Abstract
Introduction:
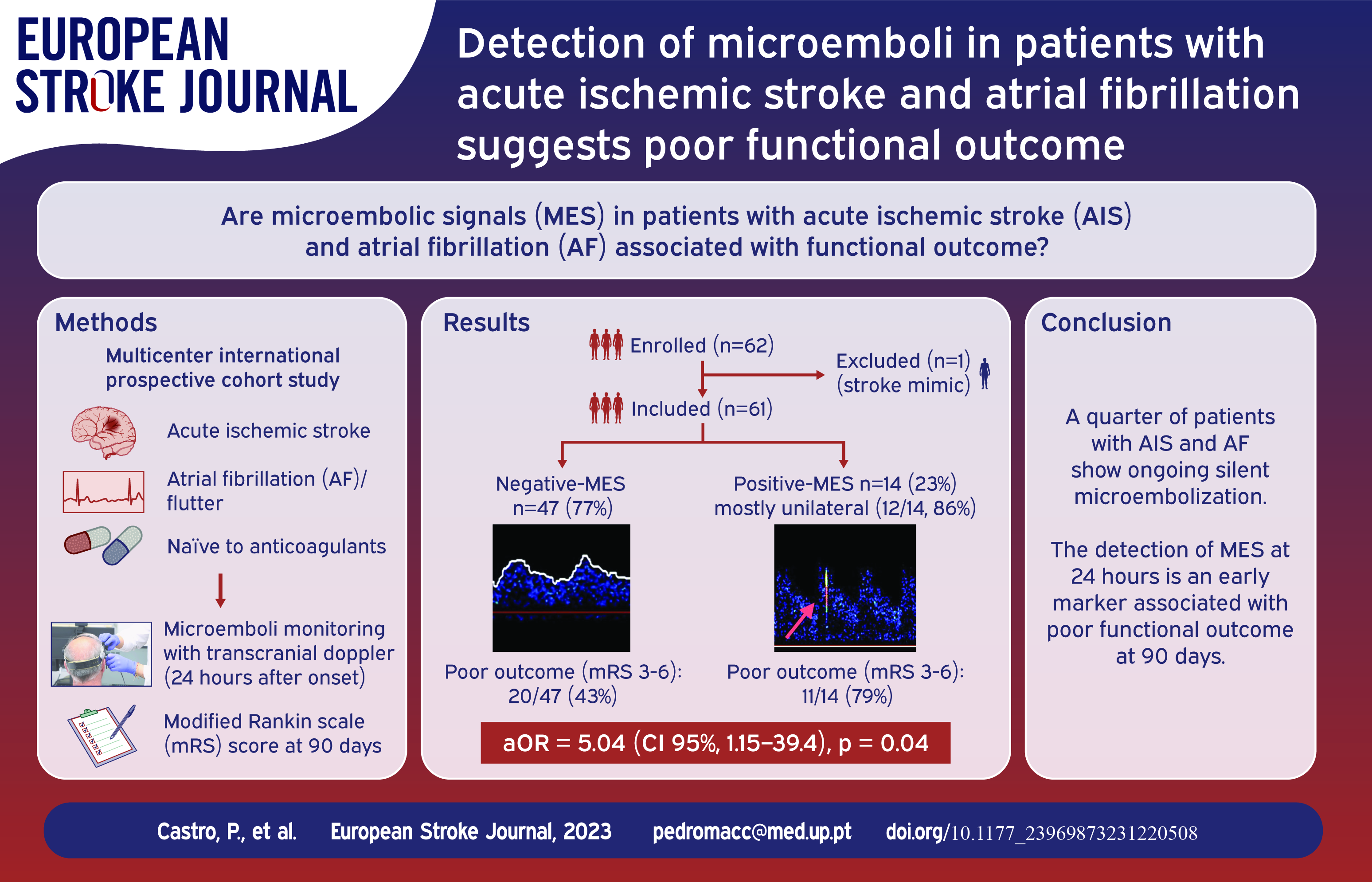
We investigated the burden of microembolic signals (MES) in patients with acute ischaemic stroke (AIS) and atrial fibrillation (AF), assessing their impact on functional outcomes.
Patients and methods:
This multicentre international prospective cohort study involved patients with AIS and either a known or newly diagnosed anticoagulant-naïve AF. All centres utilised the same transcranial Doppler machine for 1-h monitoring with bilateral 2 MHz probes within 24 h of symptom onset. Recordings underwent MES analysis by a blinded central reader. The primary objectives were to ascertain the MES proportion and its association with functional outcomes assessed by the modified Rankin scale (mRS) score at 90 days.
Results:
Between September 2019 and May 2021, we enrolled 61 patients, with a median age of 78 years (interquartile range 73–83) and a median stroke severity score of 11 (interquartile range 4–18). MES were observed in 14 patients (23%), predominantly unilateral (12/14, 86%), with a median rate of 6 counts/hour (interquartile range 4–18). MES occurrence was higher post-thrombectomy and among those with elevated brain natriuretic peptide levels (p < 0.05). A worse mRS score of 3–6 was more frequent in patients with MES, occurring in 11/14 (79%), compared to those without MES, 20/47 (43%), with an adjusted odds ratio of 5.04 (95% CI, 1.15–39.4), p = 0.04.
