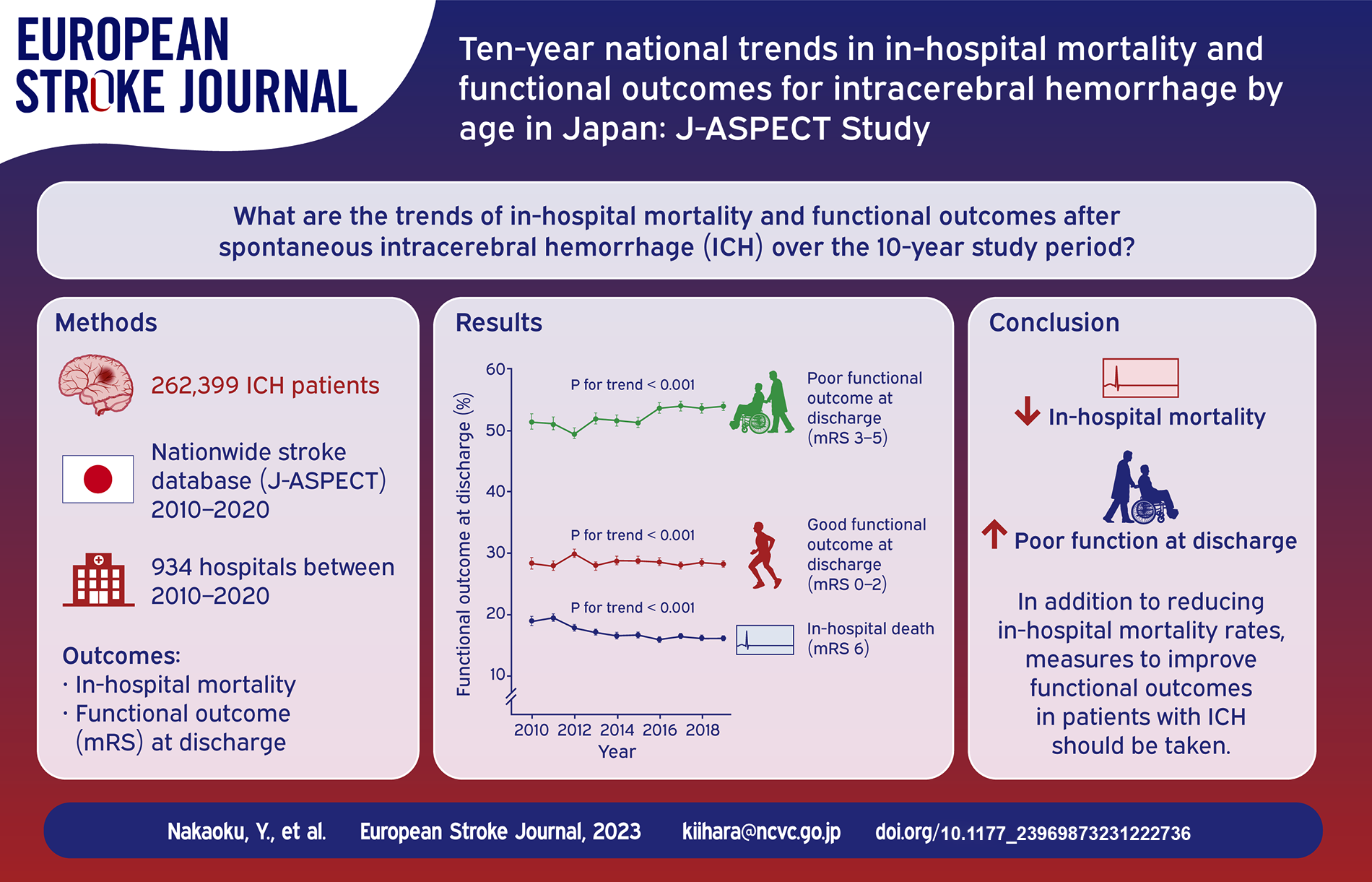
Abstract
Introduction:
National-level data on trends in the prognosis of age-stratified patients with intracerebral hemorrhage (ICH) are lacking. This study aimed to assess time trends in in-hospital mortality and functional outcomes of ICH patients by sex and age, and to explore factors associated with changes in in-hospital mortality trend.
Patients and methods:
Using the largest nationwide, J-ASPECT stroke database in Japan, this serial cross-sectional study included ICH patients aged ⩾18 years who were hospitalized for non-traumatic ICH from April 2010 to March 2020. We examined trends in in-hospital mortality and functional outcomes using the modified Rankin Scale at discharge, as well as differences in in-hospital mortality change between age groups.
Results:
Among 262,399 ICH patients from 934 hospitals, crude in-hospital mortality showed a significant decreasing time trend (from 19.5% to 16.7%), and this trend was consistent across sex and age groups. In addition, differences in in-hospital mortality change over the 10-year study period were significant between male patients aged ⩾75 years and those aged ⩽64 years (−3.9% [95% confidence interval, −5.4 to −2.4] for 75–84 years; −4.1% [−6.3 to −1.9] for ⩾85 years). On the other hand, the proportion of dependent patients (mRS 3–5) at discharge increased from 52.0% to 54.9% over the 10-year study period.
Conclusion:
The in-hospital mortality of ICH patients improved, whereas the proportion of patients with dependent functional outcome at discharge increased, over the 10-year study period. Elucidating the mechanism underlying differences in in-hospital mortality reduction in men may provide insights into effective interventions in the future.
Introduction
Although spontaneous non-traumatic intracerebral hemorrhage (ICH) is associated with high mortality (36.3% at 1 month and 50.7% at 12 months after ICH) 1 and poor functional outcomes, 2 no study has reported age group differences in nationwide trends of in-hospital mortality of Asian ICH patients. Japan, the most aged country in the world, is facing a steep surge of the aged population (the proportion of people aged ⩾65 years was 28.0% in 2020, 3 rising to 35.3% in 2040) 4 ahead of other countries. In addition, population aging explained around 2.2 million additional deaths from stroke globally between 1990 and 2017, with Japan being the most affected country. 5 We previously estimated that population aging has a major impact on the time trends of stroke deaths including hemorrhagic stroke in Japan. 6 Furthermore, the number of stroke patients is steeply increasing in Japan. 7 Therefore, analysis of recent trends in the prognosis of ICH patients in Japan by age group is important for predicting future trends in the prognosis of ICH patients worldwide.
