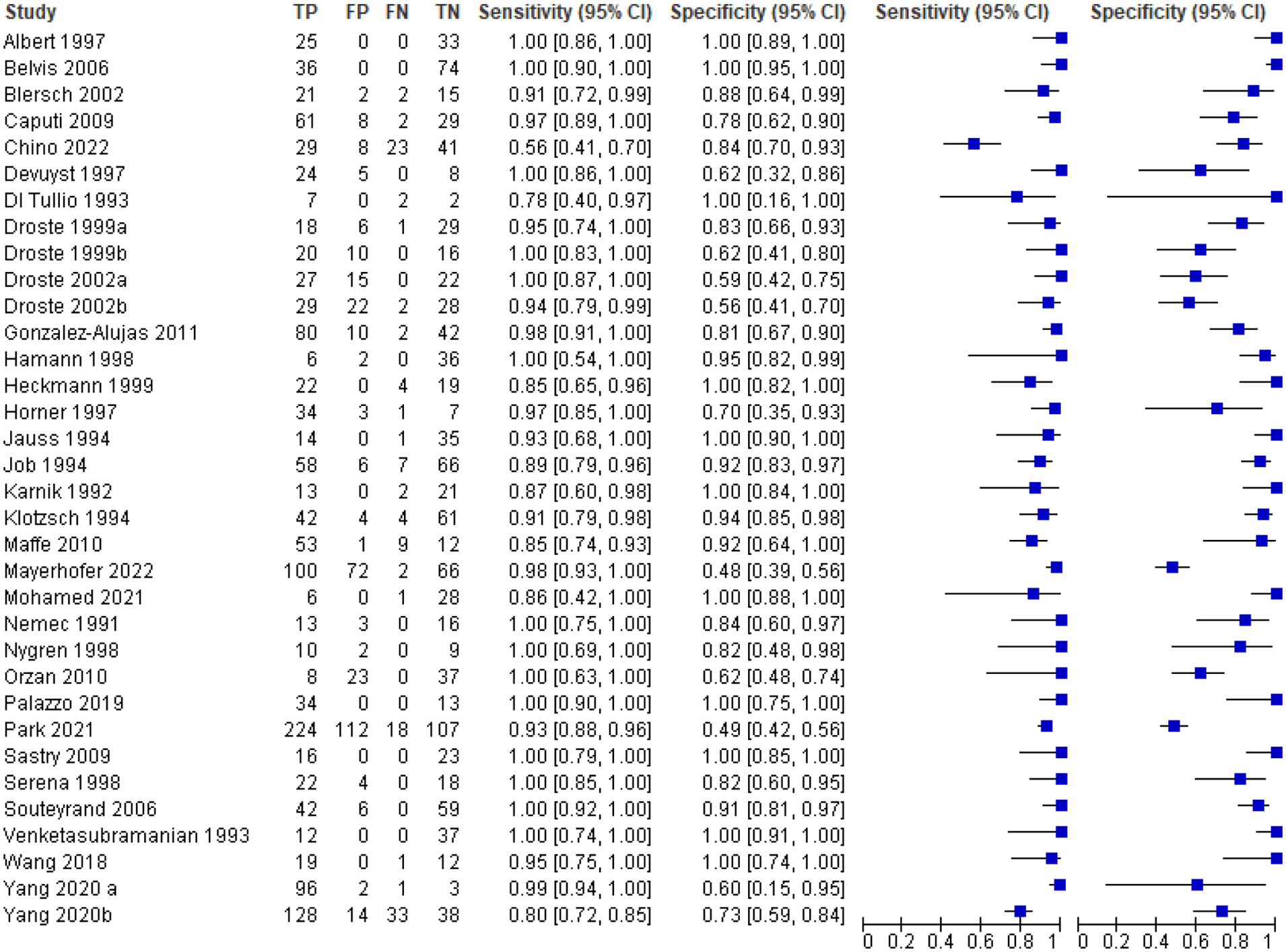
All 34 studies were included in our quantitative synthesis of sensitivity and specificity (bivariate meta-analysis; Figure 3 and GRADE Table 1). Significant heterogeneity in the included studies was identified. The pooled sensitivity of TCD was 0.96 (95% CI: 0.93–0.98). The pooled specificity of TCD was 0.90 (95% CI: 0.83–0.95). The pooled positive likelihood ratio was 9.74 (95% CI: 5.51–17.22), and the pooled negative likelihood ratio was 0.04 (95% CI: 0.02–0.07). The pooled diagnostic odds ratio for TCD was 236.99 (95% CI: 99.93–562.06). The area under the summary receiver operative characteristic (sROC) curve was 0.96 (95% CI: 0.93–0.98; Figure 4).
Random-effects meta-analysis comparing the assessment of a
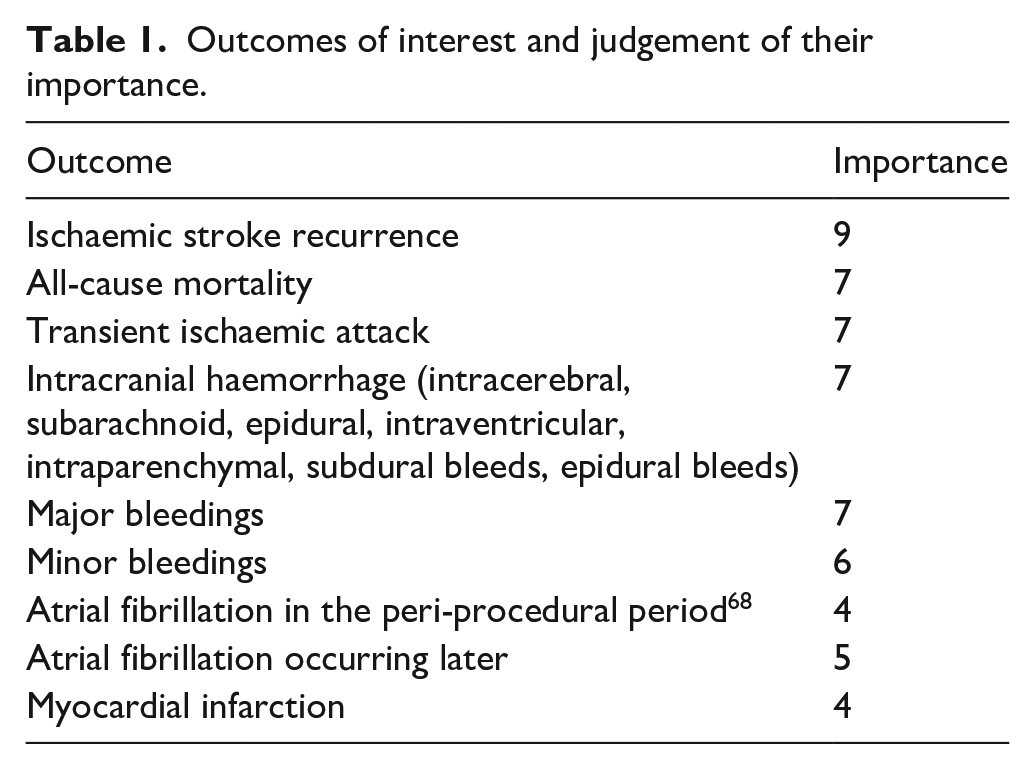
Outcomes of interest and judgement of their importance.
GRADE evidence profile for PICO 1.1 on TCD detection of a right-to-left shunt compared to c-TOE as reference test.

Table shows the number of included studies and participants contributing to the estimates of sensitivity and specificity.
Downgraded one level for risk of bias because high proportion of the studies have issues with unclear information on either ‘Index Test’, ‘Reference Standard’ or ‘Flow and Timing’.
Downgraded one level for inconsistency of results from the included studies’ results, especially for analysis of the specificity.
Downgraded one level for imprecision because high variation in the included studies characteristics.
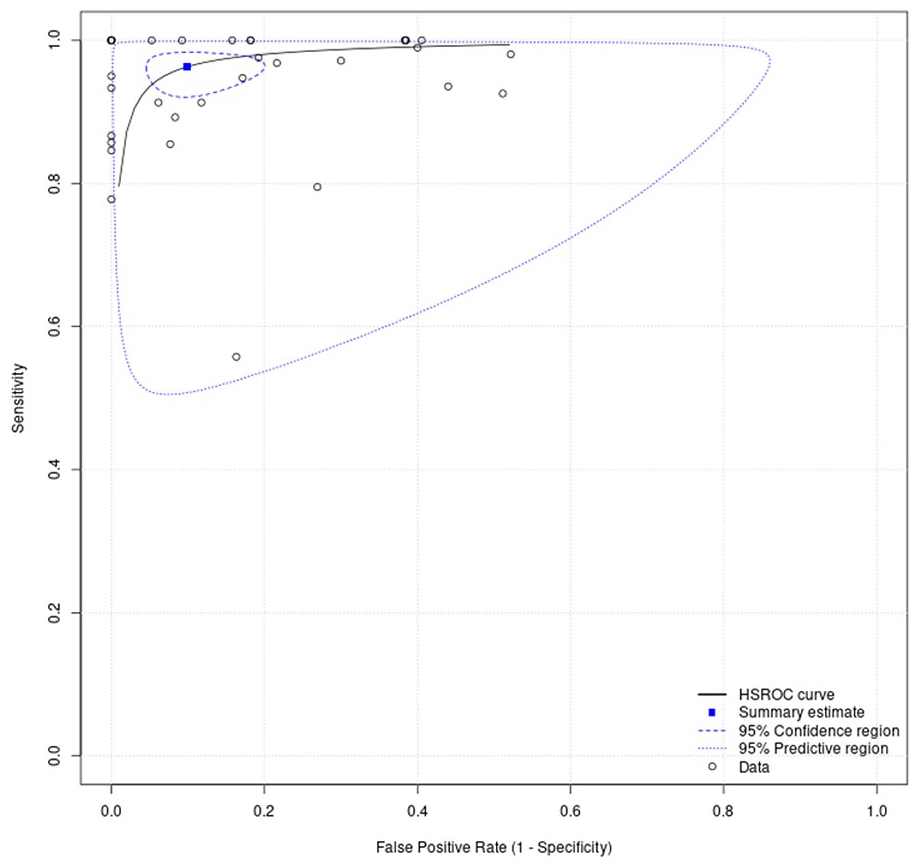
Area under the summary receiver operating characteristic curve (sROC), reflecting the diagnostic accuracy for c-TCD and ROC curve displaying the average value of sensitivity of c-TCD over all possible values of specificity. The area under the ROC curve (AUC) is 0.96.
In brief, due to its high sensitivity (96%),
Additional information
Studies have reported variations in the RLS diagnosis rate when using different contrast agents.37,69
Compared to contrast agents and AS, adding blood increases the sensitivity of c-TCD to as high as 100%. 70 However, the blood amount in the AS is still controversial, given clinical safety and practices. Gentile et al. 71 reported that 1 mL of blood in agitated saline significantly improved RLS results. Conversely, Hao et al. 72 reported that adding one drop of blood significantly improved RLS diagnosis of c-TCD by producing more microbubbles. In another study by Li et al., 73 the detection time was much shorter when the BAS compared to AS. There was a trend of more positive and higher RLS levels when using the 10% BAS; however, the differences between the 5% and 10% BAS were not statistically significant. 73
Droste et al. 37 ranked the performance of TCD with and without echovist by the number of microbubbles detected; the order from highest to lowest was as follows: Echovist with a Valsalva manoeuvre, Echovist with coughing, Echovist with a standardised Valsalva manoeuvre, saline with a Valsalva manoeuvre, Echovist alone, and saline alone.
Analysis of current evidence
The literature search identified 19 observational studies28,30,38,47,49,59–61,74–84 comparing the diagnostic accuracy of TTE with TOE as the conventional reference for the diagnosis of RLS and PFO since 1991 (Supplemental Table 3). No RCT was identified. Seven studies were based on prospective cohorts.38,62,74,75,78,79,82 All studies used c-TTE and six studies used harmonic imaging-enhanced TTE to detect RLS.28,47,80,81,84 Characteristics of the included studies are presented in Supplemental Table 3. Six studies were conducted before 2000, eight before or on 2010 and only five after 2010. Six studies included patients with acute ischaemic stroke and/or TIA.30,49,74,76,79,82
The studies used to address this PICO question correspond to a total population of 2575 patients with a mean age of 50 ± 7 years with 979 (38%) of the patients being women. A total of 1601 (62%) patients had an acute ischaemic stroke, 202 (8%) had a TIA and 20 (0.8%) had clinically silent cerebral infarcts. Three hundred six (12%) patients had a migraine and 16 (0.6%) had vertigo (Supplemental Table 3). In this population, PFOs were detected in 1029 (40%) patients using the conventional standard method of TOE.
Risk of bias analysis is presented in Figures 5 and 6. The risk of bias in flow and timing between index test (TTE) and reference test (TOE) was unclear in 11 (58%) of the 19 studies.

