Abstract
Introduction:
Hemorrhagic stroke may cause changes in intracranial pressure (ICP) and cerebral perfusion pressure (CPP), which may influence the prognosis of patients. The aim of this study was to investigate the relationship between early ICP, CPP, and 28-day mortality in the intensive care unit (ICU) of patients with hemorrhagic stroke.
Patients and methods:
A retrospective study was performed using the Medical Information Mart for Intensive Care (MIMIC-IV) and the eICU Collaborative Research Database (eICU-CRD), including hemorrhagic stroke patients in the ICU with recorded ICP monitoring. The median values of ICP and CPP were collected for the first 24 h of the patient’s monitoring. The primary outcome was 28-day ICU mortality. Multivariable Cox proportional hazards models were used to analyze the relationship between ICP, CPP, and 28-day ICU mortality. Restricted cubic regression splines were used to analyze nonlinear relationships.
Results:
The study included 837 patients with a 28-day ICU mortality rate of 19.4%. Multivariable analysis revealed a significant correlation between early ICP and 28-day ICU mortality (HR 1.08, 95% CI 1.04–1.12, p < 0.01), whereas early CPP showed no correlation with 28-day ICU mortality (HR 1.00, 95% CI 0.98–1.01, p = 0.57), with a correlation only evident when CPP < 60 mmHg (HR 1.99, 95% CI 1.14–3.48, p = 0.01). The study also identified an early ICP threshold of 16.5 mmHg.
Discussion and conclusion:
Early ICP shows a correlation with 28-day mortality in hemorrhagic stroke patients, with a potential intervention threshold of 16.5 mmHg. In contrast, early CPP showed no correlation with patient prognosis.

Introduction
Hemorrhagic stroke, which includes intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH), is a major contributor to the global neurological disease burden. 1 Statistical data indicate that approximately 50 million strokes occur annually worldwide, of which 9.6 million are classified as hemorrhagic. The mortality and disability rates associated with hemorrhagic stroke greatly exceed those associated with ischemic stroke. Particularly in low- and middle-income countries, the incidence and mortality rates associated with hemorrhagic stroke are significant and increasing. 2 As a result, the development of effective treatment approaches and prognostic assessments for hemorrhagic stroke has become paramount.
In hemorrhagic stroke, intracranial bleeding can cause a rapid increase in intracranial pressure (ICP). This is primarily due to the fact that the cranial cavity is a confined space. The formation of hemorrhage and edema within the brain occupies additional space, leading to an increase in ICP. 3 In addition, cerebral perfusion pressure (CPP) represents the difference between mean arterial pressure (MAP) and ICP and serves as an indicator of the primary pressure responsible for providing oxygen and nutrients to the brain. An increase in ICP can potentially lead to a decrease in CPP, causing cerebral tissue ischemia and hypoxia, exacerbating cerebral injury. 4 Accordingly, monitoring and regulation of ICP and CPP have significant implications for improving the prognosis of patients with hemorrhagic stroke.
However, the majority of consensus and guidelines for the management of ICP and CPP are based primarily on traumatic brain injury (TBI) research.5,6 The Brain Trauma Foundation’s TBI guidelines suggest an ICP treatment threshold of 22 mmHg, while maintaining CPP in the range of 60–70 mmHg. 7 However, compared to TBI, research specific to ICH and SAH remains limited, with most therapeutic threshold recommendations derived from TBI guidelines. In addition, although studies have examined the relationship between ICP, CPP and hemorrhagic stroke prognosis,8–11 reports on the association between their early variations and patient outcomes, particularly short-term intensive care unit (ICU) mortality, remain scarce.
Investigation of early variations in ICP and CPP may contribute significantly to identifying high-risk patients, guiding therapeutic decisions, and ultimately predicting and improving the prognosis of patients with hemorrhagic stroke. Therefore, the primary objective of our research is to investigate the relationship between early ICP and CPP fluctuations and 28-day ICU mortality in patients with hemorrhagic stroke. At the same time, we aim to identify thresholds for ICP and CPP to provide a more scientifically sound basis for early treatment strategies and prognostic assessment of hemorrhagic stroke.
Methods
Data source
This retrospective cohort study was based on the Medical Information Mart for Intensive Care (MIMIC-IV) (version 2.2) and the eICU Collaborative Research Database (eICU-CRD) v2.0. The MIMIC-IV database contains information on all patients treated at Beth Israel Deaconess Medical Center from 2008 to 2019. 12 The eICU-CRD is a multicenter telemedicine database containing data from more than 200,000 patients admitted from 335 ICUs at 208 hospitals in the United States between 2014 and 2015. 13 The databases contain comprehensive information such as length of stay, laboratory tests, medication management, and vital signs for each patient. To protect patient privacy, all personal information was de-identified and replaced with random codes to conceal patient identity. Therefore, this study did not require patient consent or ethics approval. The researcher (Zhang) has completed the training program provided by the collaborating institution (Certificate No. 53496787) and is qualified to use the database and extract data.
Study population
Patients diagnosed with non-traumatic SAH and non-traumatic ICH according to the International Classification of Diseases, 9th and 10th revisions (ICD-9 and ICD-10) were included in the analysis. The following patients were excluded: (1) patients younger than 18 years; (2) patients with multiple ICU admissions (only first admission considered); (3) patients with ICU stays of less than 24 h; (4) patients without ICP monitoring records.
Data extraction and processing
Patients’ ICP and CPP recordings during the first 24 h were extracted and median values were calculated. Missing CPP values were imputed by subtracting the median ICP from the median MAP within 24 h.
The primary outcome variable was 28-day ICU mortality, and secondary outcome variables included hospital mortality, ICU length of stay, and total hospital length of stay.
Other covariates included: (1) demographics; (2) median vital signs within 24 h of ICU admission; (3) initial laboratory values within 24 h of ICU admission; (4) sequential organ failure assessment (SOFA) and Glasgow coma scale (GCS) scores, within 24 h of ICU admission; (5) comorbidities (hypertension, diabetes, congestive heart failure, chronic obstructive pulmonary disease, chronic kidney disease, acute kidney injury); (6) use of mechanical ventilation (MV), vasopressors, sedatives, mannitol, hypertonic saline, within 24 h of ICU admission and whether craniotomy was performed. Variables with more than 20% missing data were excluded. For continuous variables, outliers and obviously inconsistent values were treated as missing data. Missing values were imputed using multiple imputation with the “missForest” package. 14
Statistical analysis
Continuous variables were described as median with interquartile range. The Mann-Whitney U test was used for statistical comparisons between two groups. Categorical variables were reported as counts and percentages, with the chi-squared test or Fisher’s exact test used for comparisons between groups. About 28-day ICU survival curves were constructed using the Kaplan-Meier method and compared using the log-rank test.