Several population-based studies on time trends in mortality after ICH have reported inconclusive results,8,9 with only a few studies reporting on time trends by age group. 8 To the best of our knowledge, no study has examined time trends in in-hospital mortality of Asian ICH patients by sex and age. A recent study from Canada reported reduced 30-day case fatality across all age groups for both women and men. 8 Data from the Dijon Stroke Registry showed a decrease in the rate of independent walking at discharge after ICH from 64.1% in 1985–1993 to 39.6% in 2003–2011, with an accompanying decrease in acute phase mortality. 9 In a recent study in Japan, a combined analysis of ICH patients of all ages showed a decrease in in-hospital mortality over the past 20 years up to 2019 only in women. 10 In terms of time trends in mortality of ICH patients, overall mortality at 1 month has remained constant over the past few decades (−0.4% per year, 95% confidence interval [CI]: −1.0 to 0.2, p = 0.155); 1 however, even the aforementioned meta-analysis did not report time trends by age group. 1
While mortality has often been used to assess the prognosis of ICH survivors, functional outcomes of these patients have rarely been reported. Nationwide trends in functional outcomes remain unclear, and in fact, a recent meta-analysis of 122 eligible cohort studies of ICH patients found only four papers reporting functional outcomes. 11 These studies of ICH survivors showed independent functional outcomes only in 32.8%–42.4% at 6 months and 16.7%–24.6% at 1 year. 11
In contrast to the rapid advances in the acute treatment of ischemic stroke, there have been no highly effective acute treatments for ICH. Prevention remains the most effective approach to reducing the burden of ICH, and the current priority is to improve primary prevention by treating modifiable risk factors. Although nationwide trends in in-hospital mortality of ICH patients should be stratified by modifiable risk factors for ICH, few studies have stratified by modifiable risk factors.
Therefore, larger-scale studies with adequate power to detect trends in ICH case-fatality within specific subgroups (e.g. stratified by age group) are needed. 12 In 2010, we launched the J-ASPECT (Nationwide survey of Acute Stroke care capacity for Proper dEsignation of Comprehensive stroke cenTer in Japan) study, a nationwide survey of real-world settings for stroke. The J-ASPECT study group used nationwide stroke registry data to gain new clinical insights into stroke and neurosurgical practices.13 –15 Using data of ICH patients from the J-ASPECT study, the present study aimed to examine trends of in-hospital mortality and functional outcomes after spontaneous ICH and identify subgroups with improved in-hospital mortality trends, which will provide valuable information for future prophylactic management and treatment.
Methods
Study population
This serial cross-sectional study was based on multicenter nationwide data from a 10-year survey (J-ASPECT study) of ICH patients in Japan. Participation of hospitals in the J-ASPECT study was voluntary. Enrollment in the J-ASPECT study has increased progressively from fiscal year 2010 to 2019. Of the training institutions certified by the Japan Neurosurgical Society, the Japanese Society of Neurology, and the Japan Stroke Society, a total of 934 hospitals agreed to participate in the J-ASPECT study from April 2010 to March 2020. In Japan, there were 975 facilities certified as primary stroke centers in April 2020. The J-ASPECT database includes data obtained from the Diagnosis Procedure Combination (DPC)-based payment system that was launched in 2002 by the Ministry of Health, Labour and Welfare (MHLW) of Japan. 16 The DPC system was initially introduced mainly at university hospitals, but 1757 hospitals with a total of 483,180 beds adopted the DPC-based payment system in April 2020. 17 The J-ASPECT database includes clinical data of all patients with stroke admitted to participating hospitals. Disease classification is coded using the International Classification of Diseases, 10th revision (ICD-10). Detailed information regarding the J-ASPECT database has been provided elsewhere. 18
We identified adult patients (aged ⩾18 years) hospitalized for nontraumatic ICH using the recommended ICD-10 codes for the diagnosis of ICH (I61.0-9). 19 Since no information was available to differentiate between first-ever versus recurrent ICH in the J-ASPECT database, the present analyses included both ICH events. We excluded patients with hemorrhagic transformation from ischemic stroke and those with traumatic intracranial injury. We further selected patients who had been urgently hospitalized between April 1, 2010, and March 31, 2020 (i.e. fiscal year 2010–2019).
The present study was conducted in accordance with the Declaration of Helsinki and Ethical Guidelines for Medical and Biological Research Involving Human Subjects. The research protocol was approved by the Ethics Committee of the National Cerebral and Cardiovascular Center (#M29-161-9). Participant written consent was waived because the data used in this study were de-identified before provision and anonymously managed at all stages, including data cleaning and statistical analysis.
Clinical data collection
We extracted from the J-ASPECT database information on patient age on admission, sex, unique hospital identifier, year (fiscal year) of hospital admission and medical treatment/management; comorbidities on admission (atrial fibrillation, hypertension, diabetes mellitus, hyperlipidemia, and renal diseases); level of consciousness on admission according to the Japan Coma Scale (JCS; the most widely used grading scale for impaired consciousness in Japan, 20 as described in Supplemental Table 1); hematoma removal with craniotomy, endoscopic surgery, or stereotactic aspiration; CSF drainage coded with Japanese original K-codes; in-hospital mortality; length of hospital stay; and modified Rankin Scale (mRS) scores at discharge (Supplemental Table 2).21,22 Initial ICH severity was presented by JCS scores on admission. Post-stroke functional outcome of patients was evaluated using mRS scores at discharge. A poor functional outcome was defined as mRS 3–6.11,22 We included mRS 3 in the dependent category (i.e. mRS 3–6) since mRS 3 can be considered to represent an impaired functional outcome. In the J-ASPECT database, hypertension, diabetes mellitus, and hyperlipidemia were defined as present if these diseases were registered as a comorbidity on admission, or if any medication specific to these diseases was prescribed. On the other hand, other comorbidities, such as atrial fibrillation, renal diseases, heart failure, liver diseases, and cancer, were defined as present only if these diseases were registered as a comorbidity on admission, as there is no specific medication used to treat these diseases.
Statistical analyses
Continuous data are reported as medians with interquartile ranges (IQRs), and categorical data are presented as numbers with percentages (%). Crude in-hospital mortality, poor functional outcome, and their 95% CIs were calculated according to age group and year and stratified by sex, assuming a Poisson distribution.
Analyses of time trends in in-hospital mortality were performed using the generalized estimation equation (GEE) model to account for hospital, sex, and age. The GEE approach using a normal distribution with identity link was used for estimating adjusted risk differences through robust sandwich standard error estimation.23,24 We used the “geepack” package of R statistical software version 4.2.0. All p values were two-sided, with p < 0.05 considered statistically significant. In order to investigate whether changes in in-hospital mortality from fiscal year 2010 differed by age group, we used the aforementioned GEE approach, modeling continuous fiscal year, age groups, JCS scores, and interaction terms between age groups and continuous fiscal year as independent variables. Beta coefficients of the interaction terms represented differences in in-hospital mortality change between age groups (⩽64 [Ref.], 65–74, 75–84, and ⩾85). We adjusted for JCS scores on admission, because the level of consciousness on admission is the most consistent outcome predictor. 25 Furthermore, to identify subgroups with improved in-hospital mortality trends, we examined in-hospital mortality change stratified by age group in subgroups according to the presence of each cerebrovascular disease (CVD) risk factor or each level of consciousness on admission. Because multiple comparisons were performed in six stratified analyses by CVD risk factors and four stratified analyses by level of consciousness, significance levels were corrected by Bonferroni correction (CVD risk factors, p < 0.0083 = (0.05/6); level of consciousness, p < 0.0125 = (0.05/4)). Based on mRS scores at discharge, trends of functional outcomes were assessed using the GEE model to account for hospital, sex, and age.