Risk of bias of individual studies used for the analysis of PICO 1.2 using the QUADAS-2 tool.
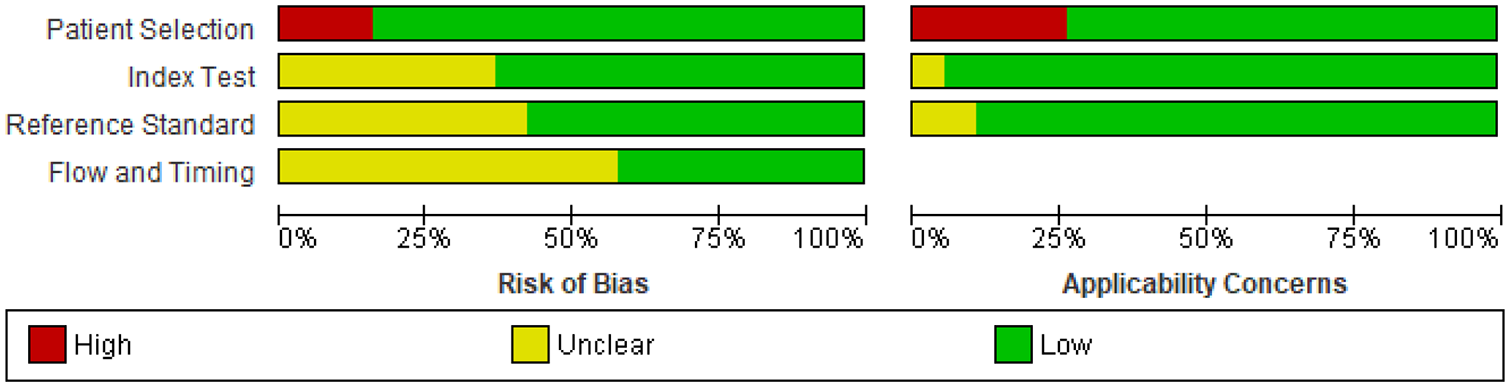
Risk of bias summary.
For our quantitative synthesis (bivariate meta-analysis), significant heterogeneity in the included studies was identified in this setting, c-TTE had a pooled sensitivity of 0.71 (95% CI: 0.50–0.86) Figure 7. The pooled positive likelihood ratio was 60.77 (95% CI: 10.97–336.6) and the pooled negative likelihood ratio was 0.29 (95% CI: 0.16–0.54). The pooled diagnostic odds ratio for c-TTE was 208 (95% CI: 44.8–967.6). The summary area under the ROC curve (sROC) was 0.71 Figure 8, indicating an overall modest diagnostic accuracy of TTE. These results suggest that TTE provides a technique for initial screening of RLS but a poor rule out test (modest sensitivity) for PFO detection in patients with cryptogenic stroke. However, this meta-analysis is limited by the serious inconsistency and severe heterogeneity across studies (especially for sensitivity), which results in a very low overall quality of evidence (Table 3). A previous systematic review analysed 13 studies with 1436 patients to evaluate the accuracy of TTE in detecting RLS compared to TEE, the reference standard. TTE and TEE used a contrast agent and a manoeuvre to provoke RLS. The findings showed that TTE had a weighted mean sensitivity of 46% and a specificity of 99%. The positive likelihood ratio was 20.85, and the negative likelihood ratio was 0.57. 70
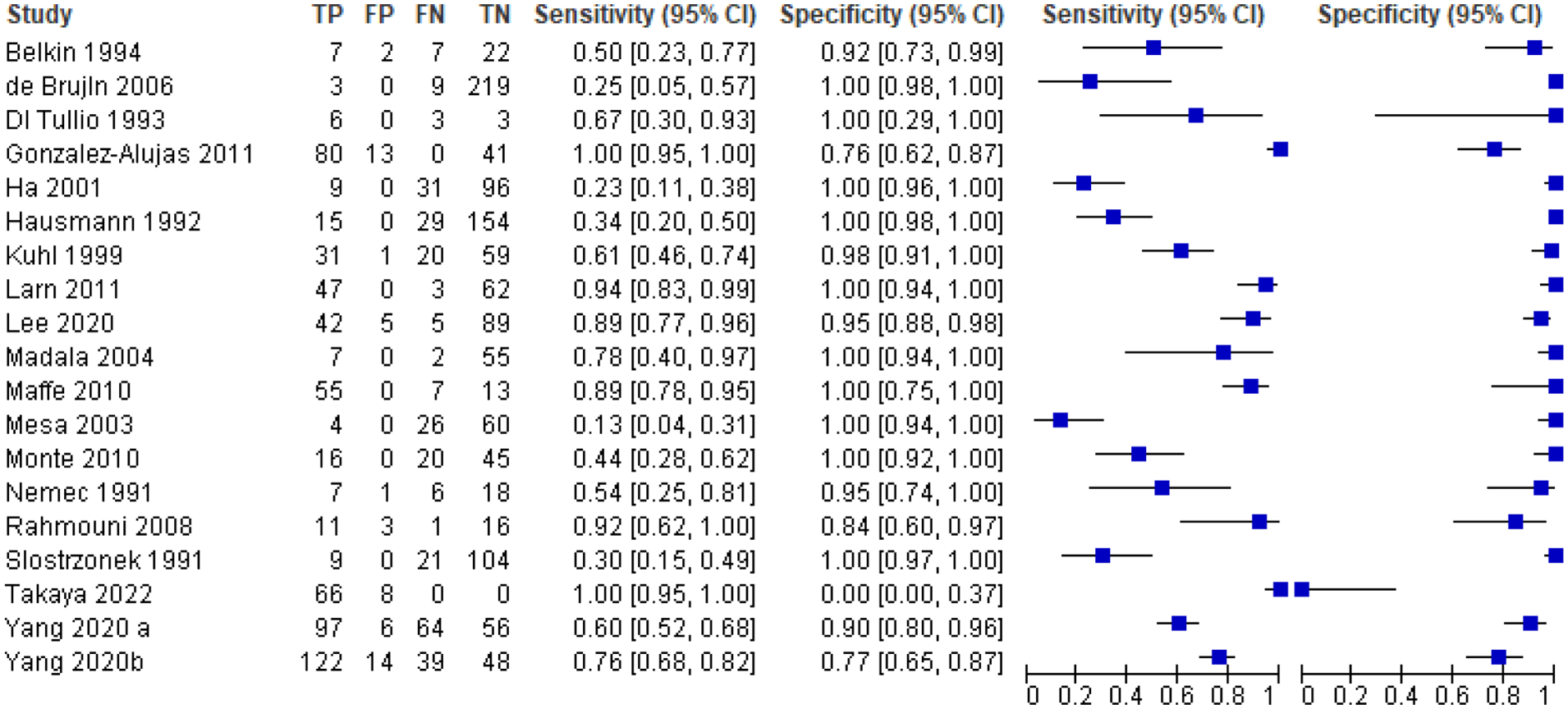
Random-effects meta-analysis comparing the assessment of a
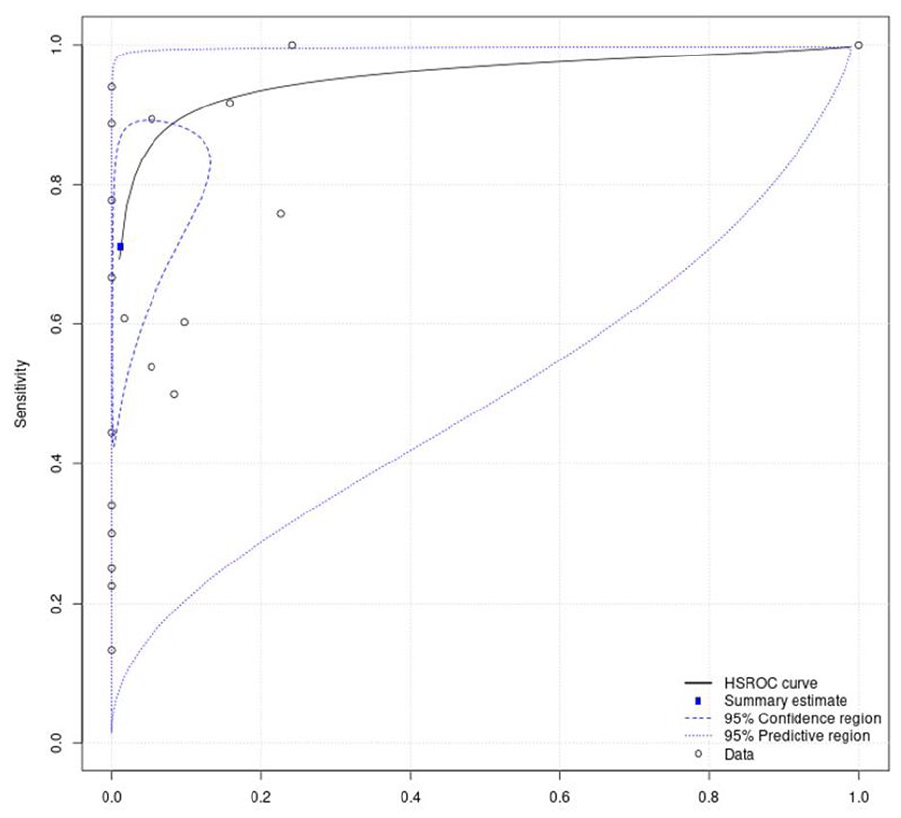
Area under the summary receiver operating characteristic curve (sROC), reflecting the diagnostic accuracy for c-TTE and ROC curve displaying the average value of sensitivity of c-TTE over all possible values of specificity. The area under the ROC curve (AUC) is 0.71, indicating an overall modest diagnostic accuracy of c-TTE.
GRADE evidence profile for PICO 1.2 on c-TTE detection of a RLS using c-TOE as conventional reference test.

Table shows the number of included studies and participants contributing to the estimates of sensitivity and specificity.
Downgraded one level for risk of bias because high proportion of the studies have issues with unclear information on either ‘Index Test’, ‘Reference Standard’ or ‘Flow and Timing’.
Downgraded one level for inconsistency of results from the included studies’ results, especially analysis on the sensitivity.
Downgraded one level for imprecision because high variation in the included studies characteristics.
Another systematic literature review analysed 35 studies with 3067 patients; TCD’s sensitivity was 96.1% (95% CI: 93.0–97.8) and specificity at 92.4% (95% CI: 85.5–96.1). In contrast, TTE’s sensitivity was 45.1% (95% CI: 30.8–60.3) with a specificity of 99.6% (95% CI: 96.5–99.9). TTE showed a higher positive likelihood ratio (LR+ = 106.61, 95% CI: 15.09–753.30) compared to TCD (LR+ = 12.62, 95% CI: 6.52–24.43), but TCD had a lower negative likelihood ratio (LR− = 0.04, 95% CI: 0.02–0.08) than TTE (LR− = 0.55, 95% CI: 0.42–0.72). The area under the curve (AUC) for TCD was 0.98 (95% CI: 0.97–0.99), significantly higher than TTE’s 0.86 (95% CI: 0.82–0.89), indicating TCD’s greater overall diagnostic yield in detecting PFO in these patients. 85
Recently, a prospective clinical trial investigated the effectiveness of robot-assisted transcranial Doppler compared to TTE in diagnosing RLS, including patent foramen ovale, in patients suspected of embolic cerebrovascular ischaemia. The study found raTCD significantly more effective, detecting RLS in 64% of cases compared to 20% with TTE. Notably, raTCD identified three times more RLS instances and was particularly more adept at identifying large shunts. This suggests that raTCD could offer a noninvasive, accurate, and accessible means for RLS detection, potentially overcoming the limitations of conventional TCD and TTE. 86
The recommendation for this PICO question will be summarised with the following results and can be found at the end of Part 1.
Additional information
The diagnostic accuracy of TTE as a screening test for RLS detection in cryptogenic stroke patients can be enhanced by using second harmonic imaging as suggested by a meta-analysis of 15 prospective studies corresponding to 1995 patients. The pooled sensitivity and specificity for TTE with harmonic imaging were 91% and 93%, respectively, with high inconsistency among studies. TTE with harmonic imaging may provide a useful tool for the initial screening test for RLS due to its high accuracy and non-invasiveness. However, TOE is superior to TTE in the identification of specific anatomic features of PFO that guide therapeutic decision-making and interventional procedure planning, such as an atrial septal aneurysm, a moderate or severe shunt (more than 10–20 microbubbles crossing during the first three cardiac cycles).8,13,79,87–90
Other variables that must be evaluated by TOE include PFO size, atrial septal aneurysm and the possible presence of other atrial septal defects, the size of the Eustachian valve and the possible presence of a Chiari network in the right atrium, the thickness of the septum primum and secundum and the atrial rim sizes.79,91–94
The literature search did not identify any study comparing the diagnostic accuracy of c-TCD to c-TTE using c-TOE as the conventional reference for the detection of RLS.
Additional information on TOE as a gold standard
A study-level meta-analysis of the accuracy of c-TOE in the diagnosis of PFO, and therefore of an interatrial shunt, compared to robust reference standards (autopsy, cardiac surgery and/or catheterisation) yielded a weighted sensitivity of only 89%, 70 probably due to the difficulty of performing an adequate Valsalva manoeuvre.95,96 This data highlight the lack of an available non-invasive gold standard for the assessment of PFO-related RLS and the associated challenges in assessing the accuracy of c-TTE, c-TCD and c-TOE.
Additional information on upcoming alternative diagnostic modalities
Regarding alternative diagnostic modalities, cardiac magnetic resonance imaging has been demonstrated to be significantly less effective than TOE in detecting right-to-left shunts. 97 A comprehensive meta-analysis, encompassing 5 studies with a total of 236 patients, revealed that cardiac magnetic resonance detected only 29.3% (95% CI: 23.6–35.0) compared to 53.7% (95% CI: 47.4–59.9) observed with TOE (p < 0.001). 98 Whereas, the sensitivity of single-phase cardiac computed tomography, for identifying PFO was found to be 85.3%, but its specificity was notably lower at 33.3%, accompanied by a negative predictive value of 33%. 99 Conversely, in a separate study that involved 70 stroke patients diagnosed with PFO, full-cycle CCT demonstrated enhanced diagnostic performance, boasting a sensitivity of 89.4% and a specificity of 92.3% in comparison to TOE. However, the sensitivity of single-phase CCT in this particular study was considerably reduced at 72.7% (p = 0.002), indicating a significant difference in the effectiveness of single-phase and full-cycle CCT in detecting PFOs. 100
Additional information on diagnostic after PFO-closure
The precise protocols and timing for these follow-up assessments to verify the complete PFO closure can differ among medical centres and practitioners as there is no standardised protocol. 101 A study between 2000 and 2005 involving 124 patients explored the prevalence of residual interatrial shunting after PFO closure using four types of devices. It reported an 87% closure rate at 2 years, influenced by the device type, PFO canal length, and ASA extent. 102
Another prospective cohort study of 1078 patients at a single hospital assessed the impact of residual shunts after PFO closure on the recurrence of stroke or TIA. Here, a c-TTE was performed 1 year post-procedure. 103 A single-centre study with 118 patients evaluating the clinical outcomes and quality of life reported that the total occlusion rate at follow-up (mean 50 months, range 3–100) was 98.1%. The day after closure, a c-TTE was repeated to confirm the proper positioning of the device and exclude residual shunt, at which point the patient was discharged. Clinical evaluation, complete neurological examination, c-TTE/TOE and cTCD were scheduled at 1, 6 and 12 months postoperatively and yearly after that. 104
Finally, a retrospective study on medical records and echocardiograms of patients who underwent transcatheter PFO closure, reported a 19.5% incidence of RLS at 4 months post-closure, which decreased to 8.4% by 11 months. Patients were evaluated using c-TTE 2–3 months after a medical procedure. If an RLS was still present, further evaluation occurred 6–12 months post-procedure using c-TEE, chest CT, or assessment in a catheterisation lab with intracardiac echocardiography and angiography to identify additional RLS sources. 101