Univariate and multivariable Cox proportional hazards models were used to examine the associations between variables and 28-day ICU mortality and in-hospital mortality. Variables with p < 0.05 in the univariate Cox proportional hazards models were included in the multivariable Cox proportional hazards models. Multivariable linear regression was used to examine the associations between variables and ICU length of stay and hospital length of stay.
Multivariable Cox proportional hazards models with restricted cubic splines (RCS) were used to account for potential nonlinear relationships between the variables and primary outcomes. If nonlinearity was detected, a recursive algorithm was used to calculate threshold inflection points, and two-segment Cox proportional hazards models were used to examine the relationships between the variable and primary outcomes on either side of the inflection point.
Sensitivity analysis was performed to determine the robustness of the results. Considering the impact of do not resuscitate/do not intubate (DNR/DNI) status on patient outcomes, as well as the impact of MAP on ICP and CPP, sensitivity analyses were performed specifically for these two factors. Subgroup analysis was performed according to age (<65 or ⩾65), sex, median MAP, craniotomy, vasopressors, sedation, mannitol, hypertonic saline, MV, and database. Subgroup analyses, RCS plots, and threshold assessments were performed specifically for the hemorrhagic stroke diagnostic subgroup.
All statistical analyses were performed using R 4.2.3 and STATA 15.1, with a p < 0.05 considered statistically significant.
Results
Study participants and baseline characteristics
After applying the exclusion criteria, a total of 486 and 351 patients were included from the MIMIC-IV and eICU-CRD databases, respectively (Figure 1). The 28-day ICU and in-hospital mortality rates for patients with hemorrhagic stroke were 19.4% and 28.2%, respectively. Baseline characteristics of all patients categorized by 28-day ICU mortality are shown in Table 1. When comparing the two groups, the initial 24-h median ICP was higher in the death group than in the survival group (p < 0.01), and the initial 24-h median CPP was lower than the survival group (p < 0.01).
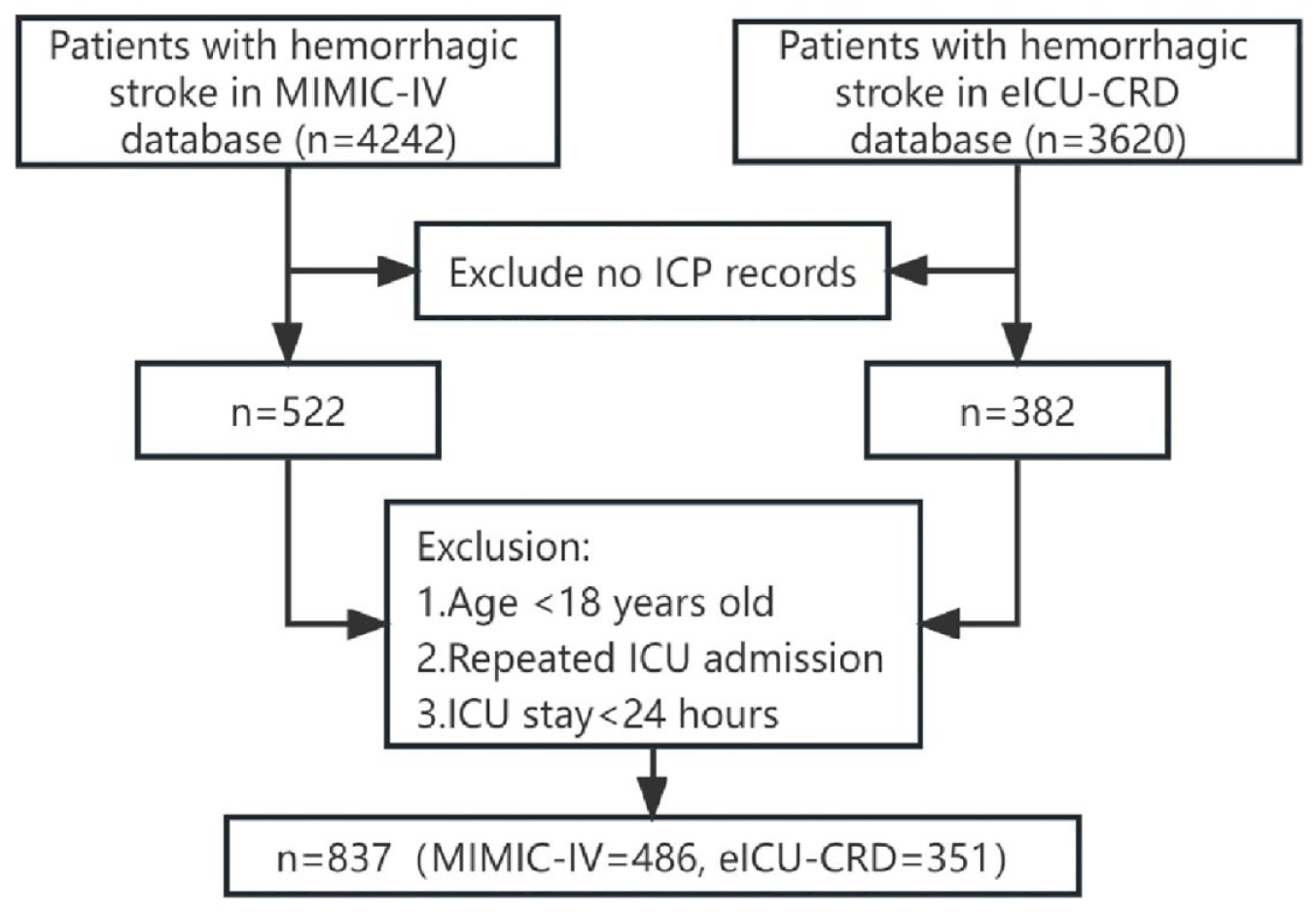
Flowchart.
Baseline characteristics categorized by 28-day ICU mortality.