Conclusions:
Nearly a quarter of patients with AIS and AF exhibited silent microembolization after the index event. Detecting MES within 24 h post-stroke (using transcranial Doppler) could signify a marker of poor functional outcomes. Subsequent trials will assess if very early antithrombotic treatment might enhance outcomes in this highly selective group of cardioembolic stroke patients. (Clinicaltrials.gov ID: NCT06018090).
Keywords
Introduction
In a patient with atrial fibrillation (AF), cerebral infarction typically arises from the embolisation of intracardiac thrombi, often formed within the left atrial appendage. The re-embolisation of residual thrombi likely contributes to the heightened recurrence of stroke in the initial hours following the index event. 1 In addition to the occurrence of new clinical events, it is conceivable that this factor contributes to new brain lesions, potentially diminishing the likelihood of achieving a favourable clinical recovery.
Recently, a large multicentre trial demonstrated a tendency towards a lower incidence of recurrent ischaemic stroke or systemic embolism within 30 days following a stroke with the early implementation of anticoagulation. However, certain limitations in the approach persist. 2 These limitations arise from the observation that stroke recurrence was not notably reduced, suggesting that additional factors might contribute to recurrent embolism. The coexistence of atherosclerosis doubles the risk of a subsequent stroke and might exhibit a more favourable response to antiplatelet therapy. 3 Other considerations persist due to potential individual variations in the effectiveness of different anticoagulants, which might account for some of the variability in outcomes.4,5 The influence of recurrent embolism on the functional outcomes of post-stroke patients highlights the significance of investigating prevention strategies and enhancing long-term prognostic methods to stratify patient risk and improve functional outcomes.6,7
For these reasons, evaluating a patient’s early embolisation activity would be beneficial in optimising management and preventing a poorer outcome. Transcranial Doppler (TCD) provides a unique opportunity to detect embolic material in the cerebral circulation at the bedside in a non-invasive manner. 8 Microembolic signals (MES) have been extensively studied in cerebrovascular disease, primarily in association with atherosclerotic conditions. 9 The estimation that MES are detectable in 15% of stroke patients due to AF is outdated, especially given the introduction of new acute treatments. Additionally, in this study, although patients taking anticoagulants at the onset were excluded, monitoring did not commence within the initial 24 h. 10
Previous studies involving patients after non-cardioembolic aetiology stroke have indicated that the presence of MES during the acute phase and following endovascular treatment correlates with recurring events, increased mortality rates, and subsequently, poorer functional outcomes 90 days post-stroke.11,12
Hence, this multicentre international prospective study examined the incidence of MES and their effect on the clinical outcome in patients with acute ischaemic stroke caused by AF.
Methods
Data availability
The data and SPSS syntax code that support the findings of this study are available from the corresponding author upon reasonable request.
Patients and participating centres
This is a multicentre international prospective cohort study involving central reading of MES events by an expert blinded to clinical data and outcomes. Five European comprehensive stroke centres participated in this study: University Hospital Centre of São João/Faculty of Medicine, University of Porto, Portugal; Hospital Centre Zagreb, Croatia; University of Padua, School of Medicine, Italy; Centre for Health Research, Faculty of Medicine, University of Ostrava, Czech Republic; and Justus-Liebig-University Giessen, Germany. The inclusion criteria comprised acute ischaemic stroke admitted to the Stroke Unit, a history of AF/flutter, or a new diagnosis of AF at the time of stroke, with the ability to be monitored within 24 h after symptom onset. Exclusion criteria included the use of anticoagulants at stroke onset, prosthetic valves, inadequate temporal bone window, and uncooperative patients.
Clinical, laboratory, and radiological assessment
Demographic and clinical data, encompassing vascular risk factors, medications, and prior cardiovascular disease, were documented upon admission. Additionally, blood pressure (BP), serum glucose, cholesterol levels, National Institutes of Health Stroke Scale (NIHSS) scores, reperfusion treatment (thrombolysis, endovascular procedures), and post-treatment grade 2b-3, as per the modified Thrombolysis in Cerebral Infarction scale (mTICI), were recorded. 13 Experienced stroke physicians reviewed radiological and ultrasound studies to identify the presence of carotid or middle cerebral artery stenosis ⩾50%. All clinical data were stored in a decentralised electronic platform (Castor®’s).
Microembolic signals detection
The presence and rate of MES were assessed by 1 h of TCD monitoring (Doppler Box X, DWL, Sipplingen, Germany) within 24 h from the last known well time. Bilateral M1 segments of middle cerebral artery were insonated at a single depth with 2 MHz transducers secured by a probe-holder with a sample volume of 8 mm and low gain. 8 MES was defined according to previous consensus 8 as a signal with (1) duration < 300 ms, (2) amplitude > 3 dB with respect to underlying flow signal, (3) unidirectionality and (4) typical audible sound (‘snap’ and/or ‘chirp’). Recordings were analysed by a single experienced reader who was blinded to clinical data and outcomes. Patients with one or more MES detected were assigned to the MES-positive group.