Part 2: Treatment of PFO-associated stroke
In this section, to inform clinicians about the different treatment options in stroke prevention in patients with cryptogenic stroke and stroke, we address the following
Analysis of current evidence and evidence-based recommendation
Patients aged 18–60 years
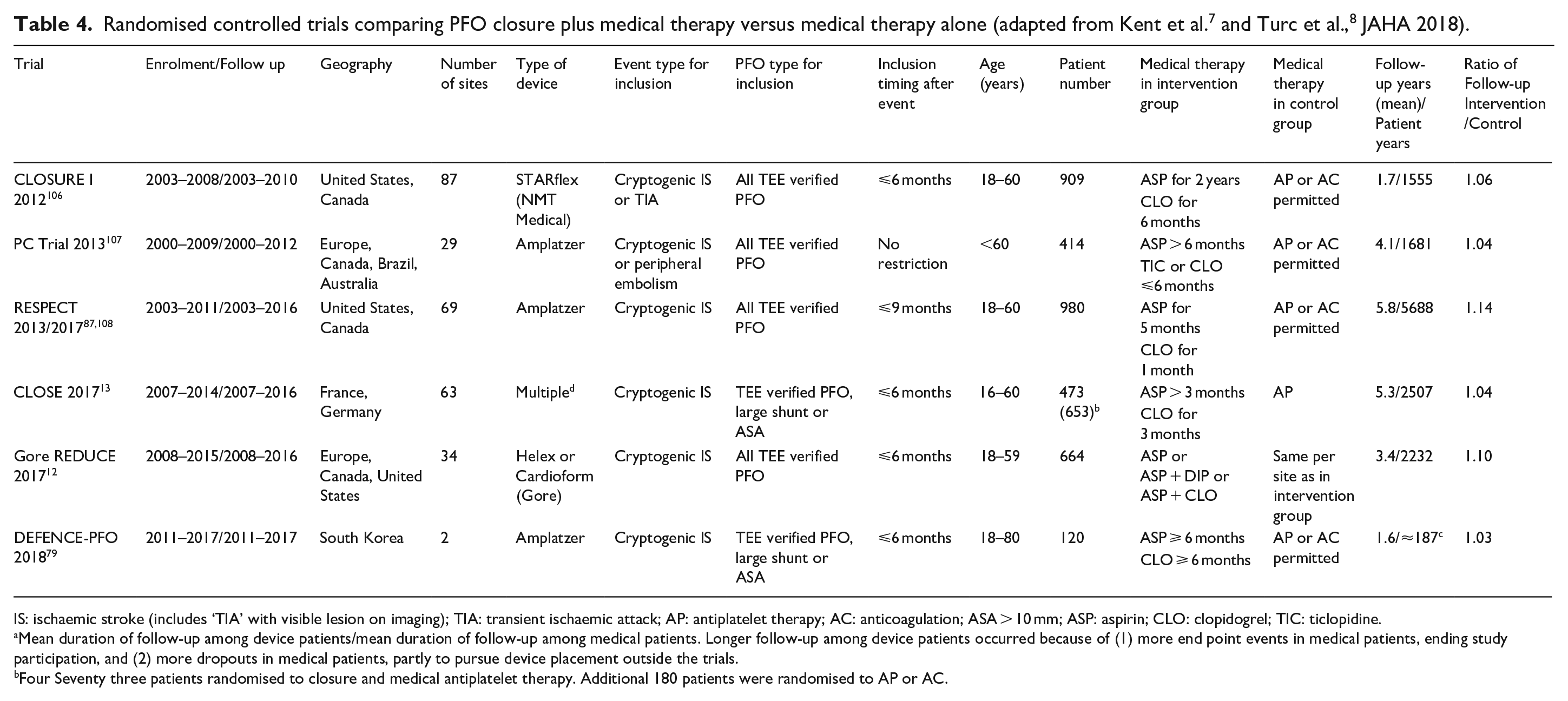
We identified 6 RCTs comparing PFO closure plus medical therapy versus medical therapy alone (Table 4), several study-level meta-analyses4,8,105 and an IPDMA by the Systematic, Collaborative, PFO Closure Evaluation (SCOPE) group, which provides a classification system (PASCAL, see Introduction) to identify which patients with cryptogenic stroke and PFO – or in pathophysiological terms PFO-associated stroke – may benefit most from PFO closure and which ones may not. 7
IS: ischaemic stroke (includes ‘TIA’ with visible lesion on imaging); TIA: transient ischaemic attack; AP: antiplatelet therapy; AC: anticoagulation; ASA > 10 mm; ASP: aspirin; CLO: clopidogrel; TIC: ticlopidine.
Mean duration of follow-up among device patients/mean duration of follow-up among medical patients. Longer follow-up among device patients occurred because of (1) more end point events in medical patients, ending study participation, and (2) more dropouts in medical patients, partly to pursue device placement outside the trials.
Four Seventy three patients randomised to closure and medical antiplatelet therapy. Additional 180 patients were randomised to AP or AC.
CLOSURE I used a clamshell umbrella device that is no longer in production. 106 Double disc devices were employed in the 5 other trials, namely PC Trial, RESPECT, CLOSE, Gore REDUCE and DEFENCE-PFO.12,13,79,87 All double disc devices are in clinical use today. Medical anti-thrombotic therapy consisted of antiplatelet therapy in Gore REDUCE and in two of the three arms of CLOSE. In the other trials, antithrombotic therapy was at the discretion of the investigators. About 66%–79% of patients received antiplatelet therapy and 21%–34% anticoagulation. 8 The trials included patients aged 18–60 years, except for CLOSE, which enrolled patients from 16 to 60 years, and DEFENCE-PFO, which included patients aged 18–80 years. 8 All studies included patients with a PFO verified by TOE. In addition, CLOSE and DEFENCE PFO only included patients with a large shunt, ASA or both.
Among the six RCTs, DEFENCE PFO, Gore REDUCE, and CLOSE were positive, while CLOSURE I, and PC Trial, and RESPECT were neutral.12,13,79,106–108 Their trends also pointed in the direction of the benefit of PFO closure and the results of extended follow up of the RESPECT trial were in favour of PFO-closure. 87
The RESPECT, REDUCE and CLOSE trials included the Amplatzer and Gore devices which are the only FDA-approved devices.12,13,87
A topical review and an IPDMA by the SCOPE consortium analysed primary and secondary efficacy outcomes of PFO closure plus medical therapy versus medical therapy alone.7,8 The median patient age in the device group [n = 1882] was 46.2 (IQR 39.0–52.7) years and 46.0 (IQR 39.0–53.0) years in the medical group [n = 1846]. The annualised incidence of stroke was 1.09% (95% CI: 0.88–1.36) with medical therapy and 0.47% (95% CI: 0.35–0.65) with device closure (adjusted HR, 0.41 [95% CI: 0.28–0.60]). All-cause mortality did not differ across treatment groups (Risk ratio 0.98, 95% CI: 0.31–3.11), while information on stroke-associated mortality was not specifically recorded. Regarding safety outcomes, the most common adverse event was AF. 7 It occurred more often in the PFO closure group (adjusted RR 4.54 [95% CI: 2.78–7.39]), and 46% of the events (50/109) were transient, occurring only in the first 45 days after randomisation. 7
The SCOPE IPD-MA represents the best available evidence on PFO closure in patients aged 18–60 years. However, SCOPE only indirectly addresses our PICO question because the comparator is not identical to the one, we have chosen (i.e. antiplatelet therapy). In several of the trials included in SCOPE, the comparator was antiplatelet therapy or anticoagulants at the discretion of the investigator (Table 4). 7 We have therefore conducted a study-level meta-analysis looking only at trials allowing the direct comparison of PFO closure versus antiplatelet therapy (Gore REDUCE and CLOSE), which is presented in Figure 9. The corresponding HR was 0.18 (95% CI: 0.05–0.63), but most patients included in the two trials had a ‘high risk PFO’ with a large shunt, an ASA or both. 7 The quality of evidence for this comparison of PFO closure versus antiplatelet therapy was deemed to be high (Table 5). The result of the overall comparison between PFO closure plus antithrombotic therapy versus anti-thrombotic therapy (antiplatelets or anticoagulants at the discretion of each investigator) alone align with those of the IPDMA [pooled hazard ratio (HR) 0.38 (95% CI: 0.19–0.77)]. Our results confirm those reported in two previously published study-level meta-analysis considering the same outcome in all the arms. 4
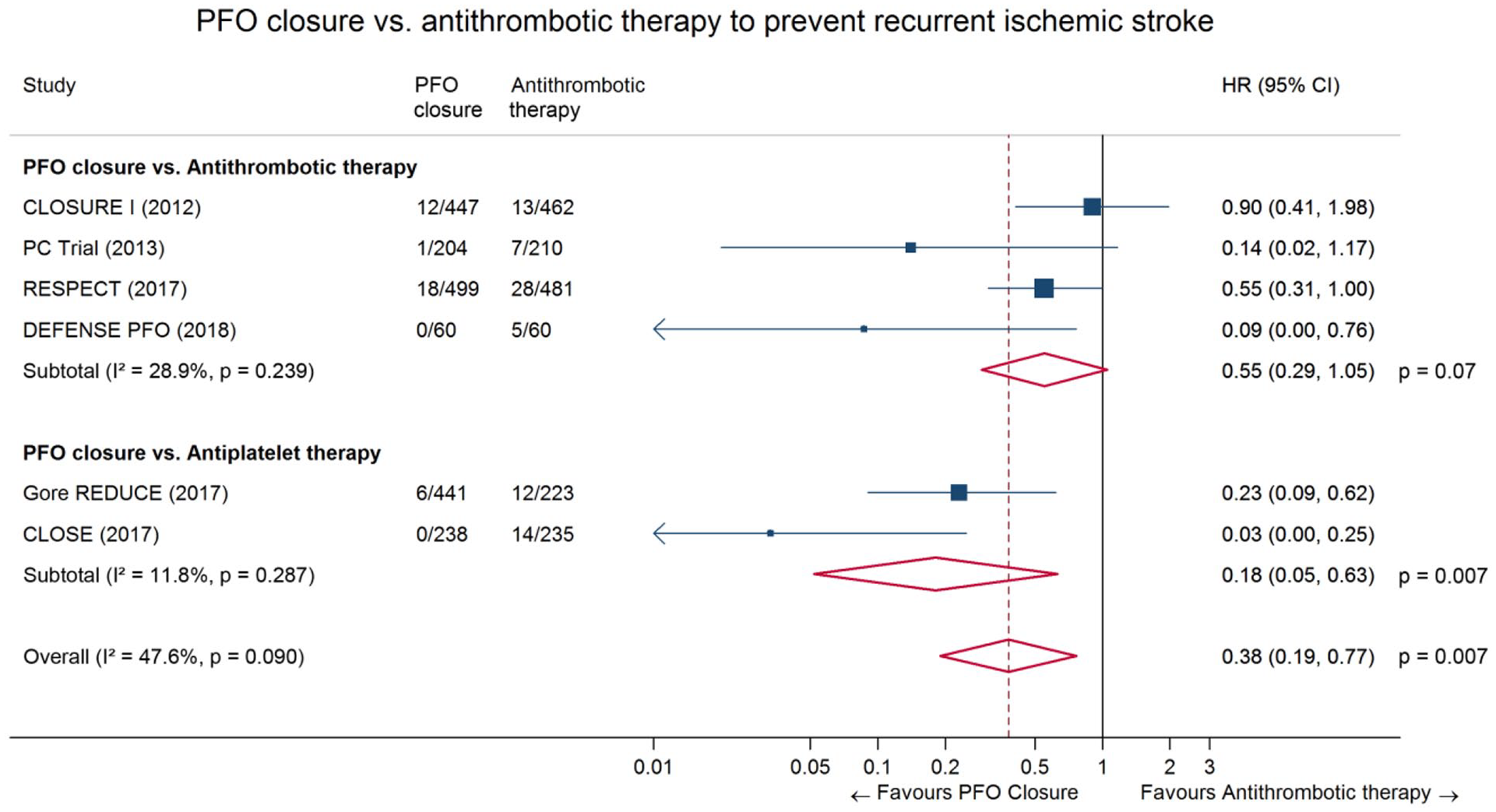
GRADE evidence profile for PICO 2 (PFO closure plus antiplatelet therapy vs antiplatelet therapy alone).
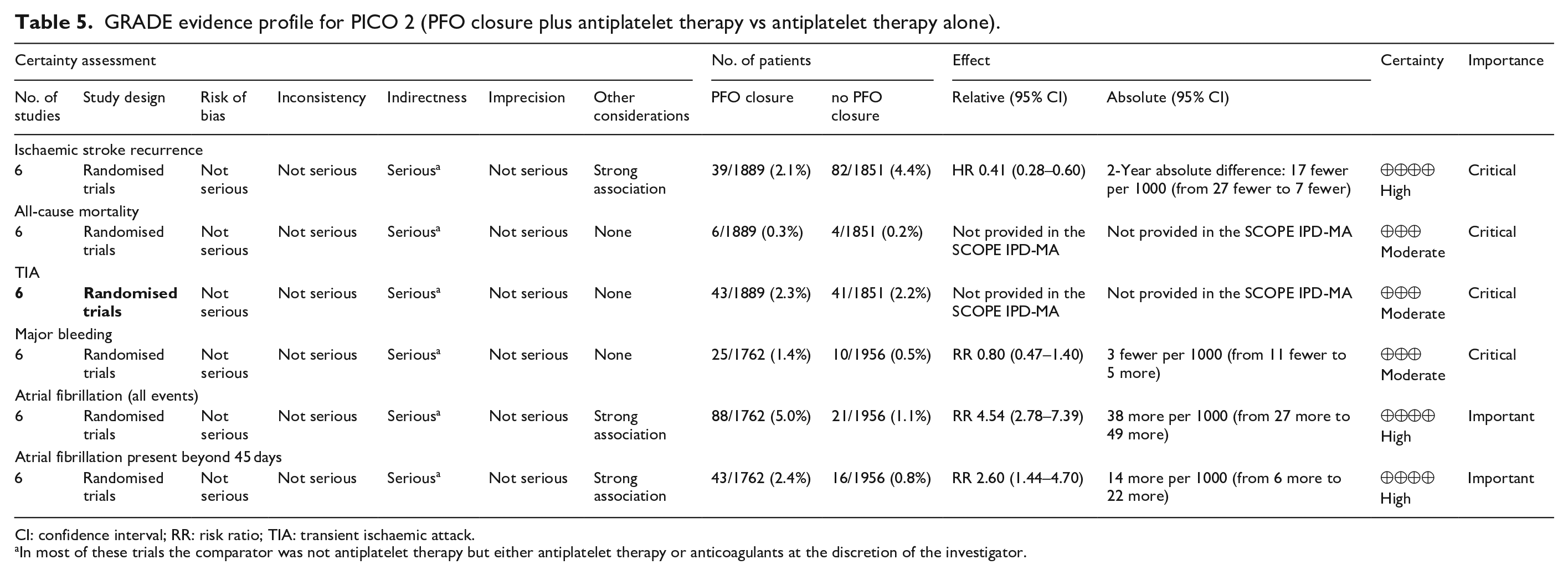
CI: confidence interval; RR: risk ratio; TIA: transient ischaemic attack.
In most of these trials the comparator was not antiplatelet therapy but either antiplatelet therapy or anticoagulants at the discretion of the investigator.
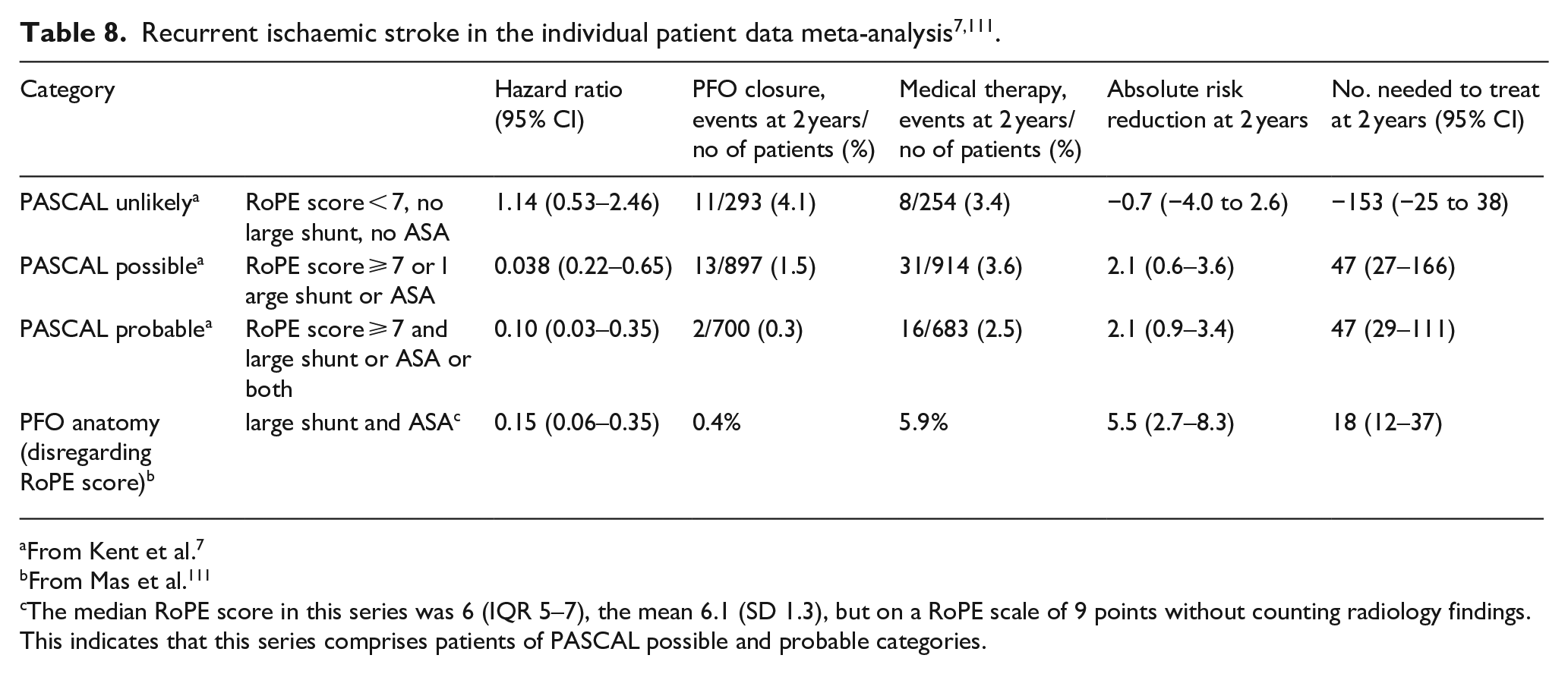
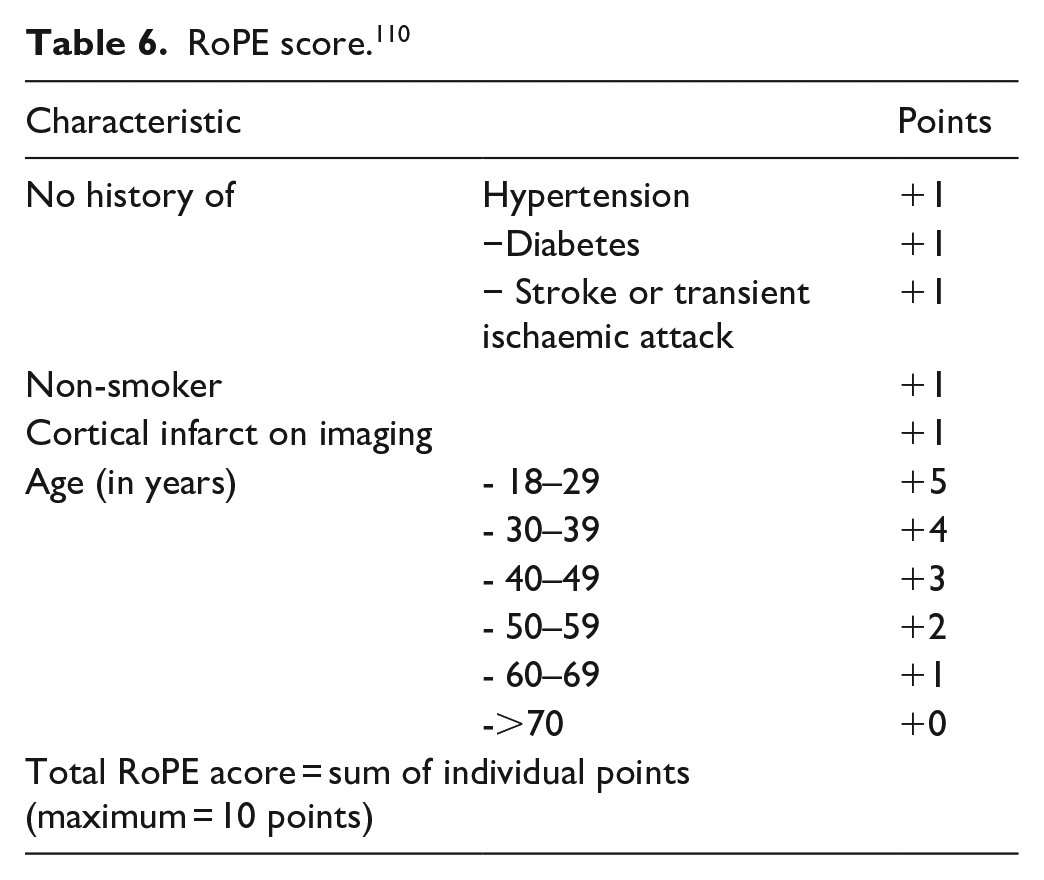
Not all strokes in patients with PFO (i.e. PFO-associated strokes) are PFO-related.4,8,105 Therefore, a clinically relevant question is whether all PFOs in patients with cryptogenic strokes are pathogenic and should be closed. The IPDMA demonstrates that the treatment effect is heterogeneous and depends on the individual patient characteristics. 7 The probability that a PFO is causally related to the stroke can be estimated with the Risk of Paradoxical Embolism (RoPE) Score and the PFO-Associated Stroke Causal Likelihood (PASCAL) classification system. The RoPE score is a 10-point scoring system, based on clinical and neuroimaging features only, in which higher scores reflect younger age and the absence of vascular risk factors (Table 6). 110 A high score of a patient with a cryptogenic stroke means a greater likelihood that a PFO is present and probably pathogenic, a lower score makes this less likely or unlikely (Table 7).
RoPE score. 110
PASCAL classification system. 7
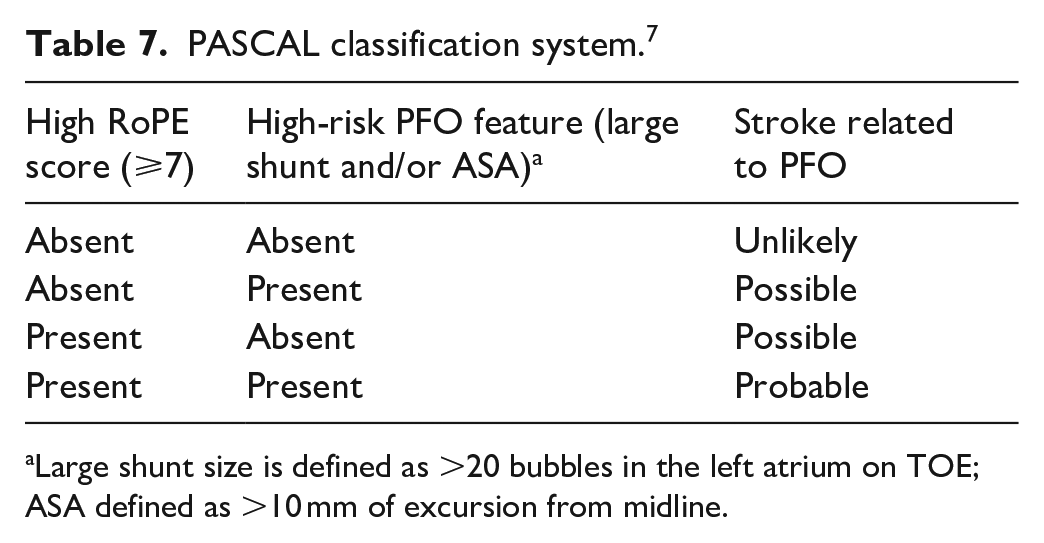
Large shunt size is defined as >20 bubbles in the left atrium on TOE; ASA defined as >10 mm of excursion from midline.
To further improve the identification of ischaemic strokes due to PFO the SCOPE IPD-MA recently proposed the PFO-Associated Stroke Causal Likelihood (PASCAL) Classification System. 7 This is different from the other three and directly germane to the current study. Among patients with no major defined cause of ischaemic stroke, the PASCAL classification system integrates information regarding: (1) presence of features that increase likelihood of PFO-stroke mechanisms (high risk PFO physiologic and structural features of large shunt or ASA) and (2) absence of features that increase likelihood of an occult non-PFO stroke mechanisms (older age, vascular risk factors, and stroke topography features) as quantified in the RoPE score. Based on this combination of factors, the original, extended PASCAL Classification System algorithmically assigns a likelihood of causal relationship among five levels: Definite, Highly Probable, Probable, Possible and Unlikely. 11 The PASCAL algorithm was developed using a mixed methods approach incorporating expert judgement, physiologic and epidemiologic data, and the validated Rope Score.
The SCOPE group derived the PASCAL classification system to assess the individual treatment effect. 7 The main results for individual treatment effects are given in Table 8.
From Kent et al. 7
From Mas et al. 111
The median RoPE score in this series was 6 (IQR 5–7), the mean 6.1 (SD 1.3), but on a RoPE scale of 9 points without counting radiology findings. This indicates that this series comprises patients of PASCAL possible and probable categories.