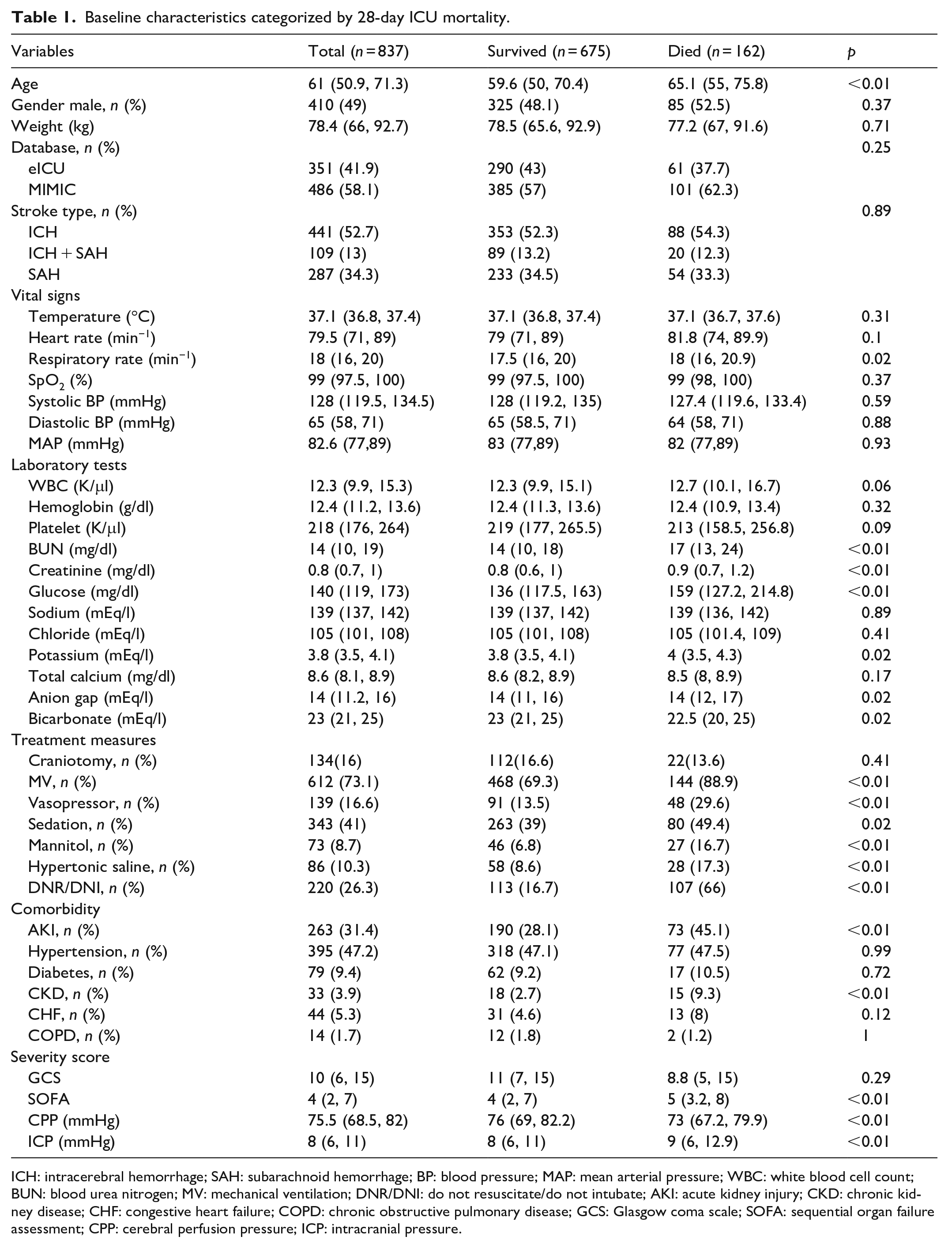
ICH: intracerebral hemorrhage; SAH: subarachnoid hemorrhage; BP: blood pressure; MAP: mean arterial pressure; WBC: white blood cell count; BUN: blood urea nitrogen; MV: mechanical ventilation; DNR/DNI: do not resuscitate/do not intubate; AKI: acute kidney injury; CKD: chronic kidney disease; CHF: congestive heart failure; COPD: chronic obstructive pulmonary disease; GCS: Glasgow coma scale; SOFA: sequential organ failure assessment; CPP: cerebral perfusion pressure; ICP: intracranial pressure.
Additionally, we performed a comparative analysis of baseline characteristics between patients excluded due to unmonitored ICP and those included in the study. The results showed that, compared to patients without ICP monitoring, those with ICP monitoring were younger (61 years vs 68 years; p < 0.01), had lower GCS scores (10 vs 15; p < 0.01), a higher incidence of MV use (73.1% vs 29.6%; p < 0.01), a higher prevalence of vasopressor use (16.6% vs 8.1%; p < 0.01), higher prevalence of mannitol use (8.7% vs 4.5%; p < 0.01), higher prevalence of hypertonic saline use (10.3% vs 4.9%; p < 0.01), longer ICU stays (292 h vs 60 h; p < 0.01), and higher 28-day ICU mortality (19.4% vs 13.6%; p < 0.01) (Supplemental Table S1).
Relationship between ICP, CPP, and outcomes
In univariate Cox proportional hazards analysis, both ICP and CPP were associated with 28-day ICU mortality (p < 0.01). Variables with p < 0.05 in the univariate analysis were age, respiratory rate, white blood cell count, urea nitrogen, creatinine, blood glucose, anion gap, blood potassium, bicarbonate, MV, acute kidney injury, vasoactive drugs, sedative drugs, mannitol, hypertonic saline, chronic kidney disease, GCS score, SOFA score, and CPP and ICP (Supplemental Table S2). When these variables were included in the multivariable Cox regression analysis, ICP was associated with 28-day ICU mortality (hazard ratio [HR] 1.08, 95% confidence interval [CI] 1.04–1.12, p < 0.01), whereas CPP was not associated with 28-day ICU mortality (HR 1.00, 95% CI 0.98–1.01, p = 0.57) (Table 2).
Multivariable Cox proportional hazards regression for 28-day ICU mortality.