Clinical endpoints
The primary objectives were to ascertain the proportion of patients with MES and to assess the association of MES with poor functional outcomes, evaluated using the modified Rankin scale (mRS) scores of 3–6 (indicating dependency or mortality) versus 0–2 (indicating independence) at 90 days. As secondary endpoints, we compared the functional outcomes between MES-positive and MES-negative groups across the entire scale using ordinal shift analysis. Additionally, we explored the relationship between MES and clinical, radiological, and laboratory characteristics.
We obtained approval from the local ethics committees of all participating institutions. Written informed consent was acquired from all participants or their guardians.
Statistical analysis
Sample size calculation
The largest study on non-valvular AF 10 estimated a 15% prevalence of MES within 24 h of stroke. However, more recent publications in the context of endovascular treatment reported higher proportions ranging from 31% to 61%.11,12,14 We estimated a proportion of 35% of patients being MES-positive, with a power of 80%, α = 0.05, and a marginal error of 10%. Additionally, based on the study by Farina et al. 11 77% of MES-positive patients experienced long-term disability after stroke (mRS 3–6), compared to only 34% of MES-negative patients. Therefore, to achieve a primary functional outcome with an odds ratio of 6.5, α = 0.05, and power of 80%, we calculated a sample size of 54. To account for a 10% loss to follow-up, we aimed to enrol a minimum of 60 patients. Sample size estimates were computed using G*Power version 3.1.9.7.
Descriptive and outcome analysis