Patients with low versus high RoPE Scores had HRs of 0.61 (95% CI: 0.37–1.00) and 0.21 (95% CI: 0.11–0.42), respectively (P for interaction = 0.02). Patients classified as unlikely, possible, and probable using the PASCAL Classification System had HRs of 1.14 (95% CI: 0.53–2.46), 0.38 (95% CI: 0.22–0.65), and 0.10 (95% CI: 0.03–0.35), respectively (P for interaction = 0.003). The 2-year absolute risk reduction was −0.7% (95% CI: −4.0 to 2.6), 2.1% (95% CI: 0.6–3.6), and 2.1% (95% CI: 0.9–3.4) in the unlikely, possible, and probable PASCAL categories, respectively. An additional analysis of the IPDMA data showed that patients with large shunts plus ASA derived the greatest benefit from PFO closure. 7 At 2 years, their absolute risk reduction of recurrent stroke was 5.5% (95% CI: 2.7–8.3) and at 5 years it was 7.1% (95% CI: 3.7–10.6), indicating an NNT of 14 (95% CI: 9–27).
Device-associated adverse events were generally higher among patients classified as unlikely; the absolute risk increases for AF beyond day 45 after PFO closure were as follows: 4.41% (95% CI: 1.02–7.80) in the unlikely PASCAL category, 1.53% (95% CI: 0.33–2.72) in the possible, and 0.65% (95% CI: −0.41 to 1.71%) in the probable PASCAL category. Major bleeding events included access site or retroperitoneal haemorrhage in 1%, pericardial tamponade in 0.17%, and cardiac perforation in 0.06%. 7
We acknowledge that the use of risk stratification tools was not prespecified in the present PICO question and therefore no systematic review was conducted with regard to the PASCAL classification system. Nevertheless, we chose to use PASCAL for our evidence-based recommendations because one of the three prespecified aims of the main SCOPE collaboration paper was to examine whether the PASCAL system was associated with a differential treatment effect, and which was clearly demonstrated by the analysis. In this guideline, the quality of evidence for using the PASCAL classification system was rated down by one level (please see the corresponding recommendation box) because this system was not used as an inclusion criterion or stratification variable in any of the six RCTs, nor was it prospectively validated.
Additional information
Patients qualified for inclusion in the DEFENCE-PFO, CLOSURE I, CLOSE and GORE REDUCE trials if their index stroke occurred 6 months before the potential intervention.12,13,79,106 However, the actual timing of inclusion varied among these studies. Specifically, the RESPECT trial saw patient enrolment at a mean time of 2.6–3.3 months post-stroke, 87 the GORE REDUCE trial around 3.4 months, 12 the CLOSE trial approximately 3.1 months, and the PC Trial at about 4.3 months following the index stroke. 107
None of the trials were powered to explore the very early timing of PFO closure.
Patients over 60 years of age
Hospital-based and population-based studies show that RLS in older patients with cryptogenic strokes are more common than in patients with known cause of stroke, similar to patients at aged 18–60 years. 4 However, data on PFO closure from RCTs are scarce in this age group. Only the DEFENCE-PFO Trial allowed randomisation of patients older than 60 years, but only 13 patients were randomised to PFO closure and 21 to medical therapy alone in this age group.8,79,112 During follow-up, no patients experienced a recurrent stroke or TIA after PFO closure, while 4 events occurred in the control group (p = 0.07). At 2 years, the event rate was 24.6% in the medical group, corresponding to a HR of 0.14 (95% CI: 0.005–3.57, p = 0.23). Because of the very low number of randomised patients, we believe that no evidence-based recommendation can reasonably be provided for this age group. It would be important to include these patients in RCTs if possible, or at least in a prospective clinical registry. Examples are the PFO Closure, Oral Anticoagulants or Antiplatelet Therapy After PFO-associated Stroke in Patients Aged 60–80 years trial (CLOSE-2, NCT05387954) or the Prospective Registry of Elderly ESUS with PFO (COACH_ESUS, NCT05238610).
Patients younger than 18 years
How to manage teenagers after PFO-associated stroke is another relevant clinical question. Patients had to be at least 16 years old to be enrolled in CLOSE and at least 18 years old in the other five RCTs. However, in CLOSE only one patient younger than 18 years, who did not have an outcome event, was included. Therefore, data from those trials do not address whether PFO closure should be recommended in patients younger than 18 years with PFO-associated stroke. A retrospective study identified 31 children who underwent PFO closure after stroke. 113 Their median age was 14.9 years and median follow-up was 568 days. No recurrent stroke occurred.
Additional information
There are many clinical variables with a putative association with paradoxical embolism. 4 None and no other abnormal clinical condition caused by PFO was part of the inclusion criteria of the RCTs and they all are omitted in the PASCAL score. Nevertheless, a history of non-cerebral embolism, deep venous thrombosis, pulmonary embolism, pulmonary arterial hypertension, history of sleep apnoea or other hypoxaemic conditions associated with PFO, Valsalva at stroke onset, recent history of prolonged immobility, recent airline travel, presence of venous thrombophilia, decompressive illness in divers, straddling thrombus through the PFO, platypnea-orthodeoxia syndrome or an Eustachian valve or other anatomical features on echocardiography possibly enhancing the risk of paradoxical embolism, may be taken in consideration when managing patients after PFO-associated strokes.7,9
Evidence-based recommendations