Results
Trends in the characteristics of ICH patients
In total, 262,399 ICH patients from 934 hospitals registered in the J-ASPECT study database between April 2010 and March 2020 were included in the entire cohort. Baseline and clinical characteristics of the entire population of ICH patients are shown in Table 1. During the 10-year study period, the number of participating hospitals and ICH patients per year increased from 267 to 653 and 12,916 to 43,558, respectively. The median age increased from 71 to 73 years (Table 1). The proportion of unarousable (JCS: 3-digit code) patients on admission gradually decreased from 24.8% to 21.9% from fiscal year 2010 to 2019. The proportion of patients having any type of surgical ICH removal decreased from 13.3% to 11.2%. Among these patients, craniotomy decreased from 10.8% to 7.7%, whereas endoscopic removal increased from 0% to 2.9%. The proportion of those having external CSF drainage decreased from 4.3% to 3.0% (Table 1). Regarding comorbidities on admission, percentages of patients with CVD risk factors or other comorbidities (heart failure, liver diseases, and cancer) are also summarized in Table 1.
Characteristics of patients with intracerebral hemorrhage in the J-ASPECT study from April 2010 to March 2020.
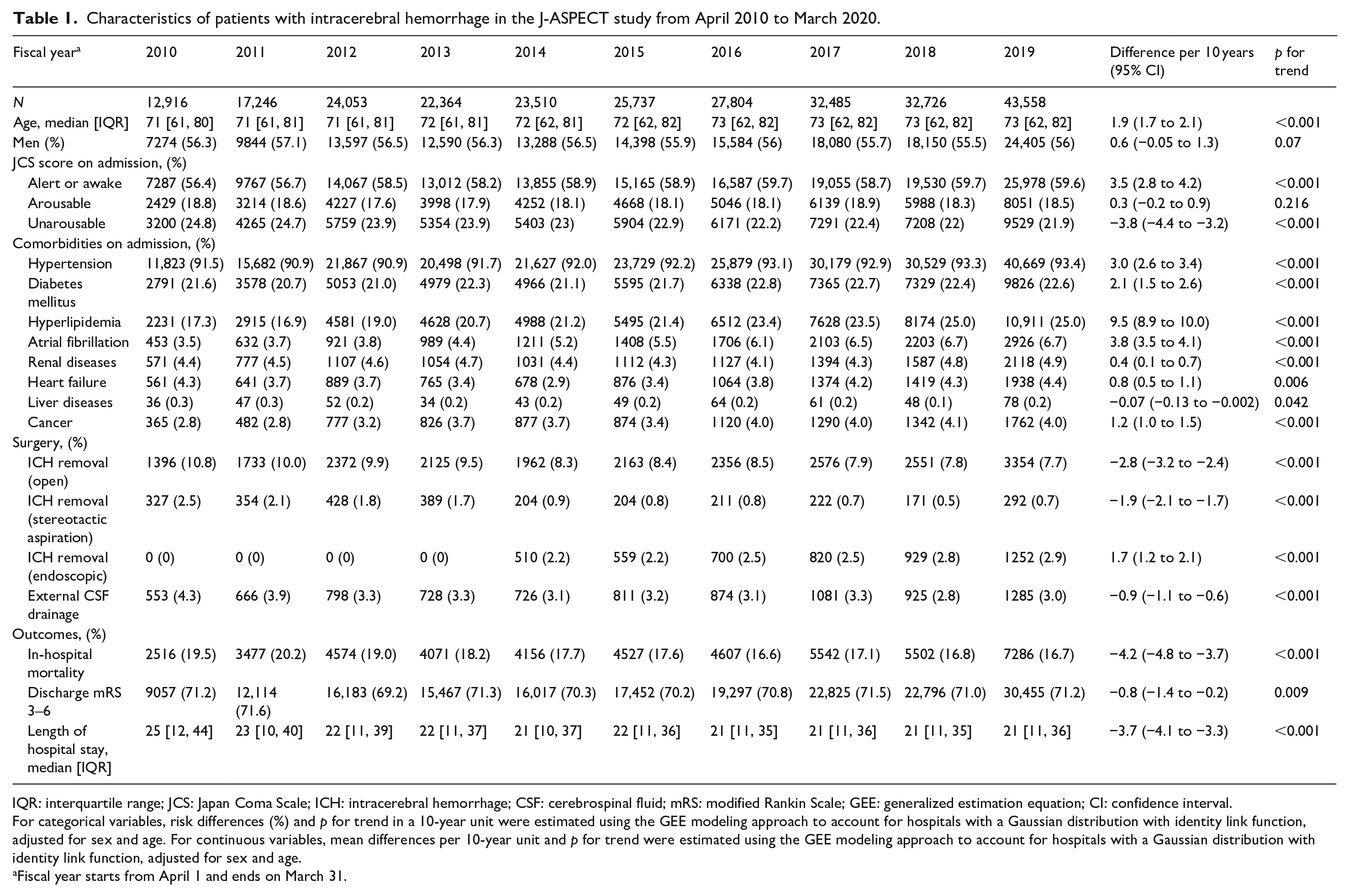
IQR: interquartile range; JCS: Japan Coma Scale; ICH: intracerebral hemorrhage; CSF: cerebrospinal fluid; mRS: modified Rankin Scale; GEE: generalized estimation equation; CI: confidence interval.
For categorical variables, risk differences (%) and p for trend in a 10-year unit were estimated using the GEE modeling approach to account for hospitals with a Gaussian distribution with identity link function, adjusted for sex and age. For continuous variables, mean differences per 10-year unit and p for trend were estimated using the GEE modeling approach to account for hospitals with a Gaussian distribution with identity link function, adjusted for sex and age.
Fiscal year starts from April 1 and ends on March 31.