The normality of continuous variables was assessed using the Kolmogorov–Smirnov test. Baseline characteristics between patients in the MES-positive and MES-negative groups were compared using chi-square/Fisher exact tests for categorical variables and Mann-Whitney tests for continuous variables. Clinical outcomes were compared between the MES-positive and MES-negative groups. Logistic regression was employed to generate odds ratios (OR) and 95% confidence intervals (CI) for poor outcome (mRS 3–6) versus good outcome (mRS 0–2) at 90 days. Additionally, outcomes were assessed using ordinal shift analysis modelling across all levels of the mRS scale after verifying conformity to the assumption of a common proportional odds.
We conducted two multivariate models to adjust analyses for all outcomes. The first model adjusted for independent variables associated with the outcome in univariate analysis, including age and NIHSS score, as major predictors of outcome. The second model adjusted for variables that exhibited imbalance between MES groups (Table 1 with p < 0.30) using a propensity score matching approach: a single propensity score between 0 and 1 was computed, encompassing brain natriuretic peptide, previous antiplatelet drugs, NIHSS, thrombectomy, anterior circulation, and carotid or middle cerebral artery ⩾50% stenosis. We performed a sensitivity analysis excluding patients with large vessel lesions and adjusted for all outcomes in two multivariate models (Supplemental Table 2).
Demographic, clinical and radiological characteristics of all patients and differences between MES-positive and MES-negative groups.
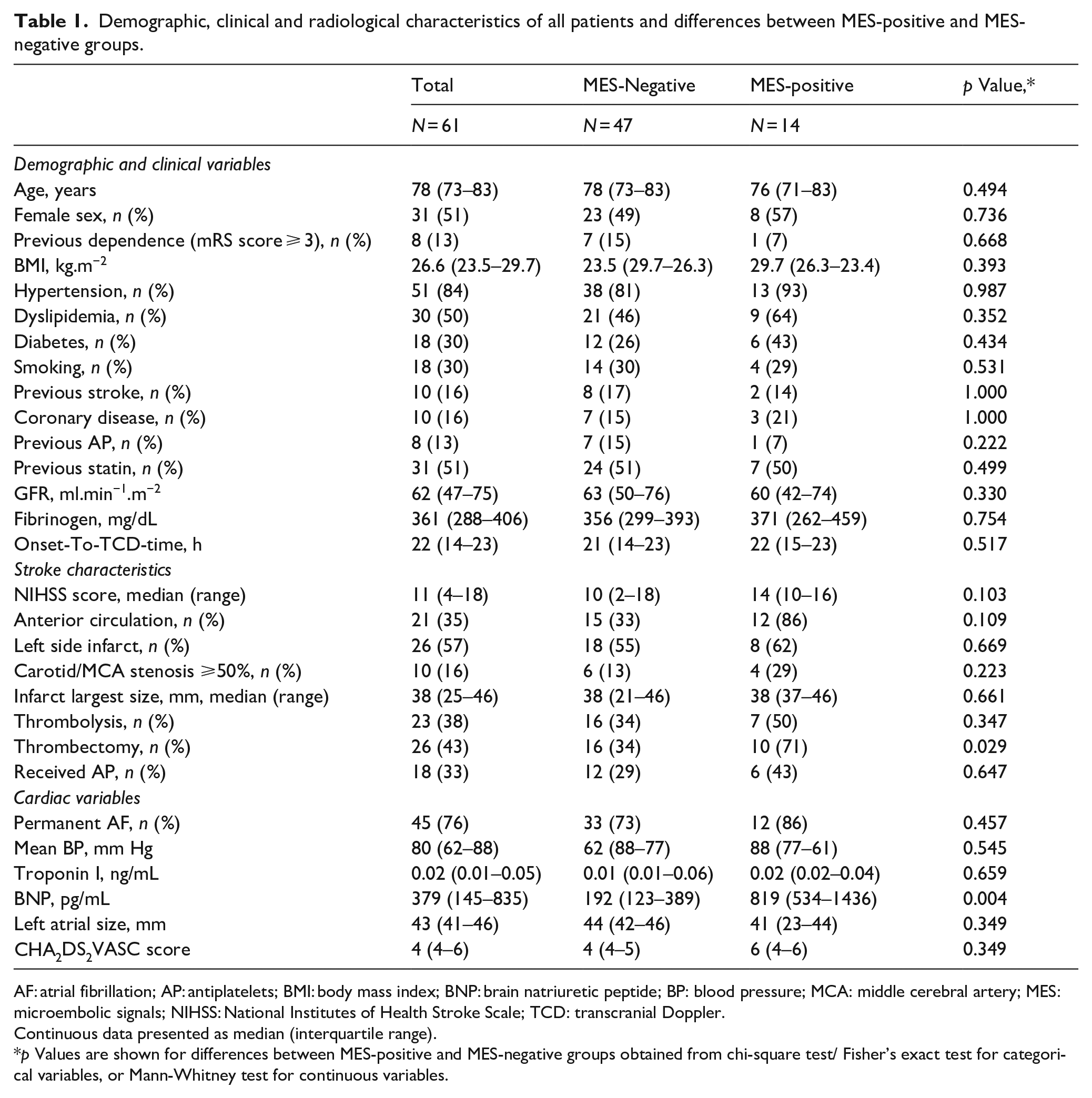
AF: atrial fibrillation; AP: antiplatelets; BMI: body mass index; BNP: brain natriuretic peptide; BP: blood pressure; MCA: middle cerebral artery; MES: microembolic signals; NIHSS: National Institutes of Health Stroke Scale; TCD: transcranial Doppler.
Continuous data presented as median (interquartile range).
p Values are shown for differences between MES-positive and MES-negative groups obtained from chi-square test/ Fisher’s exact test for categorical variables, or Mann-Whitney test for continuous variables.