Expert consensus statement


Expert consensus statement


Analysis of current evidence and evidence-based recommendation
The 3-arm CLOSE study was the only one in which patients were randomised to PFO closure or oral anticoagulation, but the head-to-head comparison of these 2 groups was not prespecified in the study statistical analysis plan (Table 9). 13 Overall, 238 patients were randomised to PFO closure and 187 to anticoagulants, of which 92,8% were vitamin K antagonists (VKA) and only 7,2% were direct oral anticoagulants (DOAC). In the anticoagulation group, 3 patients had a recurrent stroke over a follow-up of 967 patient-years, compared with none in the PFO closure group over a follow up of 963 patient-years, which were not statistically significant in the post-hoc analysis, HR 0.14; 95% CI: 0.00–1.45; p = 0.08 for log-rank test. 8 The outcome events are listed in Figures 10–12. However, CLOSE was underpowered for this comparison (Figures 13 and 14).
GRADE evidence profile for PICO 3 (PFO closure vs anticoagulation).
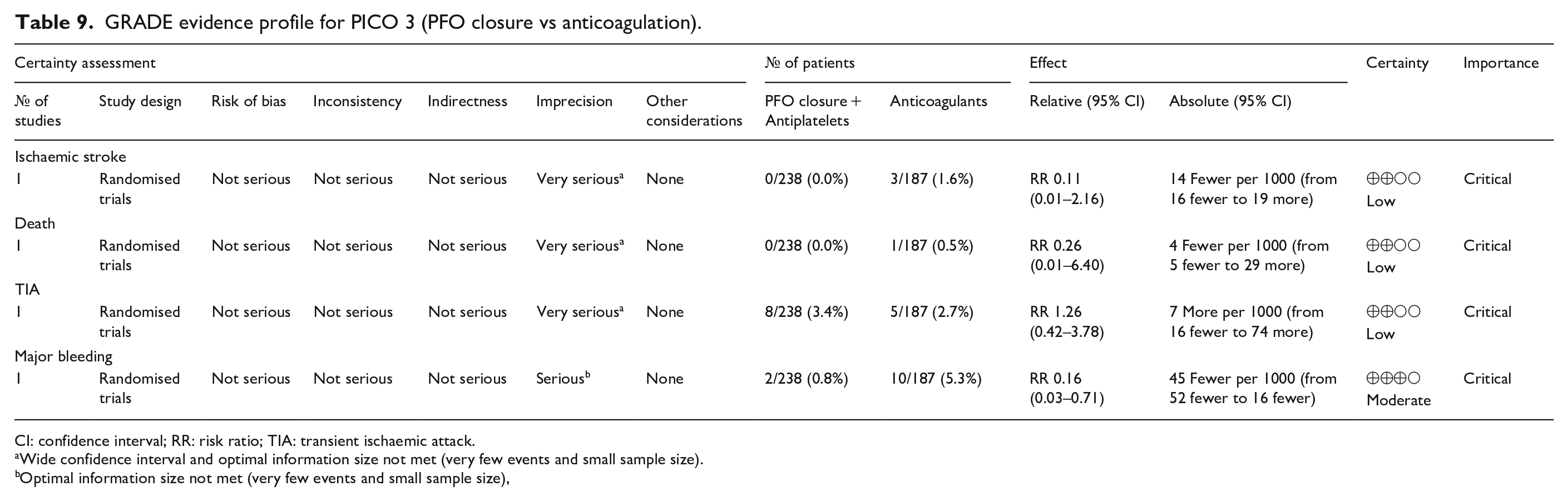
CI: confidence interval; RR: risk ratio; TIA: transient ischaemic attack.
Wide confidence interval and optimal information size not met (very few events and small sample size).
Optimal information size not met (very few events and small sample size),
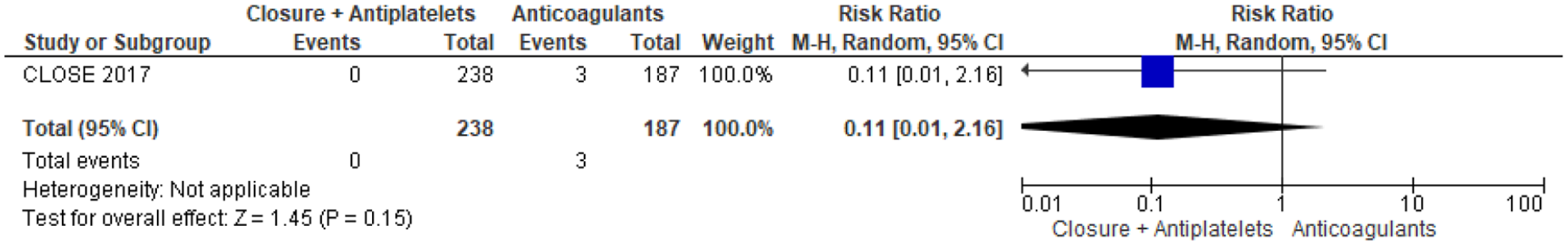
Risk of ischaemic stroke (only one study included).
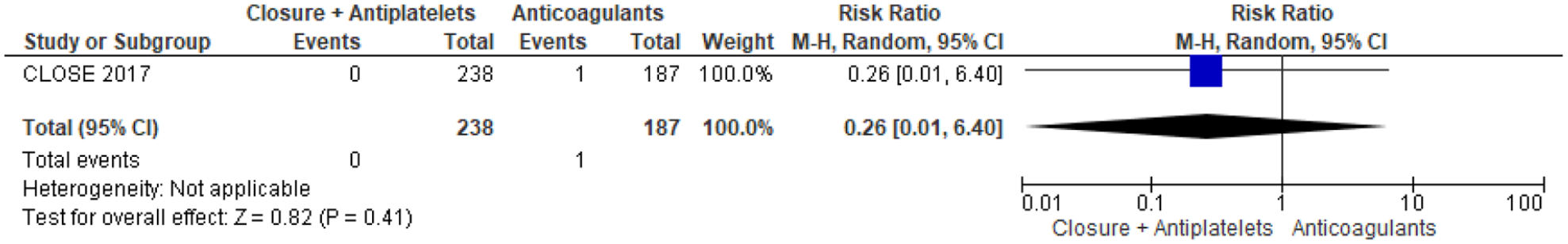
Risk of death (only one study included).
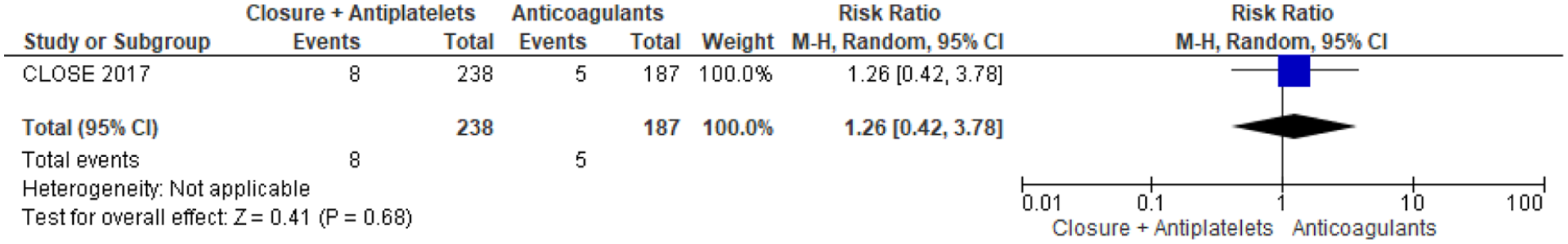
Risk of TIA (only one study included).
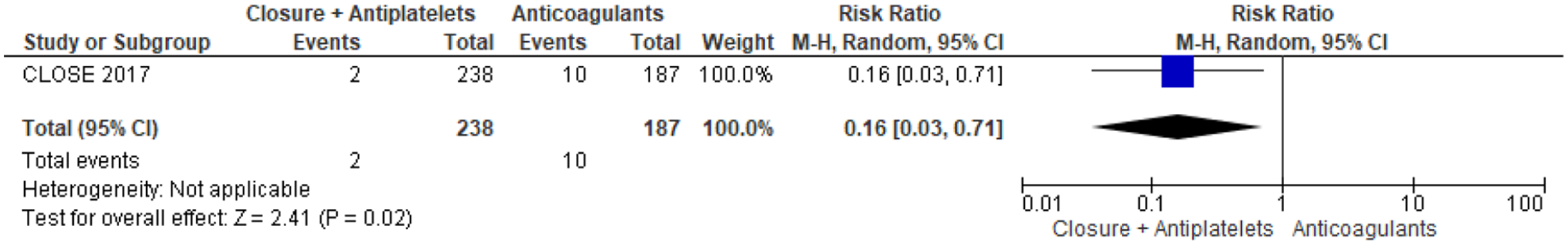
Risk of major bleeding.

Risk of bias of randomised controlled trial (outcomes: Ischaemic stroke, Death, TIA, Major bleeding).
Additional information
A non-randomised comparison between PFO closure and anticoagulation is available from the CLOSURE 106 and RESPECT trials.87,108 A meta-analysis of those data reported an OR of 1.19 (95% CI: 0.43–3.26) for stroke recurrence with oral anticoagulants. Also including data from CLOSE trial in the meta-analysis, the OR for stroke recurrence with oral anticoagulants was 0.74 (95% CI: 0.20–2.74). 4
In a prospective cohort of 591 patients with cryptogenic ischaemic stroke or TIA and PFO, 134 (22.7%) had thrombophilia [(protein C deficiency (13.4%), protein S deficiency (32.1%), Factor V Leiden (11.9%), G2021A mutation (7.5%), anticardiolipin antibody (21.6%), hyperhomocysteinaemia (23.9%), antithrombin III deficiency (11.2%), positive lupus anticoagulant (8.2%), high factor VIII levels (11.2%)]. 114 In a mean duration follow-up of 53 months, thrombophilia significantly increased the risk for recurrent events (HR 1.85; 95% CI: 1.09–3.16; p = 0.024). PFO closure was performed in 383 (65%) patients, who had a reduced risk of stroke or TIA recurrence (HR 0.16; 95% CI: 0.09–0.30; p < 0.001). Of the 134 patients with thrombophilia, 89 underwent PFO closure and 45 received only medical therapy (anticoagulants or antiplatelets), with a difference in the risk for recurrent events in favour of PFO closure (HR 0.25; 95% CI: 0.08–0.74; p = 0.012). 115