Trends in in-hospital mortality in ICH patients
Over the 10-year study period, a significant decreasing time trend in in-hospital mortality was observed from April 2010 to March 2020 (crude proportion: from 19.5% to 16.7%, in-hospital mortality change adjusted for hospital, sex, and age: −3.6% [95% CI, −4.1 to −3.0], p < 0.001); this trend was consistent across all age groups, stratified by sex. For men, in-hospital mortality decreased over time (from 15.2% to 13.2%, −2.4% [−3.5 to −1.3], p < 0.001 in those aged ⩽64 years; from 17.7% to 15.2%, −3.9% [−5.2 to −2.6], p < 0.001 in those aged 65–74 years; from 25.7% to 19.7%, −7.4% [−8.9 to −5.8], p < 0.001 in those aged 75–84 years; and from 37.6% to 29.5%, −7.1% [−9.7 to −4.6], p < 0.001 in those aged ⩾85 years) (Figure 1(a)). For women, in-hospital mortality decreased (from 11.7% to 11.1%, −3.2% [−4.7 to −1.7], p < 0.001 in those aged ⩽64 years; from 15.0% to 10.3%, −4.2% [−5.7 to −2.7], p < 0.001 in those aged 65–74 years; from 18.1% to 14.4%, −5.0% [−6.3 to −3.6], p < 0.001 in those aged 75–84 years; and from 27.1% to 22.7%, −4.6% [−6.3 to −2.9], p < 0.001 in those age ⩾85 years) (Figure 1(b)). For both sexes, trends in in-hospital mortality by age group are presented in Supplemental Figure 1. Multivariable-adjusted differences of in-hospital mortality between 2010 and each year were plotted by age group stratified by sex (Figure 1(c) and (d)). For men, changes in in-hospital mortality over the 10-year study period significantly differed in the 75–84 (−3.9% [−5.4 to −2.4], p < 0.001) and ⩾85 (−4.1% [−6.3 to −1.9], p < 0.001) age groups compared to the ⩽64 age group (Figure 1(c)).
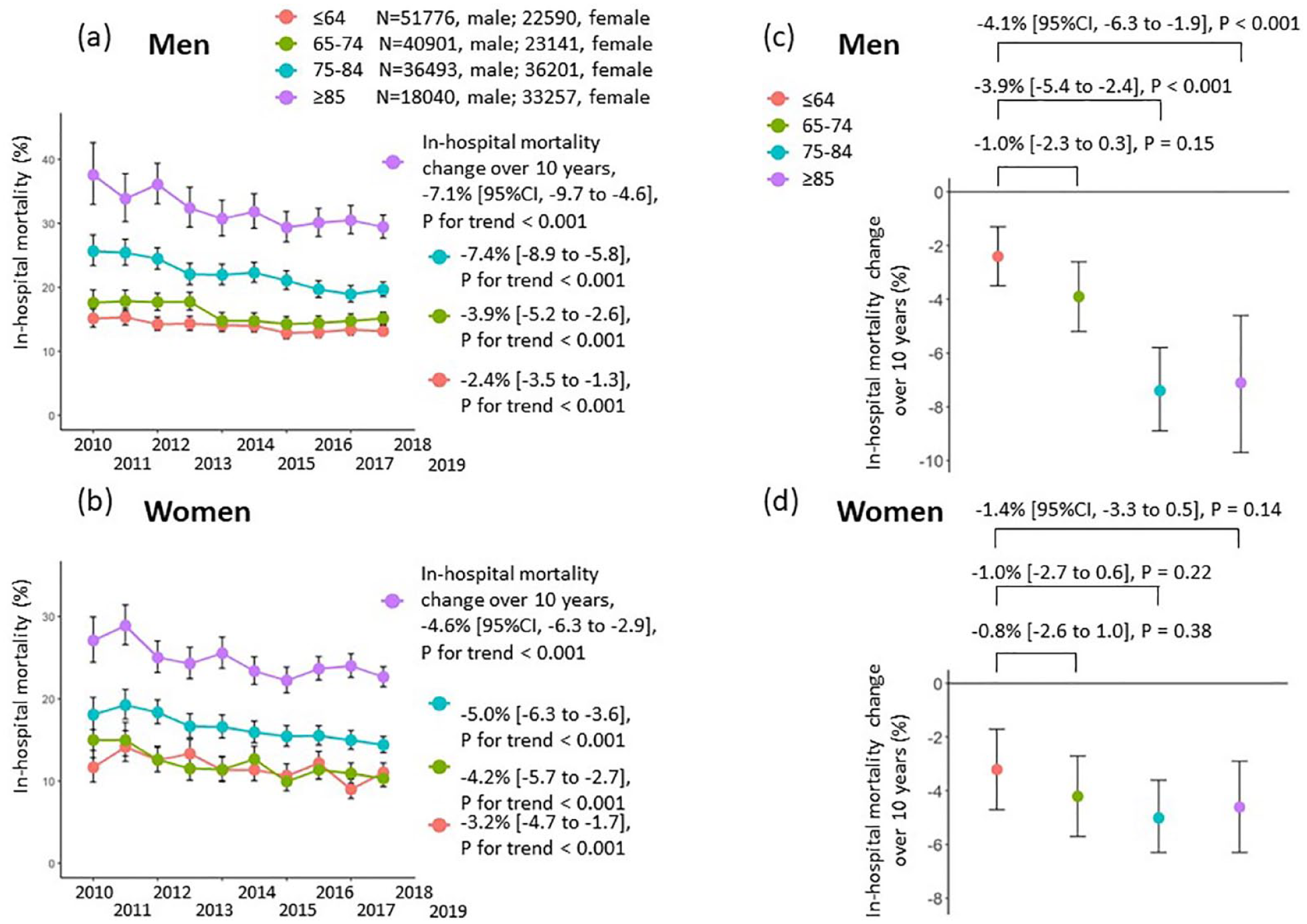
Trends in in-hospital mortality and differences in in-hospital mortality change by sex and age group, April 2010 – March 2020. (a, b) Crude in-hospital mortality and 95% CIs were calculated according to age group and fiscal year, stratified by sex. Bar lines show 95% CIs in each group. In-hospital mortality change over the 10-year study period and p-values were obtained to test year trends using the GEE modeling approach to account for hospital, sex, and age. (c, d) In-hospital mortality change over the 10-year study period, stratified by sex and age. Differences in in-hospital mortality change over the 10-year study period between ⩽64 age group and the other age groups are also shown. p-values were based on interaction terms between age groups and continuous fiscal year in relation to in-hospital mortality, adjusted for the level of consciousness on admission according to JCS scores.