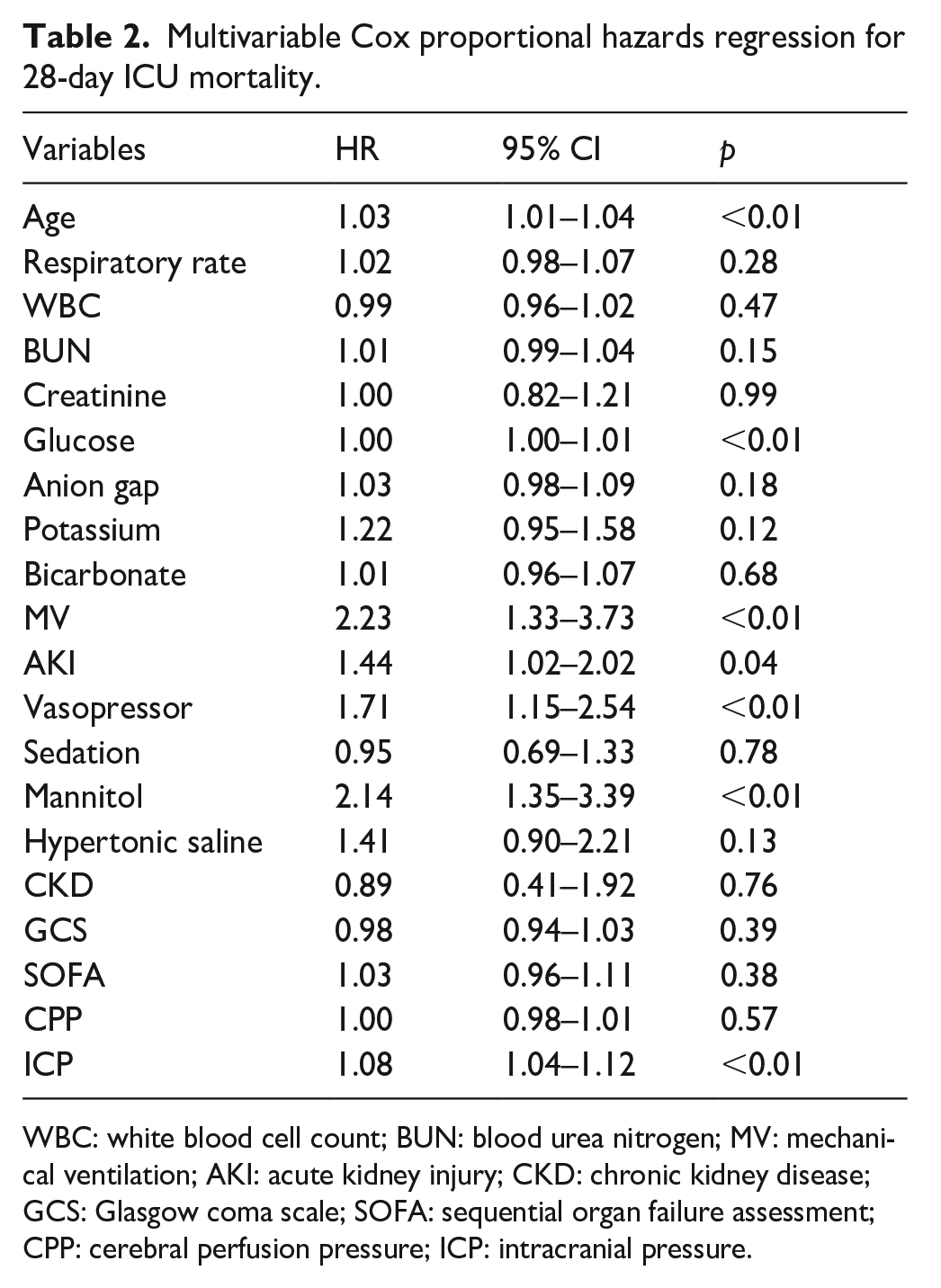
WBC: white blood cell count; BUN: blood urea nitrogen; MV: mechanical ventilation; AKI: acute kidney injury; CKD: chronic kidney disease; GCS: Glasgow coma scale; SOFA: sequential organ failure assessment; CPP: cerebral perfusion pressure; ICP: intracranial pressure.
Multivariable Cox proportional hazards analysis was performed for different ranges of CPP according to the TBI guidelines thresholds. Using 60–70 mmHg as the normal reference range, CPP < 60 mmHg was associated with 28-day ICU mortality (HR 1.99, 95% CI 1.14–3.48, p = 0.01), whereas there was no association between CPP > 70 mmHg and 28-day ICU mortality (HR 1.44, 95% CI 0.97–2.15, p = 0.07) (Supplemental Table S3).
Among secondary outcomes, ICP was associated with in-hospital mortality (HR 1.10, 95% CI 1.06–1.13, p < 0.01), whereas CPP was not associated with in-hospital mortality (HR 1.01, 95% CI 0.99–1.02, p = 0.48). ICP was not associated with ICU length of stay (p = 0.14) and CPP was not associated with ICU length of stay (p = 0.06). ICP was associated with hospital length of stay (p = 0.04), but CPP was not associated with hospital length of stay (p = 0.24) (Supplemental Table S4).