Analysis of current evidence
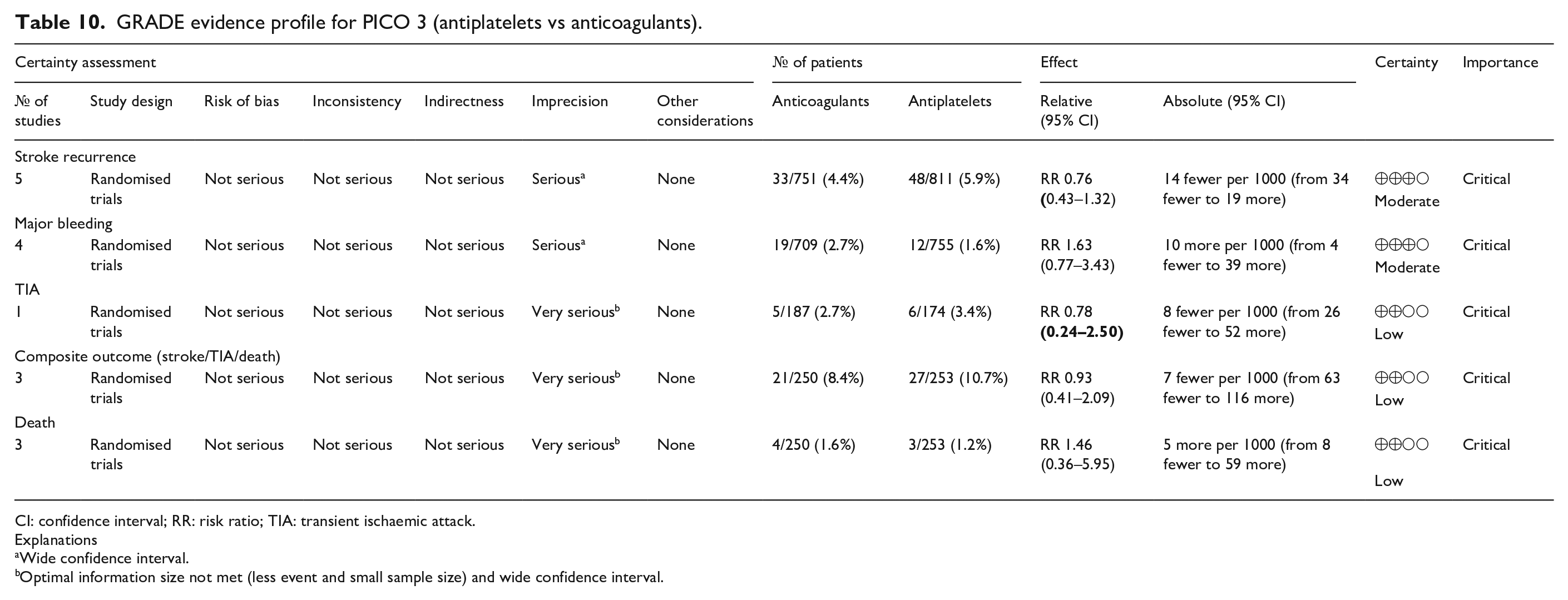
Two RCTs that enrolled patients with cryptogenic stroke and PFO and three prespecified analyses of non-randomised subgroups of patients with cryptogenic stroke and PFO enrolled in otherwise RCTs compared anticoagulant therapy and antiplatelet therapy outcomes.12,13 The duration of follow-up was different among studies: 5 years in the CLOSE trial, 13 11 months in NAVIGATE ESUS, 116 2 years in PICSS, 117 19 months in RESPECT-ESUS 118 and 13.5 months in patients receiving anticoagulants and 14.6 months in those treated with antiplatelets in the trial by Shariat et al. 119 (Table 10). In our meta-analysis of these study-level data on 1562 patients, for stroke recurrence anticoagulants had a lower recurrence risk versus antiplatelets (RR: 0.76 [95% CI: 0.43–1.32] (Figure 15). In contrast, anticoagulants had a higher risk for major bleeding; RR: 1.63 (95% CI: 0.77–3.43); for the composite of recurrent stroke/TIA and death there was a minimal difference; RR: 0.93 (95% CI: 0.41–2.09) (Figures 16–20)
GRADE evidence profile for PICO 3 (antiplatelets vs anticoagulants).
CI: confidence interval; RR: risk ratio; TIA: transient ischaemic attack.
Explanations
Wide confidence interval.
Optimal information size not met (less event and small sample size) and wide confidence interval.

Risk of bias of randomised controlled trials.
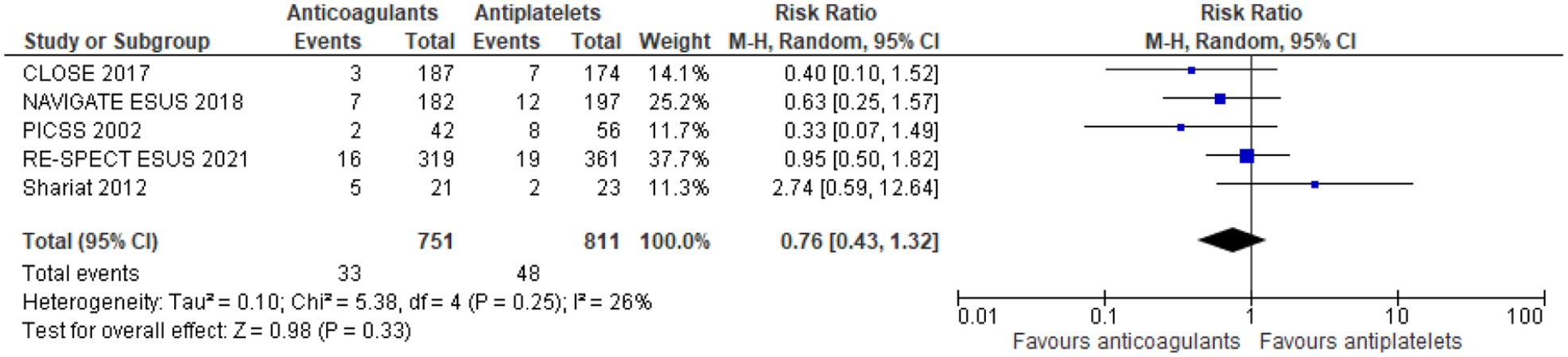
Risk of stroke recurrence restricted to RCTs (antiplatelets vs anticoagulants) stroke recurrence.
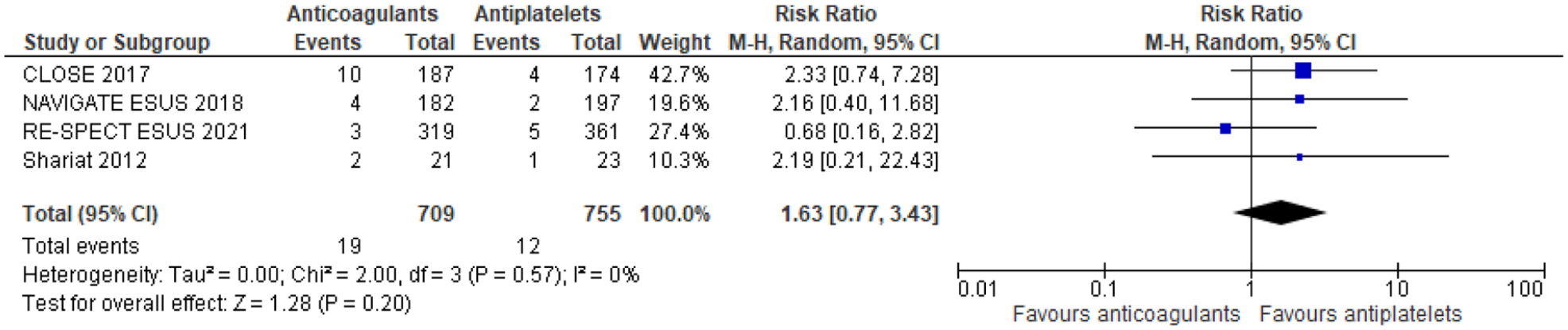
Risk of major bleeding restricted to RCTs (antiplatelets vs anticoagulants).
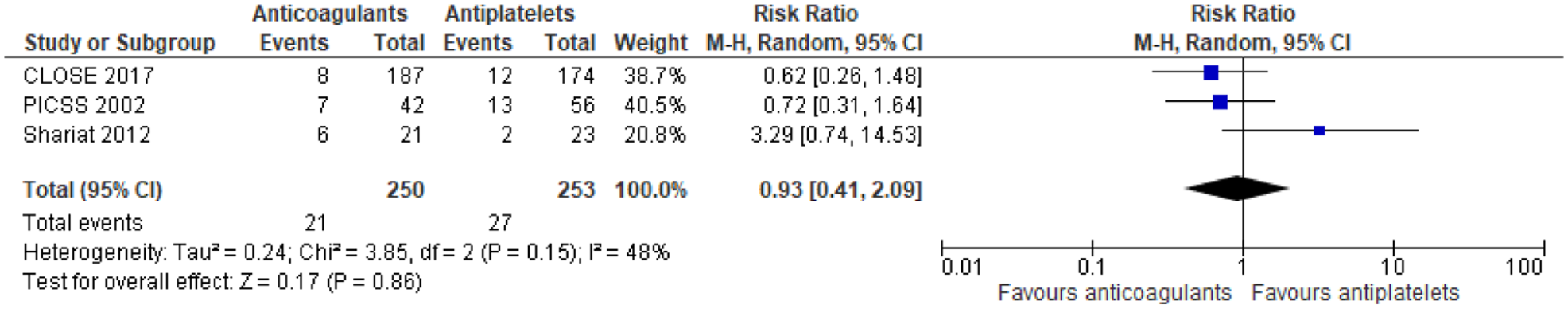
Risk of composite outcome (stroke/TIA/death) restricted to RCTs (antiplatelets vs anticoagulants).
Risk of TIA restricted to RCTs (antiplatelets vs anticoagulants) TIA.
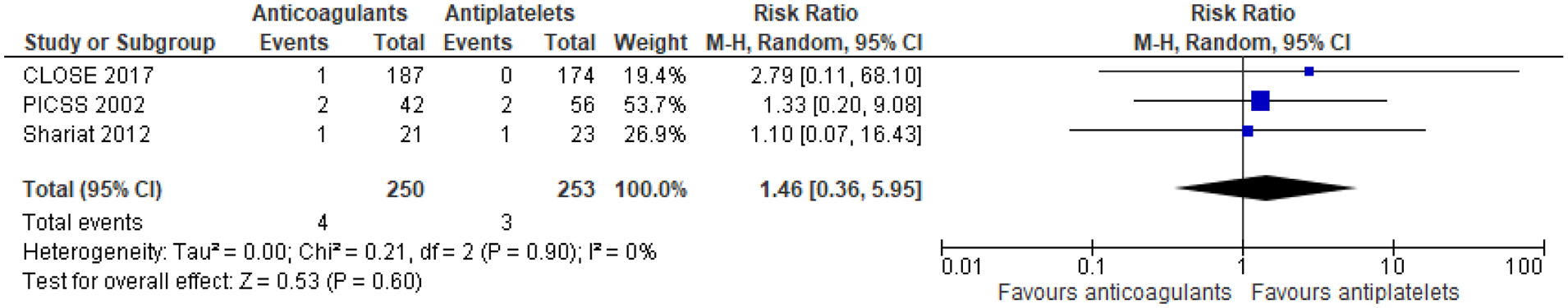
Risk of death restricted to RCTs (antiplatelets vs anticoagulants) death.
Overall, the data suggests a slightly lower risk of stroke recurrence with anticoagulants compared to antiplatelets but with a higher risk of major bleeding. The evidence for other outcomes like TIA, composite outcomes, and death is of low quality with considerable uncertainty (Figure 20).
Additional information
A previously published study-level meta-analysis including the CLOSE study as well as adjusted and non-adjusted observational comparisons found a statistically significant reduction of recurrent stroke risk in patients with PFO undergoing oral anticoagulation as compared to antiplatelet therapy (OR: 0.88; 95% CI: 0.83–0.92) at the expense of an increased major bleeding rate (OR: 4.57;95% CI: 2.10–9.93). 8 In another recent meta-analysis on cerebrovascular event recurrences and/or death in patients with PFO treated with oral anticoagulation (OAC) or antiplatelet therapy for secondary prevention of cryptogenic stroke, the efficacy endpoints were stroke recurrence and the composite of stroke, TIA, or all-cause death. Major bleedings represented the safety endpoint. Sixteen studies with 3953 patients (OAC = 1527, APT = 2426) were included. The weighted mean follow-up was 2.9 years. OAC was associated with a significant reduction in the risk of stroke compared with APT (RR: 0.65; 95% CI: 0.44–0.95; ARR 2%, NNT 49), while no difference was found regarding the composite outcome (RR: 0.78; 95% CI: 0.57–1.07) and the safety outcome (RR: 1.57; 95% CI: 0.85–2.90; p = 0.15). 120
Part 3. Post-procedural management
In this section, we address the following overarching question.