Associations of factors with change in in-hospital mortality over 10 years by age group
In order to examine factors contributing to the decrease in in-hospital mortality over the 10-year study period, we conducted a sub-analysis stratified by age group and sex, using data of ICH patients with certain comorbidities on admission (Figure 2) with a Bonferroni corrected significance level (p < 0.0083 = [0.05/6] to evaluate in-hospital mortality per 10 years). For both sexes, patients with hypertension in the ⩾65 age group showed a significant reduction in in-hospital mortality per 10 years. Patients with atrial fibrillation in the 75–84 age group for men and the ⩾75 age group for women showed a significant reduction in in-hospital mortality per 10 years. Patients with diabetes mellitus in the 65–84 age group for men and the ⩾75 age group for women also showed a significant reduction in in-hospital mortality per 10 years. Patients with dyslipidemia in all age groups for men and women showed no significant change, whereas patients with renal diseases in the 75–84 age group for men showed a significant reduction in in-hospital mortality per 10 years. In no-comorbidity group, no significant change in in-hospital mortality was observed in all age groups for both sexes.
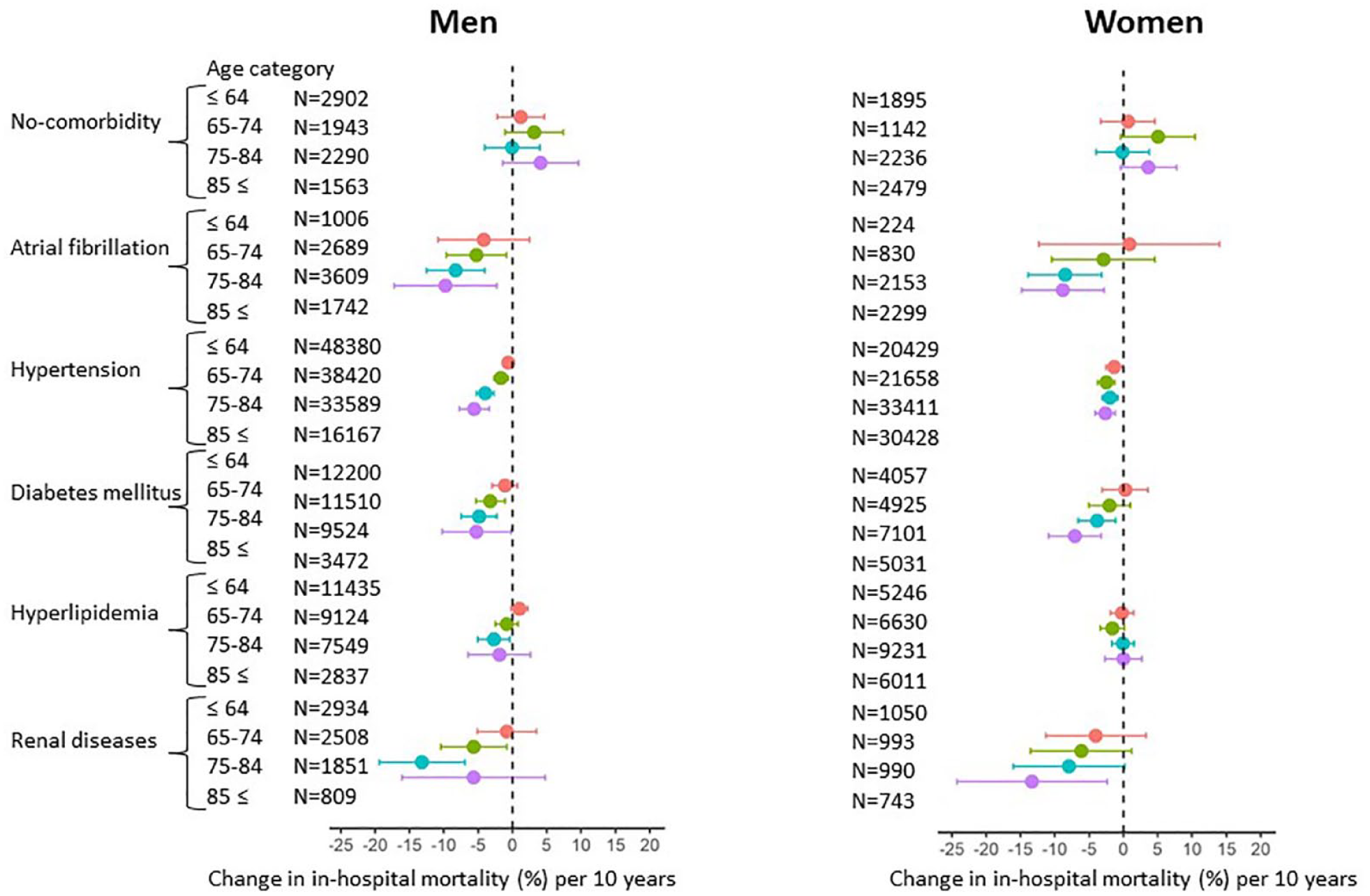
Changes in in-hospital mortality per 10 years stratified by age group in subgroups according to the presence of comorbidities on admission.
Next, we stratified patients by the level of consciousness on admission according to JCS scores and examined in-hospital mortality change per 10 years stratified by age group and sex using a Bonferroni corrected significance level (p < 0.0125 = (0.05/4) to evaluate in-hospital mortality per 10 years). In-hospital mortality was significantly reduced among elderly patients with a poor level of consciousness (Figure 3).