Analysis of nonlinear relationships
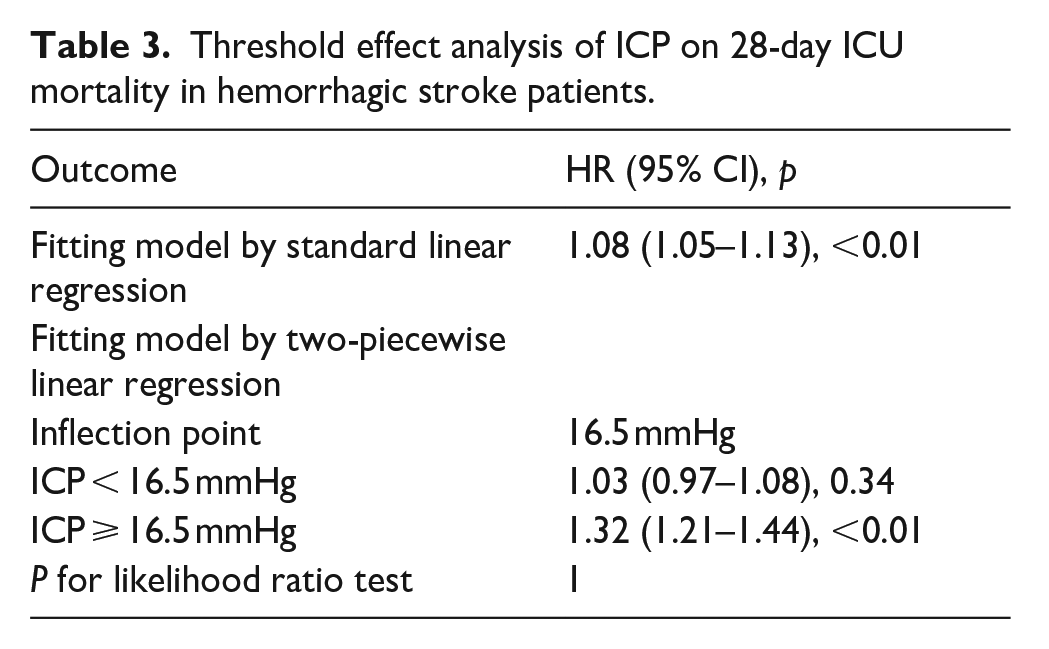
In the multivariable Cox proportional hazards model fitted with RCS, we found a nonlinear relationship between ICP and 28-day ICU mortality (Figure 2). Using a two-segment regression model, we found the threshold turning point of ICP to be 16.5 mmHg (Table 3). ICP to the left of the inflection point was not significantly associated with 28-day ICU mortality (HR1.03, 95% CI 0.97–1.08, p = 0.34), whereas ICP to the right of the inflection point was significantly positively associated with 28-day ICU mortality (HR1.32, 95% CI 1.21–1.44, p < 0.01). The Kaplan-Meier survival curve also suggested that patients with an ICP < 16.5 mmHg had a significantly higher 28-day ICU survival rate than patients with an ICP ⩾ 16.5 mmHg (Figure 3).
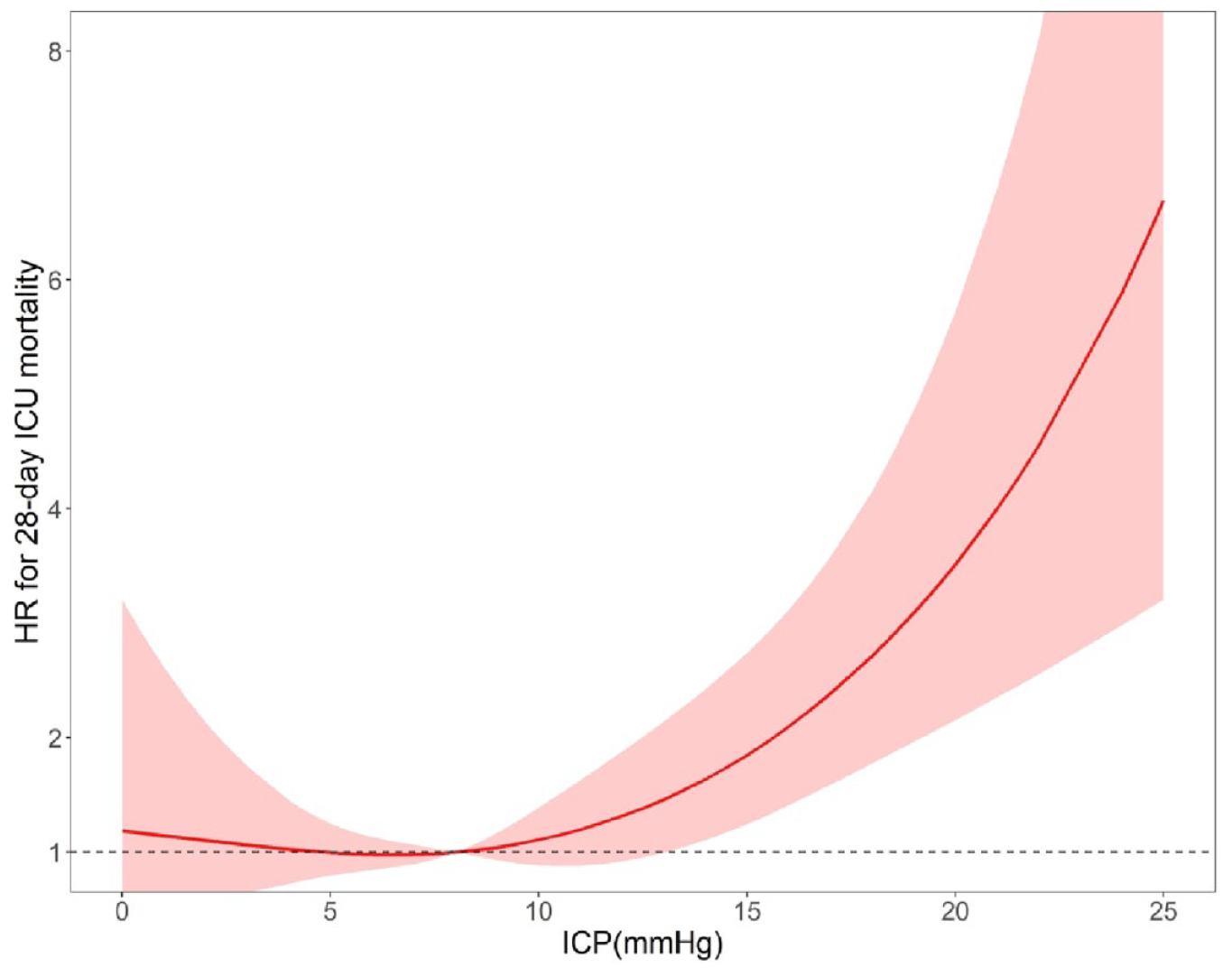
RCS plot of ICP versus 28-day ICU mortality.
Threshold effect analysis of ICP on 28-day ICU mortality in hemorrhagic stroke patients.