Analysis of current evidence
The coagulation system is part of the normal and crucial sealing process of PFO occluders by facilitating fibrous tissue growth within the meshes of the device.121,122 Therefore, it is not surprising that minor cerebrovascular events have been estimated with an incidence up to 1.0%–2.0%, within the first 4 weeks after PFO closure.123,124 Based on animal models and isolated reports in humans a complete device endothelialization is expected to be achieved within 6 months after PFO closure in the majority of patients, however the process may require up to 5 years. 125 Diagnostics cannot visualise this process on many types of implanted devices because it is a layer that is often only a few cells thick. 126 The understanding of endothelialisation, albeit incomplete, comes from the rare cases when devices were inspected at the time of autopsy or open heart surgery. These devices often had incomplete tissue covering the large surface of the device, filling the crevices at the edge of the discs, and bridging the gap between the device edge and the adjacent tissue.127,128
Antiplatelet therapy after closure may have a role to facilitate this endothelisation and to prevent ischaemic events. Our literature search failed to identify any RCT of dual antiplatelet therapy versus single antiplatelet therapy after PFO closure. Only two observational studies were found to be relevant for this comparison.129,130 These two observational studies were found to be relevant for our meta-analysis of major bleeding outcome after PFO closure following stroke (Figure 21); only one study was included in our analysis. 130 Our analysis showed non-significant benefit of single antiplatelet therapy (Aspirin or Clopidogrel) over dual antiplatelets therapy (Aspirin + Clopidogrel) in reducing the risk of major bleeding (p = 0.82, Figure 22).129,130 At the same time, no significant benefit of dual antiplatelet therapy (Aspirin + Clopidogrel) over single antiplatelet therapy (Aspirin or Clopidogrel) was observed in reducing the risk of myocardial infarction (p = 0.82, Figure 23).
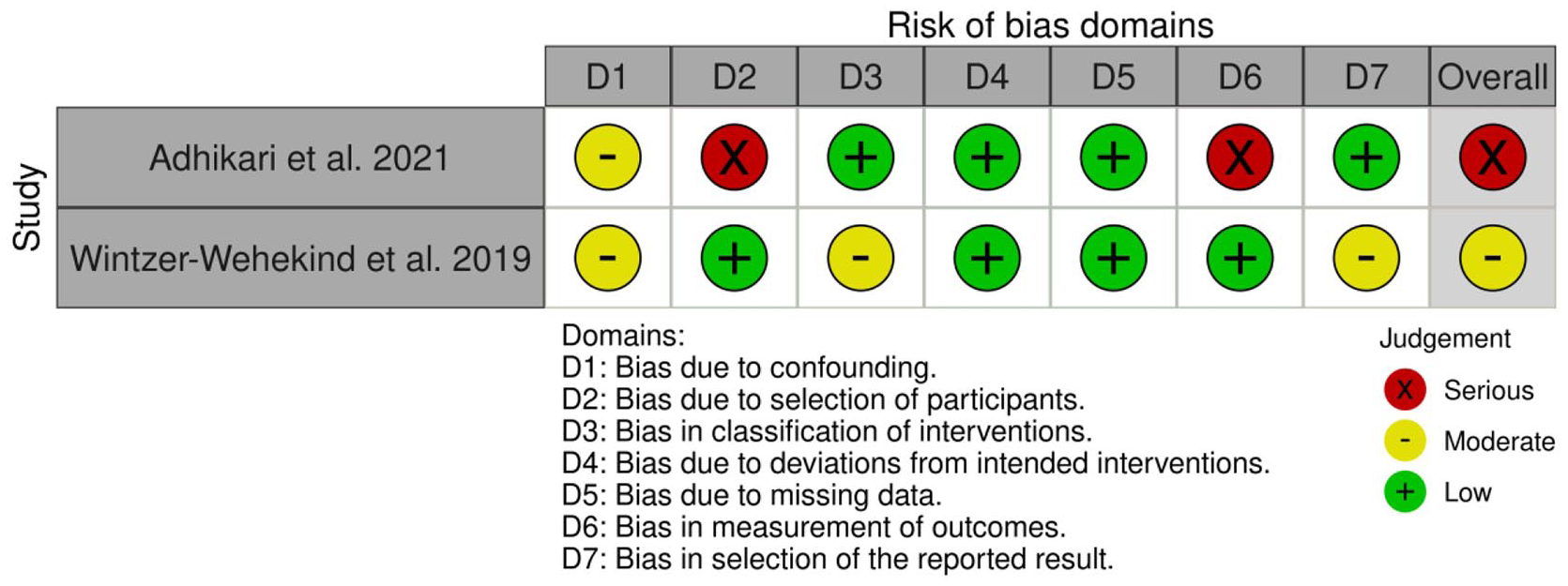
Risk of bias assessment (ROBINS-I tool) of observational studies reporting data on dual antiplatelet therapy versus single antiplatelet therapy for reducing the risk of recurrent stroke after PFO closure.
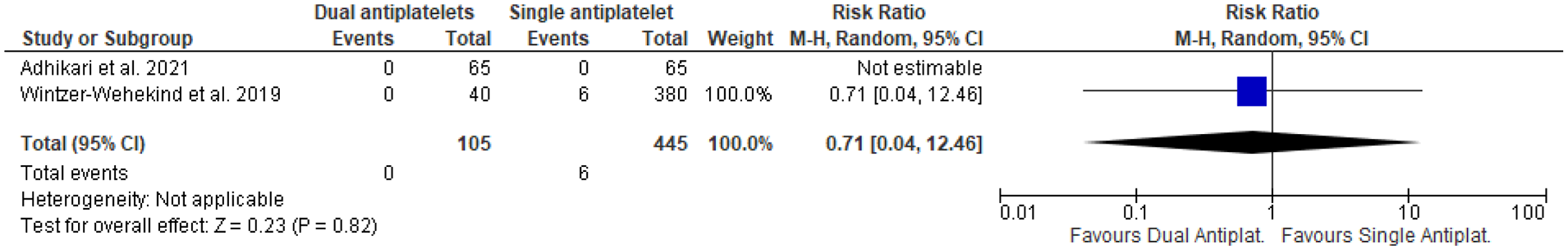
Dual antiplatelet therapy versus single antiplatelet therapy and risk of recurrent stroke.
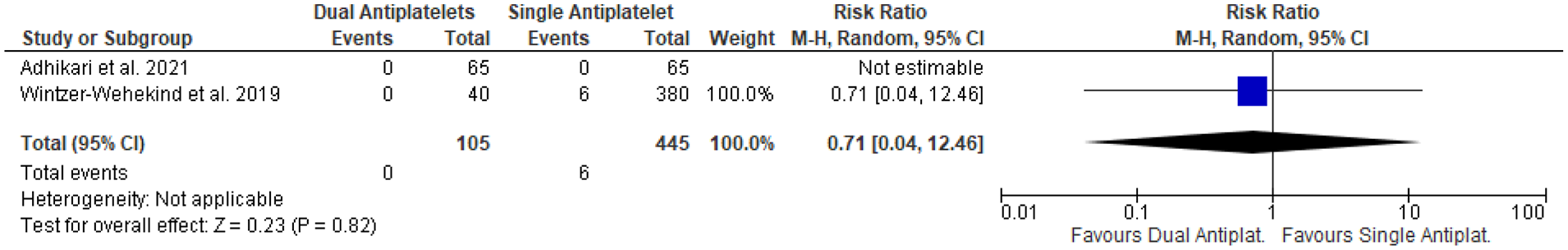
Dual antiplatelet therapy versus single antiplatelet therapy and risk of MI.
In a meta-analysis including all the six RCTs, major bleeding occurred in 1.8% and 1.7% of the patients randomised to PFO closure and medical treatment, respectively. 8 In the CLOSE trial, there was no stroke during follow-up in the closure group, but up to 24 bleeding events were described under antiplatelet therapy. 13
Additional information
In a hospital-based cohort, 453 consecutive patients who underwent PFO closure due to a cryptogenic ischaemic event were followed up and assessed at a median follow-up of 8 (IQR: 4–11) years. All patients were on antithrombotic treatment following PFO closure. Stroke and TIA occurred in 4 (0.9%) and 12 (2.6%) patients, respectively. Six percent of patients had bleeding events (major in 1.3% of patients) (Table 11). A propensity score-matched analysis of 46 patients who discontinued antithrombotic therapy within 1-year of PFO closure and 120 patients who continued antithrombotic therapy showed similar rates of ischaemic events between groups (0 vs 0.2 stroke/transient ischaemic attack per 100 patient-years in those who discontinued vs those who continued antithrombotic therapy). 130
Grade evidence profile table for PICO 5: In patients with cryptogenic stroke and PFO, does dual antiplatelets compared with single antiplatelet therapy reduce the risk of recurrent stroke after PFO closure?.
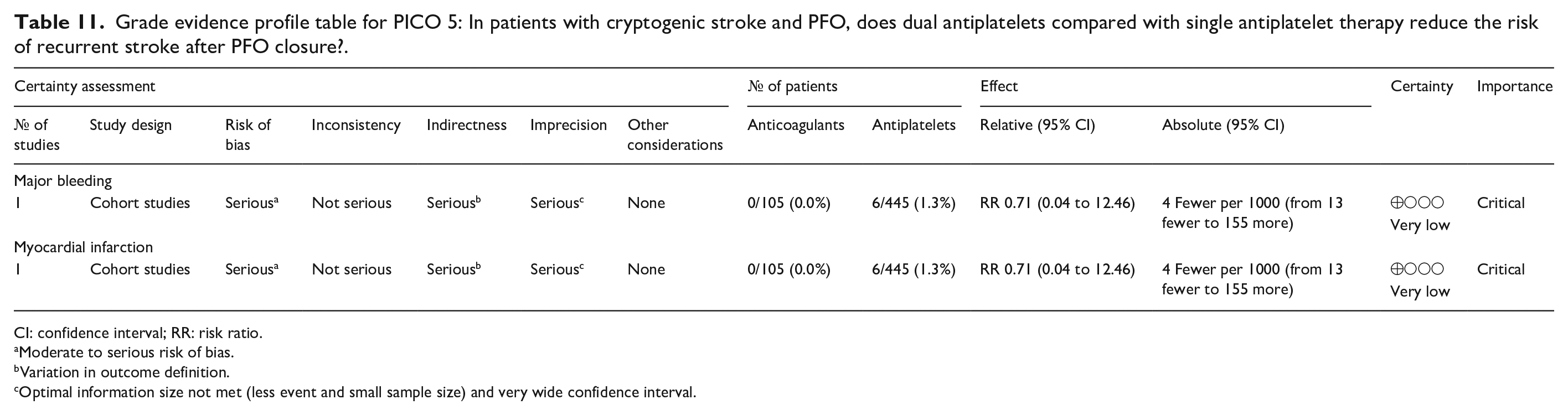
CI: confidence interval; RR: risk ratio.
Moderate to serious risk of bias.
Variation in outcome definition.
Optimal information size not met (less event and small sample size) and very wide confidence interval.


Analysis of current evidence
In RCTs, new-onset AF was 4.3 times more frequent in patients assigned to the closure group than in those assigned to the antithrombotic group. 4
In a recent meta-analysis, a 4.6% incidence of AF was reported in a follow-up period of 3.8 years. However, the Authors did not clarify whether the AF episodes were symptomatic or identified through ILR monitoring. 8 Additionally, a meta-analysis of 14 studies (13,245 patients, mean age 61.2 years), the rate of AF detection was lower in patients with stroke and PFO compared to those without (RR = 0.52, 95% CI: 0.41–0.63). In line with this, AF detection was lower in patients with PFOs found on TTE versus TOE, suggesting that the yield of AF monitoring is further reduced with increasing PFO size. 131
A meta-analysis reported that beyond 45 days after closure there was no increased risk for AF with PFO closure. 7 The incidence of these events appeared lowest with the Amplatzer and GORE occluders.4,132
Moreover, there is growing evidence from RCTs and observational studies that using data from RCTs and observational studies, the implantable loop recorder (ILR) reduces the risk of first-ever and recurrent (combined) stroke.133,134
We were unable to identify RCTs or comparative observational studies on patients with PFO closure that compared the detection of AF with prolonged cardiac monitoring compared with non-prolonged cardiac monitoring. However, our literature search identified five observational studies in which prolonged monitoring was conducted after PFO closure, without a control arm. These five observational studies involving 2076 patients were included in our meta-analysis to estimate the long-term incidence of AF after PFO closure. Of note, four of these studies had as endpoint only the detection of AF with prolonged monitoring135–138 whereas only one study included recurrent ischaemic stroke as endpoint.136–139 No recurrent stroke was recorded (Figure 24).
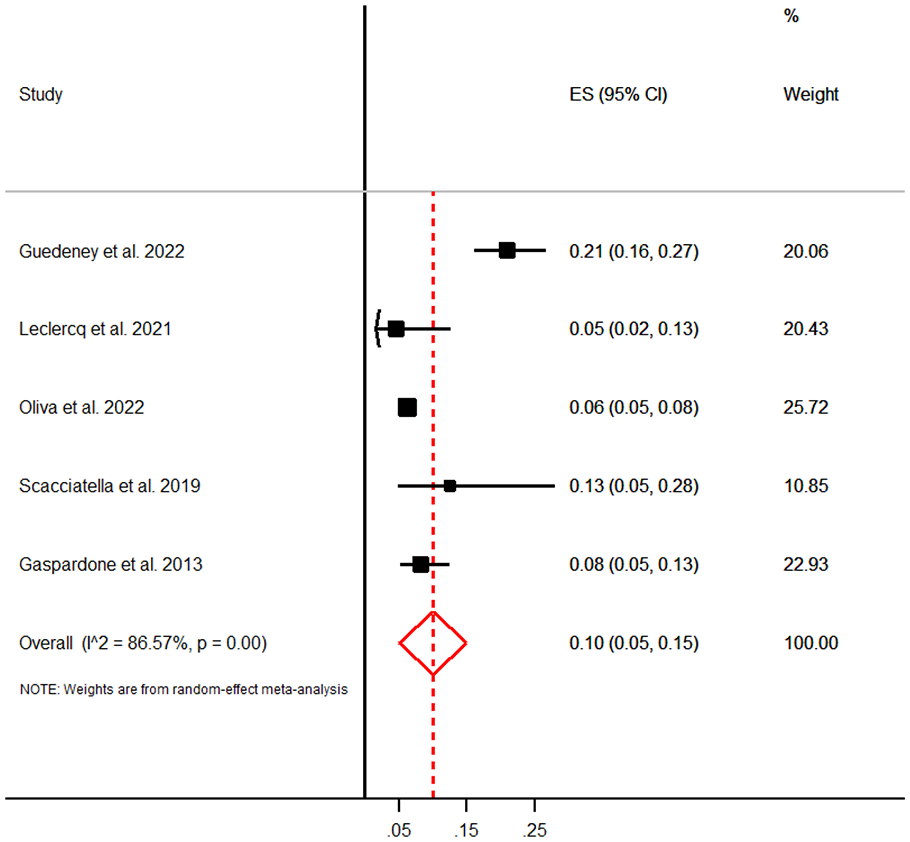
Meta-analysis of the incidence of AF after PFO closure in patients who underwent long-term cardiac monitoring.
The pooled AF detection rate was 10% (95% CI: 5–15) in patients who underwent cardiac monitoring. 140 Two studies utilised ILR in patients over 55 years and associated cardiovascular risk factors.135,138 A previously published study reported a median delay from procedure to arrhythmia of 14.0 days (IQR: 6.5–19.0 days), and one-half of these patients reported symptomatic episodes. 138 Determinants for AF were older age (adjusted OR: 1.67 per 10-year increase; 95% CI: 1.18–2.36), device left disc diameter of 25 mm compared to smaller diameter (adjusted OR: 2.67; 95% CI: 1.19–5.98) and male sex (adjusted OR: 4.78; 95% CI: 1.96–11.66). Data from Canadian administrative health data recorded new-onset AF following PFO closure in patients having diabetes and age 60 years with an HR of 2.49; 95% CI: 1.48–4.18; p = 0.001 and 2.82; 95% CI: 1.76–4.51; p = 0.001), respectively. 139
Another study also detected a statistically higher rate of AF in PFO patients independently from PFO closure. 137
Additional information
In one cohort, oral anticoagulation was prescribed in 29 (56.9%) patients. 139 Electric and pharmacologic cardioversion was performed in one and eight patients, respectively. Percutaneous ablation was performed in four patients. Among the 19 ILR-monitored patients presenting with more than 1 episode of supraventricular arrhythmia, there were seven cases of recurrence more than 3 months after the index event. One TIA occurred in one patient 1 month after admission for symptomatic flutter, despite adherence to oral anticoagulation. Among patients without supraventricular arrhythmia, 1 stroke and 1 TIA occurred in ILR monitored patients. 138 Other studies showed a statistically significant reduction of AF prevalence after percutaneous closure of PFO, suggesting some antiarrhythmic effect of the procedure. 141
Several ongoing studies are investigating the role of prolonged cardiac monitoring after PFO closure and the clinical implications of AF occurrence on the risk of recurrent stroke such as PFO-AF Observational Cohort Study (France, NCT04926142); ALFA ROMEO Observational Cohort Study (Switzerland, NCT04881578), PREDICT-AF-PFO Nonrandomized Clinical Trial (Germany, NCT04898361) and DEFENCE-ELDERLY Observational Cohort Registry (South Korea, NCT04285918).
Additional information
We decided not to develop a PICO question regarding the screening for subclinical AF in patients below 60 years of age who have experienced a cryptogenic stroke and PFO as Rubiera et al. already addressed these topics in the ESO screening guidelines. However, we listed this chapter’s relevant expert opinion statements to support physicians in the PFO management. 142


Discussion
This guideline document was developed following the GRADE methodology and aims to assist physicians in decision-making regarding cryptogenic stroke and PFO. All recommendations and Expert consensus statements are summarised in Table 12.
Synoptic table of all recommendations.