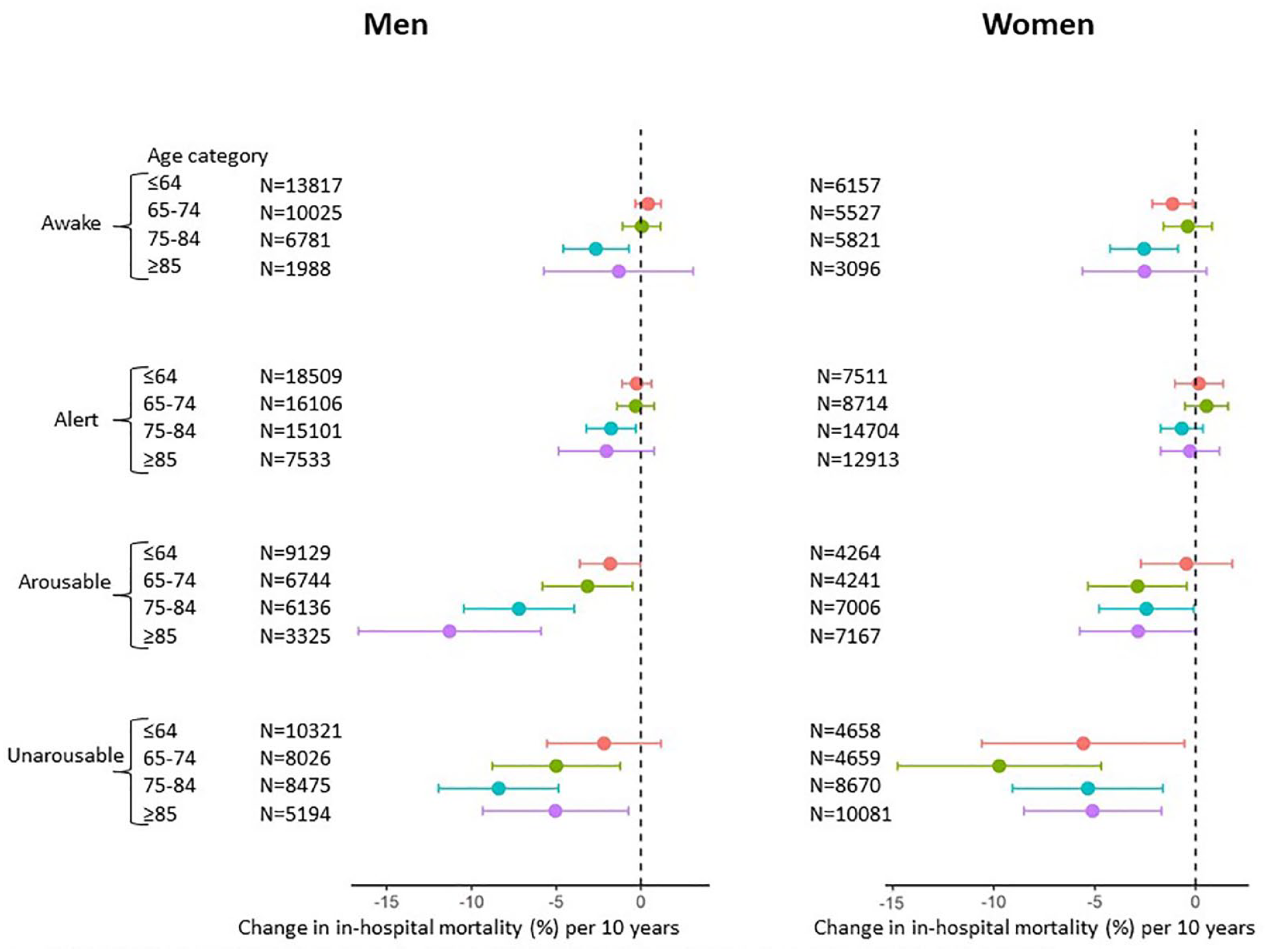
Changes in in-hospital mortality per 10 years stratified by age group in subgroups according to the level of consciousness on admission.
Trends of poor functional outcome in ICH patients
Trends of poor functional outcome (mRS 3–6 at discharge) did not change over the 10-year study period (ranged between 69.2% and 71.6%), showing only a slight decrease adjusted for sex, age, and hospital (poor functional outcome change, −1.1% [95% CI, −1.7 to −0.4], p < 0.001), which was influenced by a large decrease in in-hospital mortality (i.e. mRS 6). On the other hand, trends of dependent functional outcome (mRS 3–5) at discharge increased (from 52.0% to 54.9%), showing a significant increase adjusted for age, sex, and hospital (dependent functional outcome change, 3.2% [2.5 to 3.9], p < 0.001) over the 10-year study period (Supplemental Figure 2); a consistent trend was observed when stratified by sex (Figure 4).
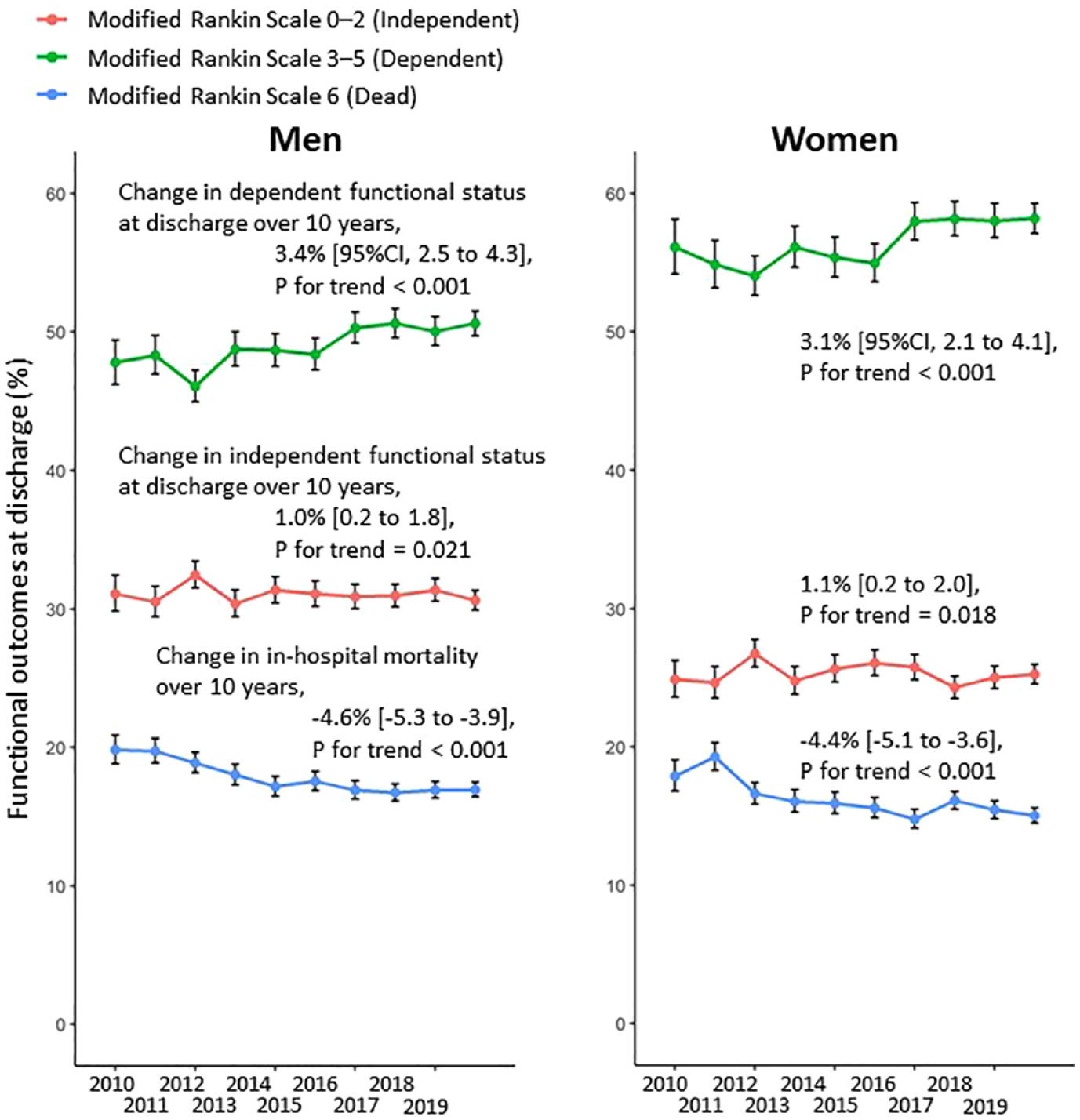
Trends in functional outcomes at discharge by sex, April 2010 – March 2020. Percentages of patients with intracerebral hemorrhage stratified by mRS score at discharge (mRS 0–2, 3–5, and 6) and 95% CIs were calculated for each fiscal year, stratified by sex. mRS 6 indicates death at discharge. Bar lines show 95% CIs in each group. Change in each functional status over the 10-year study period and p-values were obtained to test year trends using the GEE modeling approach to account for hospital, sex, and age.