All statistical analyses were conducted using IBM SPSS Statistics for Windows, version 28. Statistical significance was determined at p < 0.05.
Results
Between September 1, 2019, and May 15, 2021, we screened 491 patients and excluded 283 patients who arrived more than24 h after symptom onset. The study period coincided with the COVID-19 pandemic, and therefore, the centres faced restrictions on the use of monitoring devices and longer intervals between symptom onset and entry into the unit to facilitate monitoring. Out of 208 potential eligible patients for the study, we excluded 15 due to anticoagulant use, three non-cooperative patients, five with prosthetic valves, and 52 without a temporal bone window. One patient with a stroke mimic (epilepsy) was excluded from the final analysis, and we enrolled 62 patients (Figure 1).
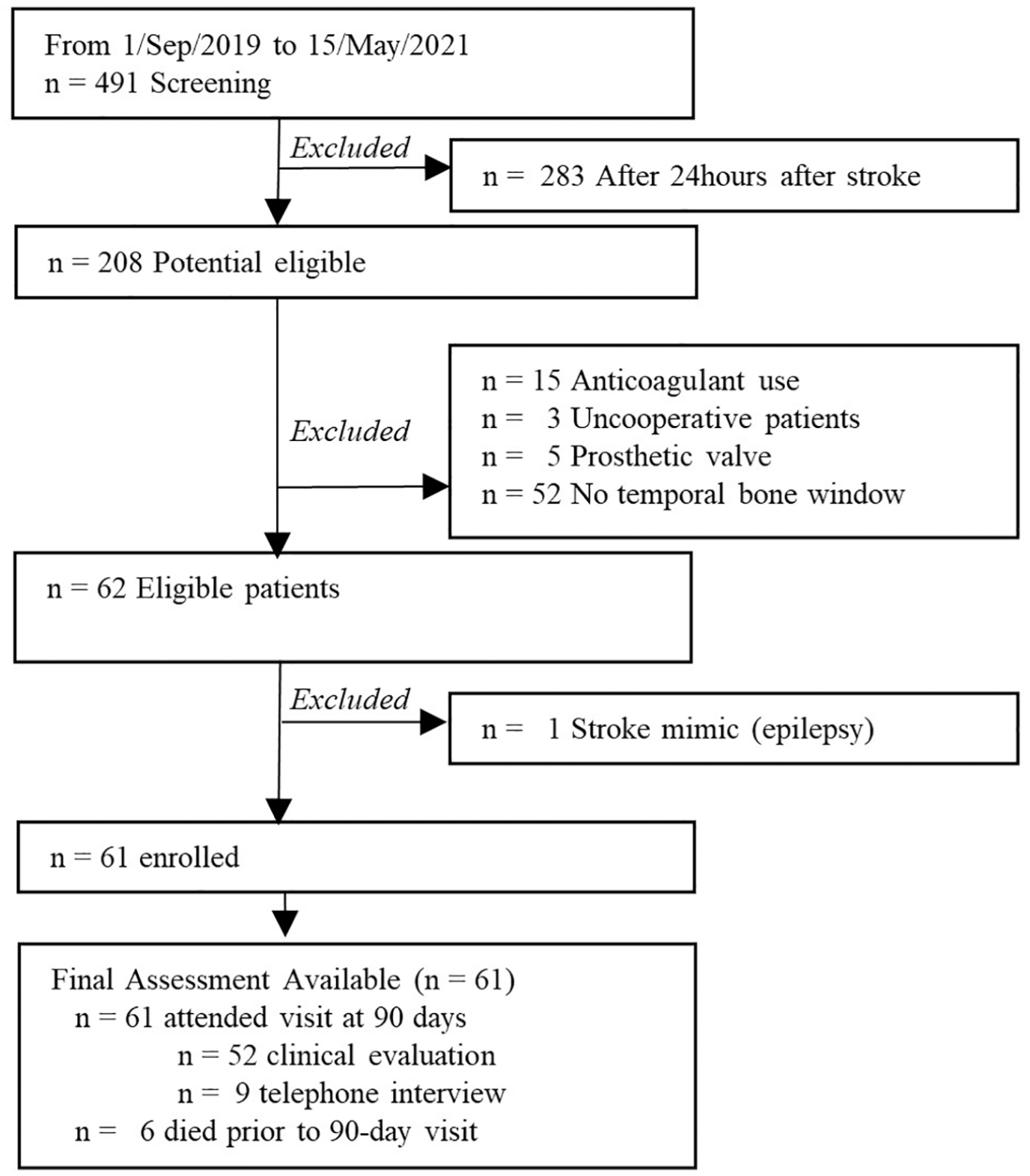
Study flow chart.
MES were present in 14 patients (23%), mostly unilateral (12/14, 86%), with a median count rate of six counts per hour (interquartile range 4–18). Among the unilateral MES-positive cases, eight occurred on the symptomatic side.
MES occurrence was significantly associated with thrombectomy and higher brain natriuretic peptide levels (Tables 1 and 2, p < 0.05). Among the 10 cases of MES-positive associated with thrombectomy, eight were on the same side as the stroke, one was bilateral, and one was detected contralateral to the stroke, p = 0.034. MES occurred more frequently with concomitant carotid or middle cerebral artery ⩾50% stenosis, higher baseline NIHSS, and previous use of antiplatelet drugs, although these associations were statistically nonsignificant (Table 1).
Outcome measures based on MES positive or negative detection.
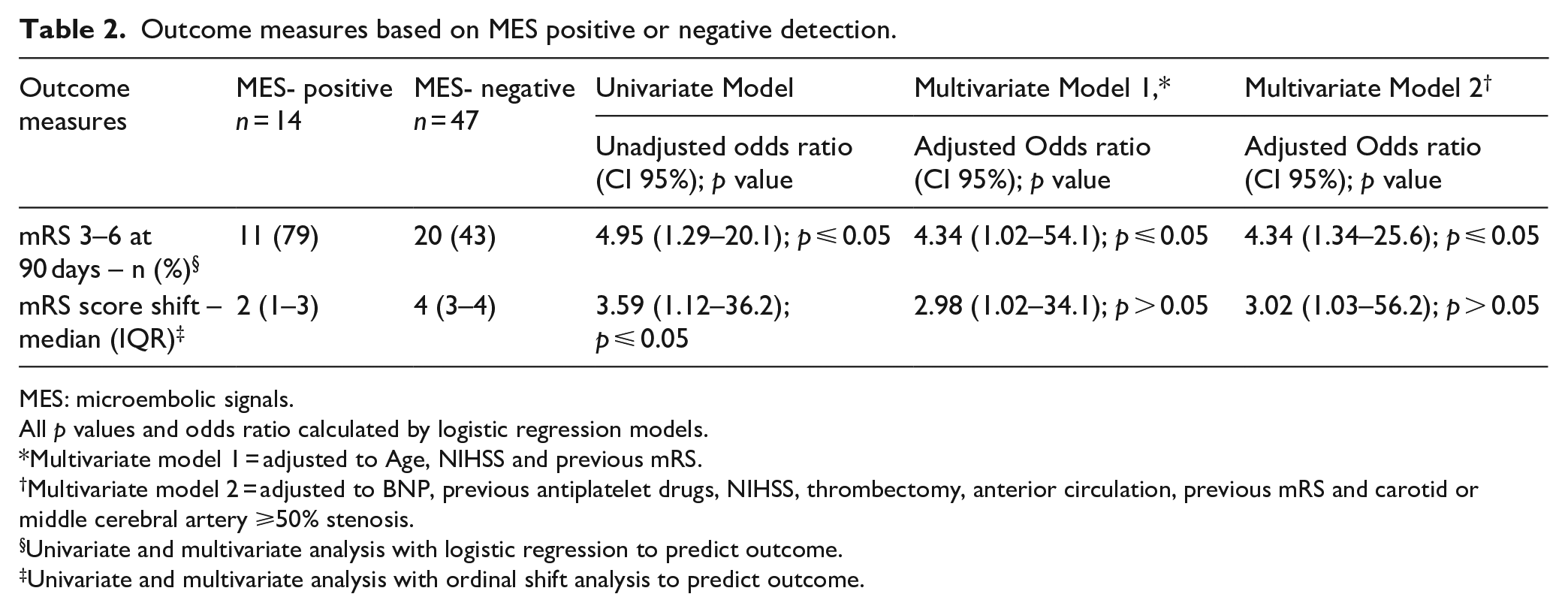
MES: microembolic signals.