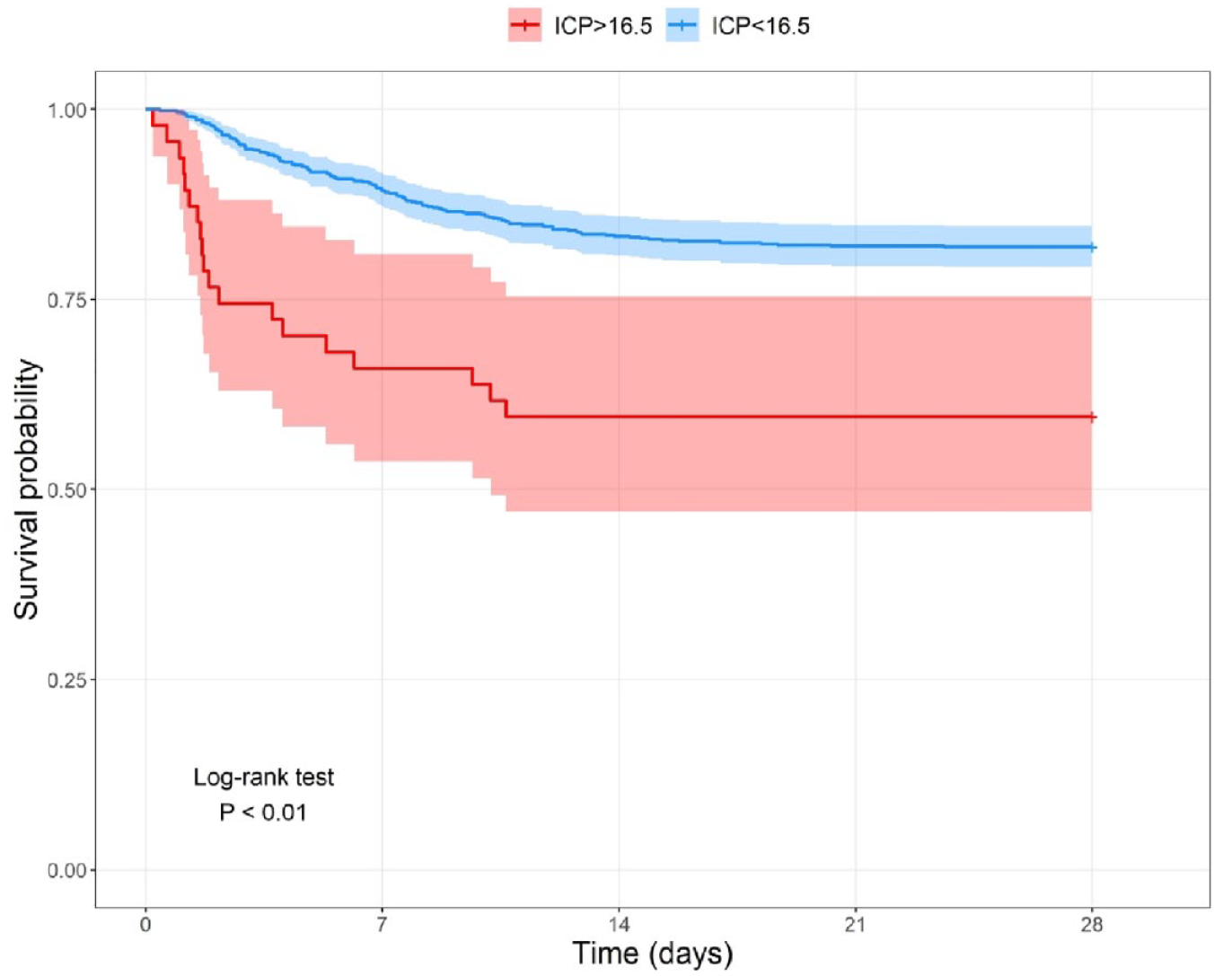
Kaplan-Meier curves for 28-day ICU mortality in the ICP < 16.5 mmHg and ICP ⩾ 16.5 mmHg groups.
Sensitivity analysis
Considering the influence of DNR/DNI status on 28-day ICU mortality, we performed two types of sensitivity analyses: one excluding DNR/DNI patients and another including DNR/DNI status as a variable in the multivariable analysis. The results of both sensitivity analyses consistently showed a significant correlation between ICP and 28-day ICU mortality, whereas no significant correlation was observed between CPP and the same outcome (Supplemental Table S5). In the analysis of different CPP ranges, an association with 28-day ICU mortality remained only when CPP < 60 mmHg (Supplemental Table S6). In the threshold analysis, after excluding patients with DNR/DNI status, the threshold for ICP was identified as 15.5 mmHg; however, when DNR/DNI status was included in the multivariable analysis, the threshold for ICP remained at 16.5 mmHg (Supplemental Table S7).
We also included MAP in the multivariable analysis. The results showed a persistent significant correlation between ICP and 28-day ICU mortality. No association was found between CPP and 28-day ICU mortality. The threshold for ICP remained at 16.5 mmHg (Supplemental Tables S5–S7).
Subgroup analysis
Subgroup analysis of the multivariable model was performed according to age (<65 years or ⩾65 years), sex, median MAP (<82.6 mmHg or ⩾82.6 mmHg), craniotomy, vasopressor, sedation, mannitol, hypertonic saline, MV, and database. The analysis results showed that there was no significant association between ICP and 28-day ICU mortality in the subgroups of males, patients with a median MAP < 82.6 mmHg, those who underwent craniotomy, those not using MV, and in the eICU database group (Figure 4). In the subgroup analysis examining the association between CPP and 28-day ICU mortality, this association was not statistically significant in any of the subgroups (Supplemental Figure S1).