Discussion
Using the largest nationwide data of 262,399 ICH patients from Japan spanning fiscal years 2010 to 2019, we found that in-hospital mortality decreased over the 10-year study period, that is, from 19.5 to 16.7%, whereas the proportion of patients with dependent functional outcome (mRS 3–5) at discharge increased, that is, from 52.0% to 54.9%. These trends in decreasing in-hospital mortality were consistent across age and sex. Despite the improvement in in-hospital mortality, increasing trends in poor functional outcomes at discharge suggest that recent advances in the acute treatment of ICH remains insufficient, and that the development of more effective acute treatments to recover functional status is needed. Another major finding of this study was that the 10-year mortality reduction was significantly greater in elderly patients aged ⩾75 years (i.e. −8% to −4%) compared with patients aged ⩽74 years (i.e. −5% to −2%) in men. Such a greater reduction in in-hospital mortality of elderly male ICH patients may provide insight into the implementation of more effective interventions, such as prevention or treatment, in the future.
Our in-hospital mortality with a median (IQR) length of hospital stay of 22.0 (11.0, 37.0) for the entire ICH cohort (17%) was in line with those of previous studies on ICH mortality. 26 A systematic review of 36 studies on ICH revealed that Asian people have a 2-fold higher ICH incidence compared with other ethnic groups, although studies in Japan reported a lower mortality at 1 month (16.7% [95% CI, 15.0–18.5]) than in other countries (42.3% [40.9–43.6]). 26 Previous reports also suggested an increase in mortality with increasing age (i.e. 28.1% in patients aged >75 years and 17.8% in younger patients). 26
Previously published trends in ICH in-hospital mortality showing stable or decreasing trends 8 are inconsistent with our findings. We analyzed 262,399 ICH patients in J-ASPECT, the largest nationwide registry in Japan, and found a significant decreasing trend in in-hospital mortality (average annual percent change of −0.4% [95% CI, −0.5 to −0.4]), consistent across age and sex. Our results differ from a recent study in Japan, which analyzed 36,014 ICH patients from 2000 to 2019 and reported that in-hospital mortality from ICH decreased only in women and remained unchanged in men. 10 Such differences may be explained by differences in the patients enrolled (e.g. number of participants and initial severity), participating departments (e.g. neurosurgery and neurology), and participating hospitals. According to an epidemiological study of ICH from Canada, in-hospital mortality decreased from 37.5% in 2009 to 27.1% in 2018 (p < 0.001). 27 A nationwide trend study of stroke from Denmark reported that, among 105,240 stroke patients aged ⩾50 years, 30-day mortality trended downward from 2005 to 2018, with an average annual percent change of −3.2% (95% CI, −4.1 to −2.4) for ICH. 28 Potential reasons for such trends include the establishment of specialized stroke units, improved acute stroke care, and less severe strokes probably attributable to better management of risk factors for stroke (e.g. hypertension, and atrial fibrillation). 29 Moreover, such trends in in-hospital mortality could be explained partly by revisions of guideline-recommended management of blood pressure (BP) in the acute phase of ICH. In 2013, the main phase of the Intensive Blood Pressure Reduction in Acute Cerebral Hemorrhage Trial (INTERACT2) demonstrated the safety of early intensive BP lowering. 30 Japanese guidelines for the management of stroke issued in 2015 revised the recommendation to early intensive BP lowering in ICH patients toward a target of systolic BP < 140 mmHg.
Although decreasing time trends in in-hospital mortality were observed in all age groups for both sexes, when comparing between age groups, there were significant differences in in-hospital mortality change over the 10-year study period between men aged ⩾75 years and those aged ⩽64 years. These differences may be explained by the fact that elderly men in the first year of this study had a higher in-hospital mortality probably due to large initial damage, whereas in recent years the prognosis may have improved probably due to better control of risk factors such as hypertension or atrial fibrillation. Atrial fibrillation is a common arrhythmia in elderly people, and people with atrial fibrillation take oral anticoagulants (OACs) to prevent stroke. However, OACs put them at risk for ICH. In the last 10 years, stroke prevention in patients with atrial fibrillation changed with the availability of direct oral anticoagulants (DOACs). 31 The downward trend in in-hospital mortality from ICH, especially in older patients, could be due to the replacement of warfarin by DOACs, which may have reduced mortality from anticoagulant-related ICH. Another explanation for decreasing trends in in-hospital mortality is recent availability of less invasive surgical treatment such as endoscopic surgery, especially for elderly ICH patients. Further studies will be needed to answer this question.
We focused on potential factors that might have contributed to the decrease in in-hospital mortality over the 10-year study period, which were assumed to vary with age. We found that the decrease in in-hospital mortality over the 10-year study period was significantly greater among those who were older and had comorbidities such as atrial fibrillation, hypertension, diabetes mellitus, and renal diseases. We also found a greater decrease in in-hospital mortality among older patients with a low level of consciousness on admission. We found an increasing trend in the proportion of patients with dependent functional outcome (mRS 3–5), regardless of their level of consciousness on admission (Supplemental Figure 3). These results suggest that, in addition to improving in-hospital mortality, measures to improve functional outcomes for ICH patients should be considered, regardless of the level of consciousness on admission. Given that the clinical importance of post-stroke rehabilitation therapy is well-established, rehabilitation should be started in a timely manner and continued seamlessly from the acute phase to convalescent and chronic phases. In Japan, the development of a seamless rehabilitation system from hospitals to the patient’s home after stroke as part of transitional care services for ICH survivors has been under way. 32
Among the reported risk factors for poor outcome after ICH, old age and male sex are nonmodifiable risk factors, whereas hypertension and coagulopathy are modifiable. 33 Diabetes mellitus has been identified as an independent predictor of mortality. 34 Previous studies used mortality itself as an individual-level outcome. On the other hand, to our knowledge, no study has reported factors associated with nationwide changes in in-hospital mortality from ICH over time. The introduction of new medications and advances in their management over the past decade may have made it possible to control diseases such as atrial fibrillation without increasing side effects, even in older patients. This in turn may have led to a significant reduction in in-hospital mortality among older patients with comorbidities.