All p values and odds ratio calculated by logistic regression models.
Multivariate model 1 = adjusted to Age, NIHSS and previous mRS.
Multivariate model 2 = adjusted to BNP, previous antiplatelet drugs, NIHSS, thrombectomy, anterior circulation, previous mRS and carotid or middle cerebral artery ⩾50% stenosis.
Univariate and multivariate analysis with logistic regression to predict outcome.
Univariate and multivariate analysis with ordinal shift analysis to predict outcome.
Table 2 presents the outcome comparisons between the MES-positive and MES-negative groups. The distribution of patients in each mRS category according to MES detection is illustrated in Figure 2. Worse clinical outcomes, defined as mRS 3–6, were more frequent in patients with MES [11/14 (79%)] than in those without MES [20/47 (43%)], adjusted odds ratio = 5.04 (95% CI, 1.15–39.4), p = 0.04 (Table 2). The presence of MES was associated with a poorer functional outcome at 90 days, even after adjusting for biased characteristics related to MES occurrence, age, and stroke severity (Table 2).
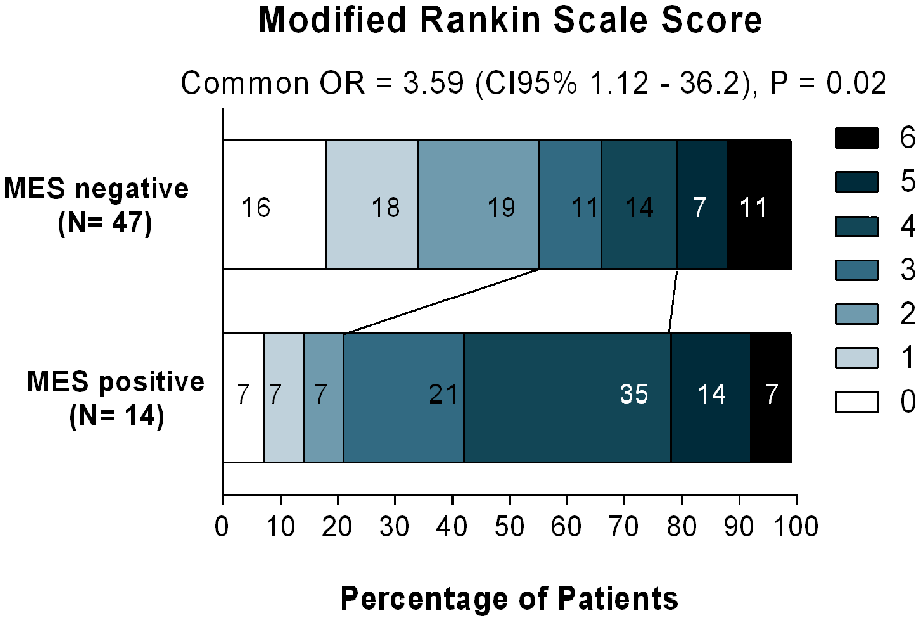
Distribution of disability scores at 90 days, as per the modified Rankin scale (range 0–6, higher scores indicating more severe disability), among patients in the MES-positive group compared to those in the MES-negative group. The figures within the bars denote the percentages of patients with each score. Statistical analysis details are provided in Table 2, including the Common Odds Ratio (OR) representing the shift analysis.
The ordinal shift analysis across mRS categories at 90 days also showed a significant difference between the MES-positive and MES-negative groups, indicating a shift towards worse functional classes at 90 days (adjusted OR = 3.59, 95% CI 1.12–36.2, p = 0.02) (Table 2).
Excluding patients with large vessel lesions, the presence of MES was still associated with a poorer functional outcome at 90 days, although without statistical significance in the multivariate model adjusted for unbalanced baseline differences between MES groups by a probability score (Supplemental Table 2).
Oral anticoagulation during follow-up did not independently associate with the outcome, but this could be due to our inability to access the timing of treatment initiation and potential interference with MES status and its consequences during follow-up.