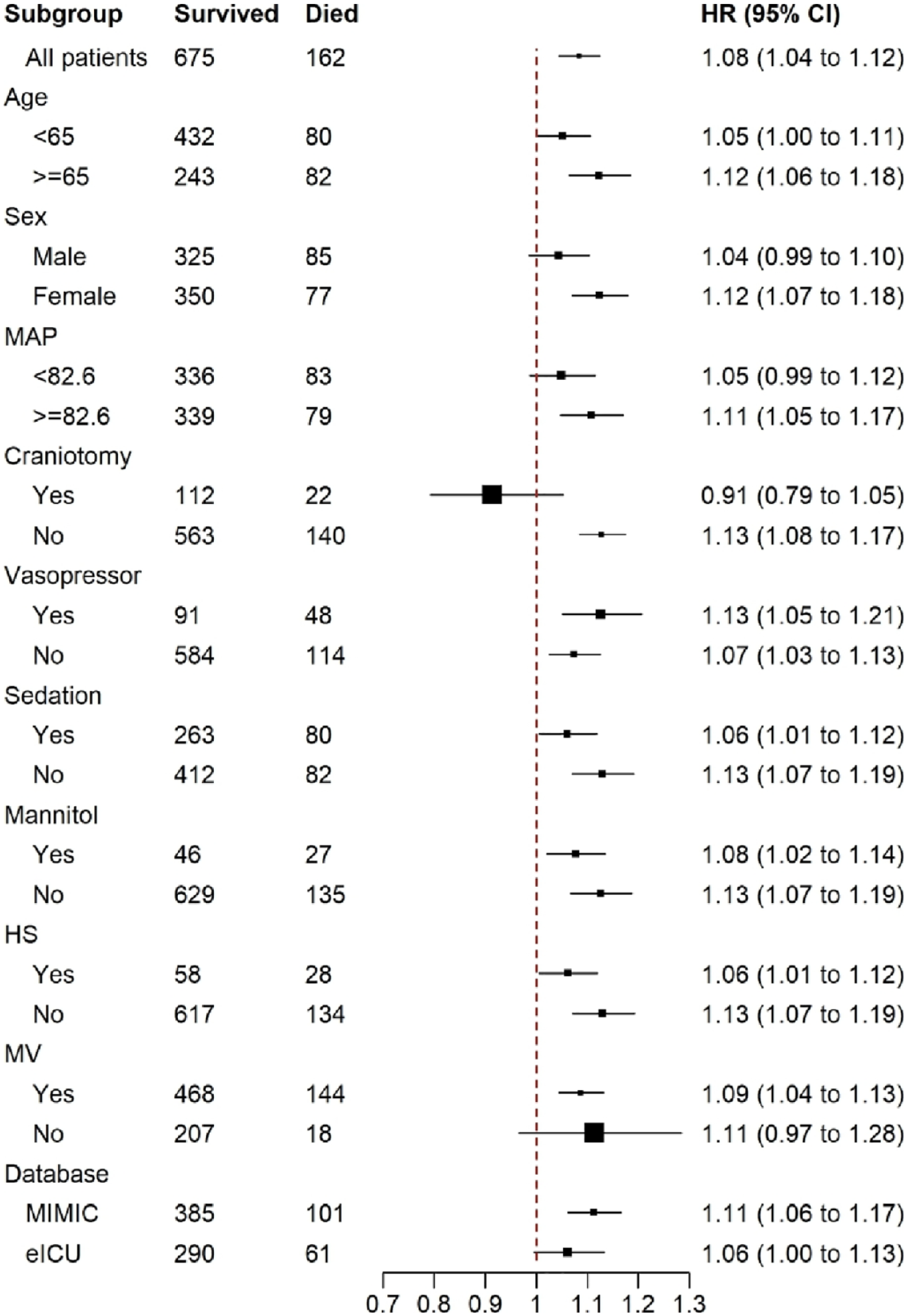
Forest plot of the subgroup analysis of ICP and 28-day ICU mortality. MAP: mean arterial pressure; HS: hypertonic saline; MV: mechanical ventilation.
In the diagnostic subgroup analysis for hemorrhagic stroke, there was an association between ICP and 28-day ICU mortality in both the ICH and SAH groups, but this association was not observed in the combined ICH and SAH group (Supplemental Table S8). No significant correlation was found between CPP and 28-day ICU mortality in any of these subgroups (Supplemental Table S8). The use of RCS fitting for the multivariable Cox proportional hazards model in both the ICH and SAH groups revealed nonlinear relationships (Supplemental Figures S2 and S3). Threshold analysis identified the ICP threshold as 15.5 mmHg in the ICH group and 18 mmHg in the SAH group (Supplemental Table S9).
Discussion
In this study, we investigated the relationship between early ICP, CPP, and prognosis in patients with hemorrhagic stroke. First, we found that median ICP within the first 24 h correlated with 28-day ICU mortality, in-hospital mortality, and length of hospital stay. Conversely, median CPP within the first 24 h showed no significant correlation with mortality and length of stay, and only when CPP fell below 60 mmHg did it show a correlation with 28-day ICU mortality. Subsequently, we identified that the intervention threshold for ICP was established at 16.5 mmHg.
The earliest documentation of ICP monitoring dates back to the 1960s, 15 although the use of invasive ICP monitoring has been controversial. 3 In a 2015 meta-analysis ICP monitoring was not found to reduce mortality in patients with TBI. 16 In contrast, in another meta-analysis for severe TBI, the use of ICP monitoring was found to reduce mortality in patients hospitalized after 2007. 17 The SYNAPSE-ICU study, a large, multicenter, prospective study, found that ICP monitoring could reduce 6-month mortality in patients with severe brain injury. 18 Recent Brain Trauma Foundation guidelines advocate ICP monitoring in all salvageable patients with severe TBI (post-resuscitation GCS score 3–8) and CT abnormalities such as hematoma, contusion, swelling, herniation or basal cistern compression. 7 There are currently no formal recommendations for ICP monitoring in hemorrhagic stroke. ICH guidelines suggest that ICP monitoring and external ventricular drainage treatment should be considered in ICH patients with clinical evidence of coma, transtentorial herniation, or severe hydrocephalus. 5 Similarly, there are no specific recommendations for ICP monitoring in the guidelines for SAH. One study focusing on hypertensive cerebral hemorrhage found that the use of ICP monitoring may improve patient outcomes. 19 However, another multicenter study, ERICH, did not support the use of ICP monitoring in cases of ICH. 20 Given the current lack of definitive monitoring standards, our research suggests that hemorrhagic stroke patients undergoing ICP monitoring typically present with more severe conditions. By contrasting the general clinical characteristics of patients with and without ICP monitoring, we found that patients with ICP monitoring were younger, had lower GCS scores, a higher rate of interventional treatments, and a poorer prognosis. These findings are consistent with the clinical characteristics reported in the ERICH and SYNAPSE-ICU studies.18,20 Therefore, the conclusions of our study may be more applicable to patients with critical hemorrhagic stroke.