Although accurate diagnosis of PFO and associated anatomical characteristics such as the presence of an ASA are very important for risk stratification and decisions on PFO closure, we were unable to provide an evidence-based recommendation on the best diagnostic strategy in patients with recent ischaemic stroke. Indeed, although TOE is usually considered to be the conventional reference to diagnose a PFO, its sensitivity against robust invasive techniques is imperfect. Bayesian methods have been suggested to deal with meta-analysis of diagnostic test accuracy in the context of imperfect reference standards but were considered to be beyond the scope of this guideline.143,144However, we provide a pragmatic expert consensus statement to use c-TOE to gain additional information about the presence and anatomy of the PFO after an initial assessment based on locally available techniques (c-TTE or c-TCD, in case TOE is not considered part of first-line investigations). In line with this pragmatic approach, we did not include recommendations on the preparation of the AS with and without blood, even if there are data showing that the addition of blood increases the sensitivity.37,73 The same pragmatic approach should be applied for longterm follow up examinations for residual RLS as no standardised protocols could be established for the lack of gold standards.
In order to increase the number of studies on PFO diagnosis, we included our study population, not only acute ischaemic strokes and TIAs, but also patients with migraine, vertigo, syncope, myocardial infarction, and peripheral embolism.
In addition, we did not include a comprehensive diagnostic protocol for patients with cryptogenic stroke to rigorously exclude other rare aetiologies beyond a PFO, as this protocol should follow international and national clinical guidelines. 145
We found high-quality evidence to recommend PFO closure plus antiplatelet therapy in selected patients aged 18–60 years in whom no other evident cause of stroke is found but a PFO (i.e. PFO-associated stroke). The PASCAL classification system can be used to select such candidates for PFO closure. Patients with both a large right-to-left shunt and an ASA benefit most from PFO closure. There is insufficient evidence to make an evidence-based recommendation on PFO closure in patients older than 60 and younger than 18 years. Whereas, for patients between 18 and 60 years whose stroke is considered unlikely to be related to PFO as per the PASCAL classification, PFO closure is generally not recommended unless there is substantial clinical evidence suggesting a causal relationship (see Expert Consensus).
We suggest against long-term anticoagulation in patients with PFO-associated stroke unless anticoagulation is indicated for other medical reasons. Moreover, our guidelines did not specifically address the management of anticoagulation therapy for concurrent deep venous thrombosis in patients with PFO and PFO related stroke. We defer to the existing specialised guidelines currently in place for the type, dosages, and duration of anticoagulation treatment. 146 These existing guidelines advocate for deferral elective procedures, such as PFO closure, to mitigate the risk of recurrent thromboembolic events. Additionally, it is important to note that there is a lack of data regarding the simultaneous management of anticoagulation treatment and PFO closure. 146
In our guidelines we did not address the issue of detection for subclinical AF in patients with PFO and cryptogenic stroke as the recent ESO guideline on screening for subclinical atrial fibrillation after stroke or TIA of undetermined origin addressed this thoroughly. 142
Regarding the detection of AF after PFO, we observed that, consistent with the IPDMA, the patients likely to derive greater benefit from PFO closure also appeared to be at lower risk of device-associated adverse events such as atrial fibrillation, making the harm benefit trade-offs of device closure more clearly favourable in the possible and probable groups according to PASCAL classification. 7 The lower risk of adverse events in the patients with potential high benefit is in line with prior evidence showing a higher risk of incident AF in patients with lower RoPE scores, 147 who are older and have more vascular risk factors. This increased risk may reflect occult AF being a more likely mechanism for the index stroke in these patients, stressing the need for an accurate AF rule-out at the diagnostic stage, but may also reflect a greater susceptibility to arrhythmogenic effects of device-tissue contact.
The MWG rated late-onset AF post-stroke as a lower priority, particularly in the context of patients who have undergone PFO when selected by PASCAL criteria. In such cases, late-onset AF detected after the cerebrovascular incident may be incidental and not directly related to the initial stroke event. To this regard, Sposato et al. reported that AF detected after a stroke is associated with a 26% lower risk of future strokes compared to AF that was known prior to the stroke. This may be because AF before a stroke is typically more severe and is detected through ECG. On the other hand, AF, identified after a stroke, is usually discovered during short-term or extended monitoring, suggesting a lesser severity. 148 Furthermore, patients who are diagnosed with AF after a stroke generally present with fewer risk factors and less cardiac disease, which contributes to a reduced risk of stroke.
Another open issue is the long-term effects of postprocedural AF on the remodelling of the left atrium, as well as the progression of left atrial fibrosis and the increased risk of AF, remain areas that require further investigation. 97 Therefore, a personalised approach to cardiac rhythm monitoring in PFO patients after closure needs to be addressed. 149
In this guideline, we have not addressed which antiplatelet agent is superior for long-term treatment as only the PC trial compared the Amplatzer PFO Occluder with any antiplatelet or anticoagulation therapy of the physician’s choice, which resulted to be acetylsalicylic acid, ticlopidine, clopidogrel and warfarin. 111 Regarding anticoagulation, the current evidence incorporated a variety of anticoagulants for comparison, such as warfarin and DOACs in order to assess the outcomes of anticoagulant therapy.13,119 Therefore, the choice of the antithrombotic agent may be personalised, based on individual clinical characteristics which may prompt to opt for a specific agent.
An open question is the management of the patients who underwent PFO and have residual RLS. The percentages and impact of RLS vary from 0% to 19%101–103 based on the different case series. Some Authors reported that residual RLS was influenced by the device type, PFO canal length, and ASA extent. 103 The incidence of right-to-left shunting (RLS) was 19.5% at a mean of 4 months’ follow-up, which reduced to 8.4% at 11 ± 2 months. 101 Moreover, in the same study alternative causes for residual RLS were detected such as pulmonary arteriovenous malformation.
We decided not to develop a PICO question regarding the screening for subclinical AF in patients below 60 years of age who have experienced a cryptogenic stroke and PFO as Rubeira et al. already addressed these topics in the screening guidelines.
There are also contrasting results on the embolic risk of residual RLS. Hammerstingl et al. 102 reported no increased risk of thromboembolic events, as 3.2% of patients experienced arterial embolic events unrelated to residual PFO shunting or device-related thrombus formation. Another single-centre study of 118 patients reported a 98.1% total occlusion rate at a mean follow-up of 50 months, with no recurrent neurological events observed. 104 Contrastingly, a large prospective cohort study revealed that patients with residual shunts had a higher rate of stroke or TIA recurrence compared to those with complete PFO closure – 2.32 versus 0.75 events per 100 patient-years, respectively. 103 These studies collectively suggest that while residual shunt post-PFO closure is possible, the correlation with recurrent thromboembolic events is inconsistent across studies. Patients should be informed about these possible risks before performing PFO closure.
We decided not to include TIA as the index event in our guideline. This decision was based on the fact that RCTs except for the Closure trial, did not include TIA as index events. 106 However, it is noteworthy that the TIA subgroup in CLOSURE 1 constituted less than 30% of the participants, and the subgroup analysis revealed no significant distinctions. 106 However, international guidance papers, including the European Action Plan and the European Position paper on the management of patients with PFO, often grouped TIAs and strokes in the same management pathway.4,145 PFO patients tend to be younger in comparison to patients with other well-defined causes of stroke. This age difference can potentially result in the overdiagnosis of TIAs due to the higher prevalence of conditions that mimic strokes in this particular age group. Conversely, relying solely on the low ABCD2 score may exclude certain patients from the diagnostic evaluation process. Therefore, based on current clinical practice, individuals presenting with sudden transient clinical symptoms such as motor deficits or speech disturbances may be considered as vascular patients when other conditions such as migraine with aura or seizures are excluded.
Moreover, we did not address sex-specific issues, such as PFO-related stroke during pregnancy. These specific concerns necessitate dedicated pathways and a multidisciplinary approach to provide comprehensive and tailored care, particularly in the context of managing delivery. In such cases, close collaboration between obstetricians, neurologists, cardiologists, and other relevant specialists is essential to ensure the safest and most appropriate management of both the pregnancy and the associated medical condition.
In conclusion, in patients with PFO-related stroke aged 18–60 years, PFO closure plus time-limited antiplatelet therapy is the recommended strategy for secondary prevention of recurrent stroke, with a low rate of complications. Further RCTs are required to test whether and in which patients over 60 years of age with stroke and PFO should be treated with PFO closure.
Plain language summary
This guideline is intended for individuals who have experienced an ischaemic stroke and have been diagnosed with a patent foramen ovale (PFO). A PFO is a heart condition where a small hole between the heart’s two upper chambers, which usually closes after birth, remains open. Most people with a PFO never know they have it, and it does not cause problems. Rarely, this hole can let blood clots travel to the brain, which might cause a stroke.
These are the treatment recommendations:
This guideline offers a structured approach for healthcare professionals to manage patients with ischaemic stroke and PFO, considering age, other medical conditions, and the potential benefits of PFO closure and medication. It emphasises evidence-based decision-making and individualised care. However, it is important to note that medical guidelines can evolve over time based on new research findings and clinical experiences.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241247978 – Supplemental material for European Stroke Organisation (ESO) Guidelines on the diagnosis and management of patent foramen ovale (PFO) after stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873241247978 for European Stroke Organisation (ESO) Guidelines on the diagnosis and management of patent foramen ovale (PFO) after stroke by Valeria Caso, Guillaume Turc, Azmil H Abdul-Rahim, Pedro Castro, Salman Hussain, Avtar Lal, Heinrich Mattle, Eleni Korompoki, Lars Søndergaard, Danilo Toni, Silke Walter and Christian Pristipino in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241247978 – Supplemental material for European Stroke Organisation (ESO) Guidelines on the diagnosis and management of patent foramen ovale (PFO) after stroke
Supplemental material, sj-docx-2-eso-10.1177_23969873241247978 for European Stroke Organisation (ESO) Guidelines on the diagnosis and management of patent foramen ovale (PFO) after stroke by Valeria Caso, Guillaume Turc, Azmil H Abdul-Rahim, Pedro Castro, Salman Hussain, Avtar Lal, Heinrich Mattle, Eleni Korompoki, Lars Søndergaard, Danilo Toni, Silke Walter and Christian Pristipino in European Stroke Journal
Supplemental Material
sj-docx-3-eso-10.1177_23969873241247978 – Supplemental material for European Stroke Organisation (ESO) Guidelines on the diagnosis and management of patent foramen ovale (PFO) after stroke
Supplemental material, sj-docx-3-eso-10.1177_23969873241247978 for European Stroke Organisation (ESO) Guidelines on the diagnosis and management of patent foramen ovale (PFO) after stroke by Valeria Caso, Guillaume Turc, Azmil H Abdul-Rahim, Pedro Castro, Salman Hussain, Avtar Lal, Heinrich Mattle, Eleni Korompoki, Lars Søndergaard, Danilo Toni, Silke Walter and Christian Pristipino in European Stroke Journal
Footnotes
Acknowledgements
We thank ESO staff Yvonne Brüchert and Sabrina Mutter for excellent administrative support as well as the Chair and Co-chair of the Guidelines Board, Terence Quinn, and Diana Aguiar de Sousa, for their support during the conduct of the guideline project.
Correction (June 2024):
This article has been updated to correct the placement of Table 1, Expert Consent Statements and Evidence based recommendations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the development of these guidelines was provided by the European Stroke Organisation, Basel, Switzerland. With the exception of the methodologists, the authors did not receive financial support for the development, writing or publication of this guideline.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
Prof. Valeria Caso
Contributorship
Valeria Caso, Guillaume Turc, and Christian Pristipino wrote and supervised the manuscript, which included overall oversight and ensuring the integrity of the work. Silke Walter, Abdul-Rahim, Pedro Castro, and Eleni Korompoki were responsible for writing PICO 1 of the manuscript. Salman Hussain and Avtar Lal performed the statistical analysis for the guidelines. Heinrich Mattle, Lars Søndergaard, Danilo Toni wrote PICO 2, 3, and 4. Valeria Caso prepared PICO 5 and 6, suggesting a focus on additional specific clinical questions or components of the research. All members developed a list of topics and corresponding questions of most significant clinical interest, identified three overarching themes (PFO diagnosis, treatment of PFO-associated stroke, and post-procedural management), and performed literature research.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.