Discussion
In this multicentre observational prospective study involving patients with acute ischaemic stroke and anticoagulant-naive atrial fibrillation, MES were detected through transcranial Doppler monitoring in nearly one-quarter of patients within the initial 24 h after symptom onset. Notably, the presence of MES significantly increased the risk of experiencing a poor functional recovery by 5-fold at 90 days.
The study’s primary strength lies in the meticulous patient selection criteria, specifically focusing on individuals with no anticoagulation, conducting recordings within 24 h after stroke onset, and excluding other stroke aetiologies. Rigorous control of potential technical confounders was implemented, including the use of a single type of ultrasound device, external offline reading, involvement of a single expert reader for MES recording, and pre-defined insonation parameters. This approach mitigated most of the confounding factors related to MES monitoring, particularly in patients with atrial fibrillation. 15
Burden of microemboli in AF and outcomes
To the best of our knowledge, this is the inaugural study investigating MES detection after acute ischaemic stroke, specifically focusing on anticoagulation-naïve AF patients, with the primary endpoint being clinical outcome. Previous studies with diverse clinical cohorts and technologies exhibited a wide variability in positive MES counts, ranging between 17% and 40% 16 According to our approach, the MES burden accounted for 23% of the total cohort. This percentage aligns with the findings of a recent meta-analysis encompassing all MES studies in acute stroke. 17 However, it must be noted that the majority of previous studies involving patients with AF did not account for the use of anticoagulants, which significantly influences the occurrence of MES.
Our data provide evidence that MES indicate a poor functional prognosis after a stroke caused by AF (Table 2). This association was significant when considering dichotomised mRS and in shift analysis across the entire scale (Figure 2). Previous studies involving patients with acute stroke of various aetiologies and those undergoing thrombectomy also observed increased mortality in cases with MES activity. 18
Several pathophysiological explanations might account for this association. Microembolic particles, as identified by ultrasound, represent the common final step in a complex sequence of preceding cellular and molecular pathways. They may indicate activated stages in the clotting system and highlight tissue surfaces vulnerable to thrombosis. 19 Indeed, MES not only serve as a robust surrogate marker for thrombogenicity but also accurately reflect actual embolic events. Interestingly, in our MES-positive cases, BNP values indicating cardiac insufficiency were significantly elevated. This may represent an additional crucial pathophysiological factor linking microembolism to a worse clinical outcome. 20
Silent embolism occurring within the initial hours after an index stroke can be linked to new ischaemic lesions and/or an expansion of the final infarct area. 21 This often results from intracranial vessel obstruction, inadequate collateral flow, and decreased embolic washout. 22 For example, Kang et al. demonstrated that 23% of patients with AF exhibited silent new ischaemic lesions on MRI conducted 5 days after a stroke. 21 MES detected through TCD monitoring can serve as a surrogate for future ischaemic lesions and subsequent ischaemic events.11,12,17 New cerebral ischaemic lesions, even if not linked to a sudden clinical event, predict new neurological deficits, including cognitive impairment. 23
We must acknowledge that the size of the study cohort was relatively small, resulting in large confidence intervals for the calculated odds ratio. This limited number precluded more robust sensitivity analyses to complement the multivariate analysis.
Numerous studies have been unable to find evidence supporting the association between MES and poor outcomes following a stroke. 17 However, many of these studies encompassed various stroke types, often including patients with small vessel disease, which clearly doesn’t align with the recording of microembolism in the primary stem of the middle cerebral artery. Most cases that tested positive for MES in prior studies were likely associated with carotid atherosclerosis, and the prognosis for these patients significantly differs from those with atrial fibrillation. Hence, research investigating the prognostic value of MES should distinctly differentiate between different stroke aetiologies and endeavour to minimise the inclusion of ‘mixed’ cohorts.
The source of MES and the cause of stroke
Our study was not intended to definitively identify the source of microemboli but could offer intriguing hypotheses. Among patients with atrial fibrillation, undissolved clots might persist within the atria and atrial appendages. A substantial presence of intracardiac clots could potentially cause blockages in major intracranial vessels, leading to more severe strokes. 17 Hence, MES showed a significant association with a higher NIHSS and mechanical thrombectomy.
Among our sample of 61 patients, 26 underwent thrombectomy (43%) (Table 1) and conducting thrombectomy on the same side was significantly linked to unilateral MES, regardless of the final TICI score. The presence or absence of a successful TICI score did not show an association with the presence of MES in our cohort. While the connection between MES and recanalisation failure might be questioned, our study failed to establish this link. Past research indicated an association between TICI 2a and MES; however, a recent study involving only TICI 2b-3 cases did not find a similar association with MES.11,12
Elevated BNP values associated with MES support this idea (Table 1). 20 However, the size of the left atrium, a more reliable indicator of advanced atrial cardiopathy, did not show a significant association with MES activity. It’s worth noting that a recent large multicentre trial demonstrated that anticoagulation provides no additional benefit over aspirin in patients with embolic stroke of undetermined source, despite the presence of atrial cardiopathy. 24
The CHA2DS2VASC score, a well-validated risk stratification tool predicting cardioembolism, also showed no association with MES activity. This could be explained by the fact that the CHA2DS2VASC score is not designed for assessment in the context of an acute stroke event, whereas our study protocol required MES monitoring within 24 h after the index event.