Extensive TBI research supports the association between elevated ICP and poor prognosis.21,22 As a result, TBI guidelines advocate a therapeutic ICP threshold of 22 mmHg. 7 However, research investigating the relationship between ICP and hemorrhagic stroke prognosis remains scarce. A retrospective study by Yang et al. 11 of 53 patients with hypertensive cerebral hemorrhage showed an inverse association between ICP and prognosis, whereas CPP was positively associated with prognosis. In addition, a study monitoring ICP in 116 patients with SAH found that increased ICP is a common complication in severe SAH patients during the first week, with mean ICP being significantly associated with the severity of early brain injury and mortality. 8 However, these studies lack robust evidence due to their limited sample sizes. As a result, relevant guidelines for SAH and ICH continue to follow the recommendations for TBI, albeit in a speculative manner. Over four editions, TBI guidelines have suggested ICP thresholds of 20, 20–25, 20, and 22 mmHg, while continuing to acknowledge the indeterminacy of the ideal value. 3 In patients with hemorrhagic stroke, the predominant causes of elevated ICP may include hydrocephalus caused by intraventricular hemorrhage or a mass effect induced by a hematoma, potentially creating pressure gradients within the brain. This differs from the mechanism of TBI-induced ICP elevation and makes uniform threshold management inappropriate.3,5 Studies of therapeutic ICP thresholds in TBI have shown that patients over 55 years of age have a lower ICP threshold than their younger counterparts (22 mmHg vs 18 mmHg). 21 Svedung Wettervik et al. 23 found that in both TBI and SAH patients, patient outcomes begin to deteriorate when ICP exceeds 15–20 mmHg. A study of hypertensive intraventricular hemorrhage found a significant correlation between ICP above 18 mmHg and early mortality. 24
Our research, which includes both ICH and SAH, has identified an early ICP threshold of 16.5 mmHg, beyond which early mortality rates increase significantly. This threshold is lower than that found in previous studies.21,23,24 A plausible explanation for this may be that the time frame of our study focused on the initial 24 h, during which the edema surrounding the hematoma and hydrocephalus may not have peaked. As a result, the final threshold found in our study is lower than that found in some previous studies. To date, no studies have specifically addressed the early ICP threshold, so the potential for improved outcomes from more stringent early ICP interventions in hemorrhagic stroke warrants further investigation. Furthermore, in our results, the threshold for ICP intervention was lower in ICH than in SAH (15.5 mmHg vs 18 mmHg). This may be due to the fact that in ICH, increases in ICP are often associated with acute hematoma expansion, which can rapidly affect cerebral perfusion.4,5 Therefore, a lower ICP threshold may reflect an increased sensitivity of brain tissue to abrupt changes in ICP, especially during rapid hematoma expansion. In contrast, ICP elevations in SAH, typically caused by hydrocephalus and vasospasm, tend to increase more gradually than in ICH.4,6,25 A higher ICP intervention threshold in SAH may indicate a greater tolerance to changes in ICP due to a slower rate of increase, allowing more compensatory mechanisms to intervene. Similarly, in TBI patients, where brain injury is usually the result of direct external force to the head, widespread brain tissue reaction and edema may occur.4,7,26 In these cases, ICP often rises slowly but steadily, allowing more compensatory mechanisms to engage. This may be one of the reasons why our study found a lower threshold than that found in TBI research.
The ideal range for CPP was originally postulated to require an increase in MAP to amplify CPP, thereby ensuring adequate cerebral blood flow. However, an excessively elevated CPP could cause congestion, increase vascular edema, trigger pulmonary complications, and promote secondary escalation of ICP. 4 The Lund concept, on the other hand, recommends reducing intravascular resistance and hydrostatic pressure, thereby reducing cerebral blood volume and facilitating increased cerebral blood flow, making a lower CPP acceptable. 27 The CPP threshold advocated in the TBI guidelines is between 60 and 70 mmHg, with hemorrhagic stroke thresholds also referring to these TBI recommendations. However, there is still some controversy regarding the relationship between CPP and prognosis, with some studies suggesting a positive correlation between CPP and patient prognosis.11,28 In contrast, in a study of TBI, CPP was found to have no effect on patient survival, 22 and some research suggests that CPP above 60 mmHg does not affect patient outcome. 29 Our study found that early CPP does not have a significant correlation with 28-day ICU mortality in patients with hemorrhagic stroke, and this lack of correlation is consistent in both the ICH and SAH subgroups. Even after adjustment for MAP, there is no significant correlation with 28-day ICU mortality. An association with short-term mortality was observed only when CPP was below 60 mmHg.
Our study has several limitations. First, inherent limitations of the public database may have resulted in the omission of some clinical features of hemorrhagic stroke, such as methods of ICP monitoring, certain neurological scoring systems, due to their absence in the database records. Second, our findings are based on analyses of the current dataset without external validation or calibration, which may affect their generalizability. Third, the period of patient enrollment in the current database spans a considerable period of time. Given the evolution of medical practices and treatment strategies over time, these temporal variations may affect the relevance and practical application of our study results. Finally, as a retrospective study, the set of inclusion and exclusion criteria inevitably introduces some selection bias. Future studies, perhaps prospective in nature or with more inclusive criteria, are needed to minimize such biases.
Conclusions
By analyzing data from two public databases, we identified an association between early ICP and 28-day ICU mortality in patients with hemorrhagic stroke and established a therapeutic threshold for ICP. In addition, we found that early CPP did not correlate with patient prognosis. These results may provide a basis for early intervention and prognostic assessment in patients with critical hemorrhagic stroke.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241232311 – Supplemental material for Correlation between early intracranial pressure and cerebral perfusion pressure with 28-day intensive care unit mortality in patients with hemorrhagic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873241232311 for Correlation between early intracranial pressure and cerebral perfusion pressure with 28-day intensive care unit mortality in patients with hemorrhagic stroke by Yang Zhang, Shuaijie Zhu, Yan Hu, Heng Guo, Jin Zhang, Tianfeng Hua, Zhongheng Zhang and Min Yang in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by National Natural Science Foundation of China (Grant No. 82072134) and the Scientific Research Fund of Anhui Medical University (No. 2020xkj036).
Ethics approval
The study was an analysis of a third-party anonymized publicly available database with pre-existing institutional review board (IRB) approval.
Informed consent
All personal information in both databases was de-identified to protect patient privacy, and patient consent was not required for this study.
Availability of data and materials
Guarantor
MY.
Contributorship
YZ conceived the idea, performed the analysis, and drafted the manuscript. MY and TH interpreted the results and helped to revise the manuscript. ZZ and JZ helped to frame the idea of the study. SZ, YH and HG helped to analyze the data. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