Another important finding of the present study is that, while in-hospital mortality decreased, the proportion of patients with poor functional outcome at discharge increased, suggesting that improving short-term functional prognosis will remain a challenge in the next decade. Only a few studies have assessed functional outcomes in ICH. A meta-analysis of 122 cohort studies identified only four studies reporting information on functional outcomes in ICH patients. 11 The meta-analysis reported that 32.8–42.4% of all ICH patients were independent (mRS 0–2) at 6 months. 11 None of these studies reported time trends in functional outcomes. To our knowledge, there is only one study that examined time trends in functional outcomes, which used the Dijon Stroke Registry in France to reveal an increase in the proportion of patients with disability at discharge (independent ambulation: 64.1% in 1985–1993, 45.9% in 1994–2002, 39.6% in 2003–2011), with an accompanying decrease in in-hospital mortality (46.3% in 1985–1993, 36.4% in 1994–2002, and 32.5% in 2003–2011). 9 The present study is the first to report a decreasing 10-year time trend in in-hospital mortality, with an increase in the proportion of patients with dependent functional outcome (mRS 3–5) in an Asian country, revealing a similar trend to that previously reported in France. 9 Although most studies to date have focused on mortality and only short-term functional outcomes among ICH survivors, a recent study showed that more than 40% recovered to good outcome (mRS 0–3) in 1 year among survivors of severe ICH with initial poor functional outcome (mRS 4–5). 35 Thus, the observed increase in the proportion of dependent ICH survivors at discharge over the 10-year study period may not necessarily be a bad trend, as more stroke survivors may recover to independent functional status from a long-term perspective.
The strength of this study is that, by using a large database including data of more than 250,000 ICH patients from more than 900 hospitals in Japan over a 10-year period, we clarified in-hospital mortality time trends of ICH patients. Furthermore, by leveraging the strengths of this dataset, we focused on changes in in-hospital mortality by sex and age, and demonstrated a great reduction in in-hospital mortality of ICH patients, especially older male patients. Our analysis is meaningful because, as recommend by Charidimou and Tirschwell, 12 examining ICH mortality trends separately by age subgroup is important.
This study also has several limitations. First, there is potential bias in the selection of hospitals participating in the J-ASPECT study. We confirmed that the trends in in-hospital mortality and functional outcomes at discharge did not differ between patients from all hospitals and those from hospitals that had participated in the J-ASPECT study in fiscal year 2010 (Supplemental Figure 4). Moreover, this is a large Japan-wide study database of ICH that encompasses most acute care hospitals in Japan. Second, important clinical data, including National Institutes of Health Stroke Scale scores, and hematoma volume and location, were not obtained. Instead, we used the JCS as a proxy for stroke severity. Third, information on premorbid medication use was available only for patients who received prescriptions in the outpatient department of the same hospital where they were admitted. A complete set of data on premorbid medications might have allowed us to examine trends in oral anticoagulation use, which is an established predictor of ICH mortality, and demonstrate a trend effect on ICH mortality improvement due to the substitution of warfarin by DOACs. Fourth, in the present study, the prevalence of comorbidities (e.g. atrial fibrillation) might have been underestimated, as we used comorbidities registered in the DPC database rather than obtaining information on medication use when defining the comorbidities. In a previous study, the frequency of anticoagulant-related ICH was reported to be 12%–23%, 36 as compared with only 4%–7% among the ICH patients with atrial fibrillation included in the present study, which is possibly underestimated. Finally, outcomes were assessed at the time of discharge from acute hospitals, as longer-term outcomes were not collected. A recent meta-analysis assessed the temporal trend of mortality and reported a significant reduction in long-term mortality after ICH. 1 However, comorbidities are common in ICH patients, especially in elderly patients, which may worsen long-term functional outcomes. 8 Therefore, post-acute transitional care for ICH survivors will be important to improve long-term prognosis. Further follow-up studies of ICH patients are warranted to clarify long-term prognosis including mortality and functional outcomes.
Conclusions
This study revealed that in-hospital mortality decreased significantly over the 10-year study period in all sex and age groups, whereas the proportion of patients with dependent functional outcome increased in Japan. We also showed that changes in in-hospital mortality differed by age group.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231222736 – Supplemental material for Ten-year national trends in in-hospital mortality and functional outcomes after intracerebral hemorrhage by age in Japan: J-ASPECT study
Supplemental material, sj-docx-1-eso-10.1177_23969873231222736 for Ten-year national trends in in-hospital mortality and functional outcomes after intracerebral hemorrhage by age in Japan: J-ASPECT study by Yuriko Nakaoku, Soshiro Ogata, Nice Ren, Tomotaka Tanaka, Ryota Kurogi, Kunihiro Nishimura and Koji Iihara in European Stroke Journal
Footnotes
Acknowledgements
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KI received grant support from Chugai Pharmaceutical Company Limited, Eisai Co., KM Biologics Company Limited, Nihon Medi-Physics, Nippon Zoki Pharmaceutical, Otsuka Pharmaceutical Company Limited, and Idorsia Pharmaceuticals Japan Limited. The other authors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Practical Research Project for Life-Style related Diseases, including cardiovascular diseases and diabetes mellitus managed by the Japan Agency for Medical Research and Development (JP19ek0210088, JP20ek0210129, JP20ek0210147, JP21ek0210147, JP 22ek0210147); Grants-in-Aid from the Japanese Ministry of Health, Labour and Welfare (H28-Shinkin-Ippan-011, 19AC1003, 21FA1010, 22FA1015); KAKENHI grants (25293314, 18H02914, 22H03191, 20K19702, 22KJ3211, 22K17821) from the Japan Society for the Promotion of Science; and an Intramural Research Fund (20-4-10) for Cardiovascular Diseases from the National Cerebral and Cardiovascular Center. The funding sources had no role in the study design, data collection and analysis, manuscript preparation, or decision to publish.
Ethical approval
Ethical approval for this study was obtained from the Ethics Committee of the National Cerebral and Cardiovascular Center (#M29-161-9).
Informed consent
Participant written consent was waived because the data used in this study were de-identified before provision and anonymously managed at all stages, including data cleaning and statistical analysis.
Guarantor
KI
Contributorship
YN, SO, NR, and KI conceptualized the present study. KI and NR contributed to data acquisition. YN and NR performed data analysis. SO and KN supervised the data analysis. All authors (i.e. YN, SO, NR, TT, RK, KN, and KI) interpreted analysis results. YN and SO drafted the manuscript, and the other authors (i.e. RN, TT, RK, KN, and KI) academically and critically reviewed the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