The presence of concomitant atherosclerotic lesions along with endovascular treatment might serve as an alternative source of MES, distinct from the heart. This study reveals that MES were more frequently detected in patients who had undergone endovascular therapy (70%vs 34%, p = 0.029, Table 1). The notably high rate of MES could be attributed to vascular/endothelial damage incurred during this procedure.11,16,25,26 In our study, the rate of MES was higher among patients with co-existing atherosclerotic carotid or intracranial stenosis on the same side as the stroke. Interestingly, patients who were already using antiplatelet drugs at the time of the stroke tended not to exhibit MES. The documented efficacy of aspirin and clopidogrel in reducing emboli originating from atherosclerotic wall disease comes from extensive large-scale randomised and case-control trials. 27
The risk of early recurrent embolism in stroke patients with AF is approximately 20 times higher than the risk of haemorrhagic transformation, 2 further emphasising the need for early treatment. Our findings support this conclusion and highlight the potential of MES monitoring to identify patients at a higher risk of a poor functional outcome.
Limitations
The commonly accepted monitoring time for detecting MES is 1 h. 8 Extended TCD monitoring might have increased detection rates, but we believe it wouldn’t have significantly altered the observed relationship with the outcome. Only the M1 segment of the MCA was monitored, potentially missing embolisms to other vascular territories, although vertebrobasilar strokes tended to be less MES-positive (Table 1). Monitoring MES with TCD, a non-invasive method, relies on the accessibility of the temporal bone window. In our sample, 25% of eligible patients had an insufficient temporal window, which is a limitation of our study.
Differences in baseline characteristics between MES groups might have biased our ability to predict outcomes. The MES-positive group tended to exhibit higher NIHSS scores and were more frequently directed to endovascular treatment. Endovascular procedures encompass various complications, such as the previously mentioned vascular/endothelial damage, potentially influencing the recorded MES rate.11,16,25,26 The variables NIHSS and thrombectomy were adjusted for in model 2 through propensity score matching to compensate for this bias (Table 2). Only 26 patients underwent thrombectomy, and a successful or unsuccessful TICI score was not found to be associated with the presence of MES in our cohort. Although adjusted in model 2 for the NIHSS of the sample, it is important to note that the average stroke severity in our sample was considerably high (NIHSS = 11). This needs to be considered when comparing it to the literature.
Conclusions
Nearly a quarter of patients with atrial fibrillation show evidence of microembolization within the first 24 h after an acute ischaemic stroke. The detection of MES within 24 h after a stroke, using transcranial Doppler, may serve as a marker for poor functional outcomes. Future studies will ascertain whether very early antithrombotic treatment can enhance the outcome in this highly selected group of cardioembolic stroke patients.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231220508 – Supplemental material for Detection of microemboli in patients with acute ischaemic stroke and atrial fibrillation suggests poor functional outcome
Supplemental material, sj-docx-1-eso-10.1177_23969873231220508 for Detection of microemboli in patients with acute ischaemic stroke and atrial fibrillation suggests poor functional outcome by Pedro Castro, J Ferreira, Branko Malojcic, Danira Bazadona, Claudio Baracchini, Alessio Pieroni, David Skoloudik, Elsa Azevedo and Manfred Kaps in European Stroke Journal
Footnotes
Acknowledgements
We extend our Gratitude to Mark Brandter and Helga Reckhaus for their exceptional assistance in establishing the decentralised digital platform and monitoring data hub for blinded reading.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ferreira, J. was funded by the Fundação para a Ciência e Tecnologia, I.P.(UI/BD/150648/2020), Av. D. Carlos I, 126 1249-074 Lisboa, Portugal.
Ethical approval
The ethics committee of Centro Hospitalar Universitário de São joão approved this study (REC number: 362/19)
Informed consent
Written informed consent was obtained from all subjects before the study.
Guarantor
P Castro
Contributorship
All authors should have made substantial contributions to all of the following:
P Castro: (1) the conception and design of the study, analysis and interpretation of data, (2) drafting the article and revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
J Ferreira: (1) acquisition of data, analysis and interpretation of data, (2) drafting the article and revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
B Malojcic: (1) acquisition of data, or analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
D Bazadona: (1) acquisition of data, or analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
C Baracchini: (1) acquisition of data, or analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
A Pieroni: (1) acquisition of data, or analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
D Skoloudik: (1) acquisition of data, or analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
E Azevedo: (1) analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
M Kaps: (1) acquisition of data, or analysis and interpretation of data, (2) revising it critically for important intellectual content, and (3) final approval of the version to be submitted.
Moreover, all authors have approved the submitted version of the manuscript.
Trial registration
Clinicaltrials.gov ID: NCT06018090
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
